Abstract
The objectives of these studies were to determine the cardiopulmonary effects of a single intramuscular administration of 1,1′-methylenebis[4-[(hydroxyimino)methyl]-pyridinium] dimethanesulfonate (MMB4 DMS) on dogs and on the central nervous system in rats. On days 1, 8, 15, and 22, male and female dogs received either vehicle (water for injection/0.5% benzyl alcohol/methane sulfonic acid) or MMB4 DMS (20, 50, or 100 mg/kg). Pulmonary function was evaluated for the first 5 hours after concurrent dosing with cardiovascular monitoring; then cardiovascular monitoring continued for 72 hours after dosing. Rats were dosed once by intramuscular injection with vehicle (water for injection/0.5% benzyl alcohol/methane sulfonic acid) or MMB4 DMS (60, 170, or 340 mg/kg). In dogs, 100 mg/kg MMB4 DMS resulted in increased blood pressure, slightly increased heart rate, slightly prolonged corrected QT, and moderately increased respiratory rate. There were no toxicological effects of MMB4 DMS on neurobehavioral function in rats administered up to 340 mg/kg MMB4 DMS.
Introduction
There is potential for exposure to organophosphorus (OP) nerve agents either on the battlefield or through terrorist attack on civilian populations. 1 The toxicity of OP nerve agents is due to the irreversible binding of the nerve agents to acetylcholinesterase (AChE), an enzyme that is responsible for terminating the transmitter action of acetylcholine (ACh) at cholinergic nerve endings and their effector organs or postsynaptic sites. The buildup of ACh at cholinergic receptor sites is equivalent to continuous stimulation of these sites by the central nervous system (CNS) or peripheral nervous systems. 2 The current standard treatment for nerve agent poisoning is administration of atropine sulfate, an anticholinergic drug that antagonizes the effects of excess ACh at muscarinic receptor sites, and an oxime, 2-pralidoxime (2-PAM), which reactivates any unaged inhibited enzyme. 1 In addition, an anticonvulsant is administered. This approach provides adequate protection against some nerve agents such as sarin and VX and is less effective against other nerve agents such as soman and Russian V-agent. 3,4 Following the reaction with nerve agents such as soman with AChE, the complex undergoes a process called “aging” that makes reactivation by standard treatment oximes, no longer possible. Consequently, 2-PAM has less than optimal efficacy against soman, 5 and a potential replacement, by developing improved oxime reactivators, has been sought after for many years. 1
One such improved oxime is 1,1′-methylenebis[4-[(hydroxyimino)methyl]-pyridinium] dimethanesulfonate (MMB4 DMS), which is being considered as a replacement for 2-PAM. The MMB4 DMS (common name) is a member of the group of bis-quaternary pyridinium aldoximes that has the potential to reactivate AChE following nerve agent poisoning. Laboratory studies indicate that MMB4 DMS may be a better reactivator of AChE than 2-PAM, especially for certain highly toxic nerve agents such as unaged soman. 5
The objectives of the present studies were to determine the cardiovascular and pulmonary effects of intramuscular administration of MMB4 DMS in the beagle dog and the CNS/neurobehavioral activity following an intramuscular injection of MMB4 DMS in Sprague-Dawley rats.
Materials and Methods
These studies were conducted according to Good Laboratory Practices in compliance with the Food and Drug Administration after approval of Battelle’s Institutional Animal Care and Use Committee, in compliance with US Department of Agriculture regulations, the sponsor’s Animal Care and Use Review Office, and The American College of Toxicology Policy on the Use of Animals.
Test Article
The test article for this study was MMB4 DMS and is described as an off-white to tan crystalline powder based on the Certificate of Analysis. Lot no. 1004 MMB4 DMS, with a manufacture date of July 2007. The source of the test article was Cambrex Charles City, Inc (Charles City, Iowa). The purity of the test article was >98%. Upon receipt, the test article was stored at room temperature with desiccant.
Animals and Study Designs
The dog study consisted of a 4 × 4 Latin-square crossover design with a total of 8 (4 male/4 female) beagle dogs (7-14 kg), aged 17 to 20 months old, instrumented with a Data Sciences International (St Paul, Minnesota) telemetry transmitter (D70-PCT or D70-PCTP), capable of collecting electrocardiogram (ECG), systemic arterial pressure, and core body temperature. The rat study consisted of a functional observational battery (FOB) performed on a total of 40 (20 males and 20 females) Sprague-Dawley rats (220-371 g) aged 10 to 11 weeks old.
Cardiovascular/respiratory study
Male and female dogs were appropriately conditioned to a restraint sling (including air dam and head dome) starting 18 days prior to dosing. Animals were progressively acclimated starting with 15 minutes of restraint on day 1 up to 6 hours by day 14. On dosing days 1, 8, 15, and 22, pulmonary function evaluation was performed concurrent with cardiovascular telemetric monitoring. Each dog was placed in a restraint sling and fitted with an air dam and a head dome. Pulmonary function was performed via head dome plethysmography. A controlled flow of fresh breathing air was supplied to the head dome. The head dome was connected via appropriate instrumentation to a Buxco BioSystem XA Data Acquisition System (version 2.5.2; Buxco Electronics, Wilmington, North Carolina). The respiratory parameters evaluated included respiratory rate, tidal volume, and minute volume. On each day of monitoring, dogs were allowed to acclimate to the sling and head dome apparatus for at least 30 minutes to allow for consistent baseline data followed by collection of 30 minutes of baseline data. The dogs then received an intramuscular administration of either vehicle (water for injection, 0.5% benzyl alcohol, and methane sulfonic acid) or MMB4 DMS at 20, 50, or 100 mg/kg. Dogs were monitored for, nominally, 5 hours following dosing while in slings. At the end of the in-sling monitoring period, the dogs were returned to their home cages, where cardiovascular telemetry data collection continued for 72 hours following dosing.
