Abstract
Carbon nanotubes (CNTs) have been introduced recently as a novel carrier system for both small and large therapeutic molecules. Biotin-functionalized single-wall CNTs have been conjugated with the anticancer agent taxoid using a cleavable linker, and multiwall carbon nanotubes (MWCNTs) conjugated with iron nanoparticles have been efficiently loaded with doxorubicin. 1,2 The MWCNTs are effective transporters for biological macromolecules and drugs to target cells and tissues, thereby attracting the attention of the biomedical industry. 3 –7 Administrating MWCNTs for medical application invariably involves intravenous administration and ultimate contact with human peripheral blood lymphocytes (HPBLs), yet toxicological studies on the effect of MWCNTs on HPBLs are lacking. Accordingly, this study evaluated the cytotoxic and genotoxic effects of MWCNTs on healthy male HPBLs. Healthy male HPBLs were treated with MWCNTs at 3 different concentrations (12.5, 25, and 50 μg/mL) for 48 hours. Under these conditions, the MWCNTs induced significant cell growth retardation, DNA damage, and cytotoxicity. The MWCNT-treated HPBLs also exhibited an increased intracellular reactive oxygen species level during the experimental period, which leads to cell damage and death, proliferation inhibition, DNA damage, and an inflammatory response.
Keywords
Introduction
With the increasing commercial usage of nanomaterials, nanotechnology has been rapidly developing and attracting scientific interest in many fields. 8,9 In particular, carbon nanotubes (CNTs) have recently become a focus of industrial attention due to their distinctive physicochemical properties, including a high electrical conductivity, low density, and high strength. 10,11 As a result, more comprehensive information is clearly needed on the potential health effects of CNTs. 12,13 Most previous CNT toxicity studies have focused on the skin and lungs, since dermal contact and inhalation are the most likely routes of CNT exposure. 14,15 Yet, once CNTs are deposited in the lungs, they can also be translocated to extrapulmonary organs via blood circulation. Since the beginning of the 21st century, CNTs have been introduced as a drug delivery system in pharmacy and medicine due to their high surface area, good chemical stability, and rich electronic polyaromatic structure. 16 The CNTs could be adsorbed or conjugated with various therapeutic molecules such as peptides, proteins, DNA, RNA, and drugs. 4,6,7,17 –24 The CNT–bioactive molecule complex has been proven to be an excellent vehicle for drug delivery by penetrating into cells and keeping the drug from being metabolized during transport. 25 –28 However, these medicinal findings are in an experimental stage and are still not applied to human medicine. 16 The biomedical application of CNTs for drug delivery through blood vessels will inevitably result in contact with human blood cells. Therefore, blood cells also need to be included as an important toxicity evaluation target for CNT exposure. Yet, despite such exposure possibilities, there is still a lack of safety evaluation studies focusing on the effect of CNTs on human peripheral blood lymphocytes (HPBLs). Accordingly, this study investigated the effect of CNT treatment on HPBLs in terms of reactive oxygen species (ROS) generation, DNA damage, cytotoxicity, apoptosis, and inflammation.
Materials and Methods
Test Materials and Dispersion
Commercially available multiwall carbon nanotubes (MWCNTs) were obtained from Hanwha Nanotech, Inc (Incheon, Korea) and have also been designated an alternative reference material by the Organization for Economic Cooperation and Development Working Party on Manufactured Nanomaterials (WPMN) representative nanomaterial safety testing sponsorship program.
29
The physicochemical properties of the MWCNTs were determined using a scanning electron microscope (JSM-6700F; JEOL, Tokyo, Japan), dynamic light scattering (DLS; ELS-8000; Otsuka Electronics, Osaka, Japan), thermogravimetric analysis (Q500; TA Instruments, New Castle, DE), and Brunauer–Emmett–Teller surface area analysis (ASAP 2020; Micromeritrics, Norcross, GA; Table 1).
29
For the biological experiment, the MWCNTs were dispersed in a 1,2-dipalmitoyl-sn-glycero-3-phosphocholine (DPPC) solution, which was prepared in Ca2+ and Mg2+-free phosphate-buffered saline (PBS; GIBCO, Grand Island, NY), pH 7.4, supplemented with 5.5 mmol/L
Characteristics of Multiwall Carbon Nanotubes (MWCNTs). 29
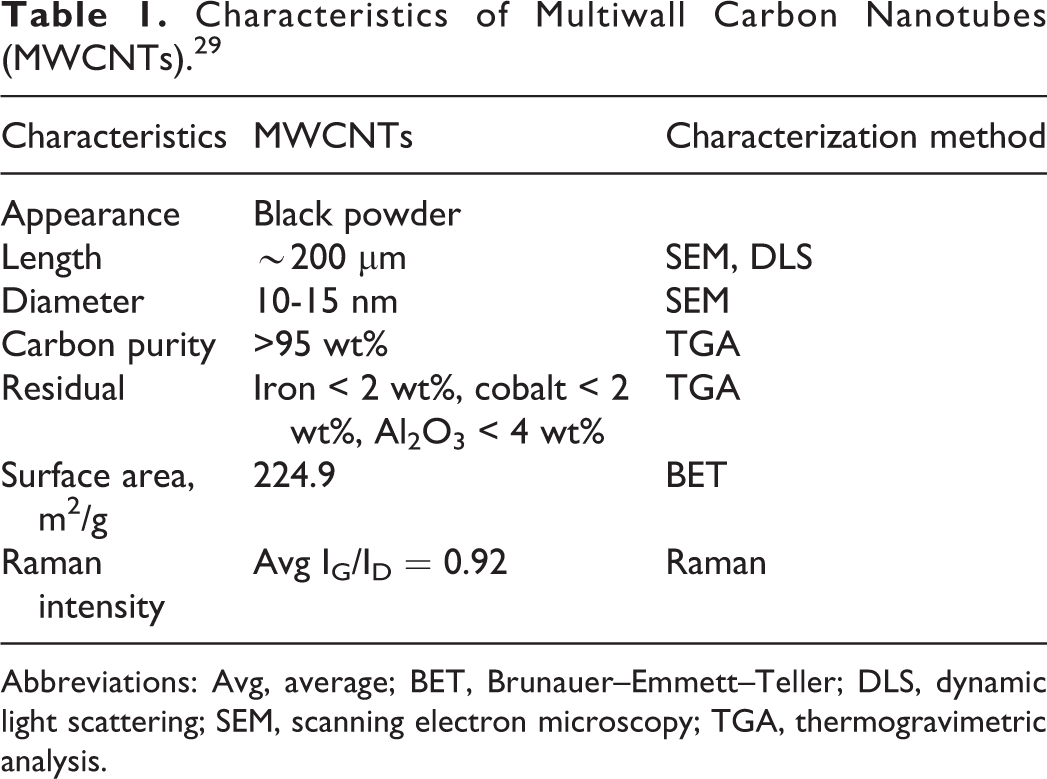
Abbreviations: Avg, average; BET, Brunauer–Emmett–Teller; DLS, dynamic light scattering; SEM, scanning electron microscopy; TGA, thermogravimetric analysis.
