Abstract
Thousands suffer poisonous snake bite, often from defibrinogenating species annually. Three rattlesnake species in particular, the timber rattlesnake, Eastern diamondback rattlesnake, and Southern Pacific rattlesnake, cause clinically relevant hypofibrinogenemia via thrombin-like activity in their venom. It has been demonstrated that iron (Fe) and carbon monoxide (CO) change the ultrastructure of plasma thrombi and improve coagulation kinetics. Thus, the present investigation sought to determine if pretreatment of plasma with Fe and CO could attenuate venom-mediated catalysis of fibrinogen via thrombin-like activity. Human plasma was pretreated with ferric chloride (0–10 μM) and CO-releasing molecule-2 (0–100 μM) prior to exposure to 2.5–10 μg/ml of venom obtained from the aforementioned three species of rattlesnake. Coagulation kinetics were determined with thrombelastography. All three snake venoms degraded plasmatic coagulation kinetics to a significant extent, especially diminishing the speed of clot growth and strength. Pretreatment of plasma with Fe and CO completely abrogated the effects of all three venoms on coagulation kinetics. Further in vitro investigation of other pit viper venoms that possess thrombin-like activity is indicated to see if there is significant conservation of venom enzymatic target recognition of specific amino acid sequences such that Fe and CO can reliably attenuate venom-mediated catalysis of fibrinogen. These data also serve as a rationale for future preclinical investigation.
Introduction
Thousands of people suffer venomous snake bite annually in the United States, 1 with rattlesnakes being the majority of identified vipers. 2 Fortunately, antivenom is readily available to most victims, 3 but there are still severe to fatal outcomes. 4 Snake venom can have many deleterious effects, which include enzymatic degradation of the coagulation system. 5,6 The enzymes involved are typically serine proteases or metalloproteinases. 5,6 In particular, a variety of rattlesnake species found in populous regions throughout North America have defibrinogenating venoms that contain thrombin-like activity. 5 However, unlike thrombin, such venom does not activate factor XIII, with a resultant weak, friable clot. 5 In particular, the venom of the timber rattlesnake (Crotalus horridus) causes a disseminated intravascular coagulation-like coagulopathy, 7 whereas the venoms of the Eastern diamondback rattlesnake (Crotalus adamanteus) 8,9 and Southern Pacific rattlesnake (Crotalus oreganus helleri) 10 primarily result in severe, sometimes fatal hypofibrinogenemia. All of these snakes have well-characterized, thrombin-like activity in their venoms. 11 –15 In sum, envenomation by these particular rattlesnakes can be successfully treated with timely administration of antivenom, but serious injury, coagulopathy, and death still occur. 4,7 –10
While antibodies to venom compounds and enzymes in antivenom can be lifesaving, the particular antivenom administered may not be effective in all cases of envenomation, and even the same species of rattlesnake may express different types of venom depending on location. 9,10,14,15 Another approach, at least for preserving circulating fibrinogen, may be to somehow render it less vulnerable to venom-mediated enzymatic attack that is not snake species-specific. One such strategy could involve changing the structural configuration of fibrinogen with recently described interactions with iron (Fe) and carbon monoxide (CO). Specifically, recent investigations have revealed that plasmatic fibrinogen exposed to Fe and CO forms thrombi that are structurally different from plasma without such exposures, and coagulation commences earlier, with greater formation velocity and final clot strength compared to clots that are Fe and CO naive. 16 CO binds to heme(s) bound to fibrinogen, 17 and Fe and heme have been found to bind to fibrinogen, 18 with chelation of Fe noted to reverse both structural and coagulation kinetic changes in plasma thrombi. 19 Fe and CO enhance fibrinogen by separate mechanisms; Fe decreases the time to onset of coagulation and increases the velocity of clot formation without affecting final clot strength, whereas CO increases the velocity of clot formation and increases clot strength without affecting the time to onset of coagulation. 16 When combined, the velocity of clot formation increases in an additive manner by both these agents. 16 Also of interest, exposure to CO resulted in the inability to recover significant portions of the γ chain after endopeptidase treatment of purified fibrinogen during mass spectroscopic analysis, 17 indirect evidence of molecular conformational change of fibrinogen. Taken as a whole, these data suggested that it may be possible to change the three-dimensional configuration of fibrinogen with Fe and CO such that thrombin could interact with it as an enhanced substrate, but perhaps thrombin-like activity in snake venom could not.
