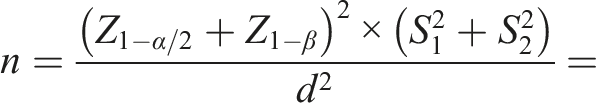Abstract
Introduction
Vitamin D overdose, often stemming from excessive supplementation rather than dietary intake. It has been associated with various conditions such as cardiovascular disorders. This study aimed to investigate the effects of vitamin D toxicity on cardiac tissue.
Methods
Sixteen Wistar rats (250 ± 50 g) were randomly divided into two groups: the control group and the high-dose vitamin D group (40,000 IU/kg). Vitamin D was administered via gavage for 8 weeks. The expression of sirtuin 1 (SIRT1), the peroxisome proliferator-activated receptor gamma co-activator 1-alpha (PGC1-α), B-cell lymphoma 2 (Bcl-2), and Bcl-2-associated X protein (Bax) genes in cardiac tissue was evaluated. Blood samples were analysed for lactate dehydrogenase (LDH) levels. Moreover, oxidative stress markers, including malondialdehyde (MDA) and superoxide dismutase (SOD), were measured in tissue samples. Histopathological evaluations were also conducted.
Results
The expression of SIRT1, PGC1-α, Bcl-2, and the SOD levels were significantly decreased in the vitamin D-treated group. In addition, the values indicated a significant increase in the expression of Bax along with LDH and MDA levels in the vitamin D-treated group compared to the control group.
Discussion
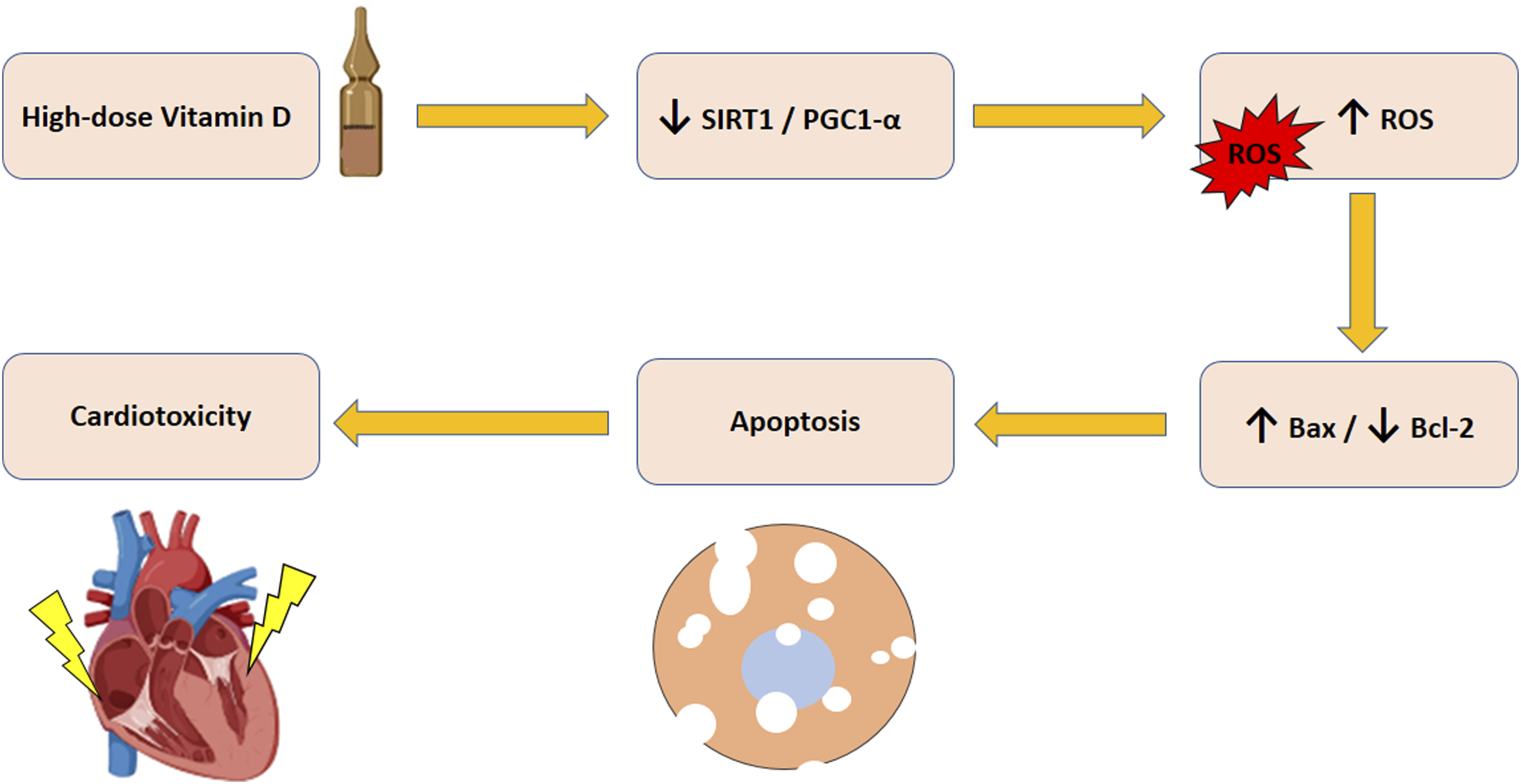
Long-term administration of high-dose vitamin D significantly increased oxidative stress and apoptosis in cardiac tissue, likely mediated by the SIRT1/PGC1-α pathway.
