Abstract
There are limited data regarding effect of trastuzumab on radiation-induced cardiovascular toxicity when used sequentially or concomitantly. This experimental study aims to investigate effect of trastuzumab on radiation-induced cardiovascular toxicity with respect to the treatment sequence. One hundred and eight female Wistar albino rats were divided into six groups (G): G1 was control, G2 was trastuzumab, and G3 was radiotherapy (RT); G4 and G6 were sequential RT and trastuzumab; and G5 was concomitant RT and trastuzumab groups, respectively. Rats were killed at 6th h, 21st and 70th days after RT; thoracic aorta and heart samples were obtained. Transthoracic echocardiography and functional studies evaluating relaxation of thoracic aorta were performed. Subendothelial edema scores of thoracic aorta samples at 21st and 70th days were higher in RT groups (G3, G4, G5, and G6) (
Introduction
Adjuvant therapies for the treatment of breast cancer including radiotherapy (RT), chemotherapy (CT), targeted therapy, and hormonal treatment significantly decrease the risk of recurrence and increase the survival. However, cardiovascular problems including treatment complications caused by many of these treatment modalities are responsible for most common cause of non-cancer mortality in breast cancer survivors. 1 Although the heart was initially thought to be relatively radioresistant, cardiovascular disease resulting from chest RT for therapeutic purposes is now clearly recognized. 2 The majority of radiation-induced cardiovascular disease has been reported in patients previously treated for Hodgkin’s disease and breast cancer. Estimated relative risk of fatal cardiovascular events after RT for Hodgkin’s disease and left-sided breast cancer range between 2.2–7.2 and 1.0–2.2, respectively, compared to healthy controls. 3 Risk is life long and absolute risk appears to increase with length of time since exposure. Radiation-associated cardiovascular toxicity may in fact be progressive. 3
Numerous studies of radiation-induced toxicity show that endothelial cell injury is the key point in most tissues even though the endothelial cells compromise only a minor fraction of cardiac cells. 4 –9 The sequence of endothelial injury, cell detachment, thrombosis, and fibrosis results in significant tissue injury that often limits radiation oncologist in attempting to deliver curative doses to a nearby tumor. Steward and Fajardo have demonstrated that damage to the myocardium develops through three phases of injury. 3,10,11 The acute inflammation phase occurs about 6 h after RT, and a neutrophilic infiltrate develops involving all layers of heart. The second phase is also known as latent phase in which a slight progressive fibrosis begins about 2 days after exposure. However, electron microscopy of the myocardial capillary endothelial cells demonstrates progressive damage leading to obstruction of the lumen and thrombi of fibrin and platelets. Although healthy endothelial cell replication in the vicinity occurred, it is generally inadequate and an inevitable ischemia leads to progressive fibrosis. Animals begin to die at approximately 70th day due to extensive fibrosis. The hallmark of this late stage is extensive fibrosis.
High-quality randomized trials have demonstrated that adjuvant trastuzumab concomitant with or following CT in node-positive and high-risk node-negative patients with Human Epidermal Growth Factor receptor-2 (HER-2)-positive early stage breast cancer improves disease-free and overall survival. 12 –17 Although generally well tolerated, the incidence of cardiac toxicity with trastuzumab, especially when combined with anthracyclines, is a consistently recognized adverse effect. 12 –14 On the other hand, especially patients treated with internal mammary chain (IMC) RT are candidates for vascular damage. Trastuzumab-induced cardiac toxicity is characterized first by an asymptomatic decline in left ventricular function, and ultimately, symptomatic cardiac failure is seen. 12 –14,18,19
The risk of toxicity of combined trastuzumab and RT on cardiovascular structures has not yet been evaluated in detail. In our previous study, we have demonstrated that when combined with high-dose RT, trastuzumab may lead severe vascular damage. 20 In clinical practice, trastuzumab is used in neoadjuvant or adjuvant setting depending upon the treatment schedule of the patient. Although as previously demonstrated the interaction between RT and trastuzumab seemed to be at least additive in concomitant usage. 20 We have no data about the effects of the sequence of trastuzumab and RT on cardiovascular structures. Therefore, we aimed to clarify the effect of trastuzumab on radiation-induced cardiovascular toxicity when used either sequentially or concomitantly.
