Abstract
Polyhexamethylene guanidine hydrochloride (PHMGH) is used worldwide as an antimicrobial agent with broad spectra of activity and also for treating pool water. This non-GLP preliminary study aims at investigating in a subchronic toxicity study possible effects at supra-optimal doses of this biocide. Both acute and subchronic toxicity studies were conducted. LD50 for PHMGH was estimated to be 600 mg/kg (ie LC50 2 ml of 7.5% solution) when administered as a single dose by gavage via a stomach tube in accordance with the expected route of administration. The acute studies showed that the median lethal dose (LD50) of 600 mg/kg was accompanied by signs of neurotoxicity. Haematological and biochemical parameters of subchronic toxicity studies were non-significant. Subchronic doses of 0.006 mg/kg, 0.012 mg/kg and 0.036 mg/kg were administered. 20% of the animals at a dose of 0.006 mg/kg and 0.036 mg/kg showed mild degrees of hydropic changes in proximal tubules while 10% of animals at all the doses had their liver tissues showing local areas of mild pericentral hepatocytes degeneration. PHMGH did not produce any major organ defect with regard to the kidney, heart, and liver. The LD50 was much higher than the recommended dosage by a factor of about 50,000. The recommended residual concentration is far less than the median lethal dose using rats as test subjects. These results could serve as a basis for investigating the full toxicological profile if it is to be used for the treatment of raw water to make it potable.
Introduction
Polyhexamethylene guanidine hydrochloride (PHMGH) is a chemical-based disinfectant and a member of the polymeric guanidine family which has proved to be a potent virucide and in vitro bactericide. 1,2 It is odorless, noncorrosive, and has been shown to be nontoxic in an in vitro cytotoxicity study involving low concentrations (0.04% and 0.005%, w/v). 2,3 It is highly soluble in water. 4,5 Polyhexamethylene guanidine hydrochloride is also used as an effective sporicidal disinfectant. 3,4 In an in vitro study, 4 it killed all spores at a concentration corresponding to 0.52% (w/v) within 90 seconds of contact and 0.36% (w/v) for 3 minutes. It has thus been recommended for use in hospitals, laboratories, food industries, and households. 4 Polyhexamethylene guanidine hydrochloride has gained interest, wide acceptance, and it is been exploited for potential multipurpose use.
Currently, PHMGH is recommended as a bactericidal and fungicidal disinfectant for the treatment of harvested cocoa beans, cooling systems, and also for the treatment of raw water to make it potable (ie, good for drinking without fear of poisoning or disease) 5 -8 Its mode of action has been predicted to be related to cell wall destruction with subsequent leakage of the cell contents. 4
In Ghana, regulatory concerns are being raised with regard to its safety on human health after prolonged or cumulative doses, especially its use in the treatment of raw water to make it potable. Studies have proved that a structurally related compound, polyhexamethylene biguanide with similar use as PHMGH as a disinfectant, has a median lethal dose (LD50) estimated between 500 and 1000 mg/kg. 8
Administration of PHMGH in both rats and humans has been associated with the potential for hepatic, renal gastrointestinal tract (GIT), and lung effects. 9,10 Although officially not used in the treatment of water, there have been instances where toxic effects have been experienced in certain individuals. For example, in the period from August 2006 to May 2007, more than 12 500 patients were admitted to hospital with a history of drinking illegal cheap “vodka” in 44 different regions in Russia, of whom 9.4% died. In reality, the “vodka” was an antiseptic liquid composed of ethanol (≈93%), diethyl phthalate, and 0.1% to 0.14% PHMGH (“Extrasept-1”). 9
Studies concerning the safety assessment of PHMGH is very rare. One study has reported LD50 value for PHMGH to be 450 mg/kg in mice and 630 mg/kg in rats. 10 This non-good laboratory practice investigation was intended to provide a preliminary evaluation of the toxicological potential of PHMGH if used in treating raw water to make it potable in both acute and subchronic oral administration to male and female Sprague-Dawley (SD) rats.
Materials and Methods
Test Substance
Polyhexamethylene guanidine hydrochloride 30% with specific gravity of 1.0 and pH between 6.1 and 6.4 was purchased from Agrimat (Ghana) with 2 years term of storage. Ten working solutions were prepared from the 30% PHMGH by diluting with deionized water to give final concentrations ranging from 0.1% to 8% PHMGH. In the case of 7.5% PHMGH, this was achieved by taking 12.5 mL of the 30% PHMGH solution and adding up to 50 mL. This gives 50 mL of 7.5% PHMGH. These were used for the acute toxicity studies. The working solution of 0.1% concentration of PHMGH, which is equivalent to 1.0 mg/mL of PHMGH, was used for the subchronic toxicity studies. Fresh dilutions were always made before administration of the test substance throughout the experiment.
Animal Husbandry and Groupings
Ten-week old SD rats (240-260 g body weight [bwt]) of both sexes were acquired from Noguchi Memorial Institute for Medical Research, housed in cages in rooms with regulated room temperature of 26°C and humidity ranging between 40% and 60%, and were used in the experiment. The animals were assigned to 5 groups of 10 animals each for the acute toxicity test. For the subchronic study, 4 groups of 10 animals each were used. They were marked for easy identification and placed in individual cages for 7 days to acclimatize prior to treatment. The animals were exposed to 12-hour light–dark cycle. Each group consisted of 5 males and 5 females. Females were nulliparous and nonpregnant. Equal numbers of animals of each sex were used at each dose level of PHMGH tested. The protocol was approved by the ethical and protocol committee, University of Ghana, College of Health Sciences. A certified animal feed with very low contaminant from Kosher Feed Mills Ltd, Osu, Accra, and distilled water were given ad libitum, but feed was withdrawn 8 hours prior to treatment to ensure effective absorption from the GIT after oral administration during both the acute and the subchronic studies. Feed was, however, reintroduced 30 minutes after treatment. All animals have been fed with this type of feed from birth.
Acute Toxicity
Ten-week old SD rats of both sexes with weight ranging from 240 to 245 g were used. Polyhexamethylene guanidine hydrochloride was administered in a single dose by gavage via a stomach tube in accordance with the expected route of administration. There was an initial administration of 3000 mg/kg in one rat and 400 mg/kg in another rat. Thereafter, doses of 8.0, 400, 600, 2000, and 3000 mg/kg were administered in order to determine 50% death of the animals at the doses tested. In this particular case, since the maximum volume of liquid to be administered was 1 mL/100 g of bwt, 11 higher concentrations were prepared to avoid exceeding the allowable dose of 2 mL/200 g volume for administration. Control animals received equivalent volume of deionized water. The animals were observed individually twice every 30 minutes, then every 4 hours for the first 24 hours. Thereafter they were observed daily for 14 days. The dose that resulted in 50% death (LD50) was calculated from the first 24 hours observations postdosing. The surviving animals were observed daily for up to 14 days after which they were euthanized. Necropsy was performed on all the animals, and the following organs, heart, liver, and kidneys, were harvested and weighed. The organs were fixed in 10% buffered formalin as soon as necropsy was performed. The samples were dehydrated in graded alcohols, cleared in xylene, and embedded in paraffin wax. Multiple sections of 4 µm were cut using a microtome, and the specimens stained with hematoxylin and eosin.