Clinical observations were performed periodically for abnormal indications, prior to and after dosing both while in restraint slings and home cages.
Neurobehavioral study
On each dosing day, rats were removed from the home cage, dosed once by intramuscular injection at 0.1 mL/injection site in the hind limb(s), and administered either vehicle (water for injection/0.5% benzyl alcohol/methane sulfonic acid) or MMB4 DMS at 60, 170, or 340 mg/kg.
Dosing was ordered so that all rats began FOB testing approximately 15 minutes following dosing. Approximately equal numbers of animals from each treatment group were dosed on each day of testing. Each animal was dosed only once. The FOB measurements were first carried out in the home cage. The observer, blinded to the dose level, recorded each rat’s posture, tremors, convulsive activity, lethargy/arousal, and eyelid closure. The observer rated ease of removal, handling, and hand-to-hand transfer. The observer then performed a detailed observation, checking general condition/appearance, salivation, piloerection, fur appearance, vocalization, lacrimation, palpebral reflex responsivity, pupillary diameter test, and pupil response. The rodent was then transferred to a technician (blinded to the dose level administered) for performance of the open-field portion of the FOB conducted in a room with a white noise generator. The rat was placed in an open-field arena observation chamber. During a 2-minute period, animal locomotion was measured by counting the number of squares that both forepaws of the rodent entered. Other notable events recorded included rearing (supported and unsupported, not differentiated), posture, tremors, convulsive activity, arousal, and gait. At the end of 2 minutes, the number of fecal boluses and urine pools on the flooring were recorded. Stereotypy, bizarre behavior, and vocalizations were also noted. Reflex testing then followed, consisting of recording each rodent’s responses to the approach of a blunt object, a touch of a blunt object to the rump (touch response), a startle response, tail pinch, paw placement on a grid, grip strength, and righting reflex (air and surface). Finally, rectal temperature was taken, and the animal was returned to its home cage. All animals were single housed, offered food and water ad libitum (except when removed from the cage during protocol-driven activities) in a room in which temperature and humidity was monitored, and observations were performed twice daily to detect any clinical abnormalities.
Data Analysis Methods
Cardiovascular/respiratory study
ECG intervals (PR, QRS, QT, and RR) were measured and averaged from at least 2 hours prior to each dosing event through 24 hours following each dosing event. Nominally, 100 beats were analyzed from each 5-minute increment. The QT interval was normalized for changes in heart rate (RR interval) by conversion to the corrected QT (QTc) interval using Fridericia formula QTc = QT/(RR)1/3 as described in the work of Hanson et al. 6
Group means and the standard error of each mean were presented for all quantitative data, including blood pressures, heart rate, ECG, body temperature, respiratory rate, tidal volume, and minute volume. Unless otherwise specified, all statistical tests were performed at the .05 significance level. All parameters were analyzed using a Latin-square design analysis of variance (ANOVA) model as described by Neter et al. 7 The ANOVA was used as an overall test to evaluate whether there was any difference among the treatment groups at specified time points following dosing. Pairwise post hoc comparisons between the treatments and the vehicle were done at a given time point only if the overall ANOVA test was significant at that time point. A Bonferroni correction 8 was used to adjust for multiple comparisons for the pairwise comparisons.
Neurobehavioral study
For categorical variables, a mean response model was used to predict the likelihood of observing a particular pattern of responses among the possible response categories, as a function of dose group. This model was fitted to categorical data using the CATMOD procedure in the statistical analysis system (SAS). 9,10 A chi-square test was used to test the significance of the dose group effect. When this effect was significant at a .05 level, additional chi-square tests were performed within the model fit to compare each of the 3 dose levels of MMB4 DMS to vehicle. Each comparison was done at a .05/3 = 0.0167 significance level, to control the overall error rate across all the 3 comparisons to not more than .05. 8
For variables classified as having continuous responses, a 1-way ANOVA model 7 was fitted to the observed responses using the MIXED procedure in the SAS. 10 The model included dose group as a fixed effect and assumed that the residual error (ie, difference in observed and model-predicted values) followed a normal distribution with zero mean. An F test was used to test the significance of the dose group effect. When this effect was significant at a .05 level, additional t tests were performed within the model fit to compare each of the 3 dose levels of MMB4 DMS to vehicle. Each comparison was done at a .05/3 = 0.0167 significance level, to control the overall error rate across all the 3 comparisons to not more than .05. 8
Results
Due to the large number of parameters in this study, only selected data figures are included to represent the results.
Cardiovascular/Respiratory Study
The MMB4 was associated with an increase in blood pressure (systolic, diastolic, and mean) in both the sexes in a dose-related manner (mean arterial blood pressure maximal increase of 25.6 ± 3.5 mm Hg for mid dose males and 26.3 ± 1.2 mm Hg for high dose females approximately 3 hours after dosing), and of statistical significance through approximately 5 hours postdose initiation (Figure 1A and B). Female dogs appeared to be more sensitive to MMB4 blood pressure alterations, exhibiting significant increases at all dose levels, whereas infrequent statistical significances occurred in the low-dose male dogs. Blood pressures appeared to return to vehicle levels by 16 hours after dosing for both males and females.
