Abstract
Hypofibrinogenemia is an important clinical consequence following envenomation by Lachesis muta muta, usually attenuated or prevented by administration of antivenom. The venom of L. m. muta contains both a metalloproteinase fibrinogenase and a serine protease thrombin-like enzyme, and exposure of fibrinogen to iron (Fe) and carbon monoxide (CO) has been demonstrated to decrease its catalysis by such enzymes. Using thrombelastographic analytical techniques, it was determined that this venom displayed weak procoagulant effects combined with fibrinogenolytic effects, and pretreatment of plasma with Fe and CO markedly attenuated venom-mediated effects. Additional experiments involving heparin exposure and varying calcium concentrations demonstrated that modification of fibrinogen with Fe and CO in human plasma rendered fibrinogen not recognizable to the fibrinogenolytic metalloproteinase but did not prevent polymerization by the thrombin-like serine protease. Lastly, when venom was exposed to CO in isolation and then placed in plasma, the fibrinogenase was inhibited but the thrombin-like enzyme was not inhibited. In sum, utilizing relatively facile modifications, we demonstrated with thrombelastography that Fe and/or CO addition can protect human plasmatic coagulation from fibrinogenase activity but not the effects of the thrombin-like activity of L. m. muta venom.
Introduction
Envenomation by bushmaster vipers (Lachesis species) has long been a source of significant morbidity and occasional mortality in South and Central America. 1,2 Systemic consumption of fibrinogen is a significant result of envenomation by bushmasters, 1,2 with this specific venom known to be composed of both thrombin-like serine proteases and fibrinogenolytic metalloproteinases. 3 –6 The Lachesis genera are an ancient one, with these snakes have been posited to have given rise to the Agkistrodon and Crotalus genera, 7 which typically have either thrombin-like enzymes or fibrinogenases. 8 –11 In sum, Lachesis vipers are an important source of envenomation with venom that has a complex nature.
Of interest, it has recently been demonstrated that exposure of fibrinogen in plasma to small quantities of ferric iron (Fe) and carbon monoxide (CO) modifies the molecule such that it is catalysed by either fibrinogenase or thrombin-like snake venom enzymes in a significantly slower manner as determined by coagulation kinetic changes assessed with thrombelastography. 8 –11 The venoms tested thus far were collected from North American Agkistrodon and Crotalus species. 8 –11 Further, we have demonstrated in unpublished work under consideration elsewhere that snake venom metalloproteinases, with either fibrinogenase or thrombin-like activity, can be directly inhibited by exposure to CO. Considered as a whole, there was a possibility that modification of fibrinogen with Fe and CO and inhibition of metalloproteinase activity could attenuate the deleterious effects of Lachesis venom on plasmatic coagulation.
Thus, one of the objectives of this study was to assess whether pretreatment of plasma with Fe and CO would attenuate venom-mediated degradation of coagulation with Lachesis muta muta venom. The other objective was to determine if exposure of this venom in isolation to CO would attenuate its effects on plasmatic coagulation.
Methods
Plasma, venom and chemicals
Pooled normal human plasma (George King Bio-Medical, Overland Park, Kansas, USA) anticoagulated with sodium citrate (9 parts blood to 1 part 0.105 M sodium citrate) stored at −80°C was utilized in all subsequently described experiments. Fifty milligram of lyophilized L. m. muta was obtained from the National Natural Toxins Research Center at Texas A&M University (Kingsville, Texas, USA). The venom was reconstituted in calcium-free phosphate-buffered saline (PBS, Sigma–Aldrich, St. Louis, Missouri, USA) at a concentration of 50 mg/mL, aliquoted, and stored at −80°C until experimentation. Ferric chloride (FeCl3, 99.9% pure), tricarbonyldichlororuthenium (II) dimer (CORM-2, a CO-releasing molecule), calcium-free PBS and dimethyl sulfoxide (DMSO) were obtained from Sigma–Aldrich.