Blood samples were drawn from the descending aorta for hematological and clinical chemistry. Hematological analysis was done using SysMex K-21 (Sysmex Corporation, Japan), whereas clinical chemistry was done using Microlab 300 equipment (2003; Sysmex Corporation).
Subchronic Toxicity
For the subchronic toxicity studies, doses were administered based on the recommended dose from the manufacturer. The intended dose to be used for the treatment of raw water to make it potable (recommended dose) from the manufacturer was 0.012 mg/kg (3 mg/L).
Thus, 3 dosage levels of PHMGH and a control were administered to groups of SD rats as single oral dose daily for a period of 90 days. Manufacturer’s recommended dose was 3.0 mg/L (0.012 mg/kg). Doses were always administered according to changes in the body weight of the animals throughout the experiment. Group 1: 0.006 mg/kg (ie, 1.5 mg/L). Group 2: 0.012 mg/kg (ie, 3.0 mg/L). Group 3: 0.036 mg/kg (ie, 9.0 mg/L).
Control: deionized water.
In all groups, the animals were initially observed every hour for the first 4 hours and subsequently every 6 hours for the next 3 days and daily thereafter for 90 days. Apart from food and water consumption, the animals were also monitored for clinical signs including respiratory distress, frequency of urination, swellings, abnormal gait, and defecation rate. They were also weighed every 3 days to monitor food and water consumption together with any weight changes.
Laboratory Examinations
At the end of the dosing period, the animals were euthanized by exsanguination under ether anesthesia, and blood samples were collected from the descending aorta for hematological analysis and clinical chemistry such as alanine aminotransferase (ALT) or serum glutamic pyruvic transaminase (SGPT) levels, alkaline phosphatase (ALP), serum glutamic oxaloacetic transaminase (SGOT) or aspartate transaminase, and γ-glutamyl transpeptidase (GGT) was measured as the liver function tests to give an indication of the state of the liver, while creatinine and urea levels together with serum electrolytes were measured as the renal function tests to give an indication of the kidney. Lipid profiles detailed as high-density lipoproteins, low-density lipoproteins, triglycerides (TGs), and cholesterol were also measured.
Hematological analysis, including hematocrit (HCT), hemoglobin (Hb), lymphocyte percentage, neutrophils (NEU), mean corpuscular hemoglobin (MCH), mean corpuscular hemoglobin concentration (MCHC), mean corpuscular volume, red blood cells (RBCs), and white blood cells (WBCs), was done using SysMex K-21 (2003; (automated hematology analyzer; KX-2IN; Sysmex Corporation), whereas clinical chemistry was done using Microlab 300 equipment. Animals were autopsied immediately, and all visible organs and tissues were macroscopically examined and harvested. Harvested organs were stored in formalin. Selected organs (liver, kidneys, and heart) were examined histologically for animals in the lethal dose group (LD50) and all groups in the subchronic toxicity studies. The experiment was conducted in accordance with the internationally acceptable guidelines for evaluating the safety of chemicals. 12,13
Statistical Analysis
Statistical analysis was done using Graphpad Prism 5. Means ± standard error of the means (SEMs) were determined for quantitative variables. Analysis of variance was used to determine statistical significance invariables among the groups at P values ≤0.05. This was used for the subchronic studies, while unpaired t test was used for the analysis of the acute toxicity study results.
