Abstract
Ketamine abusing is associated with ulcerative cystitis, but the mechanisms remain unclear. This study aimed to investigate the existence of ketamine-induced symptom in a rat model and evaluate the underlining mechanisms. Sprague-Dawley rats were chosen and randomly divided into 12 groups (n = 8), such as the control group, low dose of ketamine (10 mg/kg/day), middle dose of ketamine (30 mg/kg/day) and high dose of ketamine (50 mg/kg/day) groups. The experimental groups were administrated ketamine i.p. daily, whereas the control groups were administrated with saline. After 1, 3, and 6 months of treatment, the bladder tissues were collected. Haematoxylin and eosin (HE) staining and a transferase-mediated dUTP-biotin nick end labeling (TUNEL) assay were used to evaluate the bladder epithelium pathology and urothelial apoptosis, respectively. The protein expression levels of LC3, p62, Beclin1 were assessed by Western blotting. HE staining results of the experimental rats showed the bladder tissue denudation of the urothelial epithelium with edema and congestion compared with the control groups. TUNEL staining showed a significantly higher number of apoptotic cells in experimental groups than in the control groups. The protein LC3 and Beclin1 had significantly higher levels compared with control groups. The protein p62 had lower levels compared with control groups. The expression levels correlated with contraction of ketamine and treatment time. HE staining, TUNEL staining and Western blot results showed dose-dependent, time-dependent autophage in ketamine-treated rats. All the results suggested that autophagy proteins might be involved in inflammatory response in rats.
Introduction
Ketamine has been used as a general anesthetic for several years, which is a non-competitive N-methyl-D-aspartic acid (NMDA) receptor antagonist.1,2 Recently, the abuse of ketamine has increased dramatically, especially in nightclubs and dance parties.3,4 Many clinical studies have shown that long-term ketamine abuse can markedly affect the urinary system, causing lower urinary tract syndromes, such as urgency, frequency, suprapubic pain, hematuria, and severe dysuria.5 –7 Tsai et al. indicated that ketamine could induce papillary necrosis as well as urothelial inflammation, hemorrhages, and erythematous mucosa. 8 Chu et al. 5 suggested that ketamine and its metabolites had a direct toxic effect on the bladder epithelial cells and lead to chronic submucosal and detrusor muscle inflammatory response.
The rat models and mouse models of ketamine-induced urinary tract syndrome in vivo study have been published.4,9 –11 In Yeung et al.’s 4 mouse study, ketamine injection (30 mg/kg/day) induced submucosal infiltration of mononuclear inflammatory cells; what’s more, the urothelium became thinner and also the number of nerve fibers was reduced following 30 days of ketamine treatment. In another study, after ketamine injection for four months in the rat model, the bladder histological examination demonstrated hyperplastic urotheliums and inflammatory cell infiltration. 10
Some mechanisms have been proposed to explore the potential relationship between ketamine and urinary tract damage. One mainly refers to the direct toxic effect of ketamine and its metabolites on the bladder interstitial cells, which can cause papillary necrosis with interstitial fibrosis and structural damage that will result in organ impairment. The second mechanism is microvascular changes in the bladder causing endothelial cell injury of microvessels. The third hypothesis involves an autoimmune reaction of the bladder urothelium and submucosa triggered by urinary ketamine or its metabolites. 12 The pathogenesis of ketamine-related cystitis is complicated and involves many different pathways. 13 For most animal models, ketamine-induced cystitis may elicit from the direct toxicity of ketamine and its metabolites.
In this study, histopathological changes of bladder were observed in the rat mode to evaluate the possible mechanism.
Materials and methods
Animals and ketamine administration
Adult Sprague-Dawley rats, weighting 180–220 g, were housed in a 12 h light-dark cycle with free access to food and water. Before experiments, the animals were allowed to habituate to the housing facilities for a week. This study was carried out in strict accordance with the recommendations in the Guide for the Care and Use of Laboratory Animals of the National Institutes of Health (NIH Pub. No. 85-23, revised 1996). The protocol was conducted with approval from the Animal Ethics Committee of The Affiliated Yantai Yuhuangding Hospital of Qingdao University. Rats were randomly divided into 12 groups (n = 8), namely the control group, low-dose of ketamine (10 mg/kg/day), middle-dose of ketamine (30 mg/kg/day) and high dose of ketamine (50 mg/kg/day) groups. The experimental groups were injected ketamine intraperitoneal (i.p.) daily for 1, 3, or 6 months, whereas the control groups were administrated with saline.
