Abstract
This study investigated the protective effect of vitamin D against carbon tetrachloride (CCl4)-induced nephrotoxicity in rats. Adult male Wistar albino rats were divided into four groups ((A) control; (B) 10-week exposure to CCl4; (C) 10-week exposure to CCl4 + vitamin D treatment; and (D) 10-week exposure to CCl4 + 12 weeks of vitamin D treatment). The CCl4 dose (1.5 ml kg−1) was injected subcutaneously twice a week, while the 0.5 mg kg−1 dose of vitamin D was administered intraperitoneally every day, as appropriate for each group. Whole animal and kidney weights as well as serum urea, creatinine, and glucose levels were measured. Kidney tissue sections were stained with hematoxylin–eosin, Masson’s trichrome, and periodic acid–Schiff. Tubular and glomerular degeneration were detected in the kidney tissues of CCl4-treated rats, together with dilatation and vacuolization within the tubules and hemorrhage in the intertubular region. In the kidney glomeruli; congestion, atrophy, and adhesion to parietal layer were observed. Tissue disorganization and aggregation of Bowman’s capsules were noted. Mononuclear cell infiltration was observed between the glomeruli and the tubules. In contrast, the kidney sections and functional parameters of vitamin D-treated rats were similar to the controls, suggesting that vitamin D treatment is able to reduce renal damage.
Introduction
Carbon tetrachloride (CCl4) has been manufactured commercially for over 100 years, and although it is not found in the nature, it may be produced in the troposphere by photochemical conversion of chlorinated alkenes. 1,2 Under ambient conditions, it is somewhat volatile, having an odor similar to that of chloroform. 3 Humans can be exposed to CCl4 in drinking water, air, foodstuffs, and industrial waste water since it has been used as a dry cleaning agent, grain fumigant, and solvent. 4 In the past, CCl4 was also used in some medicines to treat intestinal worms and in the production of some polymers, refrigerants, fluorocarbons, pesticides, and pharmaceuticals. 5 Nowadays, although it is still exploited industrially as a petrol additive and in some plastics and paints, the toxicity of CCl4 has restricted its use. It is absorbed during ingestion, by inhalation and, more slowly, from direct contact with the skin. After absorption, it may spread to organs that have a high concentration of fat and accumulate there. Depending on the dose, CCl4 may be carcinogenic in humans, while acute exposure to high concentrations through ingestion or inhalation damages the liver and kidney. 6 CCl4 is activated by drug-metabolizing enzymes in the endoplasmic reticulum and is metabolized by cytochrome P450 (CYP-450) to produce trichloromethyl radicals, which are then oxidized to form the more reactive trichloromethylperoxyl radicals. 7 This metabolic reaction results in covalent binding to macromolecule, causing lipid peroxidation. Prolonged exposure to CCl4 may induce liver, kidney, and central nervous system injury. Liver is the most sensitive organ, while damage to the kidney can lead to increased levels of waste products in the blood. Kidney damage may occur within 2–3 weeks after exposure. 6,7 CCl4 is used in experimental research as a model for chemical toxins that induce renal injury. Exposure to CCl4 significantly increases serum urea and creatinine levels as a result of kidney damage, 8 which can be observed in the form of histopathological changes such as glomerular and tubular degeneration. 9 CCl4 induces oxidative stress, which results in nucleic acid and protein modification, affects cell membrane permeability, increases free calcium, and causes cell destruction by lipid peroxidation. Cells possess both enzymatic and nonenzymatic antioxidant systems that protect tissues against oxidative stress, and such systems may help to prevent lipid peroxidation and other forms of renal damage. 8 –10 Vitamin D, sometimes called the sunlight hormone, behaves like a steroid hormone, regulating cell growth, cell differentiation, and the immune system and prevents neoplastic transformation. It also plays an important role in regulating the reconstruction of bone and the secretion of insulin from pancreatic beta cells. Vitamin D is highly soluble in lipids and is transported to the blood by vitamin D binding protein. The most active form of vitamin D, 1,25-dihydroxyvitamin D3 (1,25(OH)2D3), is produced in the proximal tubules of the kidney after the hydroxylation of 7- and 8-dehydrocholesterol. 1,25(OH)2D3 has a beneficial role in calcium homeostasis. 11,12 Recent studies report that vitamin D and its analogs have protective effects in renal and cardiovascular diseases and that patients with chronic renal diseases have insufficient vitamin D. 13 The protective mechanism of vitamin D in renal diseases is mediated by various receptors and is regulated by the renin–angiotensin system (RAS). 14 In this study, we aimed to examine the protective effect of vitamin D treatment on CCl4-induced nephrotoxicity in rats using histopathological parameters.