Results
Acute Toxicity Study
Death resulted within a few minutes after administering an initial dose of 3000 mg/kg to a male rat and 3 hours after administering 400 mg/kg to another male rat. However, 50% death, LD50, resulted from administering a high dose of 600 mg/kg (ie, at a concentration of 2 mL of 7.5% solution) of PHMGH. One notable observation was that all the deaths in the 600 mg/kg consisted of 4 males and 1 female.
Clinical manifestations
Deaths within the first 5 minutes were due to tonic–clonic convulsions and/or respiratory distress. Abnormal gait, excessive grooming, and repetitive circling with arched-back posture were also observed (see Table 1).
Mortality Data Sheet for Oral Administration of PHMGH to SD Rats During Acute Toxicity Studies.

Abbreviations: PHMGH, polyhexamethylene guanidine hydrochloride; SD, Sprague-Dawley.
Hematological Analysis
Animals treated with the lethal dose of 600 mg/kg had their WBC reduced by 18.14% (4.733 × 103/µL) while NEUs increased by 18.2% (22.9167%) compared to the controls. Hematocrit, Hb, and RBCs all reduced by 7.34% (32.72%), 7.6% (10.05 g/dL), and 16.7% (5.73 × 106/µL), respectively. Lymphocyte percentage was reduced by 22.3% (74.7167%), while other parameters remained relatively unchanged (Figure 1A). It must be noted that these were not statistically significant.
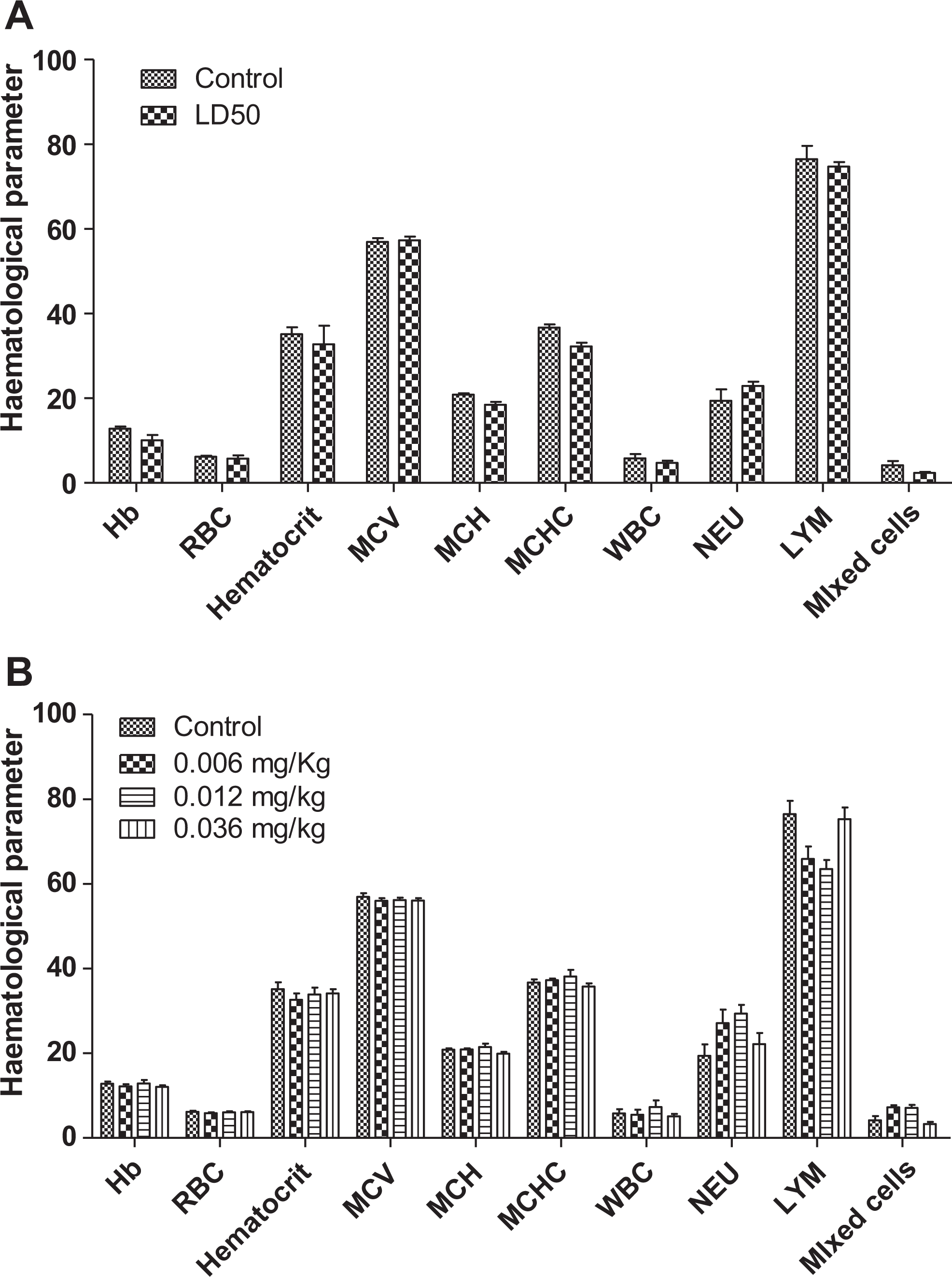
A, Changes in hematological parameters during acute toxicity studies of PHMGH (median lethal dose [LD50]) in Sprague-Dawley rats. Values are expressed as mean ± SEM, n = 5. The differences among the mean were analyzed using unpaired t test and values considered significant when P < 0.05. No significant changes in the parameters observed. B, Changes in hematological parameters during a 90-day administration of PHMGH in Sprague-Dawley rats. Values are expressed as mean ± SEM, n = 10. The differences among the mean were analyzed using 2-way analysis of variance (ANOVA) and values considered significant when P < 0.05. No significant changes in the various parameters observed. There was a slight increase in the neutrophil count. PHMGH indicates polyhexamethylene guanidine hydrochloride; SEM, standard error of the mean.
Table 2 shows the biochemical parameters observed during the acute toxicity studies of rats treated with the lethal dose of 600 mg/kg. These were blood samples taken at the end of the 14th day of observation. The LD50 animals registered an increase in ALP by 176.7% (P < 0.0037), while ALT (SGPT), AST (SGOT), and GGT increased by 5.1%, 7.82%, and 10%, respectively. There were rather increases in the levels of urea (20.5% ± 0.77%) as was observed in the treated animals, while creatinine levels increased by 137.2% ± 25.99% (P < 0.02). There were no significant changes in the lipid profiles.
Blood Biochemistry at the Lethal Dose Level (LD50) During Acute Toxicity Studies Using PHMGH in Sprague-Dawley Rats.a
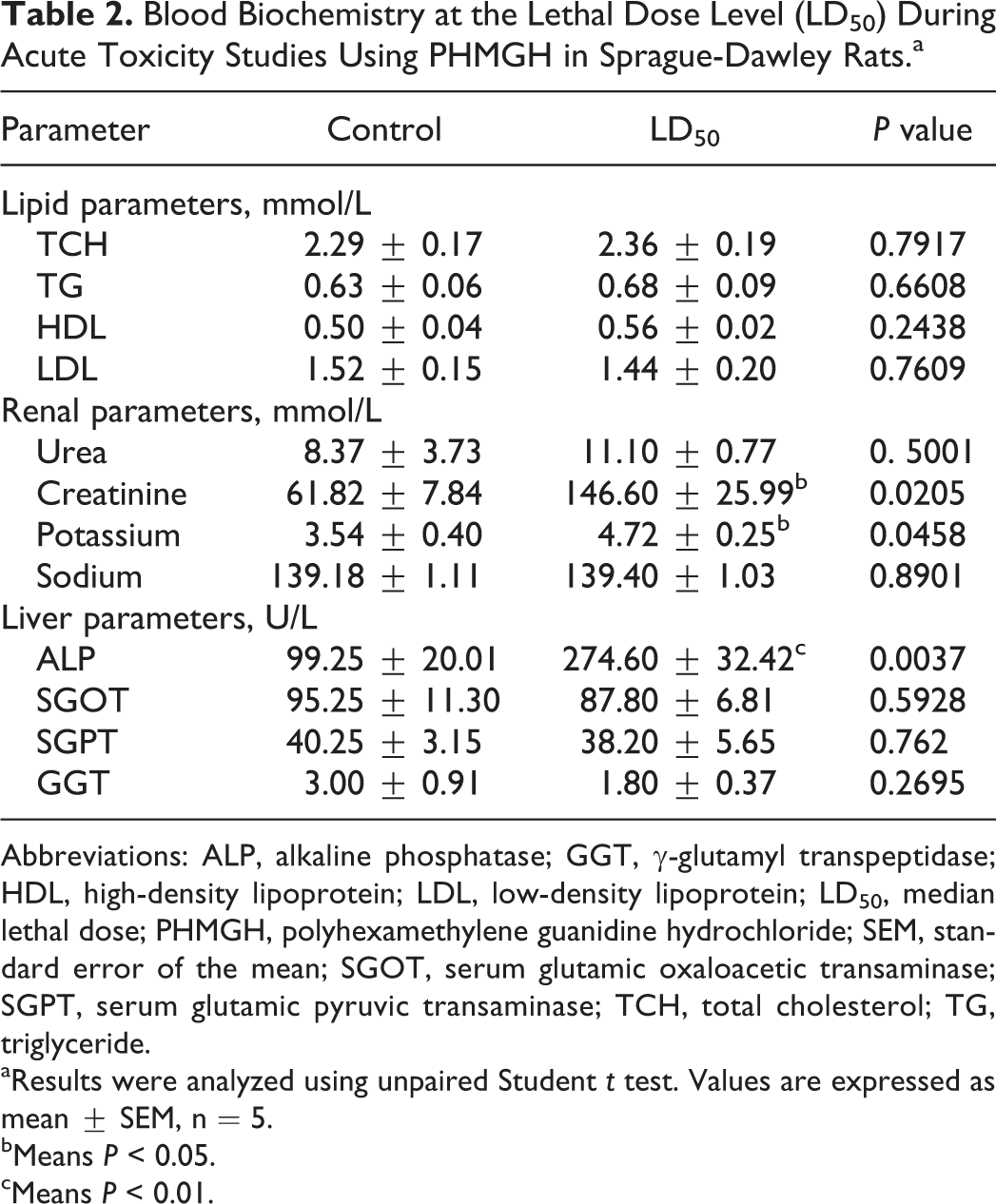
Abbreviations: ALP, alkaline phosphatase; GGT, γ-glutamyl transpeptidase; HDL, high-density lipoprotein; LDL, low-density lipoprotein; LD50, median lethal dose; PHMGH, polyhexamethylene guanidine hydrochloride; SEM, standard error of the mean; SGOT, serum glutamic oxaloacetic transaminase; SGPT, serum glutamic pyruvic transaminase; TCH, total cholesterol; TG, triglyceride.
aResults were analyzed using unpaired Student t test. Values are expressed as mean ± SEM, n = 5.
bMeans P < 0.05.
cMeans P < 0.01.