Human Peripheral Blood Lymphocyte Isolation and Culturing
The human peripheral blood was taken from a healthy volunteer (36-year-old male donor, nonsmoker, no alcohol consumption, and not taking any medication) into heparinized vacutainers after providing informed consent. The lymphocytes were then isolated from the fresh blood using Ficoll-Paque PLUS (Amersham Bio-Sciences, Sweden) according to the manufacturer’s instructions. Briefly, the diluted blood sample (4 mL) was carefully layered on the Ficoll-Paque PLUS (3 mL) in a centrifuge tube and then centrifuged at 400g for 30 minutes at 20°C. Thereafter, the lymphocyte layer was transferred to a clean centrifuge tube and washed twice in 6 mL of PBS at 100g for 10 minutes at 20°C, followed by resuspension in RPMI-1640 (GIBCO) media supplemented with 10% fetal bovine serum (Hyclone, Logan, UT), penicillin/streptomycin (100 units; GIBCO), and 1% phytohemagglutinin (PHA; Sigma) for cell proliferation induction. Finally, the lymphocytes were cultured in a 5% CO2 atmosphere at 37°C. Triplicate cultures were used for all the experiments.
Trypan Blue Dye Exclusion Assay
Trypan blue dye exclusion assay is based on the principle that live cells possess intact cell membranes that exclude certain dyes, whereas dead cells do not. In this test, a cell suspension is simply mixed with dye and then visually examined to determine whether cells take up or exclude dye. This assay procedure is as follows.
The isolated HPBLs were seeded in a T75 flask (2 × 105 cells/mL) and incubated with a 1% PHA solution for 24 hours at 37°C in a humidified 5% CO2 atmosphere. The lymphocytes were then treated with the MWCNTs for 24 to 48 hours in a T25 flask (2 × 105 cells/mL/5 mL). To measure the viable cells, a mixture of a 1:1 cell suspension and 0.4% trypan blue (Sigma) was collected and transferred to the edge of a hemacytometer chamber. The total number of unstained cells was then counted using a microscope (Carl Zeiss, Germany).
WST-1 Assay
Can you provide the basis for this assay as you did for the trypan blue exclusion assay? Cell viability quantification was performed using the cell proliferation reagent (4-[3-(4-Iodophenyl)-2-(4-Nitrophenyl)-2H-5-Tetrazolio]-1,3-Benzene Disulfonate)) (WST-1) (Roche, Germany). WST-1 is a ready-to-use colorimetric assay for the nonradioactive quantification of cellular proliferation, viability, and cytotoxicity. Also, WST-1 assay is low cytotoxic, more accurate, and sensitive than (3-(4,5-Dimethylthiazol-2-yl)-2,5-Diphenyltetrazolium Bromide) (MTT) and 3-(4,5-dimethylthiazol-2-yl)-5-(3-carboxymethoxyphenyl)-2-(4-sulfophenyl)-2H-tetrazolium (MTS) assay. Briefly, the isolated HPBLs were seeded in a T75 flask (2 × 105 cells/mL) and incubated with a 1% PHA solution for 24 hours at 37°C in a humidified 5% CO2 atmosphere. The lymphocytes were then treated with the MWCNTs for 24 to 48 hours in a T25 flask (2 × 105 cells/mL/5 mL). Next, 100 μL of the culture was transferred to a 96-well microplate and 10 μL of the cell proliferation reagent WST-1 added to the culture medium. The incubation was then continued for another 1 hour at 37°C in the 5% CO2 incubator. Thereafter, the samples were thoroughly shaken for 1 minute on a shaker, and the absorbance measured at 420 to 480 nm using a microplate reader (Spectra Max M2; Molecular Devices, Sunnyvale, CA).
Single Cell Gel Electrophoresis (Comet Assay)
For the first layer, the glass microscope slides were coated with 1.0% normal melting agarose and fully dried. The harvested HPBLs (2 × 104 cells/10 μL) were then mixed with 85 μL of 0.7% low melting agarose and rapidly spread on the first layer. Finally, 85 μL of 0.7% low melting agarose was spread as the top layer. The prepared slides were then soaked in an alkaline lysis buffer (2.5 mol/L NaCl, 100 mmol/L Na2-EDTA, 10 mmol/L Tris-HCl, 1% Triton X-100, and 10% dimethyl sulfoxide, pH 10) for 1 hour at 4°C. Next, the slides were washed in distilled water for 10 minutes and placed in a horizontal electrophoresis chamber, followed by electrophoresis in an alkaline buffer (1 mmol/L Na2-EDTA, 300 mmol/L NaOH, pH 13) for 25 minutes at 20 V and 275 mA. Thereafter, the slides were gently washed in a neutralization buffer (0.4 mol/L Tris-HCl, pH 7.4) and immersed in 100% ethanol for 1 hour. The slides were then stained with 100 μL of ethidium bromide (10μg/mL) and analyzed using a fluorescent microscope and image program (Kinetic Imaging, United Kingdom). 30
Micronuclei Assay
The HPBLs were seeded (provide cell #) into RPMI 1640 culture medium (GIBCO), pH 7.3, supplemented with 10% heat inactivated fetal bovine serum (Hyclone), 100 units/mL penicillin (GIBCO), and 1% PHA (Sigma). The HPBLs were then cultured in a 5% CO2 atmosphere at 37°C. After 24 hours of incubation, the MWCNTs were added to the cultured peripheral blood at concentrations of 0, 12.5, 25, and 50 μg/mL. The incubation was continued and 4 μg/ mL of cytochalasin-B (Sigma) was added after 20 hours from the start of the culture. The cultures were then incubated for an additional 28 hours, making a total incubation time of 48 hours. Next, the harvested cells were incubated with a KCl solution (75 mmol/L) for 3 minutes and then fixed in a methanol/glacial acetic acid solution (3:1, v/v). This fixation step was repeated twice, and the resulting cells dropped onto clean slides. After being air dried, the cells were stained using a Giemsa solution and then observed under a light microscope (Carl Zeiss, Germany). Micronuclei (MNs) were evaluated based on Fenech criteria 31 and measured per 1000 binucleated cells. In addition, 600 cells were scored to evaluate the percentage of cells with 1 to 4 nuclei, while the cytokinesis-block proliferation index (CBPI) was calculated using the following expression: CBPI = [MI + 2MII + 3(MIII + MIV)]/total, where MI through MIV represent the number of cells with 1 to 4 nuclei, respectively, and MIII and MIV are both considered to be in their third cycle. 32
Flow Cytometric Detection of Apoptosis
Apoptotic cells typically exhibit DNA fragmentation and lost nuclear DNA content. Thus, to evaluate apoptotic cell death, researchers generally use a propidium iodide (PI) flow cytometric assay, which can rapidly and precisely obtain the cellular DNA content using fluorochrome. 33 While additional stains are needed to confirm apoptosis, such as annexin V or caspase 3/7 activation within 24 hours, the current experiment was limited to an initial apoptosis analysis.
The isolated HPBLs were seeded in a T75 flask (2 × 105 cells/mL) and incubated with a 1% PHA solution for 24 hours at 37°C in a humidified 5% CO2 atmosphere. The lymphocytes were then treated with various concentrations of the MWCNTs for 48 hours in a T25 flask (2 × 105 cells/mL/5 mL). Thereafter, the lymphocytes were collected, washed with cold PBS, and fixed with 70% ethanol at 4°C for at least 1 hour. The fixed cells were then washed with PBS, treated with RNase (30 minutes at 37°C, 100 μg/mL; Sigma), and their DNA stained with PI (50 μg/mL; Sigma). Finally, the DNA content was measured using a FACSCalibur (BD), where a minimum of 10 000 cells were counted for each sample.