General thrombelastographic analyses
The coagulation kinetics of plasma exposed to no venom (contact protein activation via interaction with plastic cup and pin surface) was compared with plasma exposed to venom. The rationale for this approach was to permit venom time to interact with plasma prior to the biomechanical engagement of the cup and pin by the relatively weak thrombin generation via contact activation, with the resultant coagulation kinetics reflecting either degraded or enhanced venom-mediated change. The final plasma sample mixture volume for both subsequently described series of experiments was 359.6 µL. Sample composition consisted of 336 µL of plasma, 3.6 µL of PBS or venom (final concentration 2.0 µg/mL) and 20 µL of 200 mM calcium chloride (CaCl2; Haemonetics Inc., Braintree, Massachusetts, USA), which were rapidly mixed with immediate data collection commenced. This concentration of venom was chosen as it would be expected to contain the equivalent of 1–2 IU/mL of thrombin activity 3,4 so as to effect the coagulation before thrombin generation from contact activation via the thrombelastographic cup and pin (Haemonetics Inc., Braintree). The aforementioned plasma and venom mixtures were placed in a disposable cup in a computer-controlled thrombelastograph® haemostasis system (Model 5000, Haemonetics Inc., Braintree) at 37°C. The following elastic modulus-based parameters described previously 8 –12 were determined: time to maximum rate of thrombus generation (TMRTG): this is the time interval (min) observed prior to maximum speed of clot growth; maximum rate of thrombus generation (MRTG): this is the maximum velocity of clot growth observed (dyne/cm2/s); and total thrombus generation (TTG, dyne/cm2), the final viscoelastic resistance observed after clot formation; and time to 0.1 dyne/cm2/s (TT0.1): this is the time in seconds to an increase in velocity of thrombus formation of 0.1 dyne/cm2/s. The rationale for the use of TT0.1 as a new parameter was that in some cases plasma exposed to venom commenced clotting very quickly, but at a very small velocity that is not typical of samples previously studied by us, and it was viewed as a new phenomenon indicative of weak, venom-mediated thrombin-like activity. Data were collected for 30 min.
Experiments to assess the effects of pre-exposure of plasma to Fe/CO on venom-mediated coagulation
Plasma was rapidly thawed at 37°C on the day of experimentation. In this first series of experiments, plasma had 1% additions of FeCl3, in PBS and CORM-2 dissolved in DMSO; this mixture of additives was denoted as Fe/CO. The final concentrations of FeCl3 and CORM-2 were 0–10 µM and 0–100 µM, respectively; these concentrations are associated with nearly maximal augmenting effects on coagulation kinetics. 12 All plasma samples had 1% DMSO and PBS additions with or without the aforementioned chemicals, so the samples contained the vehicles used for all additions. Plasma was exposed to these various agents for at least 5 min prior to placement into plastic thrombelastographic cups (Haemonetics Inc., Braintree).
Experiments to assess the effects of Fe/CO on venom-mediated changes in coagulation after suppression of thrombin activity by heparin addition or calcium deficiency
Plasma was rapidly thawed at 37°C on the day of experimentation. In this second series of experiments, plasma had 1% additions of Fe/CO or PBS/DMSO vehicle as described previously. To separate the contribution to coagulation kinetics of thrombin from that of metalloproteases contained within this venom, another series of experiments was conducted without or with heparin and with or without calcium chloride added to plasma prior to venom exposure. Unfractionated heparin (1000 U/mL, SAGENT Pharmaceuticals, Schaumburg, Illinois, USA) was added to plasma for a final concentration of 4 U/mL (4 μL/mL addition), which is a concentration expected to engage antithrombin to the degree observed for anticoagulation during cardiopulmonary bypass. 13 In experiments assessing the effects of calcium on coagulation kinetics, either PBS or calcium chloride at subsequently mentioned concentration and volume were added to the plasma mixture. All samples had venom (2 μg/mL final concentration; 3.6 μL of venom solution added to 336 μL plasma mixture and 20 μL of 200 mM CaCl2 or PBS) added just prior to commencement of 30 min of data collection.