Materials and methods
Animals
The experimental protocol was approved by the Local Ethics Committee for Animal Experiments of Gazi University, Ankara, Turkey (Approval number G.Ü.ET. 12.010; 66332047-604.01.02/27-3959). In this study, adult male Wistar albino rats weighing 200–220 g were used. The animals were maintained and monitored in the University animal facility under standard conditions (temperature: 22°C; light cycle: 12-h light/12-h dark). Animals were fed pellets and given water.
Experimental protocol
The rats were divided into four groups with six animals in each group (i.e. a total of 24 rats). Animals in group A (control) received 1.5 ml kg−1 corn oil subcutaneously every day for 10 weeks. Group B rats were injected subcutaneously with 1.5 ml kg−1 CCl4 diluted in corn oil (1:1 ratio) twice a week for 10 weeks. Like group B animals, group C rats were administrated with 1.5 ml kg−1 CCl4 subcutaneously twice a week for 10 weeks but also received daily intraperitoneal injections of vitamin D at a dose of 0.5 µg kg−1. Group D rats were also given 1.5 ml kg−1 CCl4 subcutaneously twice a week for 10 weeks but received a 0.5 µg kg−1 dose of vitamin D through intraperitonal injections every day for 12 weeks. In other words, CCl4 injection was stopped after week 10, however vitamin D administration continued for two more weeks.
Blood and tissue collection
Twenty-four hours after the last injection, rats were anesthetized using ketamine/Rompun and blood was taken from each animal by heart puncture. The blood samples were collected in tubes without anticoagulant, and serum was obtained after centrifugation at 3000–4500 r min−1 for 10 min. The serum samples were analyzed for urea, creatinine, and glucose. Rats were sacrificed, and the kidney tissues were removed immediately. Kidneys were washed with normal saline solution and, after removal of fat, tissues were weighed on a sensitive balance. Kidney tissue was cut into small pieces and fixed in 10% formalin solution for histopathology. 15
Serum biochemical analysis
Urea was determined by enzymatic method. 16 Serum creatinine was estimated by Jaffe’s method. 17 The glucose levels of the serum was detected by glucose oxidase peroxidase method. 18 All serum samples were measured using a Cobas C 501 autoanalyzer (Roche Diagnostics GmbH, Mannheim, Germany) with commercial kits.
Kidney histopathology
Fixed kidney samples were processed for histopathological examination. Kidney tissues were fixed in 10% formalin, then dehydrated using an ascending series of alcohols and embedded in paraffin. Kidney tissue sections of 5–6 μm thickness were cut using a microtome and stained with hematoxylin–eosin (H&E), Masson’s trichrome, and periodic acid–Schiff (PAS) for histological assessment. The stained sections were examined for signs of cell degeneration under a light microscope and images were obtained using a Leica DM1000 microscope and DFC320 camera (UK).
Statistical analysis
All data were analyzed using the SPSS version 15.0 statistics package. Differences among groups were measured using Kruskal–Wallis H test. All values are given as mean ± SD for the six animals in each group. A value of p < 0.05 was considered to be statistically significant.
Results
Animal body weight and kidney weight
Animal body weight before and after the administration of CCl4 and vitamin D in all groups is shown in Table 1. Using Kruskal–Wallis H test it has been reported that there were no significant differences between experimental (and control) groups either before or after treatment, suggesting that CCl4 does not affect body weight. Whether or not vitamin D was administered did not affect this result.