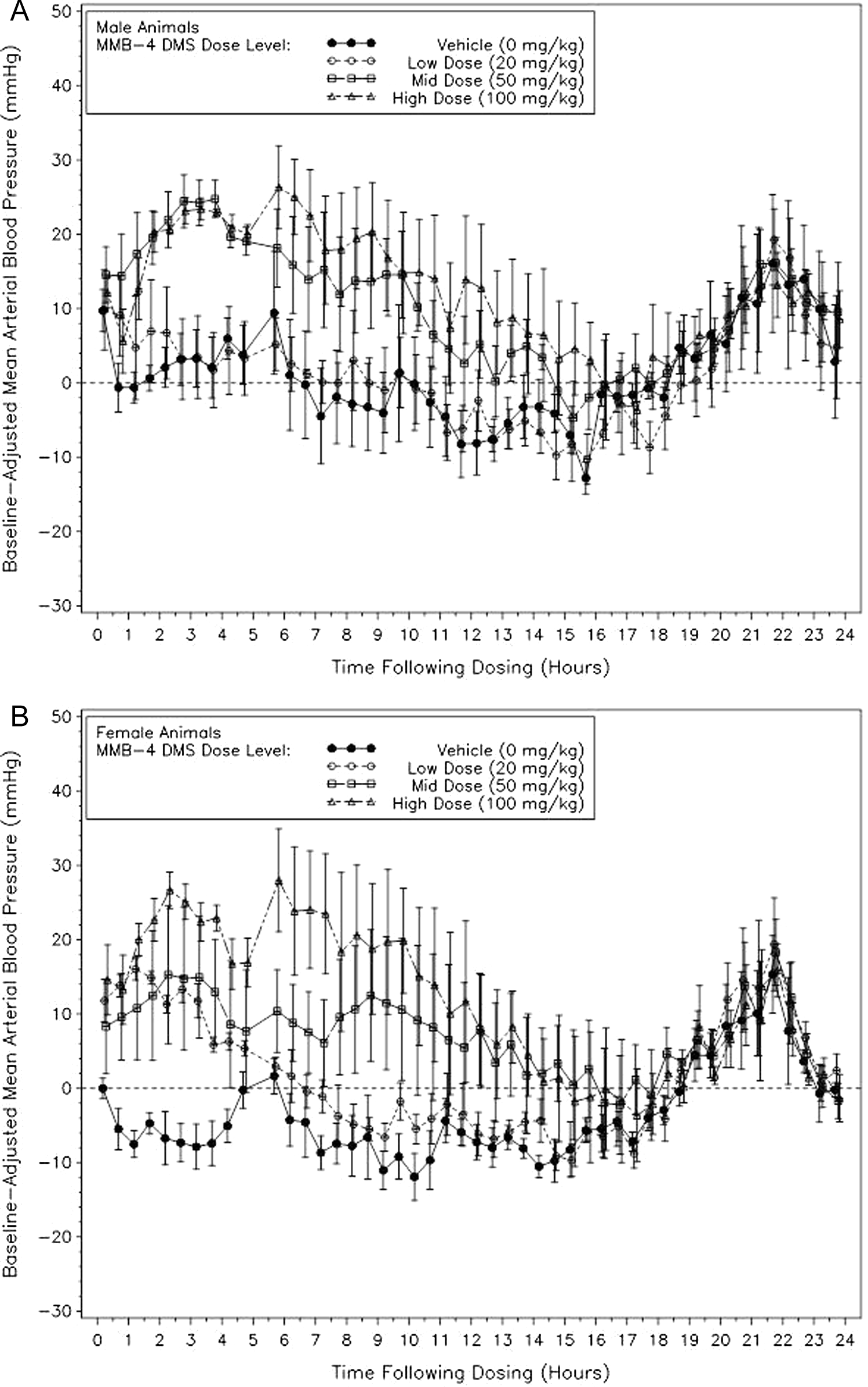
Mean arterial pressures for both the male and the female dogs demonstrated increases in blood pressure with increasing concentrations of MMB4 DMS. However, the females appeared more sensitive to MMB4 DMS, exhibiting significantly increased pressures at all the dose levels, whereas only infrequent statistically significant increases in low magnitude were noted for the low-dose males. Mean arterial blood pressure (mm Hg), dose level means (with ±standard error bars) of baseline-adjusted hourly averages following dosing with MMB4 DMS or vehicle (time 0 represents dosing and the horizontal reference line represents baseline) for 24 hours postdose. Single doses of 0, 20, 50, and 100 mg/kg MMB4 DMS were administered within a Latin-square design on days 1, 8, 15, and 22. Note: N = 4 in all but in the following cases: N = 3 for vehicle dose level at 2 hours following dosing. MMB4 DMS indicates 1,1′-methylenebis[4-[(hydroxyimino)methyl]-pyridinium] dimethanesulfonate.
Heart rates were increased more for females (44 bpm) than the males (17 bpm) at 160 to 170 minutes after dosing with 100 mg/kg of MMB4 (Figure 2A and B). Consistent with the sex differences noted for blood pressure, females appear to be more sensitive to MMB4-related alterations in heart rate with sporadic statistically significant increases noted after dosing at all the dose levels of MMB4, whereas no statistically significant increases were noted at 20 mg/kg for males. Figure 3 is a 72-hour plot of heart rate for female animals demonstrating the return of heart rate to baseline values by 15 hours after dosing, with subsequent rate increases associated with husbandry and feeding activities.
