Abstract
Venomous snake bite and subsequent coagulopathy is a significant source of morbidity and mortality worldwide. The gold standard to treat coagulopathy caused by these venoms is the administration of antivenom; however, despite this therapy, coagulopathy still occurs and recurs. Of interest, our laboratory has demonstrated in vitro and in vivo that coagulopathy-inducing venom exposed to carbon monoxide (CO) is inhibited, potentially by an attached heme. The present investigation sought to determine if venoms derived from snakes of the African genera Atheris, Atractaspis, Causus, Cerastes, Echis, and Macrovipera that have no or limited antivenoms available could be inhibited with CO or with the metheme-inducing agent, O-phenylhydroxylamine (PHA). Assessing changes in coagulation kinetics of human plasma with thrombelastography, venoms were exposed in isolation to CO or PHA. Eight species were found to have procoagulant activity consistent with the generation of human thrombin, while one was likely fibrinogenolytic. All venoms were significantly inhibited by CO/PHA with species-specific variation noted. These data demonstrate indirectly that the heme is likely bound to these disparate venoms as an intermediary modulatory molecule. In conclusion, future investigation is warranted to determine if heme could serve as a potential therapeutic target to be modulated during treatment of envenomation by hemotoxic enzymes.
Introduction
Envenomation by hemotoxic enzymes continues to be a major cause of morbidity and mortality throughout the world. 1 With regard to treatment, the gold standard to abrogate coagulopathy caused by these venoms is still the administration of antivenom 2 ; however, despite antivenom therapy, coagulopathy still occurs and recurs. With the objective of developing a therapy complimentary to antivenom administration, our laboratory has been investigating the phenomena of direct inhibition of hemotoxic enzymes by exposing the venom to carbon monoxide (CO) with in vitro experiments in human and animal plasmas 3 –8 and in vivo in a sedated rabbit model. 9 Specifically, both snake venom metalloproteinases (SVMP) and snake venom serine proteases (SVSP) obtained from venom of snakes in the families Viperidae and Elapidae exposed to CO in vitro demonstrated a marked reduction of fibrinogenolytic, thrombin-like, or prothrombin activating activity in human and rabbit plasma. 3 –9 Critically, we have recently demonstrated that a heme group was attached to a purified fibrinogenolytic enzyme isolated from Crotalus atrox venom that was inhibited by CO, strongly supporting the hypothesis that SVMP and SVSP may be modulated by heme groups. 10 In summary, substantial in vitro and preliminary in vivo investigations had demonstrated the inhibitory effect of CO on hemotoxic venom activity.
Given this robust beginning, it is of interest to continue to determine if CO-mediated inhibition has diverse application; specifically, assessing hemotoxic venom from snakes that are not yet well characterized or that do not have readily available antivenoms that are potential or actual causes of human morbidity and mortality. Several such species populate Africa, with a representative sample of nine vipers displayed in Table 1. As can be seen, a few genera (e.g. Atheris, Causus, Cerates) have not had in vitro venom characterization, with the remaining species being noted as procoagulant 11,12 or anticoagulant 12 in nature. In summary, it is of interest to thrombelastographically classify the nature of these venoms (e.g. procoagulant or anticoagulant activity) and to determine if exposure to CO could potentially inhibit venom activity as part of a supportive therapeutic regimen following envenomation.
Species of snake venoms investigated.

Considering the aforementioned, the purposes of the present investigation were to: (1) thrombelastographically define in human plasma the effects on coagulation kinetics of these various African venomous snake hemotoxic venom activities; (2) determine if CO directly inhibits these hemotoxic venom effects; (3) determine if placing a hemotoxic venom into an environment favoring metheme formation would affect venom activity; and (4) in the case of prothrombotic venoms, to utilize heparin–antithrombin anticoagulation to separate venom-mediated thrombin generation (e.g. prothrombin activating properties) from thrombin-like activity that is antithrombin resistant.
Materials and methods
Venoms, chemicals, and human plasma
Lyophilized venoms depicted in Table 1 were obtained from Mtoxins (Oshkosh, Wisconsin, USA). Venoms were reconstituted in calcium-free phosphate buffered saline (PBS, Sigma-Aldrich, Saint Louis, Missouri, USA) at a concentration of 50 mg/ml, aliquoted, and stored at −80°C until experimentation. CORM-2 (tricarbonyldichlororuthenium (II) dimer, a CO releasing molecule), dimethyl sulfoxide (DMSO), and O-phenylhydroxylamine (PHA) were also obtained from Sigma-Aldrich. Lastly, pooled normal human plasma (George King Bio-Medical, Overland Park, Kansas, USA) anticoagulated with sodium citrate (9 parts blood to 1 part 0.105 M sodium citrate) stored at −80°C was utilized in all subsequently described experiments. Changes in plasmatic coagulation kinetics, separate from the variability introduced with platelets, provides an experimental paradigm to easily assess changes in anticoagulant and procoagulant venom activity. 3 –9
Thrombelastographic analyses
Specific volumes of subsequently described whole blood and chemical additives varied by condition but summated to 360 µl. Sample composition consisted of 320 µl of plasma; 16.4 µl of PBS, 20 µl of 200 mM CaCl2, and 3.6 µl of PBS or venom (final concentration dependent on species of snake), which were placed into a disposable cup in a computer-controlled thrombelastograph® hemostasis system (Model 5000, Haemonetics Inc., Braintree, Massachusetts, USA) at 37°C, and then rapidly mixed by moving the cup up against and then away from the plastic pin five times before leaving the mixture between the cup and pin. The following elastic modulus-based parameters previously described 3 –9 were determined: time to maximum rate of thrombus generation (TMRTG): this is the time interval (min) observed prior to maximum speed of clot growth; maximum rate of thrombus generation (MRTG): this is the maximum velocity of clot growth observed (dynes/cm2/s); and total thrombus generation (TTG, dynes/cm2), the final viscoelastic resistance observed after clot formation. Data were collected for 20 min with venoms that were procoagulant, whereas venoms with anticoagulant properties had data collection for 30 min.