Rat body weight in the control and experimental groups.a

CCl4: carbon tetrachloride.
aAll values are given as mean ± SD for the six animals in each group.
b p < 0.05: significant.
Table 2 shows the kidney weight in all groups at the end of the experiment. In group B rats, which were administered CCl4 alone, a significant increase in kidney mass was observed when compared with the control group (Kruskal–Wallis H test). In contrast, although an apparent increase was seen in groups C and D, this was not statistically significant, suggesting that treatment with vitamin D can prevent or reduce CCl4-induced mass gain in the rat kidney.
Kidney weight at the end of the experiment.a

CCl4: carbon tetrachloride.
aAll values are given as mean ± SD for the six animals in each group.
b p < 0.05: significant. The significant differences is between CCl4 and control group.
Serum urea, glucose and creatinine levels
The levels of serum urea, creatinine, and glucose at the end of the experiment are presented in Table 3. The serum urea level increased significantly in CCl4-treated rats compared with the control group, while administration of vitamin D reduced serum urea to control levels. There were no differences in creatinine and glucose levels in all experimental groups. All values are given as mean ± SD for the six animals in each group.
Urea, glucose, and creatinine levels in rat serum.a

CCl4: carbon tetrachloride.
aAll values are given as mean ± SD for the six animals in each group.
b p < 0.05: significant.
Histopathological change
H&E stain
CCl4 induced histopathological changes in the kidney sections. Thus, while the control group (A) had a normal appearance in both cortex and medulla (Figure 1(a) and (b)), including the Bowman’s capsule and tubule of the cortex, the sections from the CCl4-treated group (B) showed significant renal damage. No toxic effect of CCl4 was apparent in the medulla, but the cortex was considerably damaged when compared with the control group (Figure 1(c)). CCl4-treated rats, both tubular and glomerular degeneration, were detected in the kidney tissues. There was dilatation and vacuolization within the tubules and hemorrhage in the intertubular region. In the kidney glomeruli, congestion, atrophy, and adhesion to the parietal layer were observed. Tissue disorganization and aggregation of Bowman’s capsules were noted. In addition, mononuclear cell infiltration was observed between the glomeruli and the tubules (Figure 2(a) to (e)). The administration of vitamin D (groups (C) and (D)) can lead to minimization of these histopathological changes (Figures 1(d) and 2(f)).

Photomicrograph of kidney section stained with H&E. (a) Normal morphology of control group. (b) CCl4 group showing abnormal morphology, hemorrhage, and loss of intact renal tubules. (c and d) Apparently normal morphology in kidney section from rat treated with CCl4 + vitamin D. (Original magnification, ×100.) CCl4: carbon tetrachloride; H&E: hematoxylin–eosin.

Photomicrograph of CCl4-treated rats kidney section stained with H&E. (a) Morphological alteration, including marked hemorrhage (arrows), as shown between tubular region. (b) Indicate increased connective tissue (arrow) compared with normal tissue. (c) The nuclei visible in the CCl4-treated rats are probably those of inflammatory cells (arrow). (d) Bowman’s capsule aggregation and (e) fatty material (arrows) was observed in CCl4 section. (f) Glomerular atrophy. (Original magnification, ×400.) CCl4: carbon tetrachloride; H&E: hematoxylin–eosin.
PAS stain
This shows that renal morphology was normal in the control group (A): the Bowman’s capsule basement membrane and the brush border were both PAS positive (Figure 3(a) and (b)). In contrast, sections from the CCl4-treated group (B) were graded from weakly PAS positive to PAS negative because of the disappearance of the brush border in proximal tubules. Basement membrane thickening was also observed in CCl4-treated rats (Figure 3(c)).