Graphical abstract
The illustration of the suggested mechanism underlying high-dose vitamin D-induced cardiotoxicity.
Introduction
Vitamin D is a fat-soluble vitamin that plays a critical role in the regulation of calcium and phosphorus levels, immune system modulation, and preservation of bone health. 1 It has been indicated a significant increase in vitamin D supplementation during the COVID-19 pandemic. Excessive vitamin D intake leads to vitamin D toxicity 2 Hypervitaminosis D. is associated with hypercalcemia, vascular calcification, and cardiac disorders.2–5 The growing prevalence of vitamin D toxicity underscores the urgent need to investigate its underlying pathophysiological mechanisms and to establish safe supplementation thresholds. Despite the growing clinical concern, the precise molecular mechanisms of vitamin D-induced cardiotoxicity remain poorly understood. Among potential regulatory targets, the SIRT1/PGC1-α signaling axis has emerged as a central factor in mitochondrial function, redox homeostasis, and apoptosis. SIRT1, a NAD+-dependent deacetylase, activates PGC1-α, which in turn promotes mitochondrial biogenesis and antioxidant defense. Dysregulation of this axis is implicated in cardiac ageing and heart failure.6,7 However, its involvement in vitamin D cardiac toxicity remains insufficiently explored. Thus, investigating this pathway offers a novel mechanistic perspective on vitamin D cardiac toxicity.
Sirtuin 1 (SIRT1) and the Peroxisome Proliferator-Activated Receptor Gamma Coactivator 1-alpha (PGC1-α) are crucial for maintaining mitochondrial integrity and function. 8 SIRT1 contributes to regulating mitochondrial biogenesis, energy metabolism, and cellular longevity. 9 PGC1-α plays a key role in the regulation of energy production and oxidative metabolism.10–12 In addition, the regulation of apoptosis in cardiac cells is mediated by Bcl-2-associated X protein (Bax) and B-cell lymphoma 2 (Bcl-2). Bax facilitates cell death by disrupting the mitochondrial outer membrane, leading to cytochrome c release and apoptosis.13,14 Bcl-2 mitigates the effects of Bax by maintaining mitochondrial membrane integrity and preventing apoptosis.15,16 The regulation of pro- and anti-apoptotic signals is crucial for cardiac cell survival in stress conditions. 17 SIRT1 plays a crucial role in the regulation of apoptosis through its effect on key cellular pathways. SIRT1 downregulation is associated with apoptosis by promoting Bax and suppressing Bcl-2. It leads to mitochondrial dysfunction and oxidative stress, ultimately cell death. 18 Oxidative stress and apoptosis contribute to cardiotoxicity. SIRT1 has antioxidant and anti-apoptotic effects.19,20 It enhances antioxidant defense through the production of antioxidant enzymes, such as superoxide dismutase (SOD).21,22 Furthermore, it exerts its anti-apoptotic effects by reducing the Bax/Bcl-2 ratio through deacetylation of PGC1-α.23–25 SIRT1 plays a key role in the reduction of cardiotoxicity by inhibiting oxidative stress, inflammation, and apoptosis.26,27 Given the importance of these molecular pathways in cardiotoxicity, it is imperative to understand how high-dose vitamin D-induced cardiotoxicity may interfere with these mechanisms. This study aims to investigate the toxic effects of high-dose vitamin D on the expression of SIRT1, PGC1-α, Bax, and Bcl-2 in the heart. Although vitamin D toxicity has been explored in clinical and animal models, limited studies have focused on the molecular mechanisms involved in cardiac tissue damage. Specifically, the role of SIRT1/PGC1-α signaling axis remains under-investigated in the context of vitamin D-induced cardiotoxicity.9,28,29 The current study is among the first to evaluate the impact of high-dose vitamin D on this pathway. This research provides novel insights into the cardiotoxic effects of excessive vitamin D and highlights potential molecular targets for intervention.