Methods and materials
Study design
This study included 108 female Wistar albino rats (250–300 g), the use of which was approved by the Gulhane Military Medical Academy Ethical Committee. Animals were housed four per cage in a controlled animal holding room with a 12/12-h light/dark cycle; temperature and relative humidity were continually monitored to provide standard laboratory conditions. Standard rat chow and water were allowed ad libitum. Rats were divided into six groups (G) each composed of 18 animals. G1 was defined as control group, and rats of this group were sham irradiated. G2 and G3 were trastuzumab and RT only groups, respectively. The rats in G4 (neoadjuvant trastuzumab) and G6 (adjuvant trastuzumab) were received trastuzumab 1 week before and after RT, respectively. G5 was the RT and trastuzumab group in which 15 Gy dose of RT concomitant with trastuzumab was applied (Table 1).
The abbreviations used for the study groups.

RT: radiotherapy; G: group.
Six rats in each arm were killed at 6 h, 3 and 10 weeks after irradiation. The heart and thoracic aorta tissues of each animal were dissected for light microscopy examination. In addition, a functional study for evaluating the relaxation and contraction of aortic ring-like segments mounted in isolated organ bath was performed at 21 days after RT.
RT protocol
RT was applied under general anesthesia with intraperitoneally administered 90 mg/kg ketamine hydrochloride (Ketalar®; EWL Eczacibasi Warner Lambert Ilaç Sanayi ve Ticaret A.S., Istanbul, Turkey) and 10 mg/kg xylazine hydrochloride (Rompun® 2%; Bayer Kimya San. Ltd. Sti., Istanbul, Turkey). A single dose of 15 Gy that has been shown to lead cardiovascular toxicity with 6 MV photon beams was applied via a single anterior field to 2 cm depth with source-axis distance technique; 20 1 cm elasto-gel bolus was used to build up the radiation dose and to provide contour regularity. The field size was 4 × 4 cm and included the mediastinum.
Trastuzumab protocol
Trastuzumab (Herceptin®; Genentech Inc, South San Francisco, CA, USA) dose, which was equivalent to 6 mg/kg adult dose, was calculated for each rat and injected intraperitoneally. The rats in G4 and G6 received mediastinal RT 1 week after and before trastuzumab administration, respectively. The rats in G5 had mediastinal RT 6 h of following trastuzumab administration. The rats in G1 and G2 were applied 0.5 cc 0.9% NaCl intraperitoneally.
Echocardiography
Transthoracic two-dimensional echocardiography was performed on 3rd and 10th weeks of RT. Echocardiography was applied under general anesthesia with the same dose that used during RT. Imaging was performed in the parasternal long- and short-axis views, completely by a single investigator blinded to the treatment groups using vivid S (10 MHz frequency transducer; General Electric, Connecticut, USA). An M-mode cursor was positioned perpendicular to the interventricular septum and posterior wall of the left ventricle (LV) at the level of the papillary muscles, and M-mode images were obtained for measurement of chamber dimensions throughout the cardiac cycle. LV end-diastolic dimension (LVEDD) and LV end-systolic dimension (LVESD) were assessed. During diastole, LV dimension and wall thickness were measured from the maximum chamber cavity; during systole, they were measured during maximum anterior motion of the posterior wall. The functional parameters, fractional shortening, and LV ejection fraction were defined as [(LVEDD–LVESD)/LVEDD] and [(LVEDD 3 –LVESD 3 )/LVEDD 3 ] × 100, respectively. In all cases, three beats were averaged for each measurement.