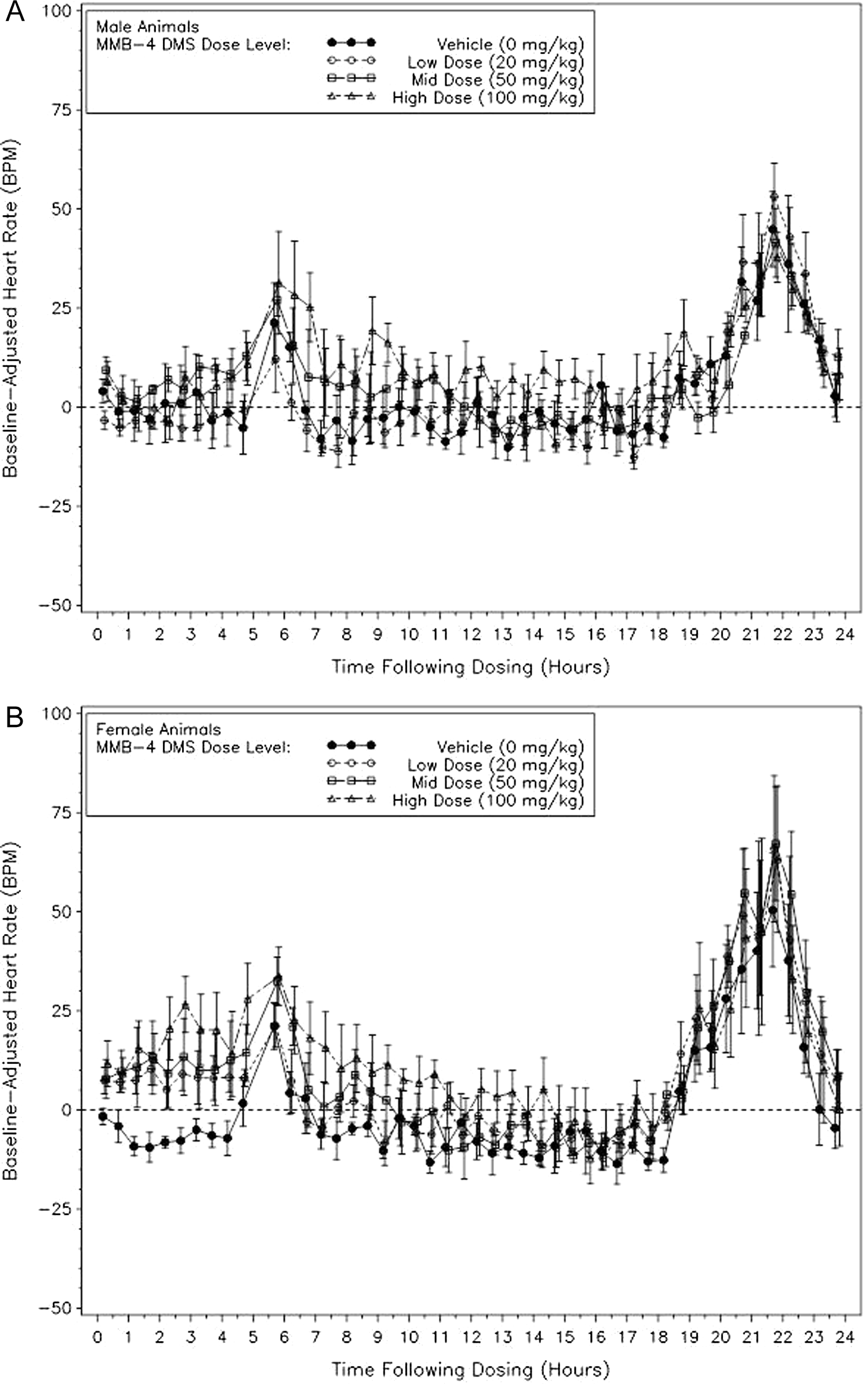
Heart rate (bpm), dose level means (with ±standard error bars) of baseline-adjusted 120-minute averages following dosing with MMB4 DMS or vehicle (time 0 represents dosing). A plot of averaged heart rates for all the doses (0, 20, 50, and 100 mg/kg MMB4-DMS) administered in a Latin-square design with dosing occurring on days 1, 8, 15, and 22, through 24 hours after dosing. Heart rate was significantly increased with all the doses for the female animals from immediately postdose through 5 hours, whereas heart rates for male animals were increased significantly for the mid and high doses of MMB4-DMS. MMB4 DMS indicates 1,1′-methylenebis[4-[(hydroxyimino)methyl]-pyridinium] dimethanesulfonate.
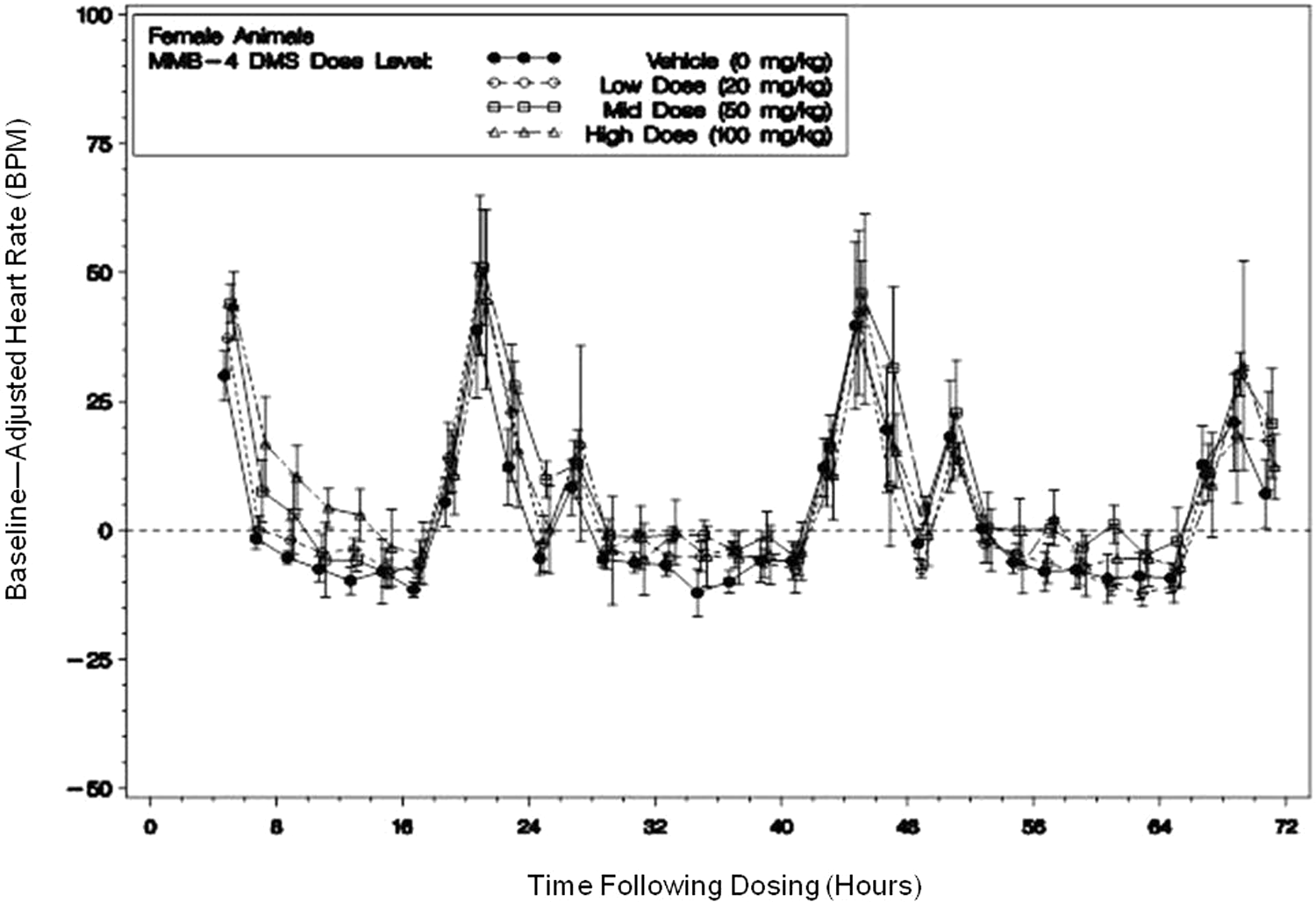
A representative plot of heart rate (bpm), for female animals. In-cage dose level means (with ±standard error bars) of baseline-adjusted 2-hour averages following dosing with 1,1′-methylenebis[4-[(hydroxyimino)methyl]-pyridinium] dimethanesulfonate (MMB4 DMS) or vehicle (time 0 represents dosing) for 72 hours after dosing. This plot shows the return of heart rate to baseline values approximately 15 hours after dosing. Peaks at 22, 46, and 70 hours were associated with lights on for each day and feeding; the smaller second increase was associated with afternoon clinical observation time frame.
Minor temperature increases persisted for both the 50 and the 100 mg/kg levels for both the sexes through, nominally, 5 to 12 hours. Temperature changes were 0.4°C or less. These apparent temperature increases were not of statistical significance.