Histopathological analysis
The rats were killed by spinal dislocation, the bladders were removed, cut open, and further fixed in 10% phosphate-buffered formalin for overnight, dehydrated and embedded in paraffin and cut on a microtome into 5 µm thick slices. 14 Sections were dried, deparaffinized in xylene, and rehydrated in ethanol, stained with hematoxylin and eosin. Briefly, they were placed in hematoxylin for 5 min, then washed in tap water and left for 5 min, followed by rinsing in 1% acid alcohol and washing in tap water. After that, sections were added in eosin solution (1%) for 5 min, followed by washing in tap water, and then dehydrated. All stained sections were assessed under light microscope. 15
Apoptotic cell staining by TUNEL assay
TUNEL staining was performed using the ApopTagkit (Millipore, USA).16,17 Briefly, sections (5 μm) were deparaffinized in xylene, rehydrated in decreasing concentrations of ethanol, pretreated slides with freshly diluted proteinase K at room temperature for 30 min. Endogenous peroxidase was blocked in 3% hydrogen peroxide. Terminal deoxynucleotidyl transferase (TdT) in reaction buffer (containing a fixed concentration of digoxigenin-labeled nucleotides) was applied to serial sections for 1 h at 37°C, and put the slides in a jar containing Stop/Wash buffer for 10 min. Following washes, HRP-conjugated anti-digoxigenin antibody was applied for 30 min at room temperature. Apoptotic cells were detected after incubation in the diaminobenzidine (DAB) chromogen (DAKO, USA) for approximately 6 min. The number of TUNEL-positive cells in 10 randomly selected non-overlapping fields in the bladder was calculated and compared between the various groups. In each experiment, negative controls with label solution (without terminal transferase) instead of the TUNEL reaction mixture were used to elucidate nonspecific immunostaining. Images were visualized using an Olympus BX51 microscope (Olympus, Tokyo, Japan), and images were captured with a JVC 3-CCD camera (Olympus) and analyzed using Image-Pro Plus software (Version 6.0, Media Cybernetics, LP, USA).
Measurement of proteins with western blotting
The bladder tissue in each group was used for western blotting, as previously described.10,18 Rats’ bladder tissues were homogenized in lysis buffer (20 mM Tris, 1% TritonX-100, 0.05% SDS, 5 mg of sodium deoxycholate, 150 mM NaCl, and 1 mM PMSF) containing protease and phosphatase inhibitor cocktail. The protein concentrations were determined using a BCA Protein Assay reagent kit (Thermo Fisher). Equal amounts of protein were separated by SDS/PAGE and transferred to PVDF membranes, the nonspecific binding of antibodies were blocked with 5% non-fat dried milk in PBS and then incubated with the primary antibodies rabbit polyclonal anti-LC3, anti-p62, anti-Beclin1, 1:600 (sigma), and mouse polyclonal anti-β-actin, 1:1000 (Santa Cruz Biotechnology) were used followed by the application of the secondary antibodies consisting of HRP-conjugated goat anti-mouse IgG, 1:4000 (Zhongshan Inc) and goat anti-rabbit IgG, 1:1000 (Zhongshan Inc). The protein band images were collected and the relative optical density (ROD) was analyzed with molecular image, ChemiDocXRS + Image System (Bio-Rad Laboratories, Hercules, CA, USA).
Statistical analysis
Data are expressed as mean ± SD. Statistical differences were evaluated by software SPSS 19.0 and Image-Pro Plus 6.0. Multiple comparisons were tested by one-way analysis of variance (ANOVA) followed by the LSD post-test (t test). A p value < 0.05 was considered significant.
Results
Histopathologic analysis
Histological features of ketamine-treated bladder damage are shown in Figure 1. In the control group, there was no marked pathological change in the urothelium (Figures 1(a)–(c)). Moreover, infiltration of mononuclear cells was found in the submucosal and muscular layer of bladder. Ketamine 50 mg/kg/day treated 30, 90 and 180 days groups (Figures 1(j)–(l)), 3 (Figure 1(h)) and 6 month (Figure 1(f)) group showed marked obviously urothelial denudation with congestion, edema, adherent hemorrhage and fibrin. Ketamine administration could impair bladder tissue in rats.

Histopathological assay of rats. Bladder slides were histological stained with hematoxylin and eosin (magnification: ×40). (a)–(c) Control groups. (d)–(l) Experimental groups. (d)–(f) Ketamine 10 mg/kg/day treated 30, 90, and 180 days, respectively. (g)–(i) Ketamine 30 mg/kg/day treated 30, 90, and 180 days, respectively. (j)–(l) Ketamine 50 mg/kg/day treated 30, 90, and 180 days, respectively.