Morphological study: Preparation of thoracic aorta rings and measurement of pharmacological responses
On the 21st day, the rats were anesthetized with 90 mg/kg ketamine hydrochloride and 10 mg/kg xylazine and killed, and then, thoracotomy procedure was performed. The thoracic aorta was dissected from the diaphragm to the aortic arc and transferred to a dish containing physiologic saline solution. Fat and connective tissues around the aorta were cleaned and 3 mm in length from aorta were prepared. Two parallel wires were placed in the lumen of the rings, and the tissues were mounted to a 20-ml organ bath filled with physiologic saline solution (in mmol/L: NaCl, 118; KCl, 4.6; NaHCO3, 25; MgSO4, 1.2; KH2PO4, 1.2; CaCl2, 1.2; glucose, 10; EDTA, 0.025) and aerated with 95% O2 and 5% CO2 gas mixture. Responses of the thoracic aorta rings were measured by a force displacement transducer and recorded by a polygraph. Aorta rings were rested for 30 min under a resting tension of 0.75 g.
Thoracic aorta segments were first contracted with 68 mM KCl and then with phenylephrine. The contraction responses of phenylephrine were taken in the increasing concentrations and were reported as the percentage of the potassium contraction. Then, aorta rings were precontracted with submaximal phenylephrine concentration. As the plateau was reached, acetylcholine (ACh) was given in an increasing order. The relaxation to ACh was recorded and expressed as percentage of phenylephrine maximum contraction.
Light microscopy
The heart and thoracic aorta samples were excised and fixed in 10% formaldehyde solution and embedded in paraffin for light microscopic examination. The slices obtained were stained with hematoxylin and eosin to evaluate the inflammation and with immunohistochemical triple staining to identify the cardiac fibrosis (CF). As a quantitative end point, extend of the radiation-induced fibrosis was graded on a scale of 0 =
Statistical analysis
The Statistical Package for Social Sciences version 13.0 was used for statistical analyses. One-way analysis of variance test was used to calculate the significance of the differences among groups. Post hoc Tukey’s HSD test was used for pair-wise comparisons. The data are represented as mean ± standard deviation. Differences yielding
Results
Thoracic aorta
Light microscopy
The mean scores of subendothelial edema, inflammation, and fibrosis for the thoracic aorta samples with light microscopy are shown in Table 2. There was no statistically significant difference between the groups with respect to the subendothelial edema scores 6 h after RT (
The mean scores of subendothelial edema, inflammation, and fibrosis for thoracic aorta samples with light microscopy.a

RT: radiotherapy; G: group; SD: standard deviation; ANOVA: one-way analysis of variance.
aANOVA test was used to calculate the significance of the differences among groups. Post hoc Tukey’s HSD test was used for pair-wise comparisons.
b
Functional results
Functional study revealed that the Ach-mediated relaxation responses of the thoracic aorta samples were significantly deteriorated in the RT groups (

The acetyl choline-mediated relaxation responses of the thoracic aorta samples.
Heart
Light microscopy
Light microscopic examination revealed statistically significant differences within the study groups with respect to CF scores during all time intervals (
The mean CF scores for heart samples with light microscopy.a

RT: radiotherapy; G: group; SD: standard deviation; ANOVA: one-way analysis of variance.
aANOVA test was used to calculate the significance of the differences among groups. Post hoc Tukey’s HSD test was used for pair-wise comparisons.