The hypothesis of the present investigation was that pretreatment of plasma with Fe and CO may attenuate the ability of the venom of the aforementioned rattlesnake species to catalyze fibrinogen.
Materials and methods
Chemicals and reagents
Pooled normal human plasma (George King Bio-Medical, Overland Park, Kansas, USA) anticoagulated with sodium citrate (9 parts blood to 1 part 0.105 M sodium citrate) stored at −80ºC was utilized in all subsequently described experiments. Two-hundred milligrams of lyophilized timber rattlesnake, Eastern diamondback rattlesnake, and Southern Pacific rattlesnake venom was obtained from the National Natural Toxins Research Center at Texas A&M University, Kingsville, Texas, USA. Venom was reconstituted in calcium-free phosphate-buffered saline (PBS, Sigma-Aldrich, Saint Louis, Missouri, USA) at a concentration of 50 mg/ml, aliquoted, and stored at −80°C until experimentation.
Fe and CO pretreatment
Plasma was rapidly thawed at 37°C on the day of experimentation. Plasma had 1% additions of ferric chloride (FeCl3, 99.9% pure, Sigma-Aldrich) in PBS) or CORM-2 (tricarbonyldichlororuthenium (II) dimer, a CO releasing molecule, Sigma-Aldrich) dissolved in dimethyl sulfoxide (Sigma-Aldrich). The final concentrations of FeCl3 and CORM-2 were 0–10 µM and 0–100 µM, respectively; these concentrations are associated with nearly maximal augmenting effects on coagulation kinetics. 15 Plasma was exposed to these various agents for at least 5 min prior to placement into plastic thrombelastographic cups (Haemonetics Inc., Braintree, Massachusetts, USA).
Experimental plasma mixtures and thrombelastographic analyses
The final volume for subsequently described plasma sample mixtures was 359.4–359.6 µl. In experiments involving venom obtained from timber and Eastern diamondback rattle snakes, the sample composition consisted of 326 µl of plasma, 10 µl of tissue factor reagent (0.1% final concentration in distilled water; Diagnostica Stago S.A.S., Asnieres sur Seine, France), and 3.4 µl of PBS or venom (final concentration 0.5 µg/ml). In pilot studies, it was determined that the effects of 5 min of incubation at 37°C with 2.5 µg/ml of either of these two snake venoms provided significant decreases in coagulation kinetic parameter values (e.g., 50% decrease in time to maximum rate of thrombus formation, 50% decrease in the speed of clot formation) before the addition of calcium to commence coagulation. Thus, it was assumed that the thrombin-like activity of these rattlesnakes was not calcium-dependent. However, venom obtained from a Southern Pacific rattlesnake had no effect on coagulation at a concentration as large as 20 μg/ml after incubation in citrated plasma. It was thus assumed that the thrombin-like activity of this snake was calcium-dependent. Consequently, 336 μl of citrated plasma was mixed with 3.6 μl of Southern Pacific rattlesnake plasma (final concentration 10 μg/ml) and calcium chloride (CaCl2) as subsequently described. These experiments with Southern Pacific rattlesnake venom had the same four conditions (plasma without addition; plasma with venom, plasma with Fe, and CORM-2; and, plasma with Fe, CORM-2, and venom) as the previous two rattlesnake venom experiments. Using the plastic cup and pin as a weak contact protein activator, sufficient time was permitted for this snake venom to affect fibrinogen in an environment with normal calcium concentrations before the biomechanical events associated with coagulation could be measured. The aforementioned plasma and venom mixtures were placed in a disposable cup in a computer-controlled thrombelastograph® hemostasis system (Model 5000, Haemonetics Inc.) and incubated for 5 min at 37°C, with addition of 20 µl of 200 mM CaCl2 as the last step to initiate clotting. Data were collected at 37°C for 15 min during experiments involving timber or Eastern diamondback rattlesnakes, whereas samples exposed to Southern Pacific rattlesnake venom had data collected until maximum amplitude was determined by the analyzer software (to allow for variability associated with a lack of a standard incubation time). The following elastic modulus-based parameters previously described 16,17,19 were determined: time to maximum rate of thrombus generation (TMRTG: this is the time interval (min) observed prior to maximum speed of clot growth); maximum rate of thrombus generation (MRTG: this is the maximum velocity of clot growth observed (dynes/cm2/s); and total thrombus generation (TTG, dynes/cm2), the final viscoelastic resistance observed after clot formation.