Subchronic Toxicity Study
Clinical manifestations
No death was recorded in any treatment group during the observation period. No normal nor debilitating clinical signs were evident. All the animals (both males and females) appeared healthy, while the rate of food and water consumption remained relatively unchanged compared to the controls.
Body Weight Changes
No significant body weight differences occurred between treated groups when compared with the controls. Growth rate and body weight changes were insignificant between all the treatment groups.
Hematology
Changes in hematological parameters were not significant. Lymphocyte percentage reduced by 13.8% (65.91%) in the 0.006 mg/kg-treated animals, 16.93% (63.50%) in the 0.012 mg/kg-treated animals, and 1.5% (75.30%) in the 0.036 mg/kg-treated animals compared to the controls (76.45%). White blood cells reduced in 0.006- and 0.012 mg/kg-treated group by 5.7% (5.4556 × 103/µL) and 12.1% (5.0833 × 103/µL), respectively, compared to the control (5.7818 × 103/µL). However, at a dose of 0.036 mg/kg, there was an increase by 26% (7.2833 × 103/µL) compared to the controls. Neutrophils increased by 39.7% (27.08%) in the 0.006 mg/kg-treated group and 14.1% (22.127%) in the 0.036 mg/kg-treated group. The 0.012 mg/kg-treated group rather increased by 51.5% (29.37%) compared to the controls (19.39%; Figure 1B).
Blood Chemistry
Results are presented subsequently.
Liver Function Tests
Alkaline phosphatase measured in the study is shown in Figure 2. The treated groups showed increase in ALP levels by 17.4% and 47.9% in the 0.006- and 0.012 mg/kg-treated groups, respectively. Mean value of 92.833 U/L was however observed in the 0.036 mg/kg when compared to the controls (99.25 U/L).
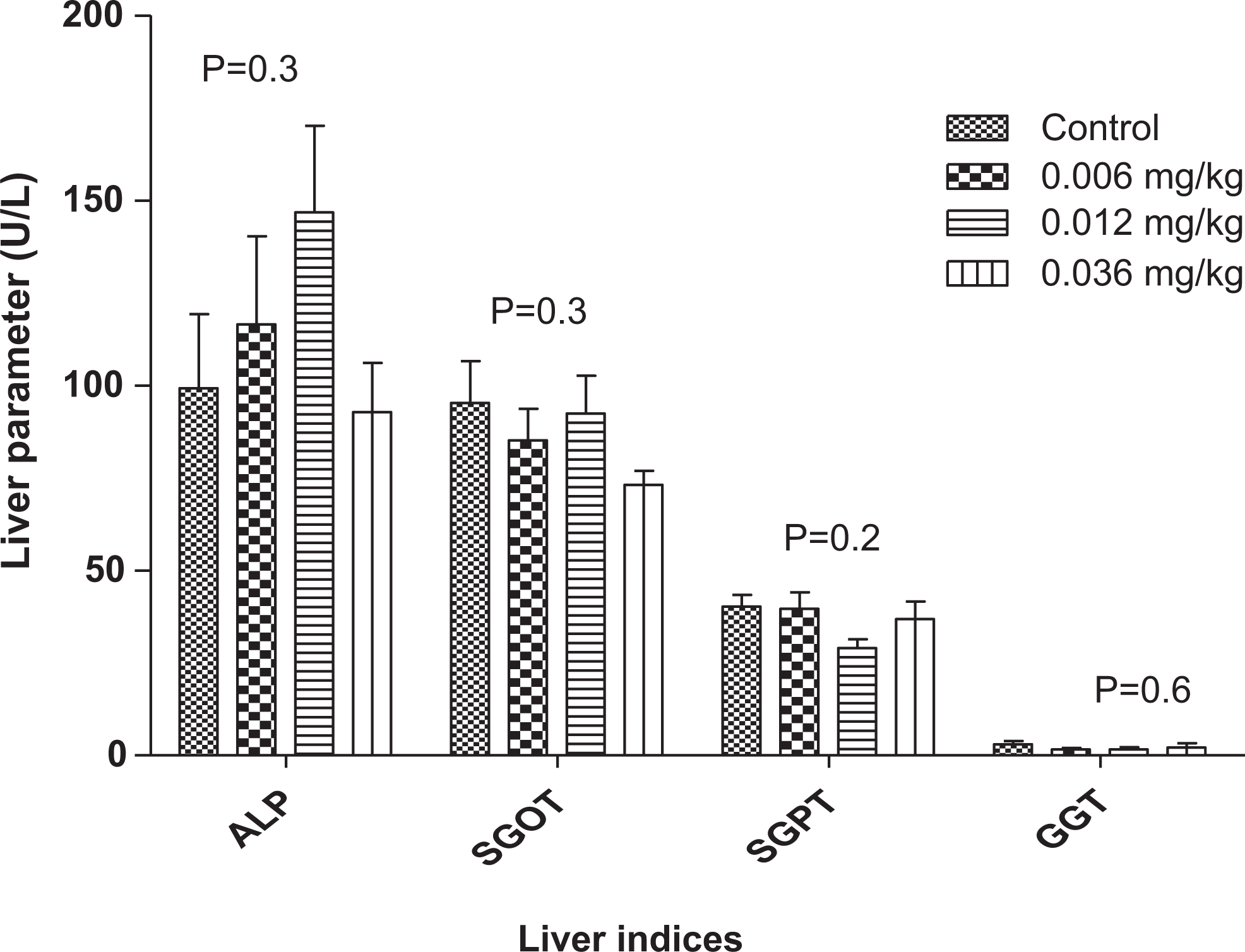
Changes in liver enzymes during a 90-day administration of PHMGH in Sprague-Dawley rats. Values are expressed as mean ± standard error of the mean (SEM), n = 10. The differences among the mean were analyzed using 2-way analysis of variance (ANOVA) and values considered significant when P < 0.05. Comparison of SGOT and SGPT (alanine aminotransferase [ALT]) in various concentrations of PHMGH and controls revealed that the differences among the mean values were nonsignificant as the P = 0.3 and 0.2 for SGOT and SGPT, respectively. Comparison of γ-glutamyl transpeptidase in various concentrations of PHMG and controls revealed that the differences among the mean values were also nonsignificant as P = 0.6. PHMGH indicates polyhexamethylene guanidine hydrochloride; SGOT, serum glutamic oxaloacetic transaminase; SGPT, serum glutamic pyruvic transaminase.
No significant changes were seen in SGPT (or ALT) levels between any treated group and the control group. The same observation was made with SGOT (or AST) and also of GGT.