Intracellular ROS Measurement
The intracellular ROS were measured using 2′,7′-dichlorofluorescin diacetate (DCFH-DA; Sigma), which undergoes an oxidative conversion to the fluorescent compound dichlorofluorescin (DCF). 34,35 Briefly, the isolated HPBLs (2 × 105 cells/mL/5 mL) were seeded in T25 flasks and incubated with the MWCNTs (0, 12.5, 25, and 50 μg/mL) for 24 and 48 hours. After removing the medium, the samples were resuspended in 200 μL of PBS and transferred to 96-well plates. Next, the samples were diluted in 2 μL of a DCFH-DA stock solution (2 mmol/L; final concentration was 20 μmol/L), incubated for 30 minutes, and the DCF fluorescence then determined at an excitation of 480 nm and emission of 530 nm using a microplate reader (Spectra Max M2; Molecular Devices). All the measurements were taken in triplicate.
Cytokine Enzyme-Linked Immune Absorbent Assays
The isolated HPBLs were seeded in a T75 flask (2 × 105 cells/mL) and incubated with a 1% PHA solution for 24 hours at 37°C in a humidified 5% CO2 atmosphere. The lymphocytes were then treated with various concentrations of the MWCNTs for 24 and 48 hours in a T25 flask (2 × 105 cells/mL/5 mL). The cells were separated from the released tumor necrosis factor α (TNF-α) and interleukin 6 (IL-6) by centrifugation at 400g for 5 minutes at 4°C. 36 The TNF-α and IL-6 levels in the supernatants were assayed using commercially available human enzyme-linked immune absorbent assay system kits (Phoenix Pharmaceuticals, Inc, California).
Statistical Analysis
The statistical analyses were performed using SPSS 12.1, and the data expressed as the mean ± standard error. A 1-way analysis of variance was applied to test all the data. A value of P < .05 indicated statistical significance.
Results
Cell Proliferation
A dose-range finding test was first conducted to determine the treatment concentrations for the cytotoxity and genotoxicity tests. Because the test substance (MWCNT) was only able to wet at below 0.5% concentration (w/v), we could use 100 μg/mL for the highest treatment concentration. Dose ranges were estimated for all the controls and cultures treated with the MWCNTs at 7 dose levels (1.56, 3.125, 6.25, 12.5, 25, 50, and 100 μg/mL) using the trypan blue dye exclusion method, and the highest treatment concentration for the experiments was determined at around 50% (50% ± 5%) of the control. As a result of the dose-range finding tests, 50 μg/mL exhibited a 50% viability for the experiment duration (Figure 1). Therefore, the remainder of this study was conducted using 50 μg/mL as the highest dose level, along with subsequent 2-fold serial dilutions of 25 and 12.5 μg/mL.
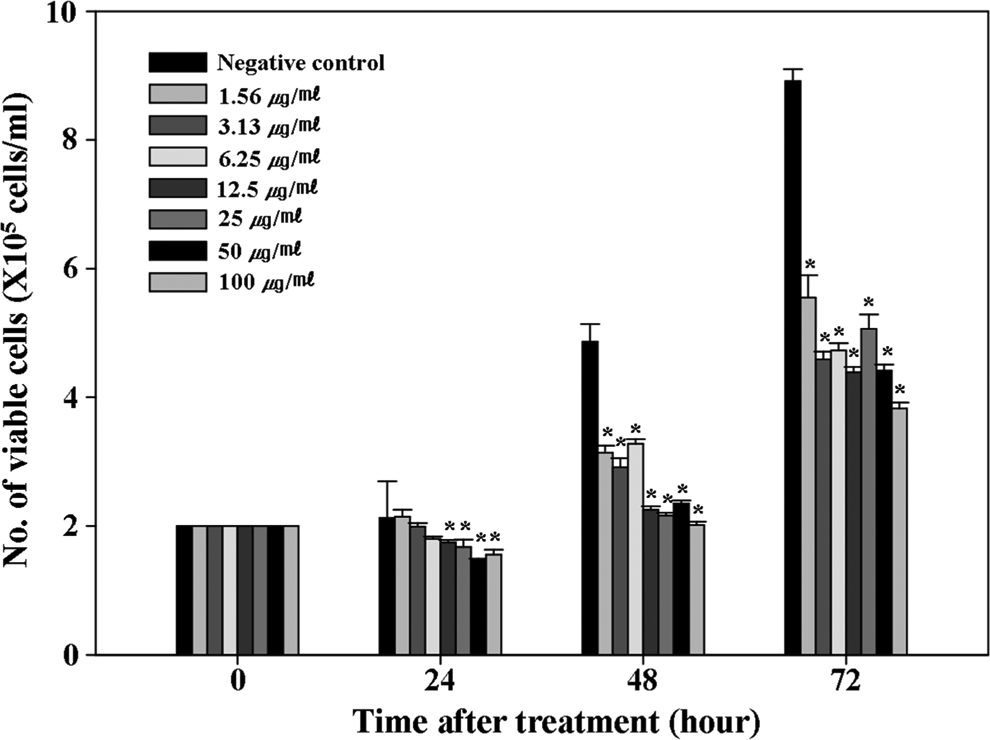
Dose-range finding test data for phytohemagglutinin (PHA)-stimulated healthy male human peripheral blood lymphocytes (HPBLs) treated with increasing doses of multiwall carbon nanotubes (MWCNTs); statistically significant difference indicated by *P < .05 when compared with the control.
To investigate the cytotoxic effect of the MWCNTs, a trypan blue dye exclusion assay and WST-1 assay were also performed. In the trypan blue dye exclusion assay, the control lymphocytes grew exponentially, whereas the MWCNT-treated lymphocytes exhibited growth retardation at all concentrations (12.5, 25, and 50 μg/mL) after 48 hours (Figure 2A). The cell proliferation rate was also measured using a WST-1 assay (Figure 2B). The MWCNTs significantly inhibited the HPBL growth at all dose levels after 24 hours of treatment (P < .05). After 48 hours of treatment, the MWCNTs induced dose-dependent cell growth inhibition. In particular, the highest treatment concentration (50 μg/mL) exhibited statistically significant toxicity when compared with the control (P < .05).