Isolated venom exposure to CO experiments
In unpublished work under consideration elsewhere by us, CO was found to inhibit snake metalloproteinase activity, presumably by reacting with the transitional Zn+2 located in the catalytic centre. To isolate the effects of CO from the carrier molecule of CORM-2, a 100 µM solution of CORM-2 was placed in a sealed plastic tube and incubated in to a 37°C water bath for 18 h in order to release CO and become inactive (iCORM-2) as has been described previously. 14 To test the effects of CO on venom activity, a 1 mL quantity of a 500 µg/mL solution of venom in PBS was prepared for each condition subsequently presented. The conditions were (1) 1% addition (v/v) of PBS and DMSO to PBS without venom; (2) 1% addition (v/v) of PBS and DMSO to PBS with venom; (3) 1% addition of CORM-2 to PBS with venom; and lastly, (4) 1% addition of PBS and iCORM-2 to PBS with venom. After 3 min of incubation at room temperature, 3.6 µL of solution from one of these conditions was placed into 336 µL of plasma in a disposable cup in a computer-controlled thrombelastograph® haemostasis system (Model 5000, Haemonetics Inc.). After test solution addition, 20 µL of 200 mM CaCl2 was immediately added. Data were collected at 37°C for 30 min.
Statistical analyses
For the first two series of experiments, data are presented as mean ± SD or as raw data in thrombus growth velocity curve figures. A commercially available statistical program was used for one-way analysis of variance followed by Holm–Sidak post hoc analyses (SigmaStat 3.1, Systat Software, Inc., San Jose, California, USA). Graphics depicting viscoelastic data were generated with a commercially available program (OrigenPro 7.5, OrigenLab Corporation, Northampton, Massachusetts, USA; CorelDRAW12, Corel Corporation, Mountain View, California, USA).
With regard to the isolated venom exposures to CORM-2, data are presented as median (first–third quartiles). All conditions were represented by n = 6 replicates. If no coagulation occurred, the sample observed was assigned a TMRTG value of 30 min depending on the experimental series, with values for MRTG and TTG designated as 0. As the data were found not to be normally distributed, Kruskall–Wallis one-way analysis of variance followed by Student–Newman–Keuls post hoc test were used for analyses (SigmaStat 3.1, Systat Software, Inc.). A p value <0.05 was considered significant.
Results
Experiments to assess the effects of pre-exposure of plasma to Fe/CO on venom-mediated coagulation
The results of these experiments are displayed in Table 1 with representative raw clot growth velocity curves depicted in Figure 1. Compared to plasma without additives, plasma exposed to venom demonstrated significantly greater TMRTG (2-fold), smaller MRTG (>10-fold) and smaller TTG (4-fold) values. However, venom-exposed plasma had significantly smaller (4-fold) TT0.1 values compared to additive naïve plasma, demonstrating a far quicker commencement of coagulation as seen in Figure 1 (panels (a) and (b)). Thrombi formed with plasma pre-exposed to Fe/CO had significantly smaller TMRTG (3-fold), larger MRTG (6-fold), greater TTG (2-fold) and smaller TT0.1 (3-fold) values compared to additive naïve plasma; further, Fe/CO pre-exposed plasma demonstrated coagulation kinetic parameters significantly different from venom-exposed plasma with the exception of TT0.1. Finally, plasma pre-exposed to Fe/CO subsequently exposed to venom still had significantly smaller TMRTG, greater MRTG, larger TTG values compared to additive naïve plasma and plasma exposed to venom alone; however, compared to plasma pre-exposed to Fe/CO alone, plasma with both Fe/CO and venom had significantly smaller MRTG and TTG values. Lastly, while smaller than additive naïve plasma TT0.1 values, plasma with Fe/CO and venom had TT0.1 values not different from the other two conditions.
Effects of plasma pretreatment of Fe/CO on venom-mediated degradation of coagulation kinetics.a

TMRTG: maximum rate of thrombus generation (min); MRTG: maximum rate of thrombus generation (dyne/cm2/s); TTG: total thrombus generation (dyne/cm2); TT0.1: time in second to an increase in velocity of thrombus formation of 0.1 dyne/cm2/s; additive naïve plasma: plasma exposed to 1% addition of DMSO/PBS; venom: plasma exposed to DMSO/PBS followed by addition of 2 μg/mL final of venom; Fe/CO: plasma exposed to 10 µM FeCl3 and 100 µM CORM-2; Fe/CO + venom: plasma exposed to 10 µM FeCl3 and 100 µM CORM-2 followed by addition of 2 μg/mL final of venom; PBS: phosphate-buffered saline; DMSO: dimethyl sulfoxide.
aData are presented as mean ± SD.
b p < 0.05 versus additive naïve plasma.
c p < 0.05 versus venom.
d p < 0.05 versus Fe/CO.

