Abstract
Rosemary (Rosmarinus officinalis; RM) and thyme (Thymus vulgaris; TV) are used as remedies. We designed this work to investigate the preventive effect of aqueous extracts of RM or TV on the gentamicin (GM)-induced hepatotoxicity and abnormalities of lipid profile in rats. Both plant extracts exhibited an in vitro antioxidant activity as determined by the 1,1-diphenyl-2-picrylhydrazyl assay. Radical scavenging activity for TV extract was 48.7% and for RM extract was 25.6%. Polyphenolic determination by high-performance liquid chromatography for both extracts revealed that catechin, coumarin, cinnamic acid and rutin were detected in both extracts. RM extract had higher values than TV extract in all except rutin. Ferulic acid and quercetin were also detected in TV extract and sinapic acid and oleuropein were detected in RM extract. In rats given GM intraperitoneally for 10 days and coadministered either RM extract or TV extract orally, both aqueous extracts demonstrated similar hepatoprotective effects manifested by approximate normalization of plasma liver enzymes (AST and ALT), bilirubin level and total protein concentration compared with the group given GM only. Abnormal lipid parameters and raised hydrogen peroxide levels were ameliorated by both extracts. Pancreatic lipase activity was markedly reduced by RM extract. Also, the atherogenic index value was significantly reduced by both extracts as compared to that of GM group. DNA fragmentation analysis confirmed tissue damage by GM and its amelioration by the extracts. These findings suggest that RM and TV extracts could be helpful in ameliorating some aspects of gentamicin toxicity.
Introduction
Dyslipidemia is considered a major risk factor for many cardiovascular and cerebrovascular complications. 1 Abnormalities in lipid metabolism or dyslipidemia are characterized by increased levels of total cholesterol, triacylglycerols (TAGs), and low-density lipoprotein-cholesterol. Meanwhile, high-density lipoprotein-cholesterol level is reduced. Many drugs produce dyslipidemia as a side effect. Drugs on administration are transferred through many barriers that are lipoid in nature, causing alterations in them with concomitant production of lipid peroxides. Antirheumatic drugs have deleterious influences on lipid profile, which may be proatherogenic. 2 Valproic acid, a commonly prescribed antiepileptic drug, causes weight gain and obesity. 3 Second-generation antipsychotics, which are useful therapies for schizophrenia, are extremely likely to cause dyslipidemia and increased cardiovascular risk. 4 Also, drugs used in highly active antiretroviral therapy have dyslipidemia as a major side effect. 5 Gentamicin (GM) is a potent aminoglycoside antibiotic that is clinically used against gram-negative bacteria. However, its uses became limited due to its documented nephrotoxicity that may be accompanied by alteration in blood lipid profile parameters. 6 –9 It was reported that acute kidney injury is strongly associated with cardiovascular disease (CVD). 10 GM can initiate oxidative stress and consequently causes lipid peroxidation. It may mediate the production of reactive oxygen species (ROS) such as hydrogen peroxide (H2O2) and hydroxyl radicals in the mitochondria that can induce injuries to tissues. 8 By causing marked changes to mitochondrial function in the liver, GM may induce hepatic damage that may progress to hepatic failure. 11
Many natural products can be used as an adjuvant with many drugs to modulate or even abolish their side effects. 12,13 Edible herbs are generally considered safe and can be used to alleviate various human ailments. They possess antioxidant activity and vital phytochemicals with either no or minimal adverse effects. 14 Thyme (Thymus vulgaris L., Lamiaceae; TV) has been used in folk medicine for the treatment of asthma and bronchitis. It is used as an antiseptic, antitussive, antifungal, antiviral, antidiabetic, and antioxidative agent. 15 TV oils contain thymol and carvacrol as their main bioactive components. Both of them may possess the same pharmacological effects as TV. TV also has many polyphenolic compounds that are responsible for its antioxidant effects such as rosmarinic acid, quercetin, eriocitrin, luteolin, and epigenin. 16
Rosemary (Rosmarinus officinalis L., Lamiaceae; RM) has been widely applied for various medicinal purposes as stimulant and analgesic. It is also effective for relieving inflammatory diseases, physical and mental fatigue, oxidative stress, and hypercholesterolemia. 17 These pharmacological properties are attributed to important phytochemicals in RM, which include phenolic compounds such as carnosic, ursolic, rosmarinic, caffeic acids and carnosol and their terpene metabolites and flavones. 18 It is possible that both TV and RM could reduce GM toxicity. However, to our knowledge, this hypothesis has not been explored previously. Therefore, the aim of the present research was to investigate the hepatoprotective effects of TV or RM against GM toxicity.