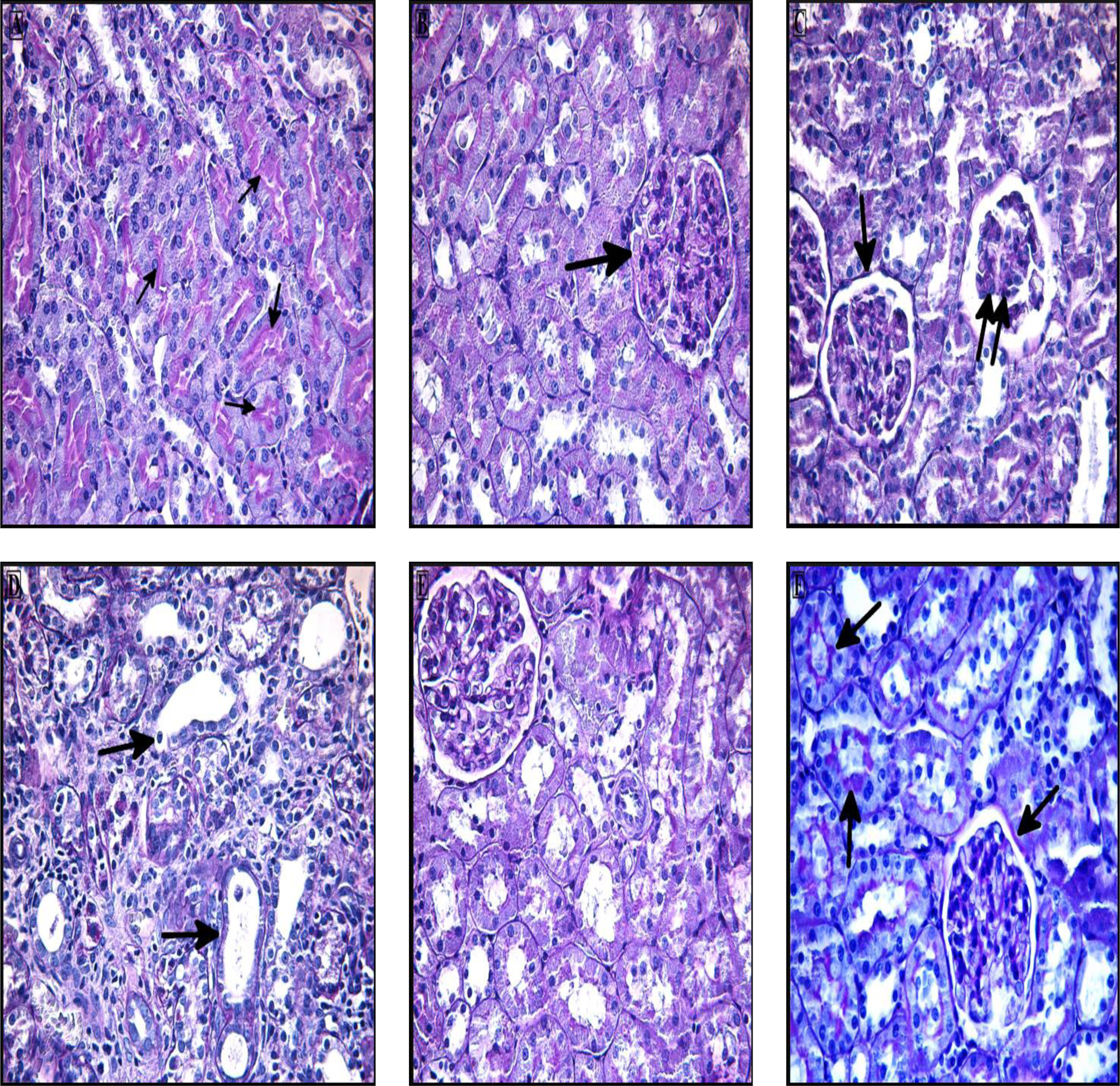
Photomicrograph of kidney section stained with PAS. (a and b) Control group showing that the tubular brush border cells and glomerular basement membrane are PAS positive (arrows). (c and d) Kidney section of CCl4-treated rats showing glomerular atrophy (double arrow) and basement membrane thickening and tubular degeneration (arrows). (e and f) Section of rats treated with both CCl4 + vitamin D had PAS positive (arrows) glomerular and tubular basement membrane and brush border cells, similar to controls. (Original magnification, ×400.) PAS: periodic acid–Schiff; CCl4: carbon tetrachloride.