Statistics and graphics
Data are presented as mean ± SD. The four conditions compared were as follows: (1) additive naive plasma, (2) plasma exposed to venom, (3) plasma exposed to Fe and CORM-2 (Fe/CO) addition, or (4) plasma exposed to Fe/CO addition followed by venom exposure. Each condition was represented by n = 6 replicate experiments. A commercially available statistical program was used for one-way analysis of variance followed by Holm-Sidak post hoc analyses (SigmaStat 3.1, Systat Software, Inc., San Jose, California, USA). Graphics depicting TMRTG, MRTG, and TTG data were generated with a commercially available program (OrigenPro 7.5, OrigenLab Corporation, Northampton, Massachusetts, USA).
Results
Data generated with additive naive plasma, plasma exposed to venom, plasma exposed to Fe and CORM-2 (Fe/CO) addition, or plasma exposed to Fe/CO addition followed by venom exposure are displayed for the three types of rattlesnake in Figures 1 to 3. With regard to plasma exposed to timber rattlesnake venom, as depicted in the top panel of Figure 1, TMRTG values were not significantly affected by venom exposure, but addition of Fe/CO significantly decreased TMRTG values in the absence or presence of venom compared to venom-naive or venom-exposed plasma samples. In contrast, as seen in the middle panel of Figure 1, venom exposure significantly decreased MRTG values when compared to venom-naive plasma; further, addition of Fe/CO significantly increased MRTG values compared to the first two conditions, and venom exposure coupled with Fe/CO addition resulted in MRTG values significantly greater than the other three conditions. Lastly, as displayed in the bottom panel of Figure 1, with regard to TTG, venom exposure significantly decreased TTG values when compared to venom-naive plasma; further, addition of Fe/CO significantly increased TTG values compared to the first two conditions, and venom exposure coupled with Fe/CO addition resulted in TTG values significantly larger than all other conditions.

Effect of timber rattlesnake venom, Fe and CO exposure on coagulation in human plasma. Data are presented as mean ± SD. Panel A: TMRTG (min); Panel B: MRTG (dynes/cm2/s); Panel C: TTG (dynes/cm2); Control = additive naive plasma; V = 2.5 μg/ml venom added, 5 min incubation prior to calcium addition; Fe/CO = 10 μM FeCl3 and 100 μM CORM-2 addition and 5 min incubation prior to calcium addition; Fe/CO/V = plasma exposed to FeCl3 and CORM-2 prior to 5 min incubation with venom before calcium addition. *p < 0.05 vs. control; † p < 0.05 vs. V; ‡ p < 0.05 vs. Fe/CO. Fe: iron; CO: carbon monoxide; TMRTG: time to maximum rate of thrombus generation; MRTG: maximum rate of thrombus formation; TTG: total thrombus generation; FeCl3: ferric chloride.