Materials and methods
Plant materials and extraction
RM and TV aerial parts were obtained from the local herbal shop at Giza, Egypt. They were authenticated by a taxonomist in phytochemistry and plant systematic department, National Research Center, Egypt. Voucher specimens have been deposited at the herbarium unit of NRC. The plant samples were finely powdered. Eight grams of each plant were boiled for 5 min in 100 ml of distilled water (8% W/V) and left for 10 min to infuse. Then the extracts were cooled and filtered. These aqueous extracts were prepared daily. The oral dose from these extracts was 10 ml kg−1 day−1. 19
Identification of phenolic compounds
Phenolic compounds were extracted from finely powdered plant materials by methanol according to Alvarez-Jubetea et al., 20 in which 1.25 g of freshly ground sample was added to 25 ml methanol, homogenized for 2 min at 24149 g using homogenizer, vortex-mixed for 20 min at 168 g, and centrifuged for 10 min at 671 g. The final extract was obtained by separating the supernatant that was then filtered. The methanolic extract was analyzed by high-performance liquid chromatography (HPLC).
HPLC analysis was carried out using Agilent Technologies (Santa Clara, CA, USA) 1100 series liquid chromatograph equipped with an autosampler and a diode-array detector. The analytical column was an Eclipse XDB-C18 (150 × 4.6 μm; 5 μm) with a C18 guard column (Phenomenex, Torrance, California, USA). The mobile phase consisted of acetonitrile (solvent A) and 2% acetic acid in water (v/v) (solvent B). The flow rate was kept at 0.8 ml min−1 for a total run time of 70 min and the gradient program was as follows: 100% B to 85% B in 30 min, 85% B to 50% B in 20 min, 50% B to 0% B in 5 min, and 0% B to 100% B in 5 min. Peaks were monitored simultaneously at 280 and 320 nm for the benzoic acid and cinnamic acid derivatives, respectively. All samples were filtered using a 0.45 μm Acrodisc syringe filter (Gelman Laboratory, Michigan, USA) before injection. Phenolic compounds were identified by comparing the retention times and UV spectra with those of authentic standards. All analyses were repeated three times.
Determination of free radical scavenging activity
The 1,1-diphenyl-2-picrylhydrazyl (DPPH) is a stable free radical that accepts an electron or hydrogen radical to become a stable molecule. The model of scavenging the stable DPPH radical is widely used for relatively rapid evaluation of antioxidant activities. DPPH RSA was evaluated using the principle proposed by Brand-Williams et al., 21 with some modification. Briefly, 700 μl of sample or methanol was added to the same volume (700 μl) of methanolic solution of a 100 μM DPPH (0.05 g ml−1). The mixture was shaken vigorously and allowed to stand at room temperature in the dark for 15 min. The decrease in absorbance (A) of the resulting solution was monitored at 517 nm. The antioxidant activity was calculated as percentage inhibition of DPPH by the following equation:
where A control is the absorbance of methanol with DPPH and A extract is the absorbance of tested extracts. All the assays were carried out in triplicate and the average values were calculated. The resulting value is indicative for antioxidant activity of the extract.
Experimental animals
Thirty-two male Sprague-Dawley rats weighing 150–200 g were purchased from Animal House of NRC, Dokki, Giza, Egypt. They were kept individually in stainless steel wire bottom cages at room temperature (25°C) under 12-h dark/light cycle. They had free access to balanced diet and water. They were included in experimental work after 1 week acclimatization period. All experimental design was approved by the Animal Care and Ethics Committee of the NRC.