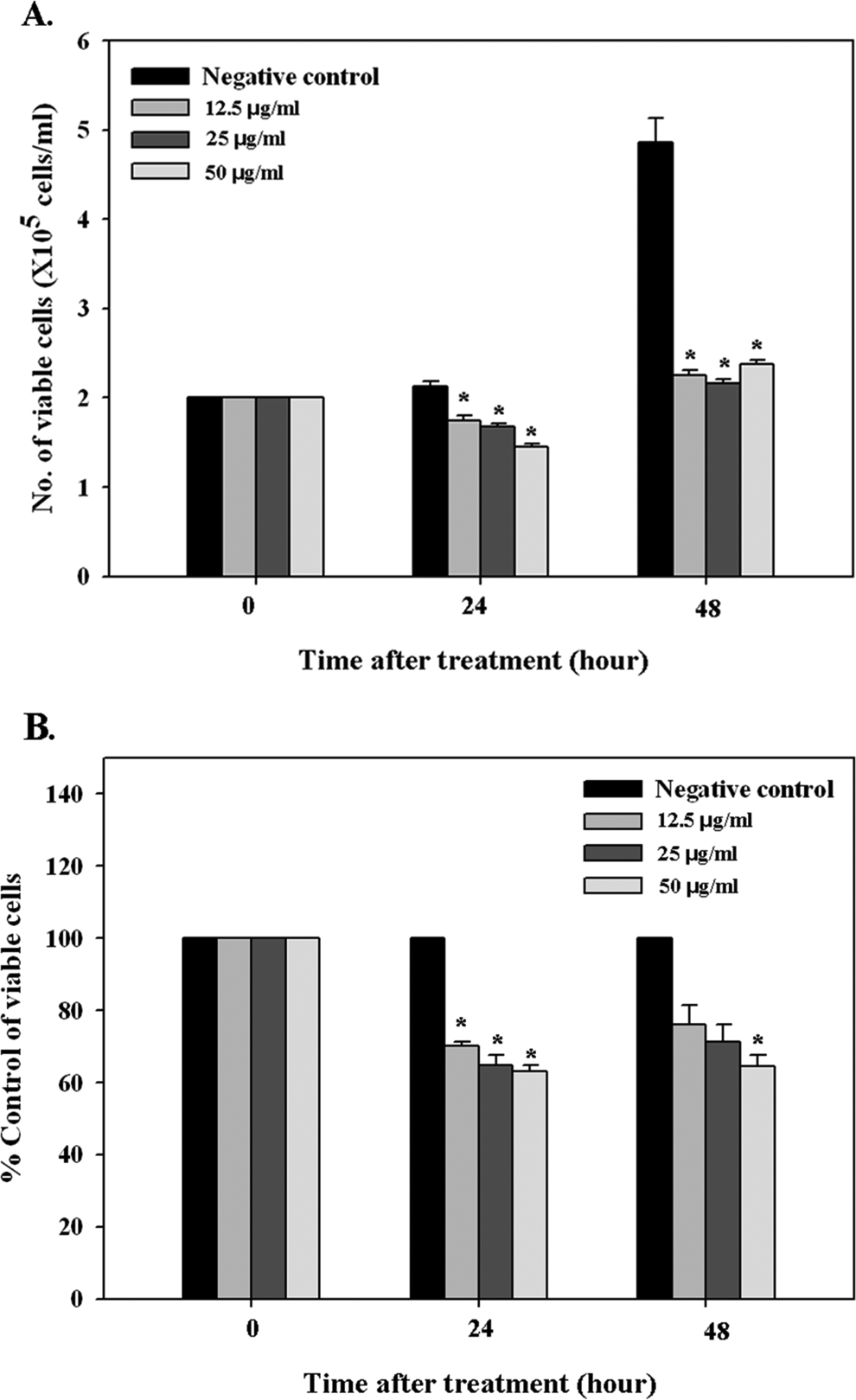
Cell proliferation and cytotoxicity measurement data for phytohemagglutinin (PHA)-stimulated healthy male human peripheral blood lymphocytes (HPBLs) treated with increasing doses of multiwall carbon nanotubes (MWCNTs). (A) Trypan blue dye exclusion assay and (B) WST-1 assay; statistically significant difference indicated by *P < .05 when compared with the control.
DNA Damage
Single cell gel electrophoresis (Comet assay) and a MN assay were performed to investigate the DNA damage effect of the MWCNT treatment. The comet assay was conducted after 24 and 48 hours of treatment with 0, 12.5, 25, and 50 μg/mL concentrations of MWCNTs. A comet assay is considered a rapid, sensitive method for detecting DNA damage, such as DNA single-strand breakages, alkali labile sites, and cross-links at the individual cell level. 30,37,38 As a result of the comet assay, the olive tail moments (OTMs) were 18.89 ± 3.57, 58.72 ± 13.28, 78.49 ± 7.47, and 64.28 ± 13.29 for the 0, 12.5, 25, and 50 μg/mL concentrations, respectively, after 24 hours of treatment (Figure 3A). Thus, the MWCNTs induced DNA damage at all the treatment concentrations (12.5, 25, and 50 μg/mL) with a statistical significance after 24 hours of treatment when compared with the control (P < .05). After 48 hours of treatment, the OTMs were 10.50 ± 2.17, 11.41 ± 3.20, 28.75 ± 6.08, and 27.47 ± 5.83 for the 0, 12.5, 25, and 50 μg/mL treatment concentrations, respectively (Figure 3B). In particular, treatment with 25 and 50 μg/mL of MWCNTs enhanced the OTMs to a statistically significant level when compared with the control (P < .05).
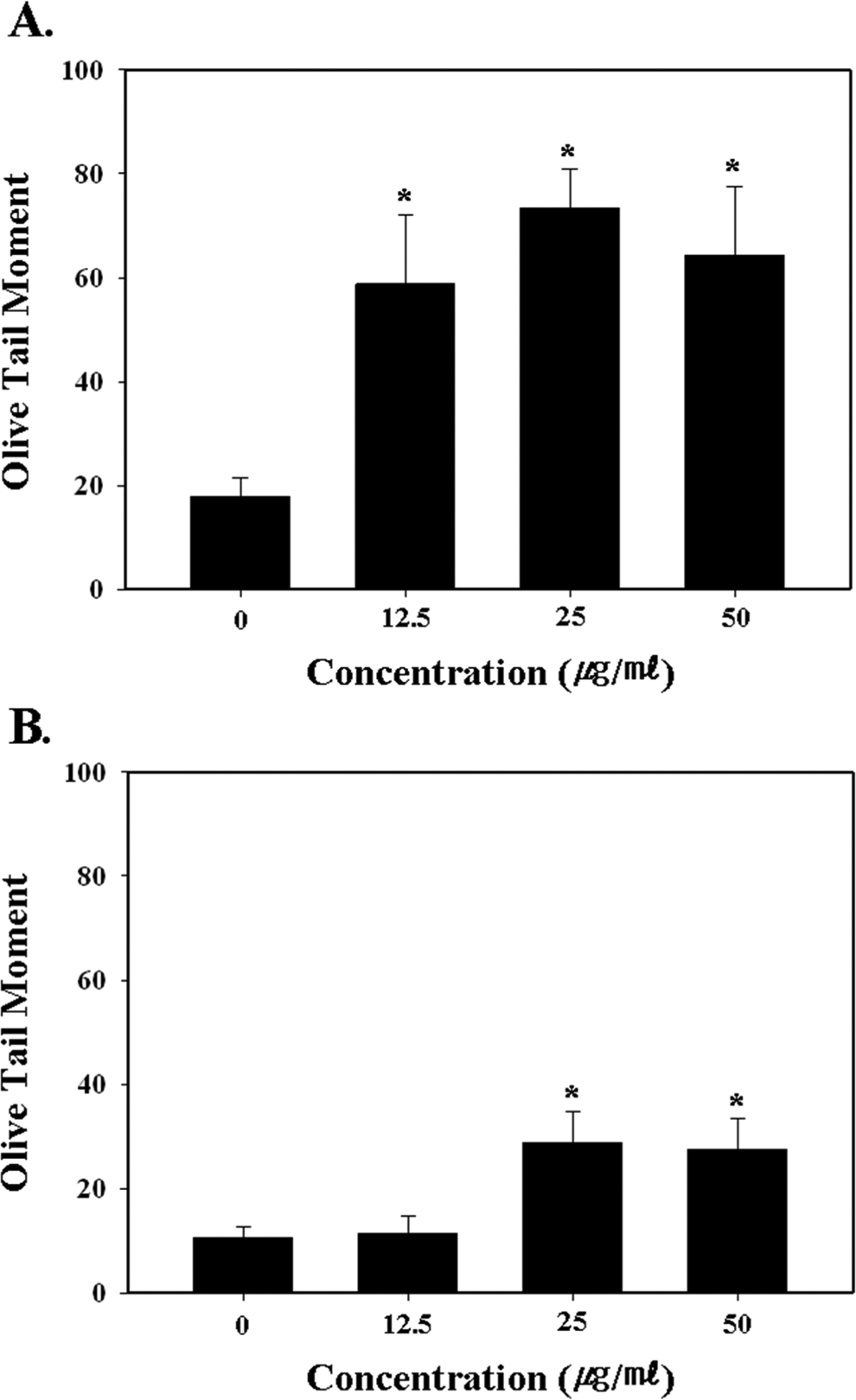
Quantitative assessment of DNA damage in multiwall carbon nanotube (MWCNT)-treated normal human peripheral blood lymphocytes (HPBLs) using single cell gel electrophoresis (Comet assay). The slides were stained with ethidium bromide (10 μg/mL) and analyzed using a fluorescent microscope and image program. (A) After 24 hours of treatment and (B) after 48 hours of treatment; statistically significant difference indicated by *P < .05 when compared with the control.