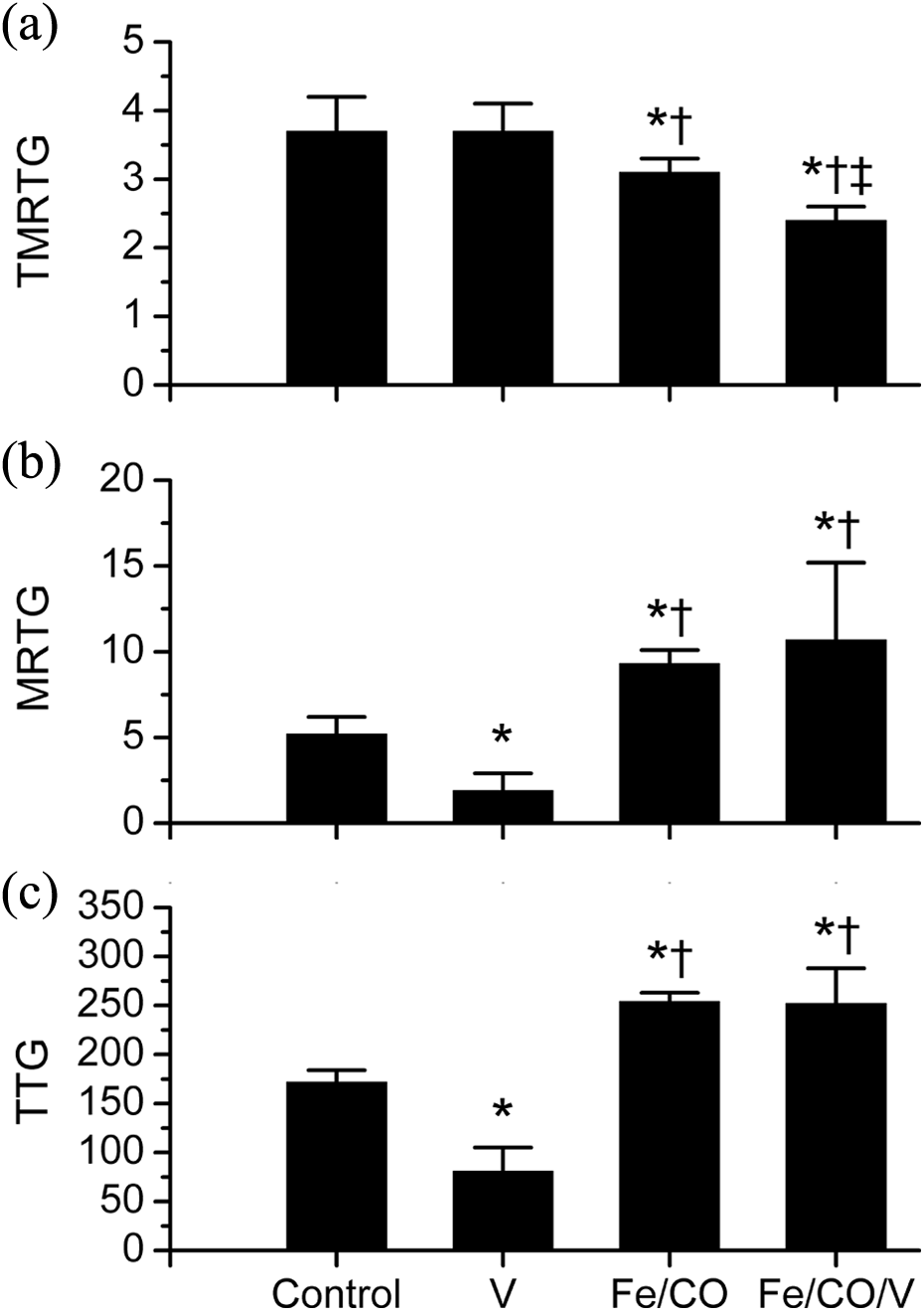
Effect of Eastern diamondback rattlesnake venom, Fe and CO exposure on coagulation in human plasma. Data are presented as mean ± SD. Panel A: TMRTG (min); Panel B: MRTG (dynes/cm2/s); Panel C: TTG (dynes/cm2); control = additive naive plasma; V = 2.5 μg/ml venom added, 5 min incubation prior to calcium addition; Fe/CO = 10 μM FeCl3 and 100 μM CORM-2 addition and 5 min incubation prior to calcium addition; Fe/CO/V = plasma exposed to FeCl3 and CORM-2 prior to 5 min incubation with venom before calcium addition. *p < 0.05 vs. control; † p < 0.05 vs. V; ‡ p < 0.05 vs. Fe/CO. Fe: iron; CO: carbon monoxide; TMRTG: time to maximum rate of thrombus generation; MRTG: maximum rate of thrombus formation; TTG: total thrombus generation; FeCl3: ferric chloride.