Treatment with vitamin D (group C) apparently prevented such structural damage as sections were similar to the controls (Figure 3(d)).
Masson’s trichrome stain
The connective tissue in kidney sections of the control group (A) was stained blue and was seen in the normal position (Figure 4(a) and (b)). However, in the CCl4 group (B), increase in the connective tissue was indicated by the Masson’s trichrome stain as a heavy blue color (Figures 4(c) and (d) and 5). In contrast, the connective tissue in the vitamin D-treated groups (C and D) was similar to that of the controls (Figure 4(e) and (f)).
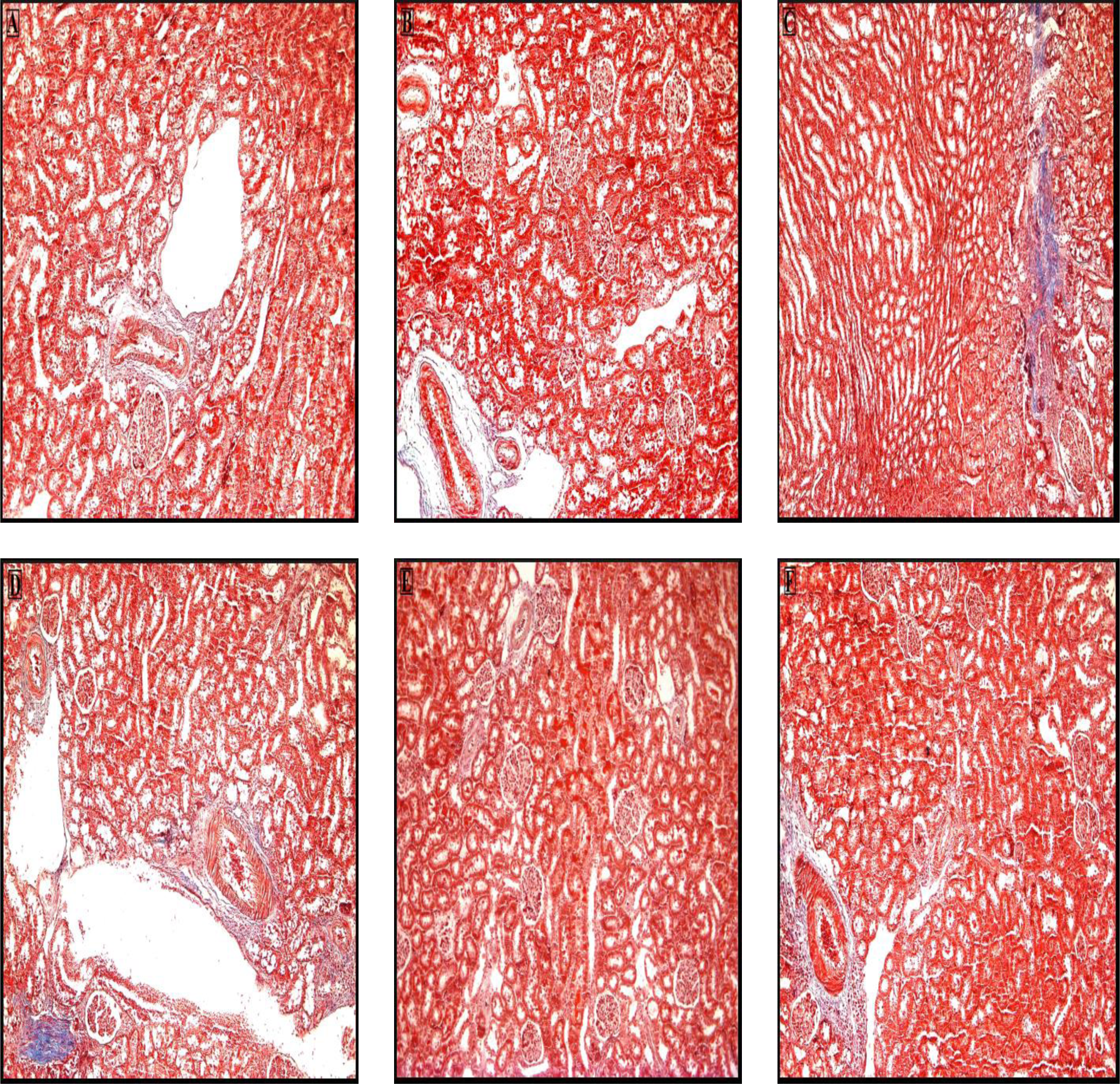
Photomicrograph of kidney section stained with Masson’s trichrome. (a and b) A representative section from the control group. (c and d) CCl4 group. (e and f) CCl4 + vitamin D groups. The extent of the connective tissue is greater in the CCl4 group compared with the control group. The kidney tissue of vitamin D groups resemble that of the control group. (Original magnification, ×100.) CCl4: carbon tetrachloride.