Determination of concentration of venom for experimentation and effects of isolated venom exposures to CORM-2 or PHA
The initial concentration for all venoms assessed was 1 µg/ml; if the onset and velocity of coagulation was procoagulant with commencement of coagulation beginning in half the time or less and/or the velocity of clot formation proceeded at twice or greater than plasma without venom addition, this concentration of venom was used in subsequent experimentation. If this was not the case, then the concentration of procoagulant venom was progressively increased until these conditions were met. If, in contrast, the venom was found to be anticoagulant in nature, the onset of coagulation had to be double and/or the velocity of clot formation half of plasma without venom addition to be acceptable to further investigate with that concentration of venom. However, if coagulation was not detectable, then the concentration of anticoagulant venom was progressively decreased until at least detectable coagulation occurred; then, this concentration was subsequently used in experimentation. In sum, this approach allowed comparison of relative potencies of the venoms in addition to determination of the overall effect of each venom on plasmatic coagulation.
With regard to exposure to CORM-2 to assess the effects of CO on venom activity, the following four experimental conditions were created: (1) control condition—no venom, DMSO 1% addition (v/v) in PBS; (2) venom condition—venom, DMSO 1% addition (v/v) in PBS; (3) CO condition—venom, CORM-2 1% addition in DMSO (100 µM final concentration; (4) inactive releasing molecule (iRM) condition—venom, inactivated CORM-2 1% addition in DMSO (100 µM final concentration). CORM-2 was inactivated as previously described. 13 Venom was placed in PBS with the aforementioned additions and after 5 min of incubation at room temperature, 3.6 µl of one of these four solutions were added to the plasma mixture in a thrombelastographic cup.
In another series of experiments prompted by resistance of Atheris chlorechis venom to 100 µM CORM-2 exposure, this venom was exposed to several concentrations of CORM-2 (0, 100, 250, 500, 750, and 1000 µM final concentration) to assess if there was a CO concentration–venom activity relationship. This range of CORM-2 concentrations corresponds to 0, 70, 175, 350, 525, and 700 µM CO as CORM-2 releases 0.7 mole of CO per mole of CORM-2. 14 Based on the results of these experiments, Causus lichtensteini venom was also exposed to 1000 µM CORM-2 and iRM when it was found resistant to 100 µM CORM-2.
Metheme-forming experiments
Other experiments involved exposure of all venoms to PHA in order to determine if their activities would change when the conditions favored the formation of metheme (Fe+3). This approach altered the heme state during investigation of fibrinogen in vitro 15 and ex vivo 16 in human plasma, decreasing the function of fibrinogen as a substrate for thrombin. Venom was placed in PBS with addition of PHA 3% addition (v/v, 30 mM final concentration) for 5 min prior to addition to the aforementioned plasma mixture in a thrombelastographic cup. The final PHA concentration in plasma after a 1% addition of the PHA-exposed venom was 0.3 mM, a concentration not expected to affect plasmatic coagulation kinetics based on previous work. 14 Results obtained from this condition were compared to the results obtained from condition 2 in the immediately preceding series of experiments.
Separation of venom-mediated thrombin generation from thrombin-like activity
To separate the contribution to coagulation kinetics of thrombin generated by prothrombin activators or other procoagulant activation dependent on human plasma proteins contained within the venoms from that of direct, thrombin-like activity found in these venoms, another series of experiments was conducted without or with heparin added to plasma prior to venom exposure. Unfractionated heparin (SAGENT Pharmaceuticals, Schaumburg, Illinois, USA), (1000 USP U/ml) was added to plasma for a final concentration of 10 U/ml, which is a concentration expected to maximally engage antithrombin and quench thrombin activity based on previous work in this laboratory. 16 Data were collected for 30 min.
Statistical analyses
Data are presented as mean ± SD. Experimental conditions were represented by n = 6 replicates per condition as this provides a statistical power >0.8 with p < 0.05 using this thrombelastographic methodology. 3 –9 A commercially available statistical program was used for one-way analyses of variance (ANOVA) comparisons between conditions, followed by Holm–Sidak post hoc analysis or unpaired, or two-tailed Student’s t tests as appropriate (SigmaPlot 14, Systat Software, Inc., San Jose, California, USA). Graphics depicting coagulation kinetic data were generated with commercially available programs, and coagulation kinetic modeling of the effects of venom exposure to various concentrations of CO was also performed with such programs (OrigenPro 2018, OrigenLab Corporation, Northampton, Massachusetts, USA; CorelDRAW X8, Corel Corporation, Mountain View, California, USA). p < 0.05 was considered significant.
Results
General observations
The coagulation parameter values for the control condition experiments were: TMRTG = 12.3 ± 2.6 min, MRTG = 3.3 ± 1.1 dynes/cm2/s, and TTG = 192 ± 19 dynes/cm2. The procoagulant kinetics of all but one venom (which was anticoagulant in nature) were sufficiently accelerated compared to control plasma without venom addition that there was no need for statistical comparison to this condition as we have done in the past. 8 The venom of Cerates cerates was anticoagulant in nature, very similar to fibrinogenolytic venoms, 3,4,6 so its kinetic profile included a comparison with control conditions. The remainder of the data are subsequently presented in Figures 1 to 4 and Tables 2 and 3. For clarity, the results of each species will be presented in a separate section.