Methods
The animals were transferred to the animal housing facility 1 week before the initiation of treatment to allow for acclimatization. The animals were housed in a temperature-controlled room (25 ± 2°C) with 50% relative humidity, under a 12-h light/dark cycle, with ad libitum access to food and water. All experimental procedures were conducted in accordance with the ARRIVE guidelines and institutional regulations for the care and use of laboratory animals. The Ethics Committee of Hamadan University of Medical Sciences (Approval No. IR.UMSHA.REC.1402.678) approved the protocol for the care and use of laboratory animals. Animals were monitored daily for signs of distress and their food, water, and bedding. Humane endpoints were predefined as severe respiratory distress (>60 breaths/min), vocalisation, inactivity, body weight loss of greater than 20% of the previous maximum, aggression, biting, scratching, pain signs and inability to eat/drink; no rats met these criteria.
The sample size was determined using the equation method based on prior studies.
30
Where S12 and S22 are means.
A total of 16 male Wistar rats, weighing 250 ± 50 g, were used in this study. The rats were randomly divided into two experimental groups (n = 8 per group), as follows: 1. Control group: the rats received corn oil via gavage daily for 8 weeks. 2. High-dose vitamin D group: the rats received 40,000 IU/kg of vitamin D dissolved in corn oil via gavage daily for 8 weeks. Investigators performing molecular, biochemical and histology analyses and data analysis were blinded to group assignments.
The primary stock solution of vitamin D (300,000 IU/kg) was obtained from Dana Pharmacy, Iran, and diluted with corn oil to achieve the target dosage of 40,000 IU/kg. Upon completion of the intervention period, the rats were anesthetized via intraperitoneal injection of 60 mg/kg pentobarbital. 31 The cardiac tissue was perfused with cold normal saline and collected for examination. The relative mRNA expression of SIRT1, PGC1-α, Bax, and Bcl-2 in cardiac tissue was assessed using real-time PCR.
Gene expression analysis by real-time PCR
Sequence of primers.

SIRT1 (Silent information regulator 1), PGC1-α (Peroxisome proliferator-activated receptor gamma co-activator 1-alpha), Bax (Bcl-2-associated X protein), Bcl-2 (B-cell lymphoma 2).
Biochemical analysis
Blood samples were obtained from the inferior vena cava. The rats were anesthetized via intraperitoneal injection of 60 mg/kg pentobarbital. Following blood collection, euthanasia was performed via incision of the chest cavity, and the heart tissue was extracted for molecular, biochemical and histological analysis. The samples were promptly centrifuged, and the serum was isolated for the quantification of lactate dehydrogenase (LDH). Cardiac tissue was homogenised to quantify oxidative stress indicators, including superoxide dismutase (SOD) and malondialdehyde (MDA).
Malondialdehyde (MDA)
MDA was quantified in homogenised cardiac tissue using the ZellBio GmbH kit (Germany), following a colorimetric technique by ELISA reader. The procedure involved preparing reagents, samples, and standards; adding 50 μL of sample and standard to designated test tubes; incorporating 50 μL of reagent R4 into the tubes followed by mixing; adding 1 mL of chromogenic solution; heating the mixture in a boiling water bath for 1 hour; cooling the tubes in an ice bath, followed by centrifugation at 3000-4000 rpm for 10 minutes; and finally, transferring 200 μL of the supernatant to a microplate and measuring absorbance at 535 nm. MDA levels were determined based on the standard OD curve.
Superoxide dismutase (SOD)
SOD activity was quantified in homogenised cardiac tissue using the ZellBio GmbH kit (Germany) by ELISA reader. The kit employs superoxide anions, converting them into hydrogen peroxide and oxygen via enzymatic processes, resulting in a color shift measurable at 420 nm at 0 minute and after 2 minutes. The final SOD activity was calculated using a formula according to the manufacturer’s instructions.