Photomicrograph of kidney section from a CCl4-treated rat stained with Masson’s trichrome. There is increase in connective tissue at different sites, especially in the corticomedulla and intertubular region. (Original magnification, ×400.) CCl4: carbon tetrachloride.
Discussion
Kidney damage by CCl4 is described in many experimental studies, with the extent of the damage depending on the duration of acute poisoning. CCl4 is one of the most extensively studied toxic agents to be found in the environment. 19 Both in vitro and in vivo studies indicate that CCl4 leads to enhanced lipid peroxidation. It is metabolized by CYP-450 to form highly reactive trichloromethyl radicals. Ganie et al. 20 reported decreased antioxidant activity in kidney tissue homogenate from CCl4-treated animals. This is similar to the results of Anand and Gokulakrishnan 21 and Cetin and Cetin 22 who found significant alterations in oxidative stress markers in CCl4-treated rats.
Suppression of RAS may be the primary mechanism for the renoprotective actions of vitamin D. 23 The present study aimed to assess the preventive effect of vitamin D in renal injury and dysfunction after chronic CCl4 administration.
Vitamin D has an important negative regulating role in renin biosynthesis and gene transcription. The deficiency of vitamin D or lack of vitamin D receptor may cause hyperreninemia, high blood pressure, and renal abnormalities. Vitamin D targets the cyclic adenosine monophosphate signaling pathway to suppress renin gene expression. 14 Our research was prompted by other works, which suggested that vitamin D treatment could block the progression of chronic renal disease by the suppression of the matrix-producing myofibroblast activation and RAS. 24,25
Our results suggest that CCl4 does not affect body weight in rats, in line with another report, which found no significant difference after 7 days of exposure. 26 Two other groups obtained the opposite result, however, reporting that administration of CCl4 can lead to weight loss. 9,27 Intriguingly, these same workers contradict each other with respect to kidney mass in CCl4-treated rats: Adewole et al. 9 measured a significant increase, in accordance with our own findings, while Manjrekar et al. 27 observed a reduction in mass. This result may be due to the low dose and short duration of the exposure to CCl4.