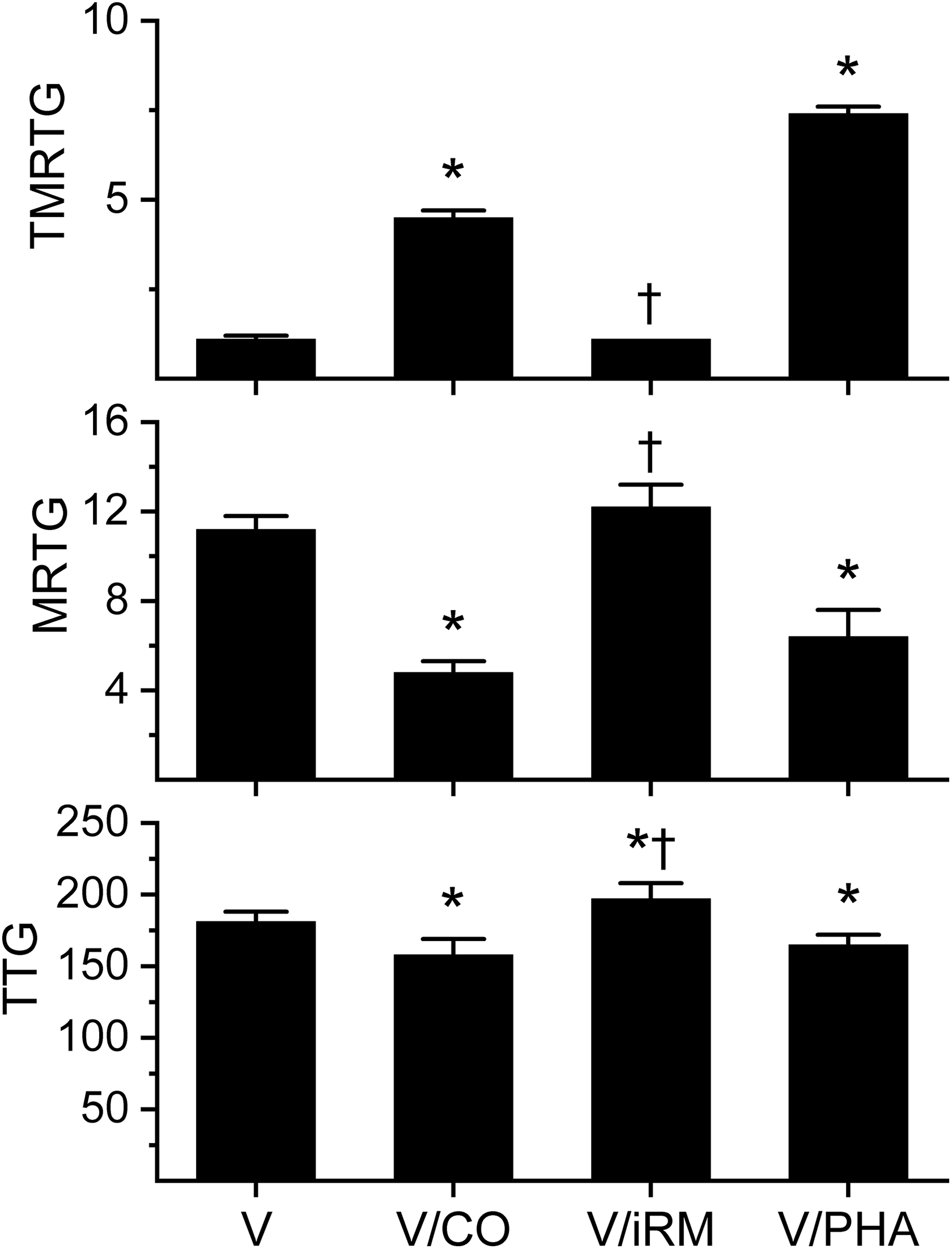
Effects of exposure to CO and PHA on Atheris broadleyi venom-mediated effects on coagulation kinetics in human plasma. Data are presented as mean ± SD. *p < 0.05 versus V; † p < 0.05 versus V/CO. V: venom without exposure to CO or PHA; V/CO: venom exposed to CO; V/iRM: venom exposed to iRM; V/PHA: venom exposed to PHA; CO: carbon monoxide; PHA: O-phenylhydroxylamine; iRM: inactive releasing molecule; TMRTG: time to maximum rate of thrombus generation (min); MRTG: maximum rate of thrombus generation (dynes/cm2/s); TTG: total thrombus generation (dynes/cm2).

Effects of exposure to a range of CO concentrations (0–700 µM) on Atheris chlorechis venom-mediated effects on coagulation kinetics in human plasma. Data are presented as raw values with best fit curve fitting with matching coefficient of determination (R 2) and statistical significance of the fit (p value). The equation describing TMRTG changes in response to CO concentration [CO] is one of exponential growth: TMRTG = 16.08( [CO]/4355) + (4.869− 10)( [CO]/31.05) − 15.15. The equation describing MRTG changes in response to [CO] is one of exponential decay: MRTG = −3.985(− [CO]/− 683.0) + 16.03. The equation describing TTG change in response to [CO] is polynomial: TTG = 184 + (0.174 × [CO]) + ((−2.825− 4) × [CO]2). CO: carbon monoxide; PHA: O-phenylhydroxylamine; iRM: inactive releasing molecule; TMRTG: time to maximum rate of thrombus generation (min); MRTG: maximum rate of thrombus generation (dynes/cm2/s); TTG: total thrombus generation (dynes/cm2).