Lactate dehydrogenase (LDH)
Serum LDH was measured using an LDH (DGKC) diagnostic kit from Pars Azmoon Co. (Iran) via a photometric method. The assay is based on LDH catalysing the conversion of pyruvate to lactate. The procedure included mixing pre-prepared reagents 1 and 2 from the kit in a 4:1 ratio. After adding the sample, absorbance was measured at 1 minute and then at 1, 2, and 3-min intervals. The average difference in absorbance was multiplied by 16,030, with measurements taken at 340 nm using an ELISA reader. Results were reported in U/L.
Histopathology
The rats were anaesthetised with pentobarbital for heart tissue sampling, followed by thoracotomy. The samples were washed with normal saline solution and placed in 10% buffered formalin for fixation. Paraffin blocks were prepared, and 5 μm sections were cut from each block. Haematoxylin and Eosin (H&E) staining was performed, and histological evaluations were carried out using a light microscope (Labomed, Los Angeles, USA). Eight rats per group were randomly selected for histological examination. At least 15 distinct non-overlapping fields were assessed per group to evaluate structural alterations in cardiac tissue (400x magnification, scale bar: 50 μm). For blind evaluation, the samples were delivered to the pathologist without group labelling.
Statistical analysis
Data are expressed as mean ± standard error of the mean (SEM). The Kolmogorov-Smirnov test was applied to assess the normality of data distribution. Moreover, Levene’s test was used to evaluate the equality of variances. An independent sample t-test was conducted to compare means between intervention and control groups. Statistical analysis was performed using SPSS version 16.0. A P-value <0.05 was statistically considered significant. 30
Results
The expression of SIRT1
The expression of SIRT1 was significantly decreased in the vitamin D-treated group compared to the control group (p = 0.037, Figure 1). This finding indicates that a high dose of vitamin D leads to a marked reduction in SIRT1 expression, potentially impairing its essential role in the regulation of antioxidant defense mechanisms and maintaining cellular redox balance. Expression levels of SIRT1 in rat cardiac tissue. Data are mean ± SEM, n = 8/group. *p < 0.05 compared to the control group. High-dose vitamin D (VitD).
The expression of PGC1-α
The expression of PGC1-α was markedly diminished in the vitamin D-treated group compared to the control group (p = 0.0003, Figure 2). This reduction suggests that vitamin D therapy adversely affects PGC1-α expression, potentially disrupting mitochondrial biogenesis and energy metabolism. Expression levels of PGC1-α in rat cardiac tissue. Data are mean ± SEM, n = 8/group. ***p < 0.001 compared to the control group. High-dose vitamin D (VitD).
The expression of Bax
Bax gene expression was markedly increased in the vitamin D-treated group compared to the control group. The control group indicated a negligible expression level of Bax, whereas the vitamin D-treated group exhibited a substantial elevation (p = 0.0007, Figure 3(a)). This indicates that vitamin D administration enhances the expression of Bax, a pro-apoptotic marker, thereby signifying an increase in cellular apoptosis. Expression levels of Bax (a) and Bcl-2 (b) in rat cardiac tissue. Data are mean ± SEM, n = 8/group. ***p < 0.001 compared to the control group. High-dose vitamin D (VitD).
The expression of Bcl-2
The expression levels of Bcl-2 were markedly reduced in the vitamin D-treated group compared to the control group (p = 0.0007, Figure 3(b)). This substantial decline suggests that treatment with high-dose vitamin D may significantly suppress the production of anti-apoptotic markers such as Bcl-2, possibly leading to increased susceptibility to cell death in cardiac tissue.