Serum urea level is the most widely measured parameter for the evaluation of kidney function. Our results showed that chronic exposure to CCl4 induced a significant increase in serum urea and this corroborates the findings of most other studies of this kind. 9,15,19,28 –31 In contrast, one report suggests that CCl4 decreases urea levels, 32 while three groups found no effect on serum urea at all. 33 –35 The creatinine level in the serum is also a measure of how well the kidneys are working. However, although several groups have reported an increase in serum creatinine level following administration of CCl4, 9,19,28 –32 we observed no such increase, in agreement with two other research teams. 34,35 In our histopathological evaluations, there was damage in the kidney cortex, while the medulla integrity was preserved. This might be related with the duration of CCl4 exposure and hence the unchanged creatinine level. Therefore, experiments with longer CCl4 exposure time are needed. We also found no change in serum glucose level, in agreement with Ramarajan et al. 15
We detected both tubular and glomerular degeneration in the kidney tissues of CCl4-treated rats. This histopathological change is frequently observed after CCl4 administration, 9,29,31,33,36 and glomerular basement thickening has also been described. 29 In addition, there are reports of moderate to severe necrosis, 9,19,29,31 while some studies detail glomerular congestion, 29 tubular vacuolization, glomerular atrophy, and parietal layer adhesion. 33 These changes were all also observed in the present study. Moreover, our data are consistent with the previous findings that CCl4 administration may stimulate connective tissue cells 33 and mononuclear cell infiltration, 9,27,29,33 which gives the tissue a foamy appearance and is indicative of inflammation. Interstitial inflammation, hemorrhage, tissue disorganization, and aggregation of Bowman’s capsules were noted in our study as well as dilatation and vacuolization within the tubules and hemorrhage in the intertubular region. We also observed moderate loss of tubular brush border after CCl4 treatment, as reported by Hamed et al. 29
The main novel result of the present study is that treatment with vitamin D can reduce the nephrotoxicity resulting from chronic exposure of rats to CCl4, and this is in line with the reports of vitamin D nephroprotectivity in other systems. For example, vitamin D significantly reduces apoptotic and histopathological changes in the rat kidney caused by ischemia–reperfusion injury. 37 One possible mechanism of this renoprotective activity, as shown for a vitamin D analog, is the suppression of myofibroblast activation. 25 Another mechanism of vitamin D treatment may be compensation for the effects of fibroblast growth factor, whose levels increase in kidney disease. This factor can suppress 1-alpha hydroxylase, which catalyzes the production of 1,25(OH)2D3, the active form of vitamin D. 38 Vitamin D treatment may also reduce fibrosis, as paricalcitol, an analog of vitamin D2, which inhibits the accumulation of fibronectin and types I and III collagen. 39 Indeed, as we and others have shown, while chronic administration of CCl4 may augment renal connective tissue, 33,40 which is an indicator of fibrosis, treatment with vitamin D eliminates this effect. Further evidence of a beneficial role of vitamin D in chronic kidney disease is demonstrated by its inhibitory effect on tumor necrosis factor and by reduced infiltration of T cells and macrophages and alters the transforming growth factor-signaling pathway into renal tissues. 41,42 Vitamin D can also reduce glomerular hypercellularity. 43 On the other hand, low and insufficient vitamin D may increase mortality in patients with end-stage kidney failure. 44 In this study, two groups of CCl4-exposed rats were given daily doses of vitamin D. In one, the animals were given both CCl4 and vitamin D for 10 weeks, while in the second, CCl4 administration was terminated at the end of week 10 and the vitamin D treatment lasted for 2 weeks longer, that is for 12 weeks.
As the histopathology suggests, there were no differences between the two groups, and it seems that the additional 2 weeks of vitamin D treatment had no noticeable effect on the toxicity induced by CCl4.
Conclusion
We conclude that vitamin D possesses good antioxidant activity in a rat model for renal toxicity, both curative and protective antioxidant effects of vitamin D were demonstrated in our experiments. Thus, vitamin D protects kidney tissue against CCl4-induced damage. From our work, and that of others with different experimental models of renal disease, it now seems clear that vitamin D has a powerful renoprotective effect. Further investigations are needed to understand the mechanism of this protective role.
Footnotes
Authors’ note
The research was conducted in Faculty of Medicine, Gazi University, Turkey and Pathology Laboratory of Etlik Veterinary Control Central Research Institute, Turkey. This work was presented as poster at the Third ‘Molecular Biology and Biotechnology Conference’, Sarajevo, Bosnia Herzegovina, 2–6 June 2014.
Acknowledgement
The authors wish to acknowledge Dr Yavuz Ulusoy (Etlik Veterinary Control Central Research Institute, Turkey) and Prof. Dr Gonca Akbulut (Faculty of Medicine, Gazi University, Turkey) for their assistance during this study.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