Clot growth velocity curves of plasma exposed to venom without or with pre-exposure of the plasma with Fe/CO. (a) Additive naïve plasma, (b) plasma exposed to venom 2 μg/mL, (c) plasma pre-exposed to Fe/CO and (d) plasma pre-exposed to Fe/CO followed by exposure to venom 2 μg/mL. Note that the Y-axis of panels (a) and (b) are 8-fold less than those of panels (c) and (d). Fe: iron; CO: carbon monooxide.
Experiments to assess the effects of Fe/CO on venom mediated changes in coagulation after suppression of thrombin activity by heparin addition or calcium deficiency
In order to determine if Fe/CO pretreatment of plasma improved coagulation kinetics in venom-exposed samples was secondary to attenuating venom-mediated degradation of fibrinogen, two methods to quench thrombin generation and factor XIII activation were utilized to reassess coagulation kinetics. As can be observed in Table 2 and Figure 2, in the presence of heparin coagulation occurred, with very small velocity of clot growth and strength. In contrast, plasma pretreated with heparin and Fe/CO subsequently exposed to venom demonstrated a significant increase in TMRTG (2-fold), increase in MRTG (8-fold) and increase in TTG (>10-fold) compared to plasma only pretreated with heparin. A similar pattern was observed, with withholding of calcium associated with a quick onset but slow growing and weak clot that became significantly faster growing and stronger following Fe/CO pretreatment. Lastly, both TMRTG and TT0.1 values were significantly greater in plasma samples exposed to venom without calcium addition with Fe/CO pretreatment compared to samples with both heparin and Fe/CO pretreatment.
Effects of pretreatment of Fe/CO in the absence or presence of heparin or calcium on venom-mediated degradation of coagulation kinetics.a
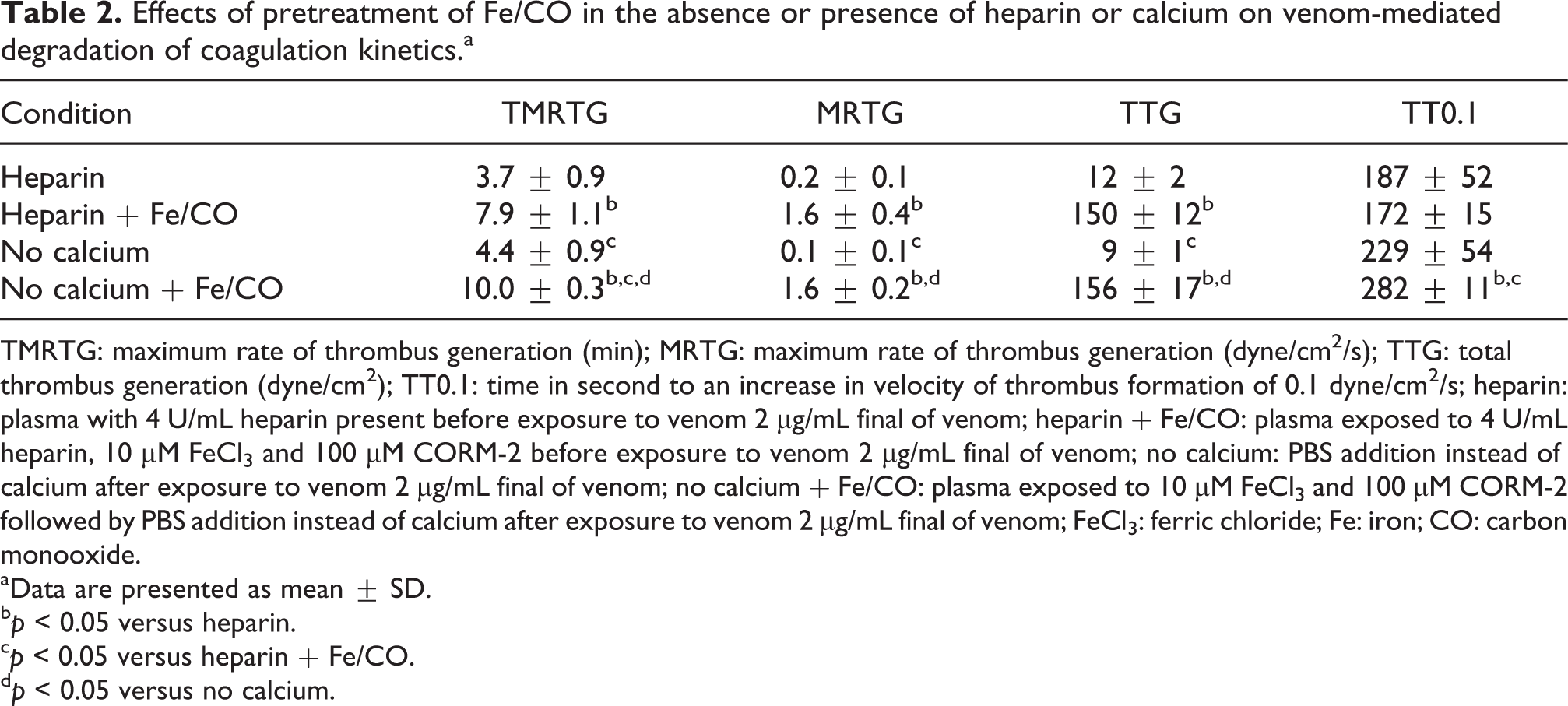