MDA concentration
MDA concentration was significantly elevated in the vitamin D-treated group compared to the control group (p = 0.0002, Figure 4(a)). The increase in MDA level reflects enhanced lipid peroxidation and oxidative stress in the vitamin D-treated group compared to the controls. Cardiac levels of MDA (a) and SOD (b) in rat cardiac tissue and serum levels of LDH in the serum (c). Data are mean ± SEM, n = 8/group. ***p < 0.001 compared to the control group. High-dose vitamin D (VitD).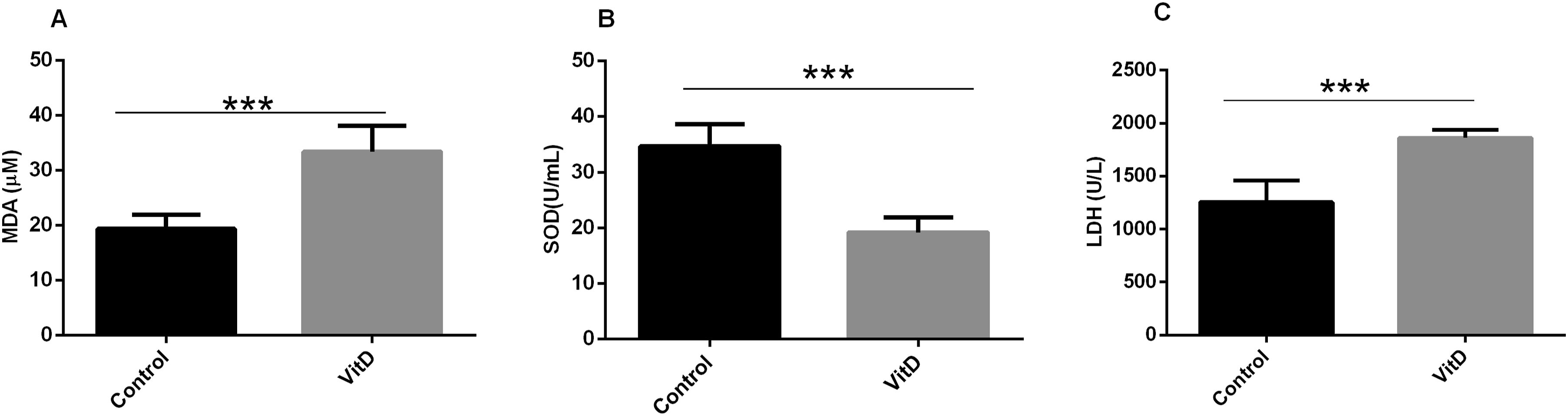
SOD level
SOD levels were markedly decreased in the vitamin D-treated group compared to the control group (p = 0.0002, Figure 4(b)). This decrease indicates that vitamin D administration notably reduces SOD enzyme activity, potentially elevating intracellular oxidative stress.
LDH level
The analysis of LDH levels revealed a significant elevation in the vitamin D-treated group compared to the control group (p = 0.0007, Figure 4(c)). The increased LDH activity suggests that vitamin D treatment may have cellular damage or stress, resulting in enhanced LDH leakage into the extracellular environment.
Histology
The histological analysis of the control group showed that the nuclei of cardiomyocytes were centrally positioned and exhibited an oval morphology (Figure 5(a)). Prolonged administration of vitamin D induced significant disarray and the formation of massive and widespread vacuoles in cardiac tissue (Figure 5(b)). Moreover, both vacuole area and vacuole number in the heart were significantly increased in the vitamin D-treated group compared to the control rats (Table 2). Hematoxylin and eosin (H&E) staining of cardiac tissue. Histopathological alterations in the heart. Control group (a); High-dose vitamin D group (b). Representative images were selected at 400× magnification (Scale bar = 50 µm). The black arrows indicate the cardiomyocytes. Vacuole area and vacuole number in the heart. High-dose vitamin D (VitD). Data are mean ± SEM, n = 8. ***p < 0.001 compared with Control group.

Discussion
The findings of this study provide valuable insights into the toxic effects of vitamin D on cardiac tissue. Our results indicate a significant decrease in the expression of SIRT1 in the vitamin D-treated group compared to the control group. SIRT1 upregulation protects the heart against oxidative stress by enhancing antioxidants such as SOD. 32 Previous studies have similarly reported that SIRT1 downregulation contributes to cellular dysfunction and apoptosis in the lung. 33 In the present study, reduced SIRT1 expression following vitamin D treatment may lead to apoptosis and oxidative stress.