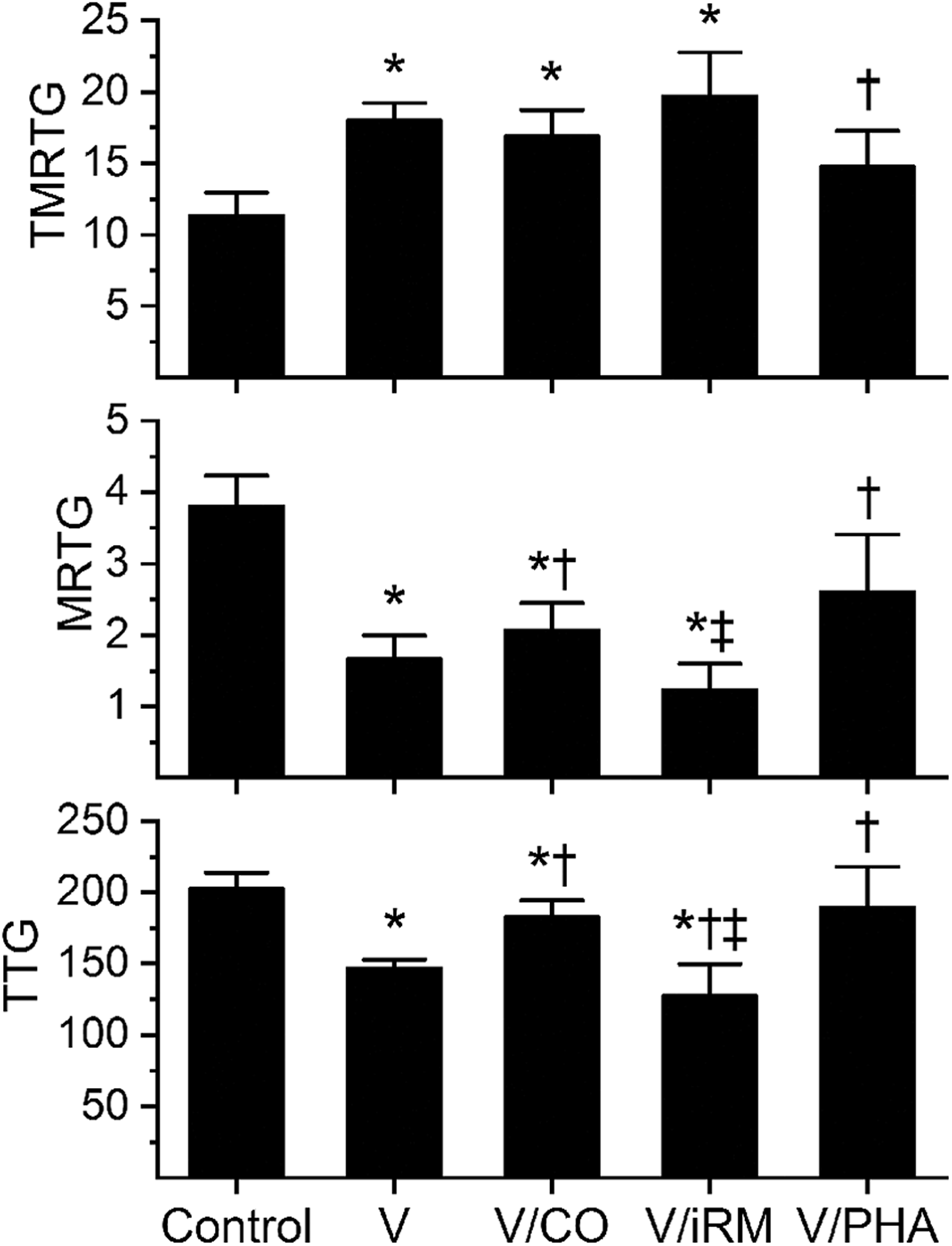
Effects of exposure to CO and PHA on Cerates cerates venom-mediated effects on coagulation kinetics in human plasma. Data are presented as mean ± SD. *p < 0.05 versus control; † p < 0.05 versus V; ‡ p < 0.05 versus V/CO. Control: plasma without any venom exposure; V: plasma with venom not exposed to CO or PHA; V/CO: venom exposed to CO; V/iRM: venom exposed to iRM; V/PHA: venom exposed to PHA; CO: carbon monoxide; PHA: O-phenylhydroxylamine; iRM: inactive releasing molecule; TMRTG: time to maximum rate of thrombus generation (min); MRTG: maximum rate of thrombus generation (dynes/cm2/s); TTG: total thrombus generation (dynes/cm2).