Apoptotic cell staining by TUNEL assay
As demonstrated in Figure 2, the TUNEL assay generated specific nuclear staining of cells with a morphology consistent with apoptosis. The number of apoptotic cells in the urothelial and suburothelial layers was significantly higher in the high dose groups (50 mg/kg/day injection for 1, 3 and 6 month) and 6-month treatment groups (10 mg/kg/day and 30 mg/kg/day injection for 6 month) than in the control groups (Figure 2(f), (i) and (j)–(l)). In the control groups, the percentage of TUNEL positive cells was less than 1%, but the apoptotic cells were significantly increased during the ketamine-treated groups (p < 0.05). The apoptotic cells also were significantly different at the same treatment period among the different dose ketamine groups (p < 0.05). The results showed that apoptosis was related to the dose and treatment time of ketamine in rats.
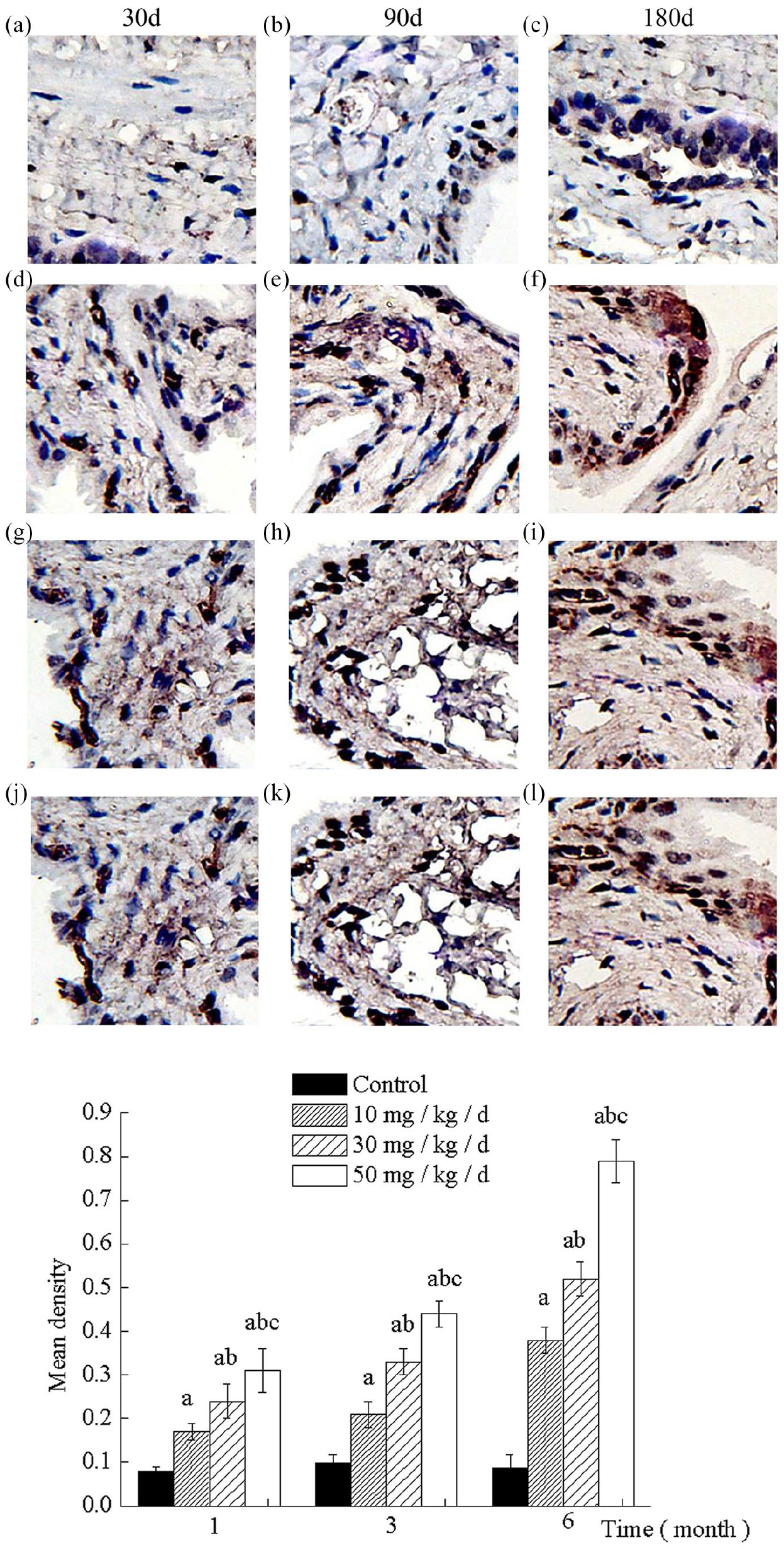
Ketamine-induced bladder cell apoptosis as assessed by the TUNEL assay. (a)–(c) Control groups. (d)–(l) Experimental groups. (d)–(f) Ketamine 10 mg/kg/day treated 30, 90, and 180 days, respectively. (g)–(i) Ketamine 30 mg/kg/day treated 30, 90, and 180 days, respectively. (j)–(l) Ketamine 50 mg/kg/day treated 30, 90, and 180 days, respectively.