This study also showed that the expression of PGC1-α and SIRT1 in the heart was significantly decreased in the vitamin D-treated group. PGC1-α regulates the expression of antioxidant genes and apoptosis through SIRT1. PGC1-α downregulation leads to oxidative stress and mitochondrial impairment, affecting cardiac cell function and accelerating cardiac damage. 34
A significant reduction in Bcl-2 expression was also observed in the vitamin D-treated group. Bcl-2 is essential for the prevention of apoptosis by inhibiting the release of pro-apoptotic factors from mitochondria. A significant decline in Bcl-2 suggests that vitamin D toxicity may impair anti-apoptotic defenses, increasing cardiac damage. Previous studies have indicated that reduced Bcl-2 expression enhances oxidative stress and apoptosis.35,36 Bax expression was markedly elevated in the vitamin D-treated group. Bax facilitates apoptosis by promoting cytochrome c release.13,14 In the present study, reduced Bax induces apoptosis in cardiac tissue, corroborating previous research.37,38
The observed decrease in SOD activity in the vitamin D-treated group supports the presence of oxidative stress. SOD neutralises superoxide radicals, preventing oxidative damage.39,40 In addition, increased serum LDH and cardiac MDA levels in the vitamin D-treated group indicate cellular damage and lipid peroxidation. Increased MDA and LDH activity reflect oxidative damage, membrane injury and cell death.41–43
Histopathological analysis confirmed these findings, revealing disorganised cardiomyocytes and increased intercellular spaces in the vitamin D-treated group. Previous research has similarly indicated that excessive vitamin D intake leads to tissue damage. 4
In conclusion, excessive vitamin D intake causes apoptosis, oxidative stress, and cardiac damage by the SIRT1/PGC1-α pathway. Our findings may inform strategies for preventing or treating vitamin D-induced cardiac complications. Despite the wide therapeutic index of vitamin D, toxicity can occur with excessive doses. The exact maximum safe daily intake is not fully established, but current guidelines suggest an upper limit of 2000 IU/day. Reported toxic doses range from 50,000 to 2,604,000 IU/day, typically leading to hypercalcemia. 44 Vieth et al. suggest toxicity may begin with chronic intake of about 40,000 IU/day. 44 In this study, treatment with vitamin D at 40,000 IU/day for 8 weeks induced cardiotoxicity, exceeding tolerable thresholds. Further studies with varied doses and measuring serum 25-hydroxyvitamin D are needed to better define human safety limits. 45
Strengths and limitations
The strength of the present study lies in its integrative approach, combining molecular, biochemical, and histological assessments to address vitamin D toxicity in the heart. However, it is important to acknowledge the exclusive use of male Wistar rats as a limitation of the present study. Sex differences in cardiovascular disease are well documented, and effects observed in males may not fully reflect those in females. Due to hormonal factors, including the cardioprotective effects of estrogen, female rats may have distinct baseline susceptibilities to cardiotoxicity. Acknowledging this limitation, we emphasize the need for experimental investigations involving vitamin D-induced cardiotoxicity in both sexes.46,47 Another limitation is the relatively small sample size, which may decrease the generalizability of the results. Moreover, solely measuring MDA and SOD provides an incomplete picture of oxidative stress. Therefore, further studies addressing the effect of excessive vitamin D intake on the heart are needed.
Supplemental Material
Supplemental Material - The role of SIRT1/PGC1-α signaling pathway in high-dose vitamin D-induced cardiotoxicity
Supplemental Material for The role of SIRT1/PGC1-α signaling pathway in high-dose vitamin D-induced cardiotoxicity by Seyed Ershad Hosseini, Elham Shiri, Shiva Nosrati, Khadijeh Ramezani-Aliakbari, Iraj Salehi, Farid Shokri and Fatemeh Ramezani-Aliakbari in Human & Experimental Toxicology
Footnotes
Acknowledgements
The authors would like to thank Hamadan University of Medical Sciences for funding. The authors thank the University for its support.
Ethical considerations
All procedures adhered to ARRIVE guidelines and were approved by the Ethics Committee of Hamadan University (IR.UMSHA.REC.1402.678). All animal procedures were conducted in accordance with the ARRIVE guidelines and approved by the Ethics Committee of Hamadan University of Medical Sciences (Approval No. IR.UMSHA.REC.1402.678). The present study complied with institutional and international guidelines for the ethical treatment of animals. Animals were housed in a temperature- and humidity-controlled environment with a 12-h light/dark cycle and free access to food and water. Humane endpoints were applied to minimize suffering.
Author contributions
SEH and FRA conceived the project. FSH, FRA, SEH, SHN, and KRA contributed to data collection, analysis, figure preparation, and manuscript drafting. FRA, ESH, and IS revised the manuscript. All authors approved the final version and declared that all data was generated independently without third-party assistance.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded by the Vice-Chancellor for Research and Technology, Hamadan University of Medical Sciences (no. 140209288729).
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
Data is available upon reasonable request. The raw histology images, real-time PCR raw cycle threshold (Ct) values, and biochemical assay readings that support the findings of this study are available from the corresponding author upon reasonable request.
Supplemental Material
Supplemental material for this article is available online
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.