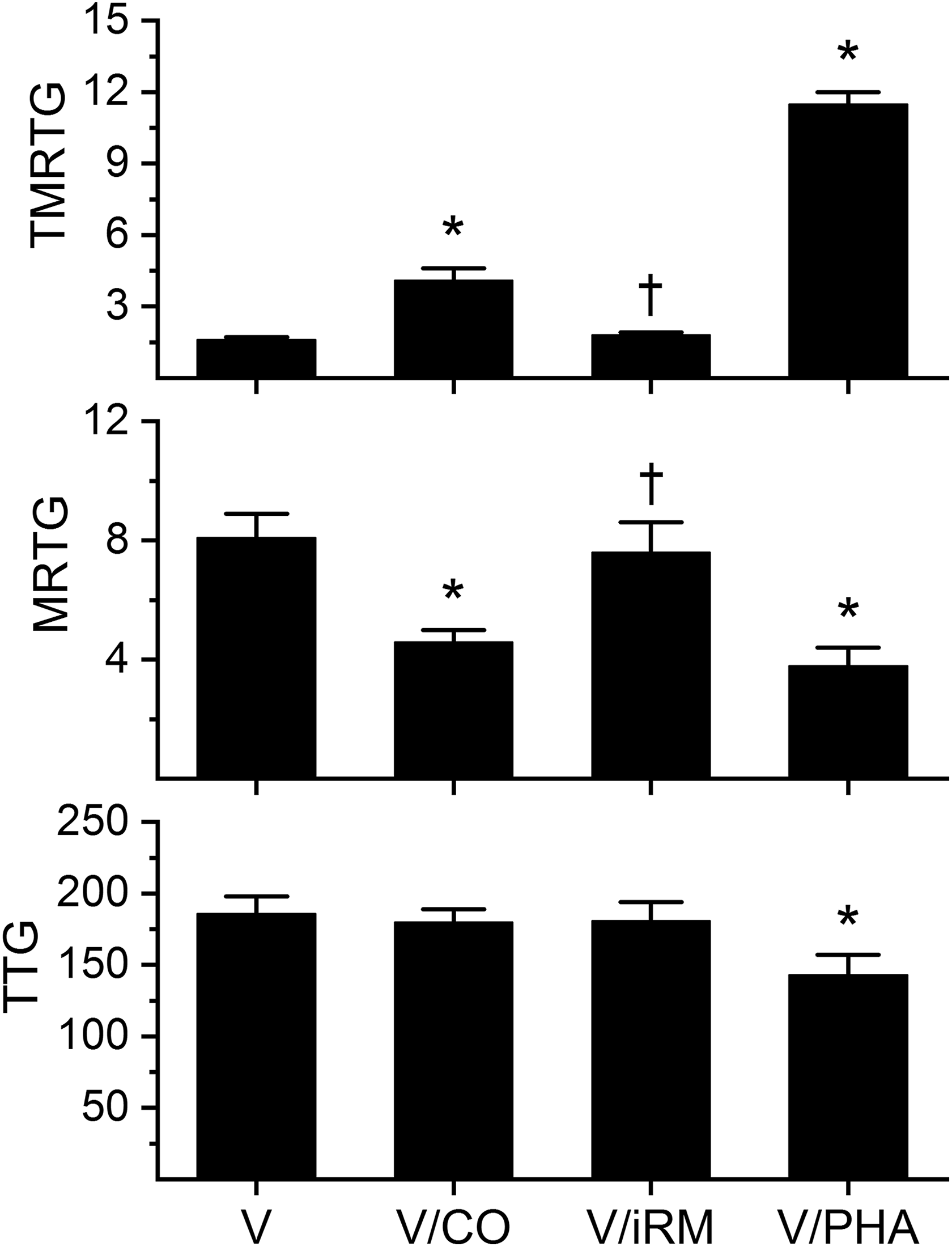
Effects of exposure to CO and PHA on Echis ocellatus venom-mediated effects on coagulation kinetics in human plasma. Data are presented as mean ± SD. *p < 0.05 versus V; † p < 0.05 versus V/CO. V: venom without exposure to CO or PHA; V/CO: venom exposed to CO; V/iRM: venom exposed to iRM; V/PHA: venom exposed to PHA; CO: carbon monoxide; PHA: O-phenylhydroxylamine; iRM: inactive releasing molecule; TMRTG: time to maximum rate of thrombus generation (min); MRTG: maximum rate of thrombus generation (dynes/cm2/s); TTG: total thrombus generation (dynes/cm2).
Coagulation kinetics of venoms ±CO or ±PHA in plasma.a

CO: carbon monoxide; PHA: O-phenylhydroxylamine; iRM: inactive releasing molecule; TMRTG: time to maximum rate of thrombus generation (min); MRTG: maximum rate of thrombus generation (dynes/cm2/s); TTG: total thrombus generation (dynes/cm2); V: venom addition without exposure to other compounds; V/CO: venom addition after exposure to CORM-2; V/iRM: venom addition after exposure to inactivated CORM-2; V/PHA: venom addition after exposure to PHA.
a Data are presented as mean ± SD.
b p < 0.05 versus V.
c p < 0.05 versus V/CO.
Coagulation kinetics of venoms ±CO exposure in plasma with heparin.a
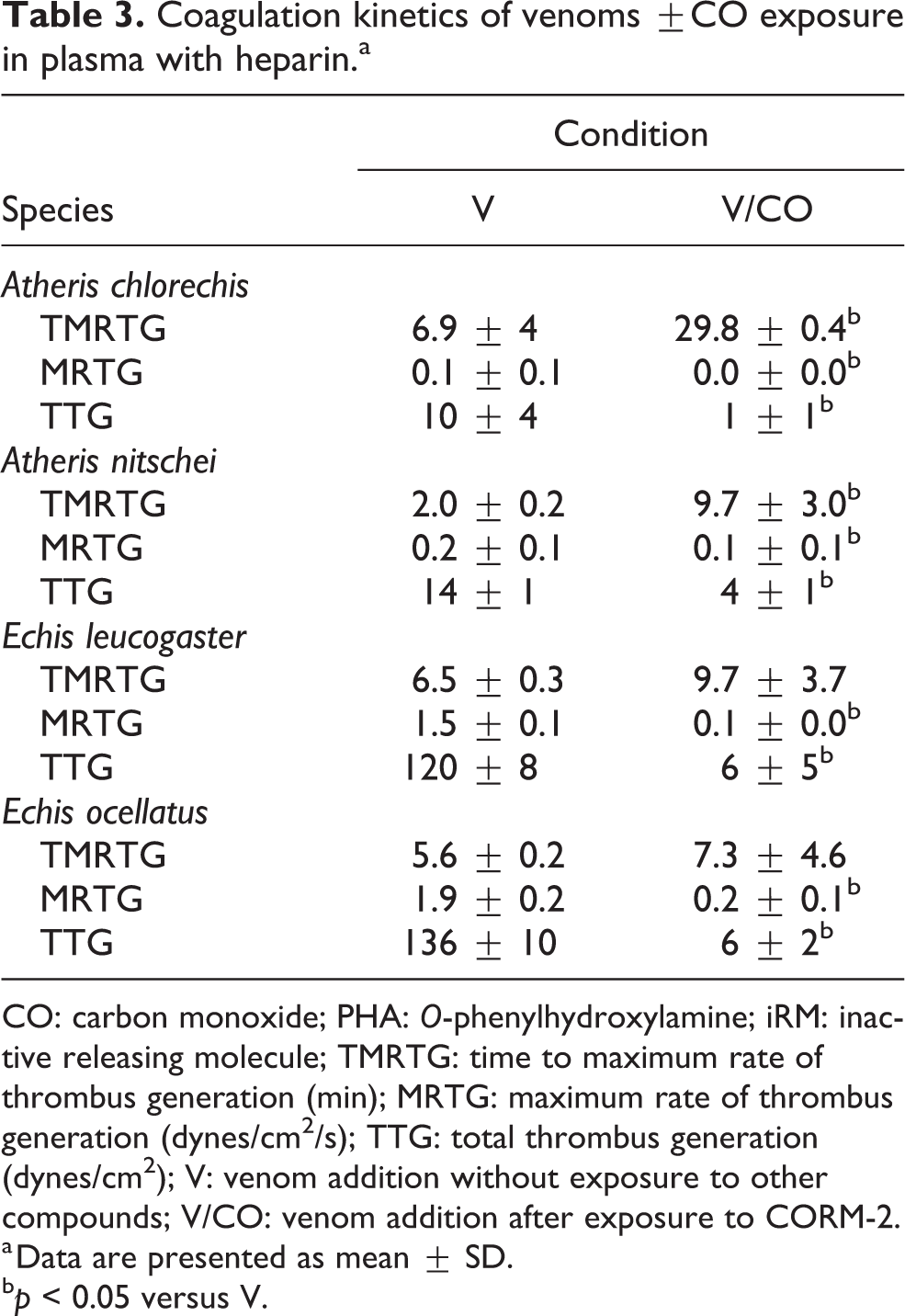
CO: carbon monoxide; PHA: O-phenylhydroxylamine; iRM: inactive releasing molecule; TMRTG: time to maximum rate of thrombus generation (min); MRTG: maximum rate of thrombus generation (dynes/cm2/s); TTG: total thrombus generation (dynes/cm2); V: venom addition without exposure to other compounds; V/CO: venom addition after exposure to CORM-2.
a Data are presented as mean ± SD.
b p < 0.05 versus V.
Atheris broadleyi
Data were generated with a venom concentration of 1 µg/ml in plasma. As seen in Figure 1, exposure of A. broadleyi venom to CO resulted in a significant increase in TMRTG, decrease in MRTG, and decrease in TTG values compared to samples exposed to venom alone. Exposure of the venom to the iRM resulted in TMRTG and MRTG values not different from CO naïve venom condition values but significantly different from venom samples exposed to CO. With regard to TTG, venom exposed to iRM had values greater than either CO exposed or CO naïve conditions. As for venom exposed to PHA, these samples had significantly greater TMRTG values, smaller MRTG values, and smaller TTG values that of PHA naïve venom samples. Lastly, when venom was added to plasma-containing heparin, there was no discernable coagulation.
Atheris chlorechis