A MN test is an effective tool for measuring cytogenetic damage in binucleate HPBLs and cultured cell lines. 39,40 In the MN test results, the frequency of MNs in 1000 binucleated cells was 1.67 ± 0.67, 11.00 ± 2.08, 14.00 ± 2.52, and 16.67 ± 1.20 for the 0, 12.5, 25, and 50 μg/mL treatment concentrations, respectively (Figure 4A). The MN frequencies induced by the MWCNT treatment increased significantly (P < .05) when compared with the control, plus the increase was dose dependent. The MWCNT treatment did not change the CBPI, which is a biological index for detecting cellular toxicity or a cell cycle delay (Figure 4B).
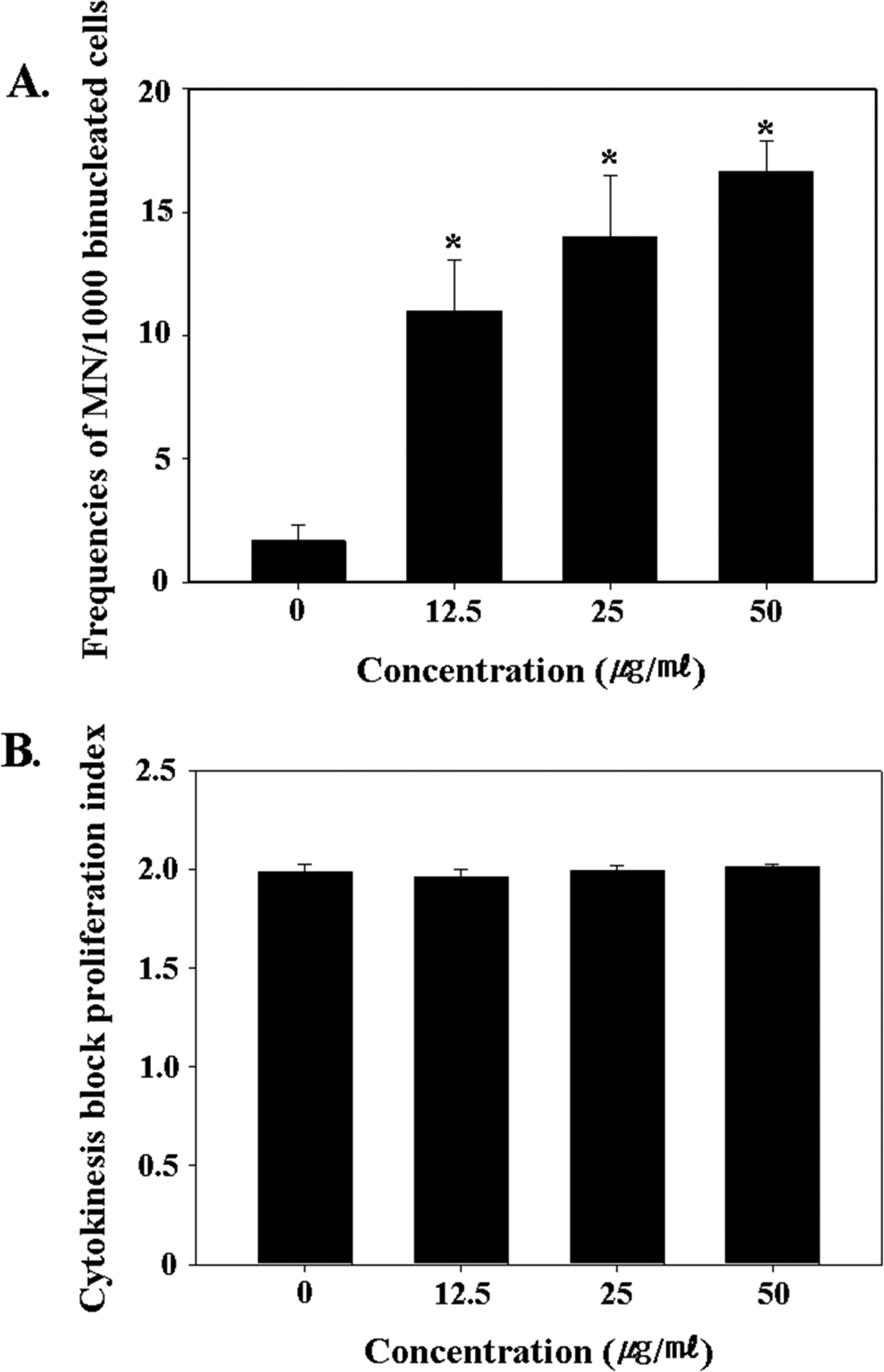
Measurement of (A) micronuclei frequencies and (B) cytokinesis block proliferation index in the control and multiwall carbon nanotube (MWCNT)-treated normal human peripheral blood lymphocytes (HPBLs). Statistically significant difference is indicated by *P < .05 when compared with the control.
Apoptosis
To examine whether MWCNT treatment induces apoptotic cell death in normal HPBLs, the apoptotic cell death of the MWCNT-treated cells was measured based on the change in the hypodiploid DNA contents in the HPBLs using a flow cytometry assay (Figure 5). Following the MWCNT treatment, the hypodiploid DNA contents in the MWCNT-treated cells were 12.63%, 45.37%, 61.74%, and 73.92% for the 0, 12.5, 25, and 50 μg/mL treatment concentrations, respectively. The apoptotic cell death in the MWCNT-treated groups ranged from 4.0- to 6.0-fold higher than that in the control group.