Lipid Profile
The lipid profile revealed no significant changes in any of the indices measured (Figure 3). There was a percentage increase in TG levels in the animals receiving 0.006 mg/kg PHMGH (0.8462-0.6300), 0.012 mg/kg-treated group (0.683-0.6300), and 0.36 mg/kg-treated group (0.6455-0.6300). High-density lipoprotein was increased by 0.5667% to 0.5000% in the 0.012 mg/kg-treated group and 0.5364% to 0.5000% in the 0.036 mg/kg-treated group.
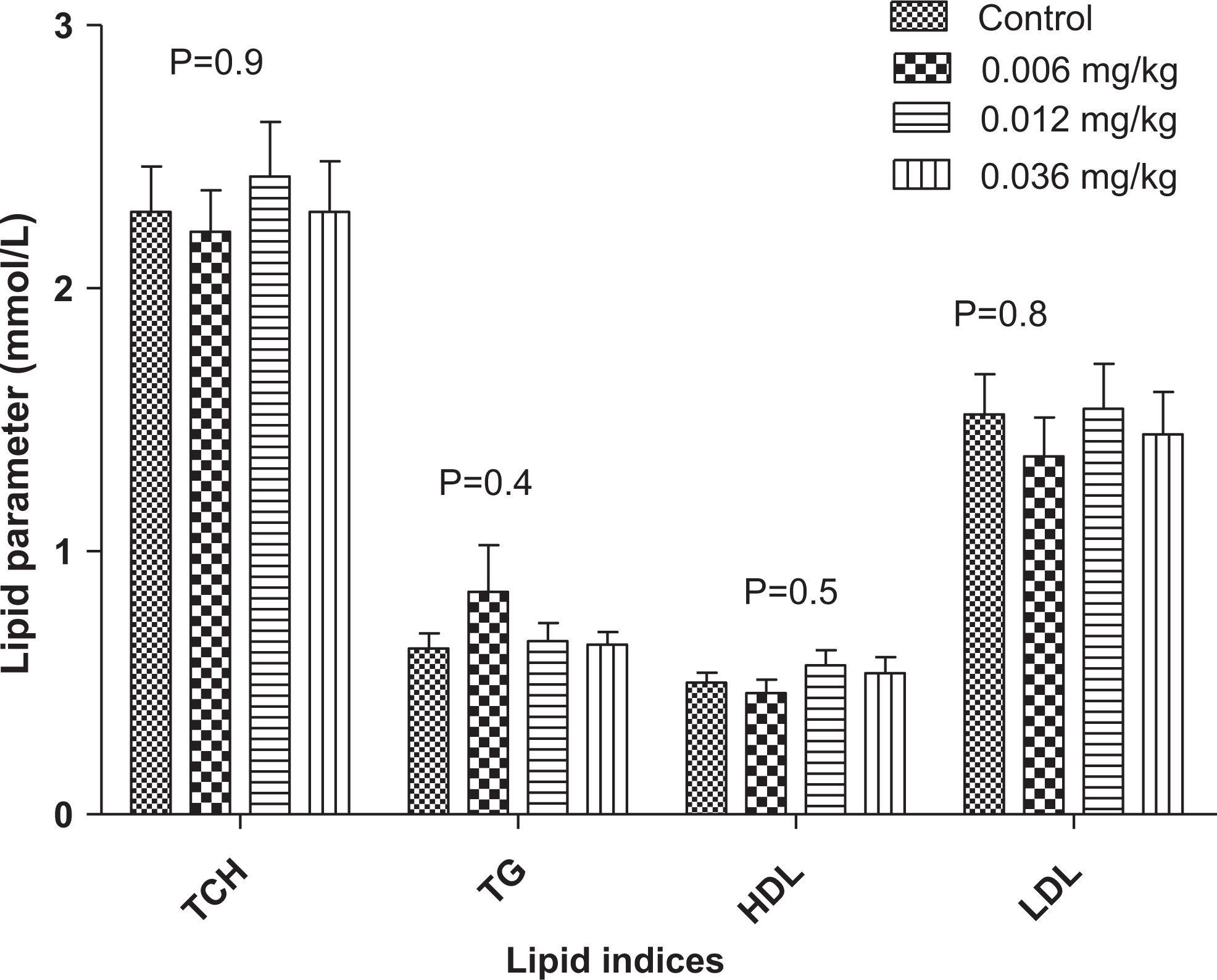
Changes in lipid profile during a 90-day administration of PHMGH in Sprague-Dawley rats. Values are expressed as mean ± standard error of the mean (SEM), n = 10. The differences among the mean were analyzed using 2-way analysis of variance (ANOVA). The differences among the mean values were considered significant when P < 0.05. The changes in lipid profile in various concentrations of PHMGH and controls also revealed that the differences among the mean values were nonsignificant as the P = 0.9, 0.4, 0.5, and 0.8 for total cholesterol, triglyceride, high-density lipoprotein, and low-density lipoprotein, respectively. PHMGH indicates polyhexamethylene guanidine hydrochloride.
Renal Function Tests
The subchronic studies revealed low levels of urea in all the doses levels tested with respect to the control group as follows: 0.006 mg/kg reduced by 32.4% (5.6615 mmol/L), 0.012 mg/kg by 38.6% (5.1417 mmol/L), and 0.036 mg/kg by 24.2% (6.3455 mmol/L; Figure 4). There were increases in creatinine levels. Creatinine levels in the 0.006 mg/kg-treated group increased by 35.3% (83.6462 mmol/L). Those in the 0.012 mg/kg-treated group had their creatinine levels increased by 2.2% (63.1500 mmol/L) and 24.3% (76.8182 mmol/L) in the 0.036 mg/kg-treated group. There was statistical difference in both potassium and sodium concentrations in all the test groups when compared with the controls.