Data were generated with a venom concentration of 1 µg/ml in plasma. As can be noted in Table 2, exposure of A. chlorechis venom to CO resulted in a small but significant increase in TMRTG; however, exposure to CO at this concentration did not significantly affect MRTG or TTG values compared to samples exposed to venom alone. Exposure of the venom to the iRM resulted in TMRTG, MRTG, and TTG values not different from CO naïve venom condition values, with only TMRTG values different from venom exposed to CO. As for venom exposed to PHA, these samples had significantly greater TMRTG values, smaller MRTG values, and smaller TTG values that of PHA naïve venom samples.
The results of experiments with A. chlorechis venom placed into heparin-containing plasma are displayed in Table 3. Preliminary experiments demonstrated that the venom had a weak, thrombin-like enzyme (TLE) activity present at a venom concentration of 1 µg/ml, so the concentration was increased to 2 µg/ml to obtain more measurable coagulation kinetic values. As can be seen, this TLE activity was significantly decreased by CO, as evidenced by significant increases in TMRTG, decreases in MRTG and decreases in TTG values in samples containing CO-exposed venom compared to samples with CO naïve venom.
As A. chlorechis venom had very little inhibition following exposure to the typical concentration of CO (70 µM), we determined if the venom was essentially unresponsive or instead needed to be exposed to greater concentrations of CO to demonstrate inhibition. As seen in Figure 2, exposure of the venom to progressively larger CO concentrations resulted in a CO concentration dependent increase in TMRTG (exponential growth pattern) and decrease in MRTG (exponential decrease pattern). In contrast, TTG values first increased and then decreased as CO concentration increased (polynomial fit). In sum, A. chlorechis venom was responsive to CO inhibition but only at large concentrations.
Atheris nitschei
Data were generated with a venom concentration of 1 µg/ml in plasma. As displayed in Table 2, exposure of A. nitschei venom to CO resulted in a significant increase in TMRTG and decrease in MRTG compared to samples exposed to venom alone. Exposure of the venom to the iRM resulted in TMRTG and MRTG values not different from CO naïve venom condition values but significantly different from venom samples exposed to CO. With regard to TTG values, there was no significant differences between the three conditions. As for venom exposed to PHA, these samples had significantly greater TMRTG values, smaller MRTG values, and smaller TTG values that of PHA naïve venom samples.