b
c
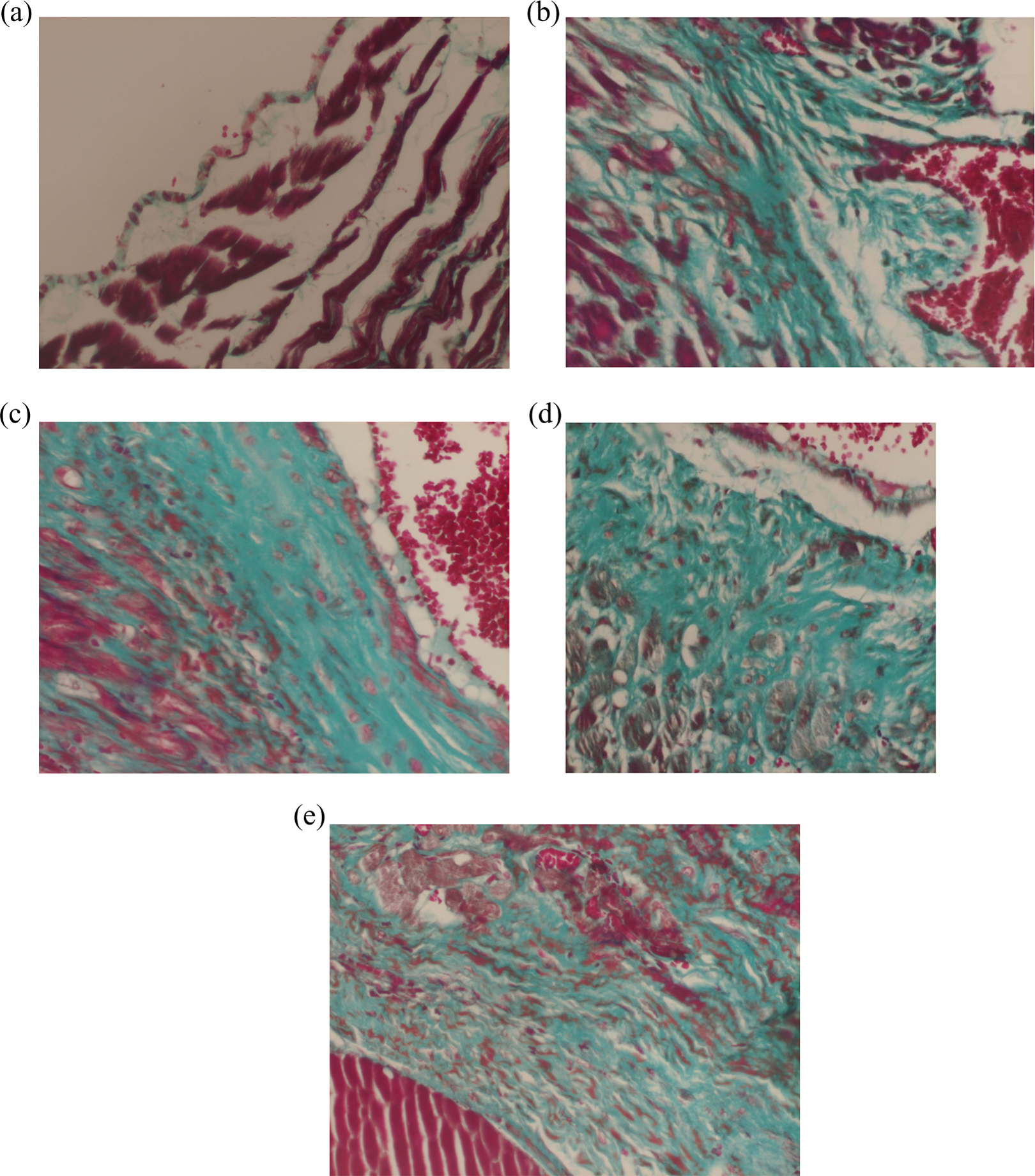
Immunohistochemical triple staining of the heart samples taken from G1 (a), G3 (b), G4 (c), G5 (d), and G6 (e) at 70th day of RT. (a) Heart sample of control group at 70th day of RT, triple staining: normal cardiac structure (×200). (b) Heart sample of RT only group 70th day of RT, triple staining: There was extensive subendocardial fibrosis (×200). (c) Heart sample of neoadjuvant trastuzumab and RT group 70th day of RT, triple staining: There is moderate thickening in the endocardium. There was extensive subendocardial fibrosis (×200). (d) Heart sample of concomitant trastuzumab and RT group 70th day of RT, triple staining: There is severe thickening in the endocardium. There was very extensive subendocardial fibrosis (×200). (e) Heart sample of adjuvant trastuzumab and RT group 70th day of RT, triple staining: There is moderate thickening in the endocardium. There is extensive subendocardial fibrosis (×200). RT: radiotherapy.
Echocardiographic examination
Echocardiographic examination revealed that after 21st and 70th days of RT, the ejection fraction, left ventricular mass, and fractional shortening were significantly decreased in the RT and trastuzumab groups (G4, G5, and G6) when compared to the control group (Table 4). This study was approved by Gulhane Military Medical Academy Ethical Committee.