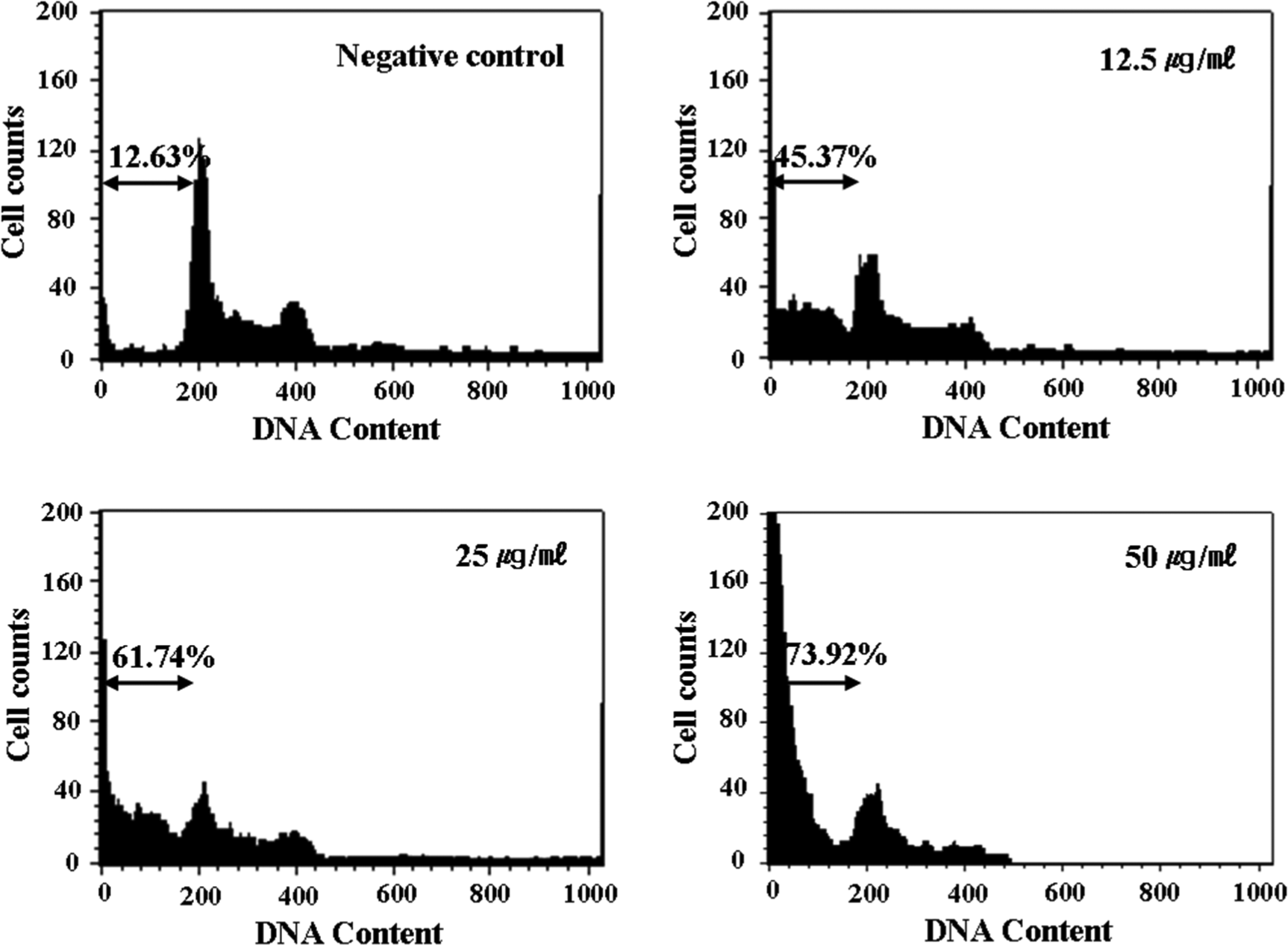
Measurement of apoptosis using propidium iodide staining in the control and multiwall carbon nanotube (MWCNT)-treated normal human peripheral blood lymphocytes (HPBLs).
Intracellular ROS Generation
It is already well known that intracellular ROS are an important signal mediator of cell proliferation or death and an inducer of genotoxicity. 41 –44 Therefore, this study examined the intracellular ROS levels resulting from each MWCNT exposure level. As shown in Figure 6, all the MWCNT-treated groups exhibited significantly increased intracellular ROS levels, about 3- to 4-fold (after 24 hours of treatment) and 2-fold (after 48 hours of treatment) higher than the levels in the control cells (P < .05). Notwithstanding, since the measurements were only taken after 24 hours, it is also possible that the measured ROS may have been the result of cell death rather than ROS in response to the MWCNT exposure.
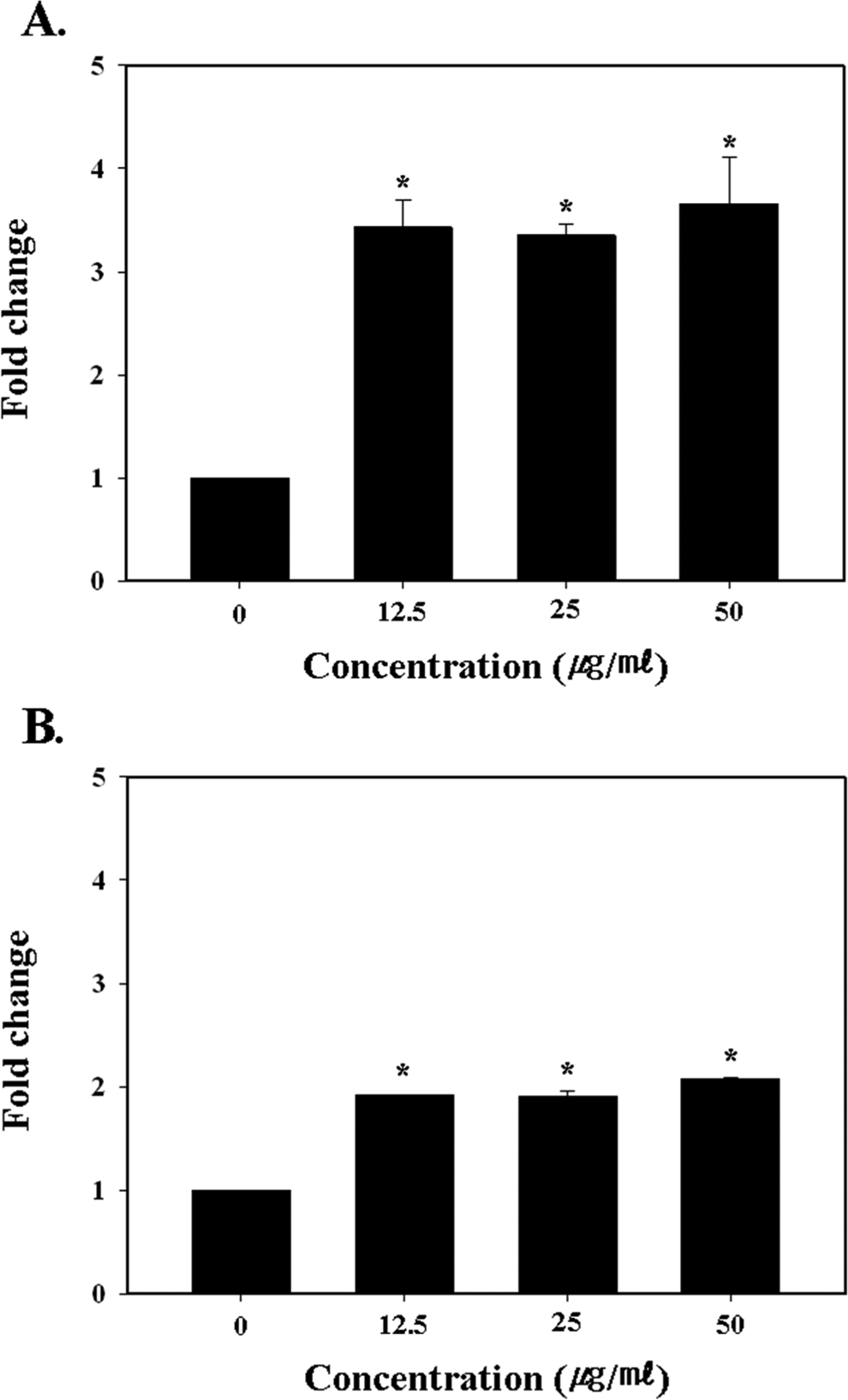
Measurement of intracellular reactive oxygen species (ROS) level in the control and multiwall carbon nanotube (MWCNT)-treated normal human peripheral blood lymphocytes (HPBLs). (A) After 24 hours of treatment and (B) after 48 hours of treatment; statistically significant difference indicated by *P < .05 when compared with the control.