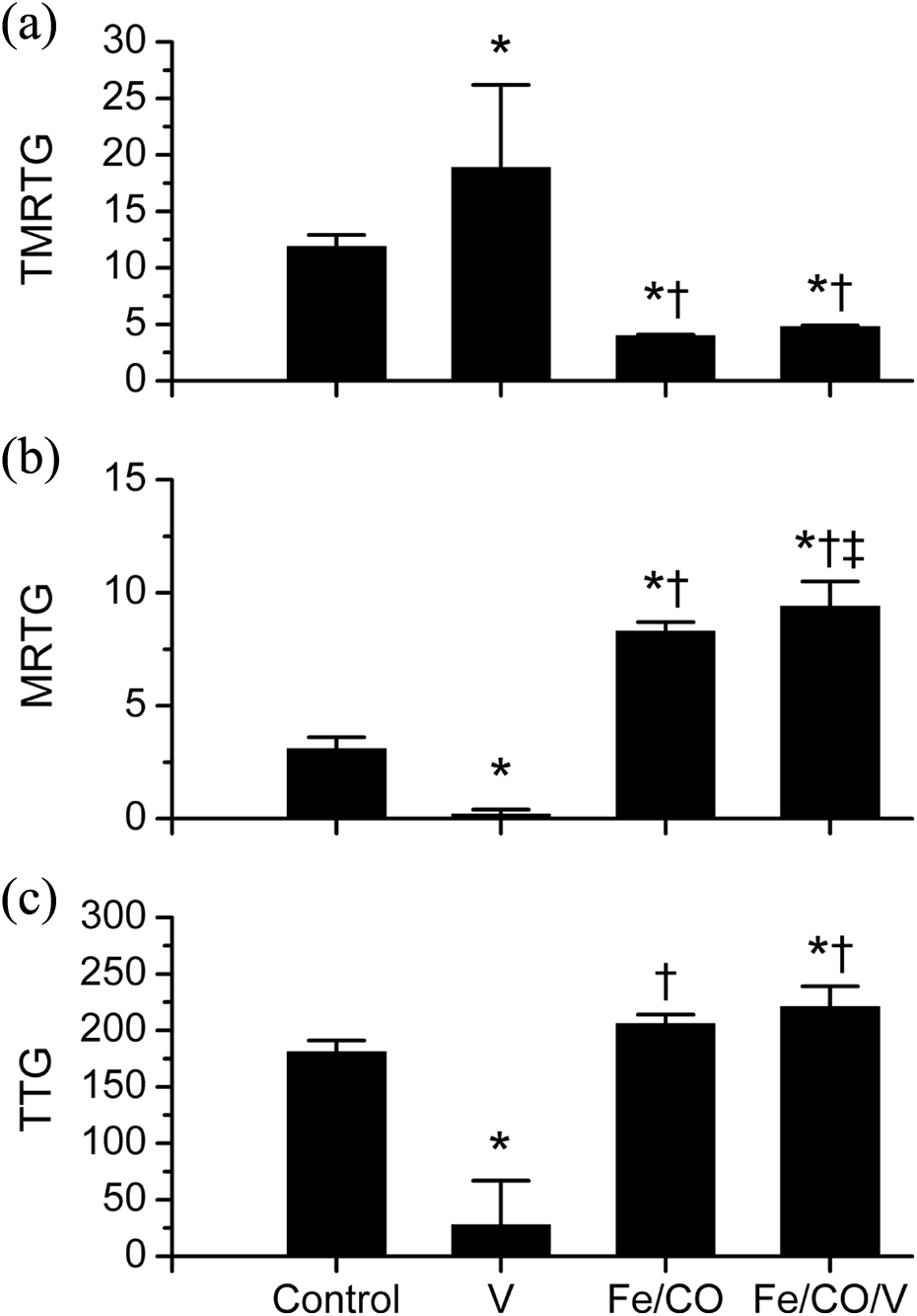
Effect of Southern Pacific rattlesnake venom, Fe and CO exposure on coagulation in human plasma. Data are presented as mean ± SD. Panel A: TMRTG (min); Panel B: MRTG (dynes/cm2/s); Panel C: TTG (dynes/cm2); control = additive naive plasma; V = 10 μg/ml venom added, just prior to calcium addition; Fe/CO = 10 μM FeCl3 and 100 μM CORM-2 addition; Fe/CO/V = plasma exposed to FeCl3 and CORM-2 prior to addition of venom and calcium. *p < 0.05 vs. control; † p < 0.05 vs. V; ‡ p < 0.05 vs. Fe/CO. Fe: iron; CO: carbon monoxide; TMRTG: time to maximum rate of thrombus generation; MRTG: maximum rate of thrombus formation; TTG: total thrombus generation; FeCl3: ferric chloride.