Results of echocardiography.a

G: group; EF: ejection fraction; FS: fractional shortening; LVIDd: left ventricle internal diameter diastole; LVPWTd: left ventricle posterior wall thickness diastole; mm: millimeter; ANOVA: one-way analysis of variance.
aANOVA test was used to calculate the significance of the differences among groups. Post hoc Tukey’s HSD test was used for pair-wise comparisons.
b
c
Discussion
HER2 receptor tyrosine kinase is known to have a critical role in cardiac development and growth, repair, and survival of adult cardiomyocytes. Neuregulin, produced by cardiac endothelial cells, binds to HER4 and leads to dimerization with HER2, and subsequent cell signaling is necessary for normal myocyte function. HER2 overexpression is reported to be around 15–25% of all breast cancer patients, and this overexpression was shown to decrease overall and disease-free survival rates in addition to diminish the response to variety of chemotherapeutic and hormonal agents. 21 –25 Trastuzumab, a humanized monoclonal antibody that specifically targets HER2-positive breast cancer cells has been shown to increase survival rates in patients with both metastatic cancer and early stage breast cancer. 13,14,26 –29 The cardiotoxic effect of trastuzumab especially when used with anthracycline CT has been well known, though the mechanism is not yet fully understood. 30 Preclinical studies indicate that the direct blockade of the HER2 receptor on myocytes has at least partially an important impact on the cardiotoxicity of trastuzumab. 31 RT on the other hand causes multiple effects on the heart as acute, subacute and chronic pericarditis, coronary vascular disease, valvular heart disease, and restrictive cardiomyopathy. 32 Radiation-induced heart disease is believed to be resulted from injury to microvasculature network and subsequent ischemia. 31
Preclinical in vitro and in vivo studies have shown that the cascade of events through the HER2 receptor is involved in tumor radiosensibility, 33,34 and application of trastuzumab concurrently with radiation thus increases the antitumor effect of radiation. There are same clinical evidences in the literature that trastuzumab also radiosensibilizes human healthy tissues, and in this way, it could increase the toxicity of the treatment. 35 Currently, the most important question remains whether the concomitant therapy with trastuzumab and RT increases cardiotoxicity of the treatment. In the literature, there are limited data about the safety of concomitant therapy with RT and trastuzumab. 33 The observation period in the studies was short, and the longest reported median observation period after the completion of concomitant treatment with RT and trastuzumab was 3.7 years. 17 Therefore, currently there is no evidence that such therapy is safe after a long observation period. 33
In our previous study, we have shown that trastuzumab, when applied concomitantly with thoracic RT, produced at least additive detrimental effect on thoracic aorta both ultrastructurally and functionally. 20 In this subsequent study, we tried to evaluate whether the severity of injury to heart and thoracic aorta induced by RT and trastuzumab might differ when used either concomitantly or sequentially. However, we used light instead of electron microscopy in this study and found that trastuzumab when added to RT though not have large effect caused consistent increase in subendothelial edema without any significant difference between the RT groups. The functional study again revealed additional relaxation defects when trastuzumab used with RT without any statistically significant difference between the groups. Although the toxic effect of trastuzumab on vascular structures has not been identified clearly yet, trastuzumab-induced vascular injury has been shown in a 51-year-old East Indian woman who developed painless prominence of the veins in her left arm and hand 3 days after her first trastuzumab injection. 36 The biopsy revealed thickened and dilated veins with considerable loss of smooth muscle fibers in the media and replacement with abundant Alcian-blue-positive ground substance and myofibroblasts. Endothelial-derived neuregulin, which is a key ligand for erbB receptors, has a protective paracrine effect on vascular smooth muscle cells as well as cardiac cells in the setting of an injury. In other words, since endothelial cells contain erbB receptors, they are targets for autocrine signaling via this pathway, an important mediator of vascular preservation. 37 The light microscopic appearance of subendothelial edema by trastuzumab in our study strengthens the clues that trastuzumab may have detrimental effect not only on cardiomyocytes but also on the vascular structures. In addition, the detrimental effect of RT on thoracic aorta in our study was so pronounced, and this may preclude the additional or supra-additional detrimental effect of trastuzumab when used either concomitantly or sequentially. On the other hand, we used light microscopy instead of electron microscopy, which was a limitation of our study. We think that electron microscopy may further enlighten the possible interaction of RT and trastuzumab in this regard.