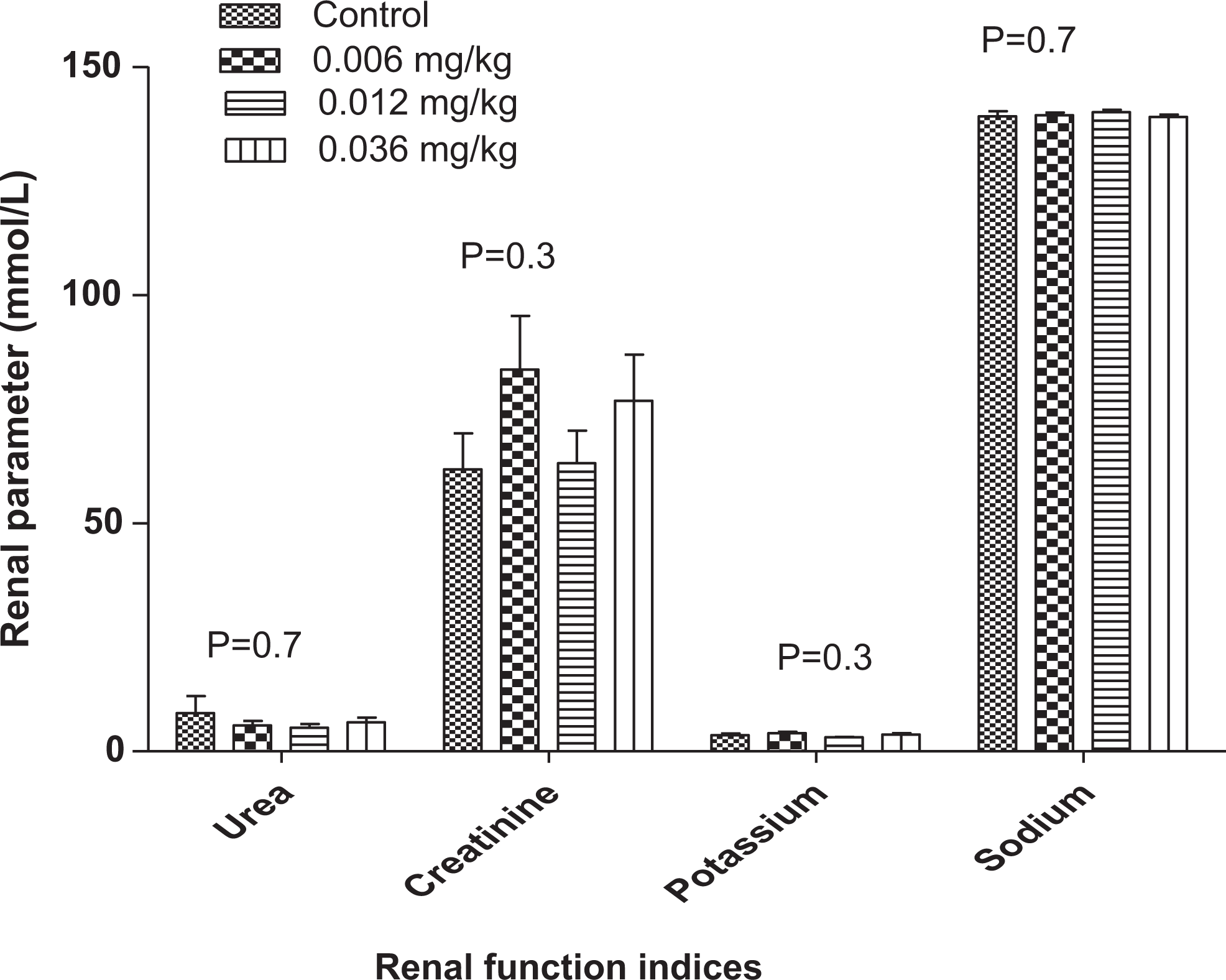
Changes in renal profile during a 90-day PHMGH administration in Sprague-Dawley rats. Values are expressed as mean ± standard error of the mean (SEM), n = 10. The differences among the mean were analyzed using 2-way analysis of variance (ANOVA) and considered significant when P < 0.05. Comparison of urea, creatinine, potassium, and sodium in various concentrations of PHMGH and controls showed that the differences among the mean values were not significant as P = 0.7, 0.3, 0.3, and 0.7, respectively. PHMGH indicates polyhexamethylene guanidine hydrochloride.
Gross Pathology
Acute toxicity study
The necropsy findings of animals dosed with LD50 (600 mg/kg) of PHMGH from the acute toxicity study showed evidence of gastroenteritis, GIT bleeding, and congestion of the lungs, liver, and kidneys. These were observed in both rats that were killed immediately after death on test and those that were euthanized at study day 14.
Subchronic toxicity study
There were no visible pathological changes in the heart, kidney, and liver samples when different dose levels of PHMGH were administered to the animals. All the organs harvested from subchronic studies were comparable with the controls in appearance, size, and consistency.
Histopathology
This was conducted on the liver, heart, and kidney of the killed animals by board-certified pathologists of the Department of Pathology, Korle-Bu Teaching Hospital. The various degree of histopathological abnormalities were grouped into mild, moderate, and severe. Representatives of each degree of tissue injury observed for both acute and subchronic toxicities are presented in the figures. It is interesting to note that all the histopathological abnormalities observed in the various organs were associated with the female rats, while death in the acute toxicity studies were associated with the male rats.
Acute toxicity study (LD50)
All the heart and kidney samples of animals dosed with 600 mg/kg examined were within normal pathological limits compared to the controls (Figure 5A and C), whereas 20% of these animals (2 males) had their liver samples showing mild hepatocytes injury in the form of mild periportal lymphocyte infiltration (Figure 5F). These corresponded to animals that died within the first 3 hours of the study. The rest showed no significant hepatic injury when compared to the controls (Figure 5E).
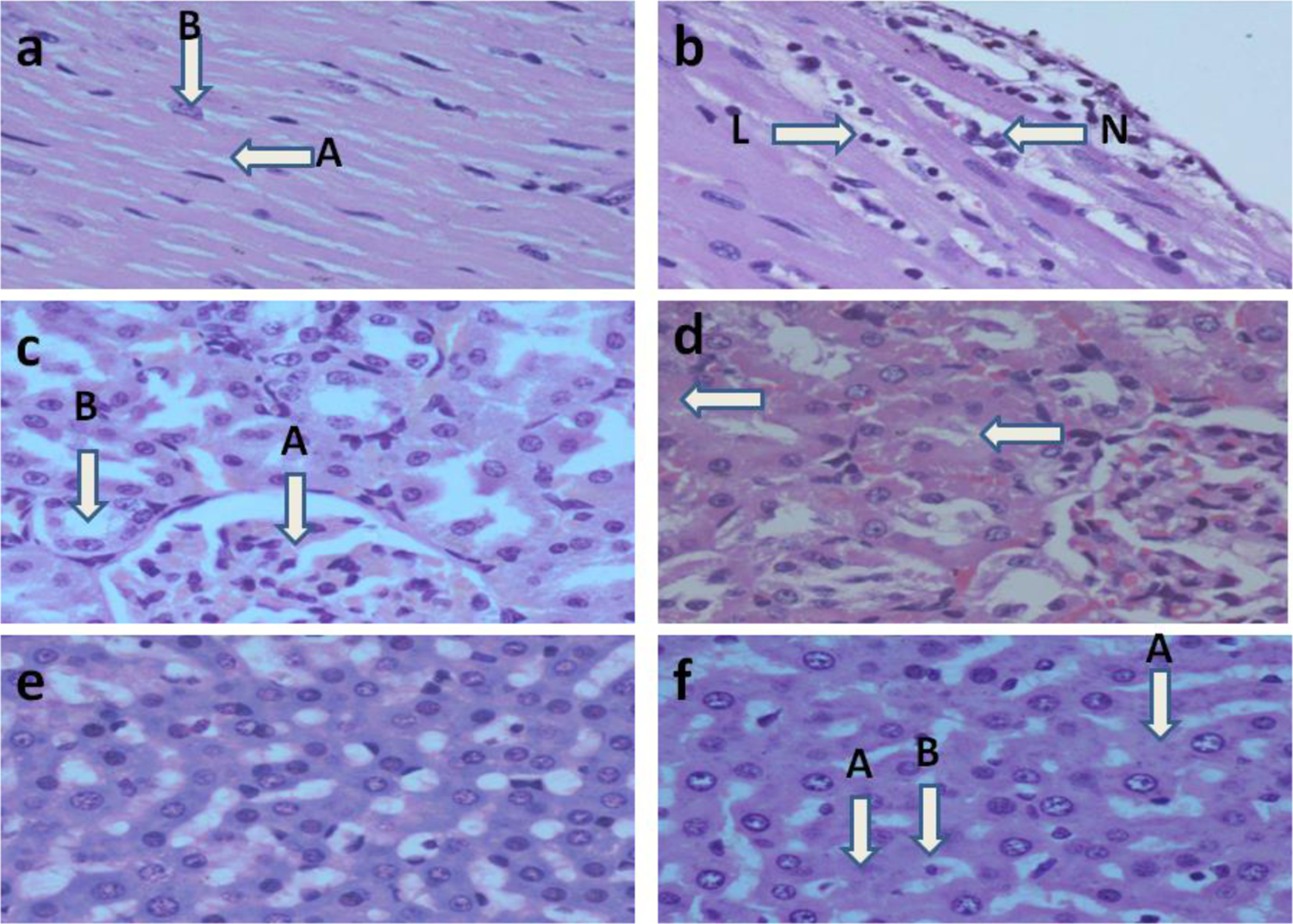
a, Representative section of the heart muscle of SD rat from the control group that received deionized water only for 90 days showing normal myocardial fibers. Note the characteristic normal branching (A) and central placed nuclei (B) of myocardial fibers. H&E stain; ×40. b, Representative section of the heart of an SD rat that received 0.006 mg/kg of PHMGH orally for 90 days showing mild myocarditis. Note the presence of few leukocyte infiltrates: lymphocyte (L) and neutrophil (N) and the apparent absence of myocardial degeneration. H&E stain; ×40. c, Representative section of the kidney of SD rat from the control group that received deionized water daily for 90 days showing normal morphology of a glomerulus (A) and tubules (B). H&E stain; ×40. d, Representative section of the kidney of SD rat that received an oral administration of 0.036 mg/kg of PHMGH daily for 90 days showing mild tubular damage. Note the presence of anucleated tubular epithelial cells (arrows) indicating mild tubular degeneration. Similar findings were observed at 0.012 mg/kg/d. H&E stain; ×40. e, Representative section of hematoxylin and eosin-stained liver of an SD rat from the control group that received only deionized water orally for 90 days showing normal hepatocytes. ×40. f, Representative section of the liver of SD rat that received median lethal dose (LD50; 600 mg/kg) single dose of PHMGH showing mild hepatocellular necrosis. Note the presence of anucleated cells (A) and cells with pyknotic nuclear (B) indicative of a mild hepatic injury. H&E, ×40. Similar findings were observed at 0.006, 0.012, and 0.036 mg/kg/d of PHMGH administered orally for 90 days. H&E indicates hematoxylin and eosin; PHMGH, polyhexamethylene guanidine hydrochloride; SD, Sprague-Dawley.
Subchronic test study
The kidney samples from the group administered 0.006 mg/kg showed no pathological lesions (Figure 5C), but 10% of the animals (1 female) had their heart samples showing mild myocarditis (Figure 5B), whereas only 10% (1 female) of the animals had their liver samples portraying mild centrilobular hepatocyte edema (Figure 5F).