The expression of proteins after ketamine administration
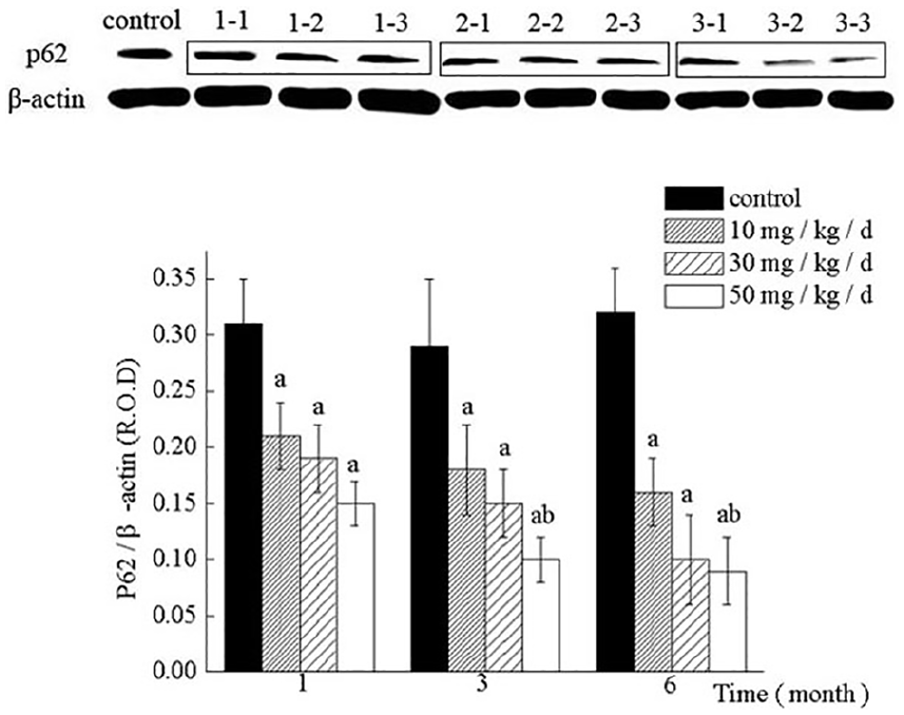
LC3, p62, and Beclin1 as markers for the autophagy, the protein expression levels reflect the upregulation of autophagy. At early time points, there were minimal amounts of LC3 and Beclin1 expression levels (Figures 3 and 4 control group), suggesting that the background autophagy at the basal level is low. The protein expression levels of LC3 and Beclin1 gradually increased after ketamine administration (Figures 3 and 4). Meanwhile, the level of p62 had a decreased trend (Figure 5). Protein LC3 and Beclin1 expression levels were significantly increased in a time-dependent and dose-dependent manner during ketamine treatment (p < 0.05). Ketamine lowered the levels of p62, noticeably. Those results shown that autophagy was highly activated with the treated of ketamine in rats, which suggested that autophagy proteins might be involved in inflammatory response in rats.

Western Blotting of LC3 expression levels in the bladder of the ketamine-treated groups. Group 1-1, 1-2, 1-3 represent ketamine 10 mg/kg treated 1, 3, and 6 months, respectively. Group 2-1, 2-2, 2-3 represent ketamine 30 mg/kg treated 1, 3, and 6 months, respectively. Group 3-1, 3-2, 3-3 represent ketamine 50 mg/kg treated 1, 3, and 6 months, respectively.