As for plasma exposed to Eastern diamondback rattlesnake venom, as depicted in the top panel of Figure 2, TMRTG values were not significantly affected by venom exposure, but addition of Fe/CO significantly decreased TMRTG values in the absence of venom compared to venom-naive or venom-exposed plasma samples, and Fe/CO addition followed by venom exposure resulted in significantly smaller TMRTG values compared to all other conditions. In contrast, as seen in the middle panel of Figure 2, venom exposure significantly decreased MRTG values when compared to venom-naive plasma; further, addition of Fe/CO significantly increased MRTG values compared to the first two conditions in the absence or presence of venom. Lastly, as noted in the bottom panel of Figure 2, with regard to TTG, venom exposure significantly decreased TTG values when compared to venom-naive plasma; further, addition of Fe/CO significantly increased TTG values compared to the first two conditions in the presence or absence of venom.
Plasma exposed to Southern Pacific rattlesnake venom had significantly greater TMRTG values (Figure 3, top panel), significantly smaller MRTG values (Figure 3, middle panel), and significantly smaller TTG values (Figure 3, bottom panel) compared to venom-naive plasma. Addition of Fe/CO to venom-naive plasma resulted in TMRTG values significantly smaller than the first two conditions, MRTG values significantly greater than the first two conditions, and TTG values greater than venom-exposed plasma. Lastly, plasma with Fe/CO exposed to venom had TMRTG values significantly less than the first two conditions, MRTG values significantly greater than all other conditions, and TTG values significantly greater than plasma samples with or without venom exposure without Fe/CO addition.
The effects of each venom on the three thrombelastographic parameters in plasma are compared in Table 1 for facile reference.
Comparison of the effects of rattlesnake venom on thrombelastographic parameters.

TMRTG: time to maximum rate of thrombus generation; MRTG: maximum rate of thrombus generation; TTG: total thrombus generation; NC: no change from plasma without venom exposure; ↑ = increased compared to plasma without venom exposure; ↓ = decreased compared to plasma without venom exposure; ↓↓: a greater decrease than that observed with the other two venoms.
Discussion
The major findings of this investigation include the documentation that plasmatic coagulation kinetics are degraded by the thrombin-like activity from venom of all three rattlesnakes tested. However, the profile of compromised coagulation varied between species (Table 1). Timber rattlesnake venom and Eastern diamondback rattlesnake venoms primarily decreased the speed of thrombus formation and strength, whereas Southern Pacific rattlesnake venom significantly delayed the onset of maximum speed of clot growth as well as significantly decreased the speed of clot growth and strength. Critically, addition of Fe/CO abrogated all venom-mediated degradations of coagulation derived from all three rattlesnakes. Further, each venom demonstrated a thrombin-like activity that positively enhanced coagulation in samples with Fe/CO addition; specifically, timber rattlesnake venom enhanced MRTG and TTG values, Eastern diamondback rattlesnake venom enhanced TMRTG values, and Southern Pacific rattlesnake venom enhanced MRTG values. Considered as a whole, these unique observations support the concept that the molecular targets of thrombin and the various venoms on fibrinogen are distinct from one another, and that their sequential activation may result in a negative or positive, additive effect on coagulation depending on the configuration of fibrinogen. Put another way, the proposed mechanism by which Fe and CO may protect coagulation from venom thrombin-like activity is by creating a “stealth fibrinogen” that is not seen molecularly by venom proteases.