The group administered 0.012 mg/kg had all the heart samples depicting no pathological lesions, but hydropic degeneration was observed in the kidneys of 20% of the animals (2 females; Figure 5D), and 10% of the animals (1 female) had their liver samples showing moderate hepatocyte necrosis. Mild pericentral hepatocytes degeneration was seen in another 10% (1 female; Figure 5F).
At a dose level of 0.036 mg/kg, there were no microscopic findings in the heart samples (Figure 5A), whereas 20% of the animals (2 females) had their kidney samples depicting various degrees of hydropic changes in proximal tubules (Figure 5D). Ten percent of the animals (1 female) had the liver showing local areas of mild pericentral hepatocyte degeneration (Figure 5F). It is interesting to note that all the animals with histopathological abnormality were females.
Discussion
Both acute toxicity and subchronic toxicity studies were conducted on PHMGH. In the acute toxicity study, the LD50 was estimated to be 600 mg/kg bwt of PHMGH, which may indicate that PHMGH has a relatively “low toxicity“ based on this LD50 value. The lowest dose for toxicity (clinical signs) was 400 mg/kg. No toxicity was however encountered at a dose of 8 mg/kg bwt. It should be emphasized that the extrapolation of findings in animal models to humans should be done with caution. Previous reports however imply that PHMGH belongs to the group of moderately toxic substances, subgroup IV-A, and does not possess species-related specificity. 10 Our LD50 results were comparable to other studies of which 50% mortality (LD50) value for PHMGH was reported as 450 mg/kg in mice and 630 mg/kg in rats. 10,14 Thus, PHMGH may be considered as a moderately toxic substance when extrapolated to humans with 70.0 kg average weight. 14
The blood chemistry carried out during the acute toxicity studies also indicated little or no adverse reaction on cellular components of the blood. Reduction in Hb, HCT, and RBCs normally indicates depression of the hematopoietic system. The very high dose of PHMGH administered may be attributed to this observation. Also, the reduction in WBC and lymphocytes normally reflects a decrease in resistance to stressful situations or diseases in the patients. It is quiet difficult to attribute these to very high dose of PHMGH since the results were not statistically significant.
A very high mean plasma potassium concentration was observed with the LD50 group compared with the control. Under usual circumstances, this could be detrimental to the heart; however, histopathological study did not confirm loss of integrity of this vital organ. Although, the LD50 was extremely higher than the recommended dose or residual dose of 1 mg/L (0.004 mg/kg) to be of any possible health risk, studies on the heart was not conducted to warrant any logical conclusion.
Sodium concentrations measured were about the same and very comparable with that of the controls. Thus, most clinical manifestations such as lethargy, weakness, and so on, usually associated with sodium imbalance, were not observed in the study. However, most of the animals that died exhibited various nervous manifestations such as abnormal gait and tonic–clonic convulsions, which may be difficult to tell at this point whether it was related to the biocidal mechanism of action of PHMGH. This observation was not supported by the electrolyte profile obtained from the blood chemistry analysis. Thus, a chronic study, which is outside the mandate of the present study, is recommended to further investigate this nervous phenomenon. It should be emphasized that mortality occurred only at very high doses other than that recommended by the manufacturer.
Blood biochemistry analyzed included AST, ALT, GGT, and lipid profile. The ALT and AST are usually found in the liver, but small quantities are found in kidneys, muscles, and pancreas. 14
From the results, it is evident that the chemical induced 3 times the levels of ALP at the LD50. This may not necessarily suggest liver damage at this dose tested since changes in ALP could also be induced by alterations in bone and GIT. Gastrointestinal tract changes were also noted macroscopically in this study. However, only 10% of the animals tested at the LD50 level had their liver showing mild periportal lymphocyte infiltrates histopathologically, thus failing to confirm any serious hepatic damage.