The results of experiments with A. nitschei venom placed into heparin-containing plasma are found in Table 3. Preliminary experiments demonstrated that the venom had a weak TLE activity present at a venom concentration of 1 µg/ml, so the concentration was increased to 4 µg/ml to obtain more measurable coagulation kinetic values. As can be seen, this TLE activity was significantly decreased by CO, as evidenced by significant increases in TMRTG, decreases in MRTG, and decreases in TTG values in samples containing CO-exposed venom compared to samples with CO naïve venom.
Atractaspis irregularis
Data were generated with a venom concentration of 20 µg/ml in plasma. As displayed in Table 2, exposure of A. irregularis venom to CO resulted in a significant increase in TMRTG, no change in MRTG, and a significant increase in TTG values compared to samples exposed to venom alone. Exposure of the venom to the iRM resulted in TMRTG values significantly different from venom that was CO naïve or exposed to CO. The MRTG values derived from venom exposed to the iRM was not different from the other two conditions. With regard to TTG, venom exposed to iRM had values significantly greater than CO naïve conditions but not different from CO-exposed venom sample values. As for venom exposed to PHA, these samples had significantly greater TMRTG values, smaller MRTG values, and smaller TTG values that of PHA naïve venom samples. Lastly, when venom was added to heparin-containing plasma, there was no discernable coagulation.
Causus lichtensteini
Data were generated with a venom concentration of 20 µg/ml in plasma. Initial experiments demonstrated that this venom was resistant to CO, so venom was subsequently exposed to 1000 µM CORM-2 or iCORM-2 for the CO or iRM conditions, respectively. As seen in Table 2, exposure of C. lichtensteini venom to CO resulted in a significant increase in TMRTG compared to CO naïve venom, whereas iRM exposed venom had TMRTG values between and significantly different from the other two conditions. With regard to MRTG values, CO-exposed venom had smaller values than CO naïve venom samples, with iRM exposed venom had MRTG values between and significantly different from the other two conditions. With regard to TTG, there was no significant difference between CO naïve venom, CO-exposed venom, or iRM-exposed venom values. As for venom exposed to PHA, these samples had significantly greater TMRTG values, smaller MRTG values, and smaller TTG values than PHA naïve venom samples. Lastly, when venom was added to plasma-containing heparin, there was no discernable coagulation.
Cerastes cerastes
Data were generated with a venom concentration of 3 µg/ml in plasma. This venom displayed anticoagulant kinetic behavior consistent with classic fibrinogenolytic venoms. 3,4,6 As displayed in Figure 3, compared to control conditions, C. cerastes venom significantly increased TMRTG values, decreased MRTG values, and decreased TTG values. Exposure of venom to CO did not significantly decrease TMRTG values, did not significantly increase MRTG values, but did significantly increase TTG values compared to CO naïve venom values; however, TTG values of this group was still less than control conditions. Exposure of venom to iRM resulted in TMRTG values not different from CO naïve or CO-exposed venom, MRTG values significantly smaller than control and CO-exposed venom values, and TTG values significantly less than CO naïve and CO-exposed venom values. As for venom exposed to PHA, these samples had significantly smaller TMRTG values, larger MRTG values, and larger TTG values that of PHA naïve venom samples.
Echis leucogaster
Data were generated with a venom concentration of 1 µg/ml in plasma. As displayed in Table 2, exposure of E. leucogaster venom to CO resulted in a significant increase in TMRTG and decrease in MRTG compared to samples exposed to venom alone. Exposure of the venom to the iRM resulted in TMRTG and MRTG values not different from CO naïve venom condition values but significantly different from venom samples exposed to CO. With regard to TTG values, there was no significant differences between the three conditions. As for venom exposed to PHA, these samples had significantly greater TMRTG values, smaller MRTG values, and smaller TTG values that of PHA naïve venom samples.
The results of experiments with E. leucogaster venom placed into heparin-containing plasma are found in Table 3. These data were generated with 1 µg/ml venom concentrations. Under these conditions, this venom behaved kinetically like an antithrombin resistant TLE, with prolonged TMRTG and small MRTG values compared with venom in plasma without heparin. However, unlike Atheris venom, it appeared that E. leucogaster venom activated factor XIII (FXIII) in a thrombin-independent manner, as the magnitude of TTG values generated by the venom require FXIII-mediated crosslinking of fibrin polymers. 17 Remarkably, CO exposure did not significantly change TMRTG values but significantly decreased MRTG and TTG values compared to CO naïve venom—but these reduced kinetic values are similar to TLE activity simply without FXIII activation when compared with the two Atheris species values (Table 3) or Crotalus simus venom. 18 Thus, while CO reduced E. leucogaster venom activity under these conditions, it may be possible that only the FXIII activating function of the antithrombin resistant TLE was inhibited.
Echis ocellatus
Data were generated with a venom concentration of 1 µg/ml in plasma and is displayed in Figure 4. Exposure of E. ocellatus venom to CO resulted in a significant increase in TMRTG and decrease in MRTG compared to samples exposed to venom alone. Exposure of the venom to the iRM resulted in TMRTG and MRTG values not different from CO naïve venom condition values but significantly different from venom samples exposed to CO. With regard to TTG values, there was no significant differences between the three conditions. As for venom exposed to PHA, these samples had significantly greater TMRTG values, smaller MRTG values, and smaller TTG values that of PHA naïve venom samples.
The results of experiments with E. ocellatus venom placed into heparin-containing plasma are found in Table 3. These data were generated with 1 µg/ml venom concentrations. E. ocellatus venom kinetically behaved essentially identically compared to E. leucogaster venom, displaying TLE-like activity combined with activation of FXIII. Again, CO exposure did not significantly change TMRTG values but significantly decreased MRTG and TTG values compared to CO naïve venom—but these reduced kinetic values were similar to TLE activity simply without FXIII activation. Thus, while CO reduced E. leucogaster venom activity under these conditions, it may be possible that only the FXIII activating function of the antithrombin resistant TLE was inhibited.