TMRTG: maximum rate of thrombus generation (min); MRTG: maximum rate of thrombus generation (dyne/cm2/s); TTG: total thrombus generation (dyne/cm2); TT0.1: time in second to an increase in velocity of thrombus formation of 0.1 dyne/cm2/s; heparin: plasma with 4 U/mL heparin present before exposure to venom 2 μg/mL final of venom; heparin + Fe/CO: plasma exposed to 4 U/mL heparin, 10 µM FeCl3 and 100 µM CORM-2 before exposure to venom 2 μg/mL final of venom; no calcium: PBS addition instead of calcium after exposure to venom 2 μg/mL final of venom; no calcium + Fe/CO: plasma exposed to 10 µM FeCl3 and 100 µM CORM-2 followed by PBS addition instead of calcium after exposure to venom 2 μg/mL final of venom; FeCl3: ferric chloride; Fe: iron; CO: carbon monooxide.
aData are presented as mean ± SD.
b p < 0.05 versus heparin.
c p < 0.05 versus heparin + Fe/CO.
d p < 0.05 versus no calcium.

Clot growth velocity curves of plasma exposed to venom without or with pre-exposure of the plasma with Fe/CO in the presence of heparin or absence of calcium. All samples had venom 2 μg/mL added after plasma was exposed to: (a) heparin 4 U/mL, (b) heparin 4 U/mL and Fe/CO, (c) PBS, no calcium addition and (d) PBS, no calcium addition, Fe/CO. Note that the Y-axis of panels (a) and (c) are 10-fold less than those of panels (c) and (d). Fe: iron; CO: carbon monoxide; PBS: phosphate-buffered saline.
Isolated venom exposure to CO experiments
The results of these experiments are displayed in Table 3. Compared to plasma exposed to PBS/DMSO, plasma exposed to venom had significantly prolonged TMRTG values (50% increase), decreased MRTG values (>10-fold) and decreased TTG (4-fold) values. Addition of venom exposed to CORM-2 in isolation to plasma resulted in TMRTG values significantly less, MRTG values significantly greater and TTG values significantly greater than that observed after CORM-2 naïve venom was added to plasma. However, the MRTG and TTG values observed after addition of venom exposed to CORM-2 in isolation were significantly less than those obtained with addition of PBS/DMSO to plasma. Addition of venom exposed to iCORM-2 in isolation to plasma resulted in coagulation kinetics not significantly different from that observed with addition of CORM-2 naïve venom, but significantly different from the two other conditions.