Western Blotting of Beclin1 expression levels in the bladder of the ketamine-treated groups. Group 1-1, 1-2, 1-3 represent ketamine 10 mg/kg treated 1, 3, and 6 months, respectively. Group 2-1, 2-2, 2-3 represent ketamine 30 mg/kg treated 1, 3, and 6 months, respectively. Group 3-1, 3-2, 3-3 represent ketamine 50 mg/kg treated 1, 3, and 6 months, respectively.

Western Blotting of p62 expression levels in the bladder of the ketamine-treated groups. Group 1-1, 1-2, 1-3 represent ketamine 10 mg/kg treated 1, 3, and 6 months, respectively. Group 2-1, 2-2, 2-3 represent ketamine 30 mg/kg treated 1, 3, and 6 months, respectively. Group 3-1, 3-2, 3-3 represent ketamine 50 mg/kg treated 1, 3, and 6 months, respectively.
Discussion
Ketamine has been abused as a recreational drug since the 1980s. The common additive effects of ketamine usually last an hour or less.7,19 However, the adverse effects can be long-lasting. 20 Usually known adverse effects include cardiorespiratory effects, nausea, vomiting, hallucinations, and convulsions.21,22 Lately, since 2007, there have been reports of urinary system complications arising from the chronic abuse of ketamine. This new clinical entity, termed “ketamine-associated ulcerative cystitis,” is characterized by symptoms of lower urinary tractirritation related to ketamine use among young people. It is relatively underdiagnosed because many medical professionals are still unaware of this disease. 23 In this experiment, cystitis was induced by ketamine administration, as evidenced by inflammatory changes, for instance, ulcers, edema, hemorrhage, and inflammatory cell infiltration in the bladder. These findings were consistent with previous studies,23 –25 which suggested that the model was successfully developed in our study.
Apoptosis is a stepwise process, which is characterized by a variety of stereotypical morphological changes that eventually lead to cell death. 26 In the study, some results showed the apoptotic process was highly activated in the urothelial cells of the ketamine-treated rats. The degree if apoptosis was related to the dose and treatment time of ketamine. This study demonstrated urothelial cell apoptosis in patients which resulted from upregulation of inflammatory signals, such as p38 mitogen-activated protein kinase and TNF-α. 27 Whether this signaling pathway plays a key role in modulating urothelial in KC remains uncertain.
It is believed that autophagy plays an important role in cytoprotection and in controlling the inflammatory response by way of clearing damaged organelles and misfolded proteins. However, the role of autophage in bladder cystitis has not been clarified. 28 During cellular stress, a series of autophage-related genes are activated including microtubule-associated protein 1 LC3.12,29 LC3, synthesized as a full-length precursor pro-LC3, is immediately processed by another autophagy molecule Atg4 into the cleaved form, LC3I, and LC3 I could be lipidated into LC3II. LC3II immunoblots are often used for quantification of autophagy. The protein of p62 is associated with autophagosomes and degraded in the autolysosome.28,30 The p62 is the scaffold protein implicated in selective autophagy which is induced by the stress of cells. Beclin1 is not only a central positive regulator in the early stage of autophagy but also one of the core proteins of autophagy, apoptosis, and inflammatory reaction. 31 In this article, we demonstrated that LC3, p62 and Beclin1 in the bladder were significantly changed at each time point in ketamine-treated groups, which suggested that autophagy proteins might be involved in inflammatory response in rats.
Although ketamine-induced urinary tract syndrome in rats cannot fully replicate the pathophysiological changes in humans, our study provides a new perspective and views that autophagy could be involved in the pathogenesis of ketamine abusers. However, this model has a number of limitations which need to be addressed in the future study. This model had no provision for the measurement of changes in bladder pressure or compliance. Also, further study on the specific molecular mechanisms is required.
In conclusion, we found that in rat model of ketamine-induced urinary tract syndrome, urothelial homeostasis was severely impaired. Apoptosis and autophagy were significantly increased and decreased bladder function. This indicates that chronic inflammation in the bladder wall contributes to the unwanted urological symptoms.
Footnotes
Animal welfare
The present study followed international, national, and/or institutional guidelines for humane animal treatment and complied with relevant legislation.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethics approval
Ethical approval for this study was obtained from the Animal Ethics Committee of The Affiliated Yantai Yuhuangding Hospital of Qingdao University (No. WFPHSPF/SQ-21).
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This project is supported by National Natural Science Foundation of China (No. 81970659, 81700664, 81870525, and 81572835), Taishan Scholars Program of Shandong Province, Key Research and Development Program of Shandong Province (No. 2018GSF118118), Shandong Provincial Natural Science Foundation (No. ZR2016HP38).