Experimental design
Rats aged 2 months were assigned into four groups, each included eight rats. They were fed standard balanced diet and had free access to water. Group 1 was the negative control. Control rats were injected intraperitoneally (ip) with saline (0.2 ml/day). The influence of circadian rhythm was avoided by starting all dosing at 10.00 am. 22 Group 2 served as a positive control. Rats of this group were injected ip with GM (60 mg/kg/day). 23 Group 3 were injected ip with GM and coadministered 8% aqueous RM extract orally 10 ml/kg/day. 19 Group 4 rats were injected with GM and orally coadministered TV extract 10 ml/kg/day. The experiment lasted for 10 days through which body weights were monitored every second day for plotting growth curves. On the 10th day, the rats were fasted overnight, anesthetized by light ether, and blood samples were obtained in heparinized tubes from retro-orbital veins. Plasma was separated and stored at −80°C for biochemical analysis. Meanwhile, livers were immediately removed, washed with saline, weighed, and divided into two parts for assessment of DNA fragmentation and histopathology
DNA ladder assay
One gram of liver tissue was homogenized in 10 ml (10% w/v) of TE solution pH 8 (5 mM TrisHCl, 20 mM EDTA and 0.2% triton X-100). DNA was extracted from homogenate by using a ready-made kit (Gene Jet Genomic DNA purification kit; Thermo Scientific, Lithuania) according to the manufacturer’s instruction. Five micrograms of DNA was loaded in 1.5% agarose gel containing 1.0 μg ml−1 ethidium bromide. The electrophoresis gel was then studied under gel doc system and was photographed. 24
Histological examinations
Livers from all animals were dissected immediately after death and fixed in 10% neutral-buffered formal saline for at least 72 h. All the specimens were washed in tap water for half an hour and then dehydrated in ascending grades of alcohol, cleared in xylene, and embedded in paraffin. Serial sections of 6-µm thick were cut and stained with hematoxylin and eosin. Then, each hepatic section was observed under bright field using a Carl Zeiss Axioskop (Jena, Germany) microscope. Images were captured and processed using Adobe Photoshop version 8.0.
Biochemical analysis
Liver function was evaluated by determining plasma alanine aminotransferase and aspartate aminotransferase activities colorimetrically using a Biodiagnostic kit (29 Taheer St., Dokki, Giza, Egypt, www.bio-diagnostic.com) according to the principle of Reitman and Frankel. 25 Also, plasma total bilirubin level was assessed by applying Walter and Gerarde 26 procedure using commercial kits. The synthetic activity of the liver was determined by evaluating plasma albumin 27 and total protein concentration. 28 Total cholesterol and TAG levels were assayed enzymatically. 29,30 High-density lipoprotein cholesterol (HDL-Ch) was evaluated by an enzymatic colorimetric method after precipitation of very low-density lipoprotein cholesterol (VLDL-Ch) and low-density lipoprotein cholesterol (LDL-Ch). 31 VLDL-Ch + LDL-Ch value was calculated by subtracting HDL-Ch from the total cholesterol. Plasma phospholipids level was determined colorimetrically. 32 Atherogenic index was calculated (total cholesterol/HDL-Ch). H2O2 concentration was assayed colorimetrically, which in the presence of 3,5-dichloro-2-hydroxybenzene sulfonic acid and 4-amino antipyrine peroxidase reacts to produce a chromophere (quinoneimine dye) as cited in the Biodiagnostic kit. Finally, pancreatic lipase (PL) activity was measured calorimetrically according to Moss and Henderson 33 using Salucea kit (Haansberg 19, 4874 NJ Etten Leur, the Netherlands, www.Salucea).
Statistical analysis
Data were presented as mean ± SE. Statistical analysis of the data was performed using SPSS-PC software version 22 ( Chicago,IL). Unpaired Student’s t-test was used to compare biological differences. One-way analysis of variance was used for comparison of different biochemical values in various experimental groups. It was followed by Duncan’s multiple range tests to clarify the significance. p values less than 0.05 were considered significant.