Tumor necrosis factor α and IL-6 Release
To investigate whether MWCNTs can increase the proinflammatory cytokines release, the normal HPBLs were treated with 12.5, 25, and 50 μg/mL of MWCNTs for 48 hours. As shown in Figures 7A and C, after 24 hours of treatment, the lymphocytes did not exhibit a significant increase in TNF-α and IL-6 secretion when compared to the control. After 48 hours of treatment, the TNF-α release was also not significantly increased with any of the treatment concentrations, except for a slight increase with the highest treatment concentration (Figure 7B). In contrast, after 48 hours of treatment, the IL-6 secretion dose dependently increased and was significantly higher with the highest treatment concentration (P < .05; Figure 7D).
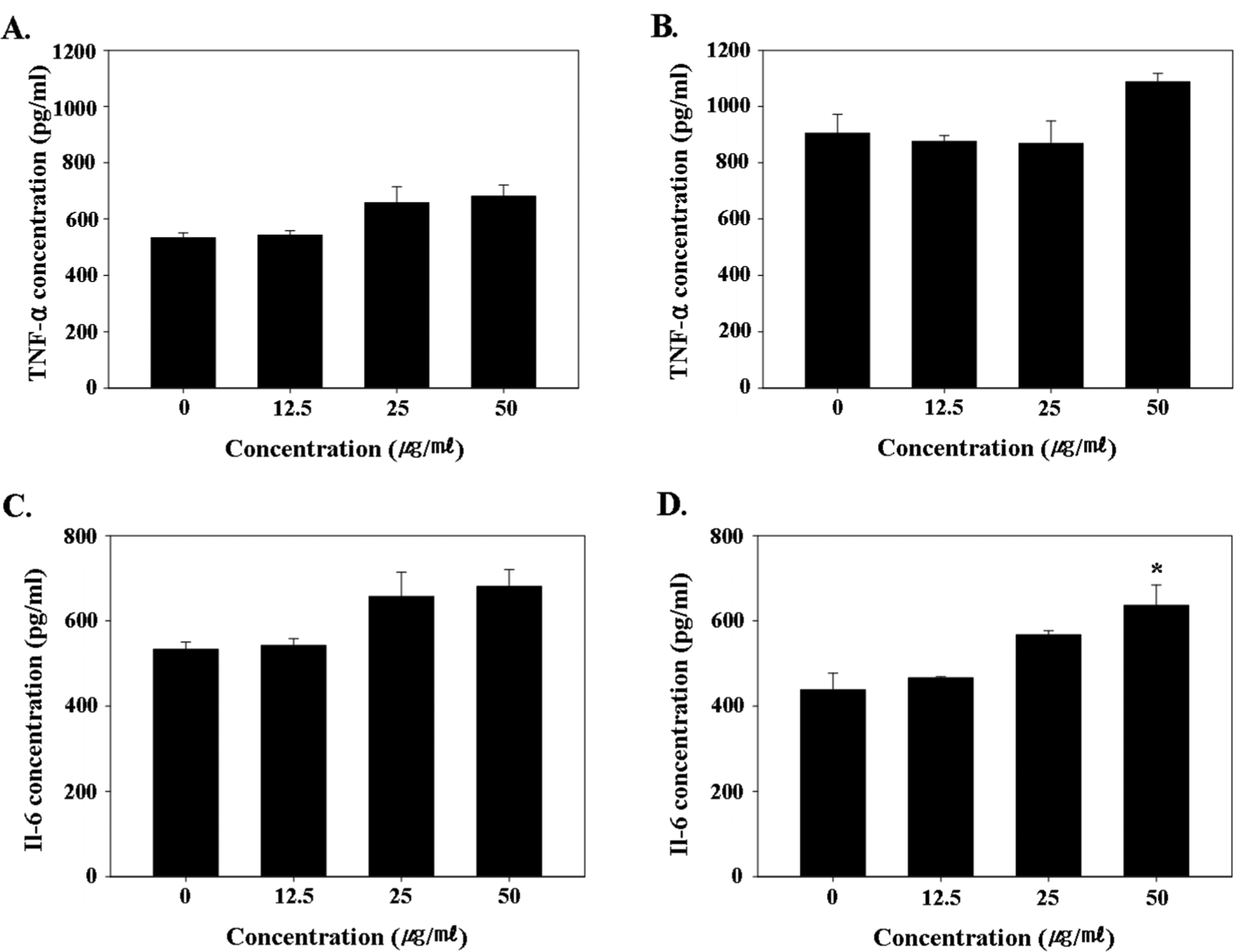
Measurement of TNF-α and IL-6 secretion by multiwall carbon nanotube (MWCNT)-treated normal human peripheral blood lymphocytes (HPBLs). (A) TNF-α level after 24 hours of treatment, (B) TNF-α level after 48 hours of treatment, (C) IL-6 level after 24 hours of treatment, and (D) IL-6 level after 48 hours of treatment; statistically significant difference indicated by *P < .05 when compared with the control. IL-6, interleukin 6; TNF-α, tumor necrosis factor.