There are limited published data in the literature with respect to the use of trastuzumab with concomitant RT particularly in patients with internal mammary chain radiation treatment (IMC RT) indication. Since trastuzumab is generally administered postsurgically for 12 months, patients receiving breast RT generally take it concurrently. Preclinical data suggest a radiosensitizing effect of trastuzumab on breast cancer cells; however, whether it causes radiosensitization of normal cells or not is unknown. 28 In a retrospective study by Shaffer et al., it was found that there was no deleterious effect of trastuzumab on acute cardiac toxicity when used with IMC RT. 38 However, only data of 59 patients were included in this study in which only 13 of them were applied IMC RT and the mean follow-up time was only 15 months. It is well known that at least 3–20 years median follow-up time is needed to detect clinical radiation-induced cardiovascular toxicity in breast cancer patients. 3 In another study, Halyard et al. assessed whether concomitant use of trastuzumab and RT increases adverse events after breast-conserving surgery or mastectomy in North Central Cancer Treatment Group (NCCTG) phase III trial N9831. 17 Their results suggested that concomitant trastuzumab with RT was not associated with increased acute adverse events including cardiac toxicity. However, the patients with IMC RT were excluded in this particular study because of questionable outcome as well as potential cardiac toxicity. In our study, we demonstrated that RT groups have higher CF scores when compared with non-RT groups. Trastuzumab, even when used alone, started to induce CF from the beginning of 21st day. By 70th day, the CF score of the concomitant trastuzumab and RT group was significantly higher than RT only group. These findings suggest that trastuzumab may have additive effect on radiation-induced cardiac toxicity, and it seems that concomitant use may cause worse damage compared to sequential use.
In a prospective single-center study, Jacob et al. assessed the outcomes and the toxicities related to the concomitant trastuzumab and RT in localized breast cancer patients. 39 The authors measured left ventricular ejection fraction by echocardiography or myocardial scintigraphy, and it was considered as impaired when below 55%. In this study, IMC RT was performed in 73.7% patients. At the end of the RT, 8.4% of patients had impaired left ventricular ejection fraction. Trastuzumab had to be discontinued due to impaired left ventricular ejection fraction in 5.8% patients. In univariate analyses, neither the treated breast side nor IMC RT caused left ventricular ejection fraction alteration. The authors concluded that the toxicities of concomitant trastuzumab with RT were acceptable. In the current study, we demonstrated that the ejection fraction, left ventricular mass, and fractional shortening were significantly decreased in the RT and trastuzumab groups when compared to the control groups. Trastuzumab when added to RT produced, though not statistically significant, a small additional detrimental effect. This deterioration was independent from either concomitant or sequential use of trastuzumab.
Our study has some limitations that should be mentioned as well. We performed functional analyses only in the 3rd week based on the findings of our previous study in which we have found that thoracic aorta relaxation responses were deteriorated by 3rd week of RT. 20 Again in the current study, we demonstrated a deterioration of Ach-mediated relaxation responses in thoracic aorta segments by 21st day in RT groups. Trastuzumab used either concomitantly or sequentially produced small but consistent increase in the relaxation damage of aortic samples, a finding that supports the histopathologic evaluation as well. Again, it seemed that both therapeutic agents caused cardiac damage, which can be either synergistic or additional. This study is an experimental study; therefore, our results should be clarified with clinical studies.
In conclusion, our results suggested that trastuzumab, when used with RT may deteriorate radiation-induced cardiovascular toxicity. The effect of trastuzumab on cardiovascular structure was independent of the timing, either concomitant or sequential. Further phase III clinical studies are warranted.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship and/or publication of this article: This work was supported by Gulhane Military Medical Academy (AR. 2010/73). There is no role of study sponsors in the study design, in the collection, analysis and interpretation of data, in the writing of the manuscript and in the decision to submit the manuscript for publication.