Results
Figure 1 and Table 1 illustrate the changes in growth and body and liver weights, respectively, throughout the experiments. The growth rate was not significantly different between experimental groups and negative controls; however, the positive controls showed a significant increase compared with the negative controls (p < 0.05).

Changes in rats weights in different experimental groups.
Body weight and liver weight following treatment with GM or GM plus coadministration of RM or TV extract.

GM: gentamicin; RM: rosemary; TV: thyme.
aValues are significantly different at p ≤ 0.05 as compared to normal control.
bValues are significantly different at p ≤ 0.05 as compared to positive control.
Body weight gain was markedly elevated in positive controls given GM only (p < 0.05), compared to negative controls. Coadministration of RM extract with GM significantly reduced this body weight gain. Coadministration of TV extract with GM reduced body weight gain slightly but not significantly. RM extract with GM significantly reduced liver weight compared to those of the positive control group given GM only. The relative liver weight in positive controls was significantly decreased compared to that of the negative controls. Coadministration of TV extract with GM, but not RM extract with GM, significantly ameliorated the GM-induced decrease.
DPPH scavenging activity and HPLC phenolic analysis
Radical scavenging activity of TV extract was higher (48.7%) than that of RM extract (25.6%). Phytochemical screening by HPLC for both extracts showed that catechin was the most predominant phenolic compound according to our available standards, while rutin was the lowest one. Sinapic acid and oleuropein were only detected in RM extract, whereas ferulic acid and quercetin were only detected in the TV extract (Table 2).
DPPH RSA and HPLC polyphenols of methanolic extracts of RM and TV.

DPPH: 1,1-diphenyl-2-picrylhydrazyl; RSA: radical scavenging activity; HPLC: high-performance liquid chromatography; RM: rosemary; TV: thyme.
The effects of various treatments on plasma liver enzymes, total proteins, albumin, and H2O2 are shown in Table 3. Our results indicated that rats treated with GM exhibited an extreme damage to the liver as manifested by significant elevation of activities of plasma liver enzymes ALT and AST, as well as the bilirubin level. The significant decrease in plasma total protein level was also consistent with liver dysfunction. H2O2 concentration was markedly elevated in GM group as compared to negative control group indicating a state of oxidative stress. Both TV and RM extracts ameliorated such deleterious effects of GM treatment to a similar extent.
Liver function markers, total protein, albumin, and H2O2 levels following treatment with GM or GM plus coadministration of RM or TV extract.a

GM: gentamicin; RM: rosemary; TV: thyme; H2O2: hydrogen peroxide; SE: standard error.
aValues are expressed as mean ± SE. Mean values in the same column sharing the same superscript letters (b, c, d) are not significantly different (p ≥ 0.05).
Coadministration of both extracts significantly decreased ALT and AST activities, and the RM extract significantly reduced the total bilirubin level, compared with the GM only positive control group. Also, through their powerful antioxidant effect, both extracts significantly reduced H2O2 levels compared to the GM only positive control group.
GM administration significantly elevated plasma total cholesterol phospholipids and TAGs as compared to the negative control group. The concentrations of various lipoproteins (HDL-Ch or LDL-Ch + VLDL-Ch) showed insignificant changes by GM treatment. However, the atherogenic index was significantly raised in GM group (1.97 ± 0.163) as compared to that of the control group (1.47 ± 0.126). Both RM and TV extracts showed comparable hypolipidemic effects as manifested by significant decrease of total cholesterol, phospholipids, TAGs, and atherogenic index. TV extract treated significantly reduced LDL-Ch + VLDL-Ch as compared to that of GM group (Table 4). Moreover, GM caused a marked induction in PL activity, which was significantly inhibited by RM extract, while TV had no effect.
Plasma lipid parameter levels, atherogenic index, and PL activity following treatment with GM or GM plus coadministration of RM or TV extract.a

PL: pancreatic lipase; GM: gentamicin; RM: rosemary; TV: thyme; TAGs: triacylglycerols; LDL-Ch: low-density lipoprotein cholesterol; VLDL-Ch: very low-density lipoprotein cholesterol; HDL-Ch: high-density lipoprotein cholesterol; SE: standard error.
aValues are expressed as mean ± SE. Mean values in the same column sharing the same superscript letters (b, c) are not significantly different (p ≥ 0.05).