Effects of venom pretreatment with CO on venom-mediated degradation of coagulation kinetics.a

TMRTG: maximum rate of thrombus generation (min); MRTG: maximum rate of thrombus generation (dyne/cm2/s); TTG: total thrombus generation (dyne/cm2); DMSO/PBS vehicle: 1% addition of each of these vehicles to PBS solution without venom addition; venom + DMSO/PBS: 2 μg/mL final in DMSO/PBS vehicle solution; venom + CORM-2 2 μg/mL final after exposure to 100 μM CORM-2; venom + iCORM-2: 2 μg/mL final after exposure to 100 μM iCORM-2; PBS: phosphate-buffered saline; DMSO: dimethyl sulfoxide.
aData are presented as median (first–third quartiles).
b p < 0.05 versus DMSO/PBS.
c p < 0.05 versus venom + DMSO/PBS.
d p < 0.05 versus venom + CORM-2.
Discussion
The present investigation identified hereto-unappreciated aspects of L. m. muta venom effects on plasmatic coagulation. First, the most significant enzyme in terms of compromise of velocity of clot growth and strength was determined to be the fibrinogenolytic metalloproteinase, which markedly diminished coagulation kinetics (Table 1, Figure 1). Second, based on coagulation kinetic changes that occurred following pretreatment of plasma with Fe/CO, it can be inferred that fibrinogen catalysis by the metalloproteinase was diminished (Table 1, Figure 1). However, in the absence of thrombin generation and FXIII activation (Table 2, Figure 2), while it can be inferred that the metalloproteinase could not destroy fibrinogen following Fe/CO pretreatment of plasma, it is apparent that the serine protease with thrombin-like activity was able to polymerize fibrinogen. It is also important to note that both venom enzymes could function without calcium, but the serine protease appeared to be more active in the presence of calcium with heparin addition (Table 2). In the last series of experiments wherein venom was exposed in isolation to CO (Table 3), partial, but not complete attenuation of the effects of venom on plasmatic coagulation was observed after CORM-2 exposure. This is consistent with the data of the first two series of experiments, as it would be possible that CO could interact with and potentially inactivate the transitional zinc catalytic centre of the metalloproteinase, as has been observed with human metalloproteinases in vitro previously. 15 Taken as a whole, our data support the conclusion that modifying fibrinogen with Fe/CO likely protects it from fibrinogenolysis by the metalloproteinase found in L. m. muta venom; that exposure of the metalloproteinase to CO can inhibit its activity; and, that the serine protease with thrombin-like activity is not directly or indirectly affected by CO or Fe/CO in plasma, respectively.
Unlike three North American viper venoms with thrombin-like activity that were found to have their degradation of coagulation attenuated by pretreatment of plasma with Fe/CO, 10 the serine protease with thrombin-like activity found in the venom of L. m. muta easily polymerizes fibrinogen modified by Fe/CO. We recently documented the prothrombotic effects of the venom of Bothrops asper with thrombelastography, which contains a serine protease with thrombin-like activity, and these effects were enhanced in plasma pretreated with Fe/CO. 16 There is known to be a great deal of variability in enzyme structure and substrate molecular target among snake venom serine proteases with thrombin-like activity, 17 which may account for these three different patterns of coagulation kinetic response to Fe/CO pretreatment by divergent serine proteases with thrombin-like activity (e.g. decreased, unaffected, enhanced catalysis with fibrinogen).
In conclusion, the action of the fibrinogenolytic metalloproteinase contained in L. m. muta venom was attenuated by either modifying its substrate, fibrinogen, with Fe/CO or by presumably inhibiting its zinc catalytic centre with CO. However, neither biochemical approach prevented the enzymatic activity of the serine protease with thrombin-like activity found in the venom. Further preclinical investigations are warranted to determine if utilization of Fe/CO and/or CORM-2 as interventions to attenuate coagulopathy until antivenom therapy can be administered.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This investigation was supported by the Department of Anesthesiology, University of Arizona, College of Medicine, Arizona, USA.