The PR intervals were shortened in males for both 50 and 100 mg/kg dose groups and shortened for females for all the dose groups. PR shortening was apparent from 1 through 17 hours and was statistically significant from 2 to 15 hours in males (Figure 4) and from 1 to 5 hours in females. Maximum PR shortening was 17 ms for males and 14 ms for females. PR shortenings were, in part, related to decreases in RR interval (increases in heart rate).
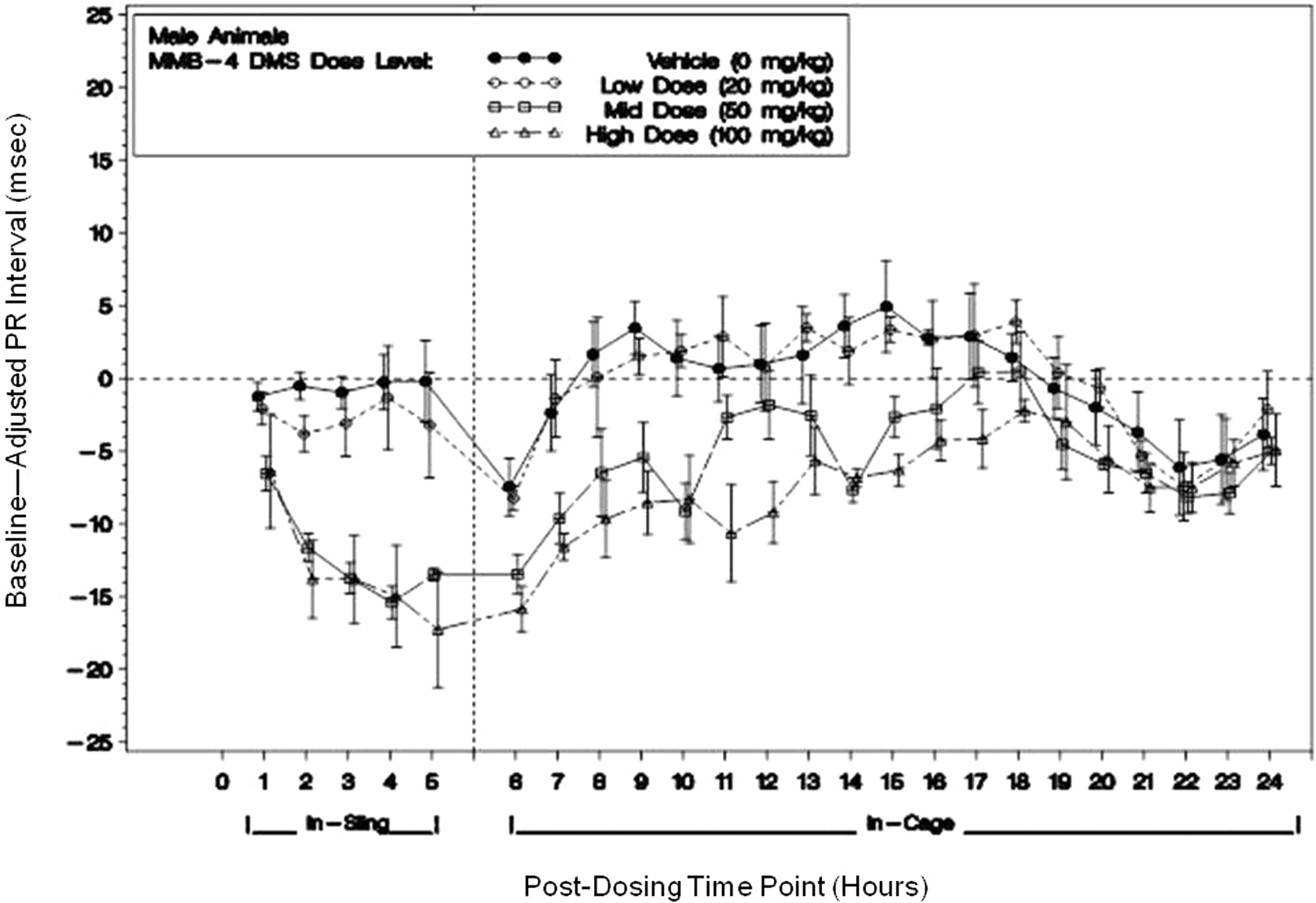
A representative figure of PR interval (ms) for this study with male animals to include both in-sling and in-cage dose level means (with ±standard error bars) of baseline-adjusted averages following dosing with MMB4 DMS or vehicle. (Horizontal reference line represents baseline; vertical reference line indicates transfer from sling to home cage.) Single doses of 0, 20, 50, and 100 mg/kg MMB4 DMS were administered within a Latin-square design on days 1, 8, 15, and 22. These PR reductions are consistent with the observed heart rate increases. MMB4 DMS indicates 1,1′-methylenebis[4-[(hydroxyimino)methyl]-pyridinium] dimethanesulfonate.
QRS durations were not remarkably altered for either sex during the 24-hour postdosing evaluation period.