On agarose gel electrophoresis, there was liver tissue DNA fragmentation by GM and preventive effects of RM and TV extracts (Figure 2). From the left, lane 2 appeared as DNA laddering pattern with smear caused by GM treatment to rats as compared to that of the negative control, lane 1. Coadministration with RM or TV extracts significantly eliminated injuries to DNA as there were intact regular DNA bounds with very faint smear (lanes 3 and4, respectively). DNA fragmentation was not present in the negative control group (lane 1).

Agarose gel showing DNA damage by GM and protective effect of RM and TV on rat’s liver of all experimental groups. Lane 1: control group, lane 2: GM-treated group, lane 3: RM-treated rats, lane 4: TV-treated rats, and lane 5: DNA molecular weight marker. GM: gentamicin; RM: rosemary; TV: thyme.
Hepatic histopathological changes in various experimental groups are shown in Figure 3. Figure 3(a) shows control hepatic tissue with normal architecture of portal vein and normal hepatocytes arranged in a form of trabeculae, around the central vein and with normal blood sinusoids. Each hepatic cell possesses a surrounding membrane, centrally placed huge nucleus and prominent nucleoli. Figure 3(b) illustrates microscopic changes in livers treated with GM. Hepatic tissues depicted marked hepatocellular alterations and almost all of the hepatocytes have lost their cellular membrane with foci of necrotic hepatic tissue. Treatments with TV or RM extracts along with GM exhibited reversal of some of the hepatic lesions produced by GM alone (Figure 3(c) and 3(d), respectively).