The AST and ALT determined from this acute study was comparable with the control, suggesting that integrity of the liver was not really compromised at this dose level. 15,16 However, 20% of animals had their liver samples portraying mild hepatocytes injury in the form of mild periportal lymphocyte infiltration (Figure 5F). It should be emphasized that these liver samples corresponded to animals that died within the first 3 hours of the acute toxicity study. Mean SGOT (AST), SGPT (ALT), GGT, and the lipid profile from the biochemical analysis indicated a trend, which suggested that there was no significant difference between the control and the treated groups, lending further credence to the assertion that the chemical may be mildly toxic to rats only at extreme high dose such as the LD50, which is evidenced by the high level of only ALT but no other biomarker. 16
Poor renal function at the LD50 is evidenced by high level of creatinine, although the high level of blood urea nitrogen was not statistically different from the controls. This suggests that at such high levels as the LD50, the kidney function was not the best; however, it should be noted that such poor renal function occurred at a dose that was far higher than (50 000 times) the dose recommended by manufacturer. Also, the histopathological examination of the kidney samples did not adequately support the poor kidney function observed at LD50.
We proceeded with subchronic toxicity studies that play a major role in the determination of safety evaluation of chemicals. Three dose levels were tested for the subchronic study, and the results treated as indicated earlier and compared to that of the controls. Clinical manifestations were absent as the animals appeared healthy during the study.
Analysis of the blood chemistry for the RBCs count, MCHC, and MCH did not show any statistical differences between the doses tested and the controls. 17 In fact, all the insignificant changes observed in the various hematological parameters (Figure 1B) were not statistically significant, suggesting that the chemical may have no adverse effect on cellular components of the blood even at the tested doses of 0.006, 0.012, and 0.036 mg/kg bwt.
This shows that, at least, for the period and dose levels tested, the chemical appeared to have no adverse effect on the hematopoietic system. These results are consistent with other studies previously done in subacute toxicity studies. 9
Biochemical analysis included AST, ALT, GGT ALT, and AST as a manifestation of liver function. 15 The results obtained suggest that the liver was in no serious toxic danger from the insult of the chemical. This was confirmed by the low level of degenerative lesions observed in the specimens at the histopathological examinations. 18 The high levels of GGT found in the 0.036 mg/kg and control groups were not statistically different from the rest of the tested doses as evidenced by the large SEM associated with these groups. Further, only 10% of the animals had their liver samples showing mild to severe hepatocyte injury relative to the controls. The lipid profile suggests normal levels for all the 3 different components of the profile in all the animals, including the controls. Normally, PHMGH have been used at various concentrations against microbes.
With respect to the electrolytes, there was no observable difference between the potassium and sodium profiles of the test groups and the controls, suggesting that the chemical does not influence the electrolyte composition of blood even when it is used subchronically.
Although the mean urea levels appeared to be lower than that of the controls, the difference was, however, not statistically significant, and the observation was not entirely supported by the histopathological evaluation of the kidney tissues. This is because only some mild degeneration lesions were observed in a few of the kidney specimens (Figure 5C). It is rather interesting to note that creatinine levels were comparable between the treated and the control groups. Further PHMGH did not exert any negative effect at normal doses on the heart except at high doses where mild myocarditis was observed. This may correspond with observations made by Kim et al 19 where dead zebra fish treated with PHMGH (0.3% final concentration) showed severe accumulation of fibrous collagen in the bulbous artery of the heart with elevation in reactive oxygen species. The above-mentioned results may give an idea of the relative safety of PHMGH when used within the recommended doses of 0.012 mg/kg in vivo in subchronic condition. However, considering this study and other studies conducted, 20 it appears PHMGH affects mostly the activity of liver, kidney, and heart and behavioral reactions suggesting that hepatotoxicity, nephrotoxicity, cardiotoxicity, and neurotoxicity may play a major role in the pathogenesis of intoxication by PHMGH. These results which mostly are not statistically significant cannot authenticate the safety of PHMGH to treat water for drinking. Recently, PHMGH was found to induce cellular toxicity through the generation of intracellular reactive oxygen species and alteration of gene expression at very low concentrations of 2 to 5 µg/mL in human alveolar epithelial A549 cells in vitro. 21 In this study, it was shown that PHMGH has cytotoxic potential. This calls for more detailed research to be conducted to obtain full toxicological profile of its safety. Further in this study, it was shown that PHMGH treatment leads to cellular toxicity in both normal and carcinoma cell lines while downregulating antioxidant-related genes. There is thus the need for further investigations to evaluate its effect especially during chronic use and potential risks associated with the lifelong exposure of humans and wildlife to this biocide 22 if it is to be used for the treatment of raw water to make it potable. Further, the insignificant change in body weight that accompanied the 90-day administration of PHMGH need to be investigated irrespective of the fact that the SD rats were near adulthood. According to other studies conducted, 10 the LD50 to minimal working dose ratio of PHMGH lies between 50 and 126 in which the authors in that study imply that tested samples of PHMGH possess average cumulative toxicity. Our acute study results also did not confirm such a claim since our LD50 of 600 mg/kg to the manufacturers working dose of 0.012 mg/kg lies beyond this range. This preliminary study has provided a background as to the safety of PHMGH during a 90-day daily administration in SD rats.
Conclusion
An acute toxicity study in male and female rats showed the LD50 to be 600 mg/kg of PHMGH. This study also identified behavioral changes associated with high doses of PHMGH.
Subchronic toxicological studies with PHMGH in male and female rats for up to 90 days at dose level of 0.036 mg/kg were not associated with mortality or visible clinical signs of toxicity. There were no observable anomalies in the hematological and biochemical parameters utilized to evaluate liver function, kidney function, and lipid profiles. However, microscopic evidence showed mild hepatocellular necrosis in 10% of animals at all dose levels administered, mild tubular damage in 20% of animals (0.012 and 0.036 mg/kg), and mild myocarditis in 10% of animals (0.006 mg/kg). Manufacturer recommends a dose of 0.012 mg/kg bwt (3.0 mg/L) to be used for water treatment process.
Limitations
Monthly urine assessment and other parameters should have been assessed in the course of the study and organ weights should also have been assessed at the end of the study. These would be carried out during our chronic toxicity studies.
Recommendation
Chronic toxicity studies using other animal species including juveniles are recommended to ensure its safety in long term if it is to be used use for the treatment of raw water to make it potable. Besides, genotoxicity, reproductive, and developmental toxicity studies of PHMGH need to be conducted including the possible mechanism of its toxicity.
Footnotes
Acknowledgments
The authors wish to acknowledge the immense contributions of Prof Yao Tettey and Prof R. K. Gyasi of the pathology department, University of Ghana Medical School, Mr Kwasi Kumi of the department of Pharmacology and Toxicology, and Prof K. A. Bugyei and the entire technical staff of the department of pharmacology, University of Ghana Medical School.
Author Contributions
I. J. Asiedu-Gyekye contributed to conception and design, acquisition, analysis, and interpretation, drafted the article, critically revised the article, gave final approval, and agrees to be accountable for all aspects of work ensuring integrity and accuracy. A. Seidu Mahmood contributed to design, interpretation, drafted the article, critically revised the article, gave final approval, and agrees to be accountable for all aspects of work ensuring integrity and accuracy. C. Awortwe and A. Nyarko contributed to analysis, critically revised the article, gave final approval, and agrees to be accountable for all aspects of work ensuring integrity and accuracy.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