Discussion
Nanomaterials have specific physicochemical properties that are attractive for many industrial and scientific applications. 45,46 In particular, nanomaterials can potentially revolutionize certain medical fields, such as diagnostics and therapy strategies, since they have the ability to reach and affect target organs and tissues at the molecular and cellular levels. 47 Furthermore, drug delivery system research has shown that nanomaterials may help solve drug solubility and stability problems and even minimize drug side effects. 48 However, medical and pharmacological researchers are only focused on the application of nanoscale materials and are not seriously considering the side effects of their usage. Plus, current information on nanotoxicity remains far from complete, warranting further thorough investigations. 46,49,50 Among nanomaterials, CNTs are known as effective transporters of biological macromolecules and drugs to target cells and tissues. 4,6,17 –24 However, this type of medical application implies intravenous administration and translocation into to the blood circulation. Therefore, this study investigated whether MWCNTs produce cytotoxic and genotoxic effects in healthy HPBLs. The test substance used in this study was commercially available MWCNTs, which have also been designated as an alternative reference material in the sponsorship program for the safety testing of nanomaterials by the OECD WPMN. 29 First, the cytotoxicity resulting from MWCNT treatment was examined. In terms of the toxic effect on HPBLs, single-wall carbon nanotubes (SWCNTs) at concentrations of 25 and 50 μg/mL have already been found to decrease the proliferation of HPBLs, yet did not induce cell death and apoptosis. 15 Also, MWCNTs have not previously been found to have a significant effect on lymphocyte proliferation or apoptosis. 24 However, in contrast to these results, the present study found that cells treated with MWCNTs showed dose-dependent growth defects and spontaneous cell death (Figure 1). In particular, the MWCNTs induced cell proliferation inhibition after 24 hours of treatment, although the cell growth inhibition recovered with continued treatment beyond 24 hours (Figure 2).
Genotoxicity testing results are very important as they provide information for the hazard identification and risk assessment of CNTs in the carcinogenesis process. 51 Therefore, the genotoxic effect of MWCNT treatment was investigated using a comet assay and MN assay. Until now, no genotoxicity studies have investigated the impact of MWCNT treatment on normal HPBLs, although SWCNTs are known to induce genotoxicity in HPBLs. 52 In the present study, the MWCNTs induced genotoxicity in normal HPBLs (Figures 3 and 4). In the comet assay, the MWCNTs induced a 3- to 5-fold higher OTM when compared with the control after 24 hours of treatment at all the treatment concentration levels. Plus, the OTM for the 25 and 50 μg/mL concentrations was about 2-fold higher than that for the control after 48 hours of treatment. In the MN assay results, the MN frequencies were dose dependently increased by the MWCNT treatment and about 10-fold higher than that in the control. It is already known that nanomaterials induce genotoxicity for 2 main reasons: ROS generation, which is caused by the high atom-to-surface ratio, and mechanical interference with cellular components, such as the nucleosome, microtubules, actin filament, and centrosome. 46,51,53 The ROS are also known to be involved in diverse physiological processes, including cell growth and differentiation, senescence, and cell death in various cell types, including prokaryotic and eukaryotic cells. 54 Plus, ROS give rise to various genotoxicities, including DNA damage, gene mutation, MN formation, and chromosome aberrations. 55 –62 Therefore, the intracellular ROS level was measured to investigate the genotoxicity due to ROS generation. The intracellular ROS level was significantly increased at all the treatment concentrations when compared with the control cells (Figure 6). Interestingly, the MWCNT-treated cells maintained similar levels of intracellular ROS after 48 hours, yet the ROS level in the cells at 48 hours was about 1.5-fold higher than that at 24 hours in the control group (data not shown). It has been reported that iron and iron derivatives (such as heme or iron-sulfur [Fe-S] clusters) are incorporated into and essential for the function of ROS-producing enzymes. 63 Iron has also been observed at the active site of the H2O2-destroying enzymes catalase and peroxisome. Thus, it has been suggested that iron-dependent ROS-producing enzymes and labile iron may contribute to ROS-dependent cell damage and death. 63 According to Shvedova et al research, a high concentration (30%) of a residual iron catalyst in CNTs increased oxidative stress, cell death, and produced morphological alterations. 64 Plus, it has been reported that ROS can produce endogeneous and exogeneous stimuli, and intracellular ROS is essential for cell cycle entry and required for cell proliferation. 65 –67 However, excessive oxidative stress is harmful to cells, and normal cells have an antioxidant capacity for appropriate redox state retention. 68 Thus, it is expected that the intracellular ROS level increased with the lapse of time, and this was a natural physiological phenomenon in the control cells. Yet, despite continuous oxidative stress, the MWCNT-treated cells were able to maintain a constant intracellular ROS level (although higher than the control cells), which was due to the existence and action of an ROS scavenger system. Oxidative DNA damage has been hypothesized as an important etiologic factor in aging and in the development of systemic diseases, such as cancer. 69,70 Therefore, to prevent these adverse effects, DNA repair enzymes continuously monitor chromosomes and correct damaged nucleotides. 71 The repair of oxidative DNA damage is mediated by both base excision repair and nucleotide excision repair mechanisms, which are vital cellular integrity mechanisms in oxidative damage. 72 –74 However, specific and bulky DNA lesions trigger apoptosis that have been identified. 74
In this study, the ROS generation and DNA damage were lower in the 48-hour treated cells than in the 24-hour treated cells (Figures 3, 4, and 6). Notwithstanding, severe apoptotic cell death was observed among the lymphocytes treated with the MWCNTs for 48 hours (Figure 5). It is believed that these results were related with the redox system, DNA repair system, and apoptosis mechanism activation. Finally, the CBPI was measured to determine any mechanical interference by the MWCNTs. As a result, the CBPI confirmed that the MWCNTs did not evoke any mechanical interference (Figure 4B).
In addition, the oxidative stress induced by particle exposure may mediate the release of proinflammatory cytokines. 50,75–76 The MWCNTs induced ROS generation, cell death, and cytokine production in A549 cells and other cell systems. 77,78 Our instillation and acute and subactue inhalation studies of MWCNTs also indicated ROS induction with DNA damages in the exposed lung cells. Tumor necrosis factor α and IL-6 are 2 major cytokines expressed during an acute inflammatory reaction and required for effective regulation of the early phase of inflammation. 78 In the current study, the TNF-α (not statistically significant) and IL-6 (P < .05) release increased with the MWCNT treatment concentration of 50 μg/mL after 48 hours treatment (Figure 7). Thus, it is speculated that a higher concentration of MWCNTs may induce an inflammatory response (Figure 7).
In conclusion, the current study demonstrated that MWCNTs induced ROS generation, cell growth inhibition, DNA damage, and apoptotic cell death in PHA-stimulated healthy male HPBLs. Thus, prudence is suggested for anyone taking MWCNTs for medical purposes, and more work is needed to validate safety of these materials.
Footnotes
Authors’ Note
J. S. Kim substantially contributed to conception or design; acquisition, analysis, or interpretation of data; and drafted the manuscript. K. S. Song contributed to conception and design, acquisition, drafted manuscript, and critically revised manuscript. I. J. Yu contributed to conception and design; acquisition, analysis, and interpretation; and drafted manuscript. All authors critically revised manuscript, gave final approval, agree to be accountable for all aspects of work ensuring integrity and accuracy.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Industrial Strategic technology development program (10047829, Development of technologies on testing evaluation method and standardization for nanoproduct and nanomaterial) funded By the Ministry of Trade, industry & Energy (MI, Korea).