QT interval in both the males and the females shortened when compared to vehicle for all the dose groups through 5 hours. QT values subsequently lengthened approaching vehicle values between 10 and 17 hours postdosing. QT shortening was periodically statistically significant in all the dose groups for both sexes during the period from 2 to 5 hours, with maximum shortening values of 26 ms. Although important to report raw QT interval data, the dependency of the QT interval to changes in heart rate results in QTc being more instructive for the purposes of evaluating change in ventricular repolarization.
Heart rate QTc was prolonged in both male and female animals from 2 to 5 hours postdosing, when compared to vehicle, in an apparent dose-related fashion. The QTc prolongation was significant in the 100 mg/kg groups with maximum increases when compared to vehicle of, nominally, 23 ms (10%) for the males and 37 ms (16%) for the females (Figure 5).
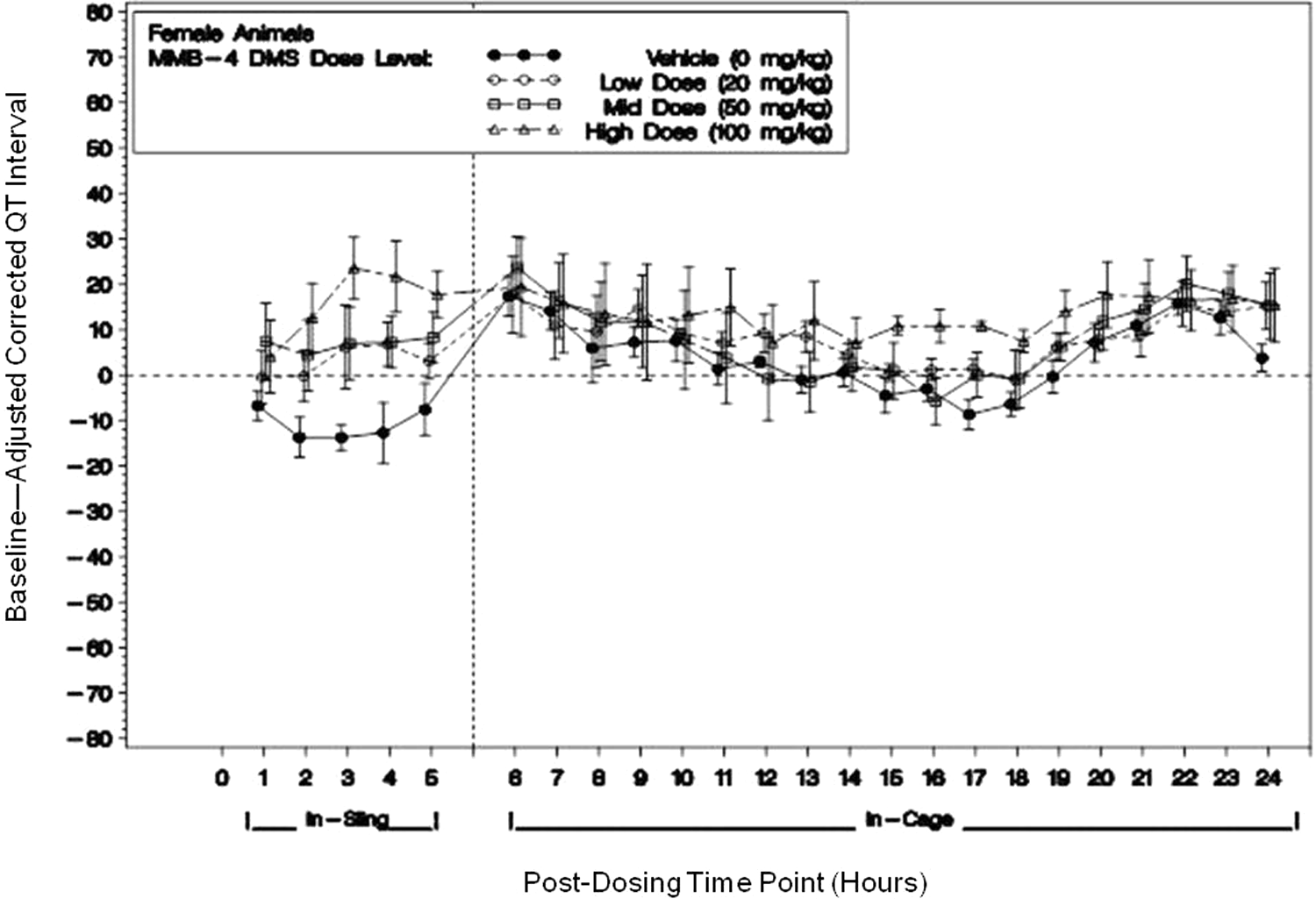
A representative figure of heart rate corrected QTc interval for female animals only during both in-sling and in-cage dose level means (with ±standard error bars) of baseline-adjusted averages following dosing with MMB4 DMS or vehicle. (Horizontal reference line represents baseline; vertical reference line indicates transfer from sling to home cage.) Single doses of 0, 20, 50, and 100 mg/kg MMB4 DMS were administered within a Latin-square design on days 1, 8, 15, and 22. The correction method used did not completely reduce the QTc values to baseline values. MMB4 DMS indicates 1,1′-methylenebis[4-[(hydroxyimino)methyl]-pyridinium] dimethanesulfonate; QTc, corrected QT.