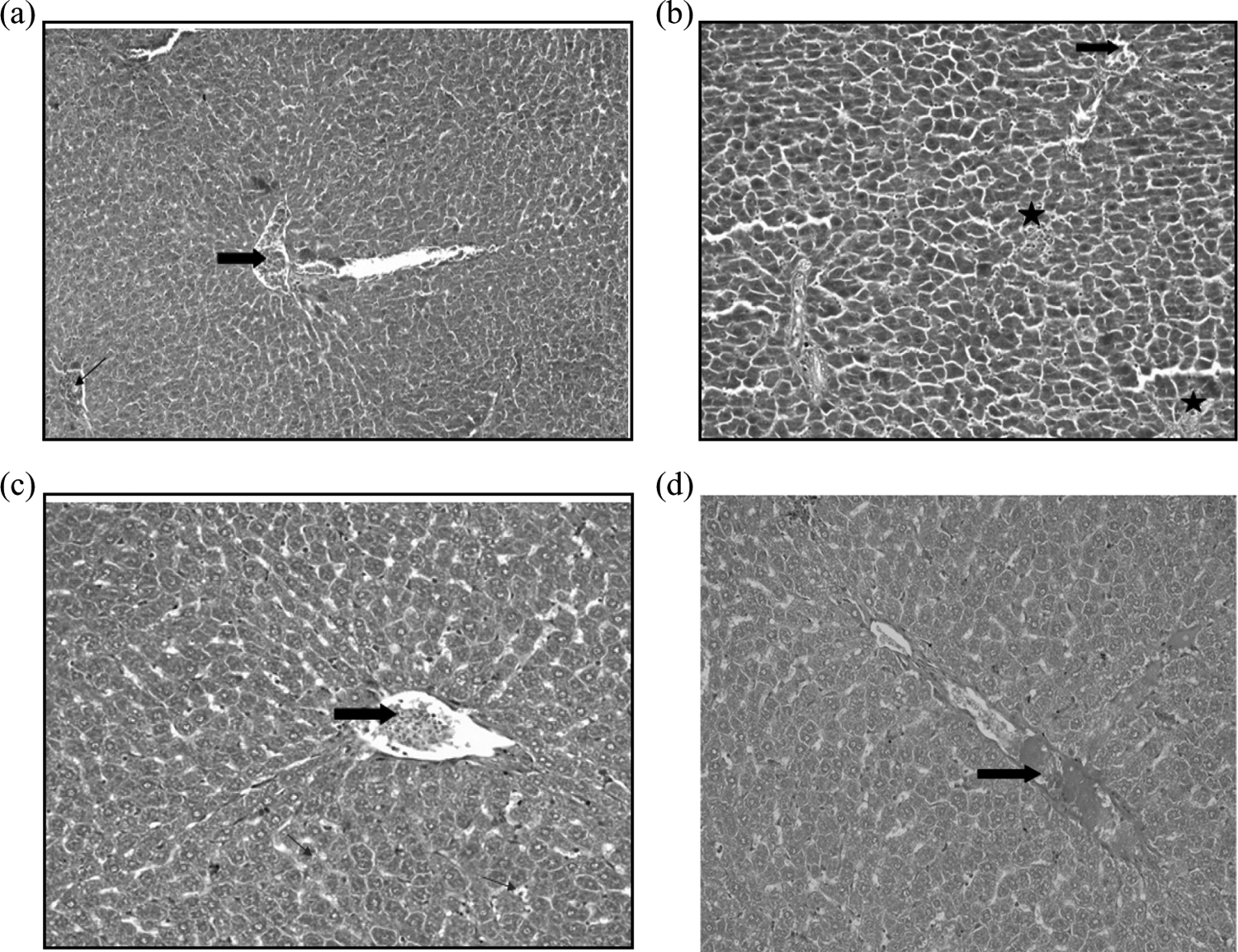
(a) A photomicrograph for (control group) showing normal liver tissue. The liver is divided histologically into lobules. The center of the lobule is the central vein (thick arrow), and at the periphery of the lobule, there are the portal triads (thin arrow) (H&E 100). (b) A photomicrograph for (GM group) showing congested blood vessels (thick arrow), almost all of the hepatocytes have lost their cellular membrane with foci of necrotic hepatic tissue (black star) (H&E 200). (c) A photomicrograph for (TV group) showing congested blood vessels (thick arrow) with mild interhepatic edema and mild ballooning in the hepatic cells (thin arrow) (H&E 200). (d) A photomicrograph for (RM group) showing normal hepatic architecture unless congested dilated hepatic vessel (thick arrow) (H&E 200). GM: gentamicin; H&E: hematoxylin and eosin; TV: thyme; RM: rosemary.
Discussion
Antibiotics are the most widely used class of drugs that prevent many infections. However, they have many adverse effects and can injure various body organs such as liver, kidney, brain, and eyes. GM uses have been limited due to its side effects. Many strategies have been applied to overcome such side effects. The coadministration of natural products with GM may be helpful. 8,34,35 The present study was designed to evaluate the protective capacity and ameliorating effects of TV and RM extracts on GM -induced liver toxicity and dyslipidemia, respectively.