Macrovipera mauritanica
Data were generated with a venom concentration of 1 µg/ml in plasma. As seen in Table 2, exposure of M. mauritanica venom to CO resulted in a significant increase in TMRTG, decrease in MRTG, and decrease in TTG values compared to samples exposed to venom alone. Exposure of the venom to the iRM resulted in TMRTG, MRTG, and TTG values not different from CO naïve venom condition values but significantly different from venom samples exposed to CO. With regard to venom exposed to PHA, these samples had significantly greater TMRTG values, smaller MRTG values, and smaller TTG values that of PHA naïve venom samples. Lastly, when venom was added to heparin-containing plasma, there was no detectable coagulation.
Discussion
This investigation achieved its stated goals. First, the venoms tested were determined in eight cases to primarily cause human thrombin generation as the major coagulation kinetic effect, with the remaining venom displaying typical fibrinogenolytic effects. It should be noted that while M. mauritanica has been documented to contain a fibrinogenolytic metalloproteinase, 19 the activity of this enzyme is overshadowed by another prothrombotic enzyme that causes thrombin generation (Table 2). Such data demonstrate the discriminating power of the thrombelastograph to determine the predominant effect of unfractioned venom, and therefore identifying the most likely enzyme type responsible for in vivo coagulopathy. Also of interest, there was at least a twenty-fold difference in potency between these venom, with the Atheris and Echis species demonstrating procoagulant kinetics equal to that of the Australian taipans recently characterized by us with the same methodology. 8 With regard to our second and third aims, all venom activities were found to a greater or lesser extent to be inhibited by CO and PHA. These data continue to indicate, using two different ways to modulate putative attached heme groups, that hemotoxic SVMP/SVSP are heme bearing enzymes that have potential as therapeutic targets in the treatment of envenomation. Lastly, two of the Atheris and both Echis species’ venoms tested also had heparin–antithrombin resistant TLE activities present that were in part similar to that thrombelastographically described in South American Crotalus simus species. 18 However, the relatively vigorous clot strength seen following the action of Echis species venom is very suggestive of FXIII activation 17 that is thrombin independent—a result not previously seen by us. In summary, using thrombelastographic methods, key characteristics of the venoms of these African viper species were defined for the first time.
The CO concentration–coagulation kinetic response of A. chlorechis venom (Figure 2) provides insight into the consequences of inhibition of complex activation sequences (thrombin cleavage of fibrinogen, thrombin polymerization of fibrin monomers, and thrombin-mediated activation of FXIII; consequent FXIII-mediated crosslinking of fibrin polymers). When considering the parameters TMRTG and MRTG, the relationship of inhibition of venom activity to increasing CO concentration was exponential, with TMRTG changes secondary to venom activity dramatically reduced at the highest concentration of CO, whereas MRTG changes were more gradual in response to increasing CO. In contrast to TMRTG and MRTG, changes in TTG are curvilinear, which is consistent with the complex interplay of progressively decreasing venom-mediated thrombin generation. In way of explanation, the relative concentrations and reaction rates of thrombin, fibrinogen, fibrin polymers, and FXIII modify the results of progressively decreased venom activity by CO. There is 100-fold more fibrinogen than FXIII in plasma, 20 and the rates of thrombin cleavage of fibrinogen and thrombin activation of FXIII are similar. 21,22 Then, even though the reaction rate of FXIII to crosslink fibrin polymers if 50-fold greater than that of thrombin cleaving fibrinogen, 23 rapid thrombin generation and consequent chaotic fibrin polymer formation may not permit the usual orderly FXIII-mediated crosslinking and ideal clot strength. This has been demonstrated using thrombelastographic methods in a previous work. 24 Thus, considered as a whole, as venom activity was diminished by CO, the time to commence clotting increased at first gradually and then quickly toward values near normal; the speed of clot formation decreased gradually; and then, lastly, clot strength at first increased with decreasing thrombin generation (allowing time for FXIII-mediated crosslinking) and then decreased as thrombin generation was reduced below an optimum rate to generate fibrin polymers and permit FXIII crosslinking.
In conclusion, the present work characterized the unique thrombelastographic signatures of the coagulopathic effect of the hemotoxic venoms of nine different African vipers. Further, the inclusion of A. irregularis in our analyses was particularly important, as this species represents the third family (Atractaspidinae) of snake found to have its venom inhibited by carboxyheme and metheme-forming agents. The inhibition of diverse hemotoxic venom activities (procoagulant/anticoagulant, SVMP/SVSP) by heme modulating agents is likely naturally achieved with either CO or nitric oxide (NO, a natural metheme-forming agent) via heme oxygenase or nitric oxide synthase activity in venom glands. Variation in inhibition to CO concentration was species-specific and potentially indicates that CO or NO may be used by the vipers to modulate different venoms. Further, there may be a difference in affinity for CO binding to a particular heme between species, or there may be more than one heme attached to an enzyme, requiring a greater concentration of CO to fully inhibit activity. From our perspective as the potentially envenomed, modulation of such heme bearing enzymes could serve a therapeutic purpose as supportive care prior to definitive therapy with antivenom. In sum, this investigation serves as a rationale for ongoing in vitro and in vivo investigation to determine the role played by heme as a therapeutic target to treat envenomation.
Footnotes
Authors’ note
Mr. Nathaniel Frank is the owner of the company who supplies the venoms for the research.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This investigation was supported by the Department of Anesthesiology, the University of Arizona, College of Medicine, Arizona, USA.