The amount of Fe and CORM-2 used in vitro in the present investigation is significantly less than has been administered in vivo 20,21 including in the clinical setting. Using a rabbit ear-bleed model, CORM-2 was injected at a dose of 10 mg/kg (equivalent to 279 µM final concentration) to effect CO-mediated resistance to tissue-type plasminogen-mediated coagulopathy without adverse effects noted. 20 As for Fe, in order to administer a dose that would result in an increase of 10 µM in the circulation, consider the following: assuming a molecular weight of 55.8 atomic mass units for elemental Fe, multiply 0.558 µg/ml by 70 ml/kg (estimated blood volume) and by 70 kg (an average weight), for a final result of approximately 2.7 mg of Fe. It is clinical practice to infuse up to 100 mg of Fe to augment red blood cell production, a value more than 30-fold of the amount required to enhance coagulation. 21 In sum, administration of Fe and CO in the quantities required to modify fibrinogen to attenuate degradation by thrombin-like activity in the venom of the rattlesnakes tested may be clinically achievable without adverse effects.
These findings also have implications for diagnostic data collection of snakebite victims. Fibrinogen concentrations are typically measured from citrated plasma, and if a venom’s activity is calcium-independent, then the concentration of fibrinogen may be artificially decreased if there is a prolonged time from collection to analysis in the clinical laboratory. In contrast, calcium-dependent venoms would not cause such an artifact; thus, one might expect that blood samples obtained by patients bitten by a timber or Eastern diamondback rattlesnake may have very small fibrinogen concentrations, especially if some delay in centrifugation or other processing steps prolonged the time to analysis. In sum, the data of the present investigation should also give clinical laboratorians pause when considering the diagnostic evaluation of plasma fibrinogen concentration for the care of snakebite patients.
It is important to consider the multiple regional and systemic effects of snake venom, which are mediated in part by zinc metalloproteinases that induce a hemorrhagic state and myotoxins that destroy vascular endothelial and muscle cells, resulting in pulmonary and circulatory failure. 22 –24 Further, the tissue damage that occurs may permanently debilitate the snakebit victim that often times requires surgical intervention that include fasciotomies for ischemic compartment syndromes. 25 It is not proposed that preserving fibrinogen with Fe and CO administration will attenuate all the other effects of crotaline venoms, but instead is possible such intervention will at least potentially diminish coagulopathy in snakebite victims. Further, administration of blood products pretreated with Fe and CO (e.g., fresh frozen plasma, and cryoprecipitate) may have a longer intravascular duration in such patients. In sum, the utilization of Fe and CO to protect fibrinogen from venom-mediated digestion is envisioned as a part of a multimodal approach to treat patients envenomed with hemotoxic enzymes.
In conclusion, these data demonstrate an attenuation of the effects of timber rattlesnake, Eastern diamondback rattlesnake, and Southern Pacific rattlesnake venom on fibrinogen activation via Fe/CO addition that is not based on immunological principles (e.g., administration of antivenom). Further investigation of other pit viper venoms that possess thrombin-like activity is indicated to see if there is significant conservation of venom enzymatic target recognition of specific amino acid sequences such that Fe and CO can reliably attenuate venom-mediated catalysis of fibrinogen. These data serve as a rationale for future preclinical investigation to see if administration of Fe/CO can attenuate venom-mediated degradation of fibrinogen-dependent coagulation. Then, perhaps, administration of Fe and CO could serve as a “bridge-to-antivenom” in settings involving prolonged patient transport, or as concurrent therapy with antivenom to treat venom-mediated coagulopathy.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This investigation was supported by the Department of Anesthesiology, the University of Arizona, College of Medicine, Arizona, USA.