Our data demonstrated that GM administration significantly damaged liver cells as manifested by a large increase in ALT and AST activities and total bilirubin level. Moreover, it caused a marked decrease in total protein concentration indicating a failure in liver synthetic capacity caused by GM administration. Our results were confirmed by hepatic histopathological examination that showed severe liver damage induced by GM. Also, DNA fragmentation studies demonstrated that necrosis and apoptosis of hepatic cells were observed to certain extent. Those data were in agreement with Ademiluyi et al. 11 and Galaly et al. 36 Moreover, dyslipidemia was induced by GM treatment as it was clear from the elevation of TAGs, cholesterol and phospholipids concentrations, and PL activity. Our data are in agreement with Abdel-Gayoum et al., 7 Abbas et al., 37 Ahmadvand et al., 9 and Ahmadvand et al. 38 Besides, GM treatment produced an oxidative stress as it was clear from the increment of H2O2 level compared to the negative control, which is consistent with the findings of Ademiluyi et al. 11
GM has been shown to increase mRNA levels for many proinflammatory cytokines such as interleukin-16 and tumor necrosis factor-α. 39 ROS and accumulation of H2O2 that resulted from oxidative stress are injurious to various biomolecules such as proteins, nucleic acids, and membrane lipids either for cell or its organelles. Destruction of cell membrane caused release of hepatic enzymes (ALT and AST). Destruction of lysosomes and mitochondrial membranes resulted in degradation of essential components in the liver, either functional or structural. Therefore, GM may damage bile duct with concomitant increase in plasma bilirubin. It can also affect erythrocyte membranes and increase their fragility, 40–41 which in turn lead to increased hemoglobin breakdown with consequent elevation in plasma bilirubin level. It can be noticed that the decrease in plasma protein level by GM may be attributed to the damage of hepatic protein synthesizing structure. Also, it may be due to increased urinary protein excretion by GM-induced nephrotoxicity. 8
CVD and kidney disease are closely correlated. 42 GM administration at high doses confirms this association. GM caused dyslipidemia, which is considered a major risk factor to CVD. Also, it is well documented that GM can induce renal damage. Thus, GM may have dual effects that could influence the induction of CVD. In our study, this was shown by the marked elevation in the atherogenic index in the GM positive control group compared with the negative control group.
GM increased PL activity, which may be attributed to its release to plasma from damaged pancreatic cells by GM. Ri et al. 43 demonstrated that GM diffused in pancreatic juice. Pancreatic lipase is a key factor for absorption of dietary TAGs. Therefore, elevated activity caused increased TAGs hydrolysis and absorption with concomitant increased TAGs plasma levels. 44 Also, ROS resulting from GM may inhibit β-oxidation of free fatty acids in the mitochondria either by structural damage to mitochondria or by inhibiting their enzymes that are involved in such oxidation. Also, ROS stimulate the production of epinephrine, 45 which in turn suppresses insulin release but promotes glucagon secretion with concomitant induction for hormone-sensitive lipase activity. The latter enzyme enhances lipolysis in both adipose tissue and liver increasing FFA. Excess FFA in circulation triggers their conversion into TAGs and cholesterol, 46 with concomitant increase in VLDL-Ch and LDL-Ch, respectively. Therefore, hypercholesterolemia may result from enhanced synthesis or impaired catabolism in the liver especially with injured liver by GM. Chisholm et al. 47 had demonstrated that drug-induced cholestasis is accompanied by the enhanced hepatic cholesterol synthesis. Also, ROS have the capability to (1) enhance 3-hydroxy-3-methylglutaryl-co-enzyme A: reductase (HMG-CoA reductase), the key enzyme for cholesterol synthesis, (2) increase esterification of cholesterol by activating acyl coenzyme A: cholesterol acyl transferase, (3) decrease cholesterol ester hydrolysis by inhibiting cholesterol ester hydrolase, and (4) inhibit cellular cholesterol efflux into HDL-Ch 48 that may be due to inhibition of the activity of lecithin cholesterol acyl transferase by ROS. 49
Regarding the elevation of phospholipids level in GM-treated rats, Carlier et al. 50 showed that aminoglycoside antibiotics inhibit lysosomal phospholipases (A1 and A2) with concomitant accumulation of lysosomal phospholipids in liver, spleen, and other tissues, a condition known as phospholipidosis. The structural features of GM are responsible for such effect as it has an aromatic ring and an amino group present at varying distance from the hydrophobic region. 51 Such deleterious effects caused by GM were considerably ameliorated by both RM and TV extracts where both RM and TV protected the liver from GM intoxication as manifested from the significant decline of AST and ALT activities and bilirubin level as compared to those of GM alone. Moreover, DNA fragmentation in liver tissue was markedly reduced by both RM and TV extract, to a similar extent. The same result was observed in the histopathological studies to the liver tissue where equivocal repair to liver tissue was performed by both extracts as compared to livers of group treated with GM. Both DNA fragmentation and histopathological studies confirmed our biochemical results. Our results are consistent with those of Ramadan et al. 52 and Rašković et al. 53 for RM and with Rana and Soni 54 and Youssef 55 for TV.