There were no changes in ECG rhythm or morphology attributable to MMB4.
Assessment of the pulmonary data of the male dogs revealed no apparent differences for any of the dose groups with the exception of rate and minute volume increases in the 100 mg/kg group from 2.5 to 3.5 hours postdosing. These changes were statistically significant and were attributed to alterations from 1 animal.
Pulmonary rate and minute volumes for the females were periodically increased with the occurrence and magnitude of the alterations being generally dose related. Rates increased nominally 124%, 150%, and 263% in the 20, 50, and 100 mg/kg dose groups, respectively (Figure 6). In all, 2 of the 4 animals exhibited large increases in respiratory rate in the 20 mg/kg group, and 3 of the 4 animals exhibited large increases in respiratory rate (albeit with larger variation) in both the 50 and the 100 mg/kg groups.
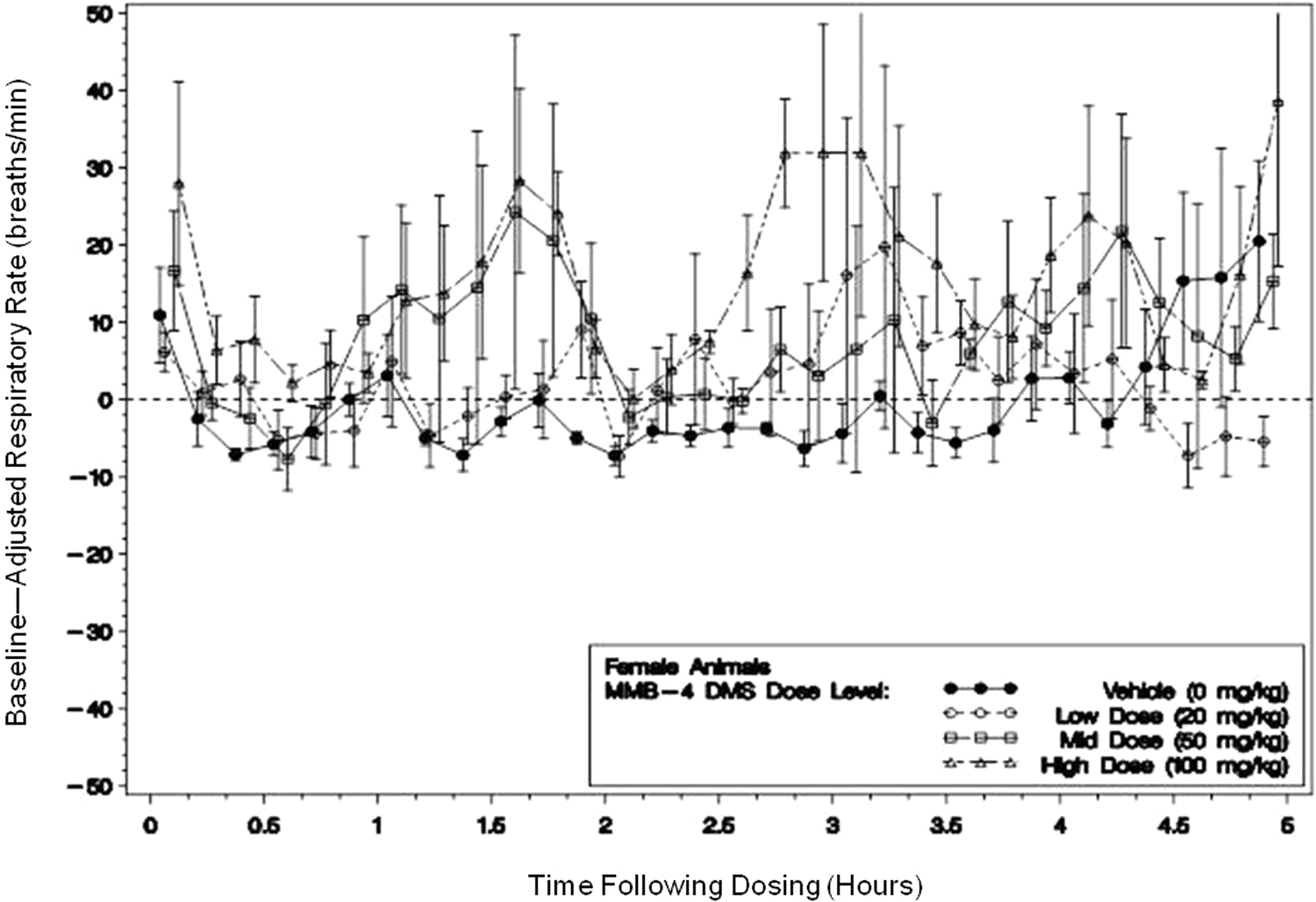
A representative figure of respiratory rate (breaths/min) for female animals during in-sling (5 hours postdose) dose level means (with ± standard error bars) of baseline adjusted 10-minute averages following dosing with MMB4 DMS or vehicle (time 0 represents dosing). Single doses of 0, 20, 50, and 100 mg/kg MMB4-DMS were administered within a Latin-square design on days 1, 8, 15, and 22. The rate increases for all the dose groups exhibited individual animal variability, especially at the 2 higher dose levels. Note: N = 4 in all but in the following cases: N = 3 for the high-dose level at 290 to 300 minutes following dosing. MMB4 DMS indicates 1,1′-methylenebis[4-[(hydroxyimino)methyl]-pyridinium] dimethanesulfonate.