The hypolipidemic effects of TV and RM in our studies were previously reported by Ozkol et al. 15 and Labban et al., 56 respectively. Both RM and TV extracts could play a role in reducing the risk of CVD, as it is evident from their reduction of the atherogenic index, compared to the GM only group.
Many mechanisms may be involved to explain the hepatoprotective and hypolipidemic effects of RM and TV. The secondary metabolite polyphenolic content of both plants that is partly responsible for their antioxidant effects may take part in such protection. This hypothesis is supported by our results showing both extracts significantly decreased H2O2 levels and both had strong in vitro antioxidant effects as detected by DPPH. TV extract in our work had higher antioxidant effects than RM extract, which was in accordance with Kim et al. 57 In addition, our HPLC analysis showed that both extracts had several phenolic compounds mainly catechin, coumarin, and cinnamic acid. Wojdylo et al. 58 demonstrated that plants that belong to Labiatae (Lamiaceae) family possess the highest antioxidant activity that is related to phenolic acids and flavonoids. Rosmarinic acid is the main antioxidant constituent in the Labiatae family, in addition to hydrocaffeic and caffeic acids, which results from enzymatic hydrolysis of rosmarinic acid. The predominant flavonoids were quercetin, luteolin, and apigenin that are abundant in Labiatae. Flavonoids RSA depends on the substitution pattern of hydroxyl group (OH), where the presence of 3-OH group or 3- and 5-OH groups giving a catechol structure in ring C is beneficial together with C2–C3 the double bond. Wojdylo et al. 58 also showed that RM had high content of flavonoids (quercetin, luteolin and kaempferol) and had high antioxidant activity. Meanwhile, TV antioxidant activity resulted from phenolic acids, caffeic, and coumarin. Such antioxidant activity of both extracts compensated oxidative stress with consequent reduction of ROS production, thus the liver tissue was protected from DNA, protein, and membrane damage. Hence, liver markers toxicity was approximately normalized. Also, RM and TV extracts were demonstrated to enhance the activity of phase I and phase II enzymes responsible for the detoxification of GM. 59–60 Moreover, both extracts exhibited anti-inflammatory, antiapoptotic, and antinecrotic effects. All such mechanisms protected the liver tissue from GM toxicity.
Concerning the hypolipidemic effects of RM and TV extracts, it may be explained by their induction of insulin secretion, 61 consequently inhibited hormone-sensitive lipase with concomitant reduction in plasma lipids. Also, our results revealed that RM significantly inhibited PL induction by GM, which contributed to decreased TAG levels. Polyphenolics in plants through their hydrophobic and hydrogen bonding have great affinity to proteins.
Thus, the RM aqueous extract could inhibit PL by aggregation of its protein. 44 Many polyphenols are active against PL, while others are weak inhibitors of PL, 62 as in TV extract in our study. Besides, the antioxidant effects of both RM and TV extracts inhibited oxidative stress induced by 3-hydroxy-3-methylglutaryl coenzyme A(HMG-CO) reductase activity, thus reducing cholesterol synthesis. In addition, polyphenols were demonstrated to increase fecal excretion of total cholesterol and bile acids. 63 Consequently, an enhanced need of cholesterol for synthesis of biliary juices and a reduced absorption of cholesterol from intestine due to lower bile acids levels lead to decreased plasma cholesterol level.
In conclusion, the present data revealed that both RM and TV extracts had free radical scavenging capacity and that of TV extract was the superior. GM administration induced liver damage as evidenced from elevated hepatic markers (AST, ALT) and total bilirubin and impaired its synthetic power as indicated from the fall in plasma total protein level and dyslipidemia. The latter was partly due to elevation of PL activity and increased oxidative stress (H2O2) by GM. However, coadministration of RM and TV extracts markedly ameliorated the deleterious effects of GM mainly through their antioxidant activity. Their effects were approximately equivalent. DNA fragmentation analysis and histopathological studies on liver tissues confirmed our data. It is possible that either RM or TV extracts may be useful as adjuvants with GM as they may reduce some of the adverse effects of the antibiotic on the liver.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.