Neurobehavioral Study
Categorical variables (score and yes/no variables)
The results of statistical analyses performed on these variables are summarized in Table 1 (for both males and females). For female rats, there were no significant differences observed. For male rats, there were significant alterations observed for hand-to-hand manipulation and approach response when compared to normal response. The hand-to-hand responses were observed in 5 of 5 animals administered 170 mg/kg dose of MMB-4 DMS (mid dose) and described as decreased resistance to being held compared to the vehicle group. There was an overall statistically significant difference in approach response; however, no significant differences between any of the 3 dose levels of MMB4 DMS and vehicle were observed.
Neurobehavioral Test Result Summary for Categorical Variables (Score and Yes/No Variables), Male, and Female Animals.
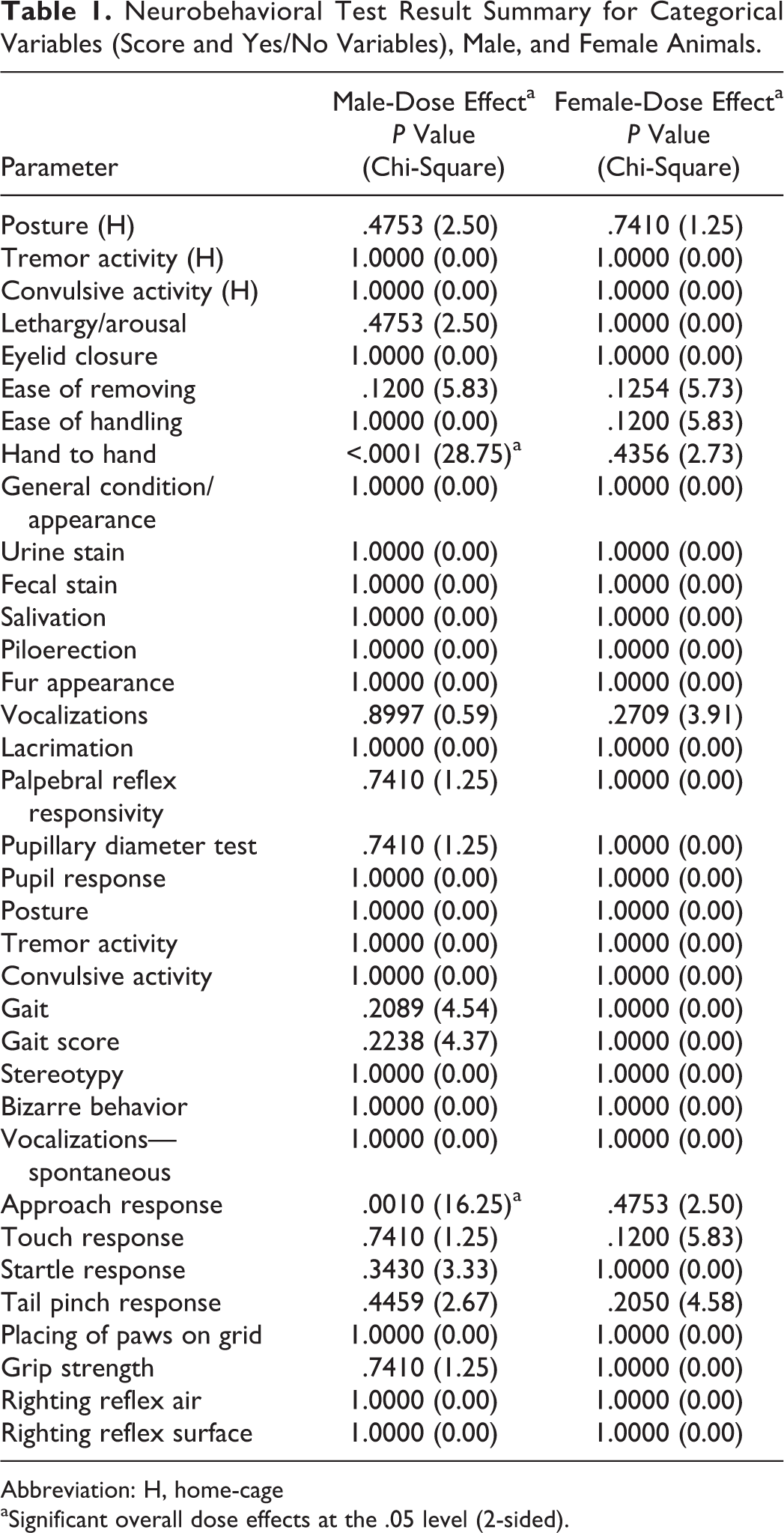
Abbreviation: H, home-cage aSignificant overall dose effects at the .05 level (2-sided).
Continuous variables
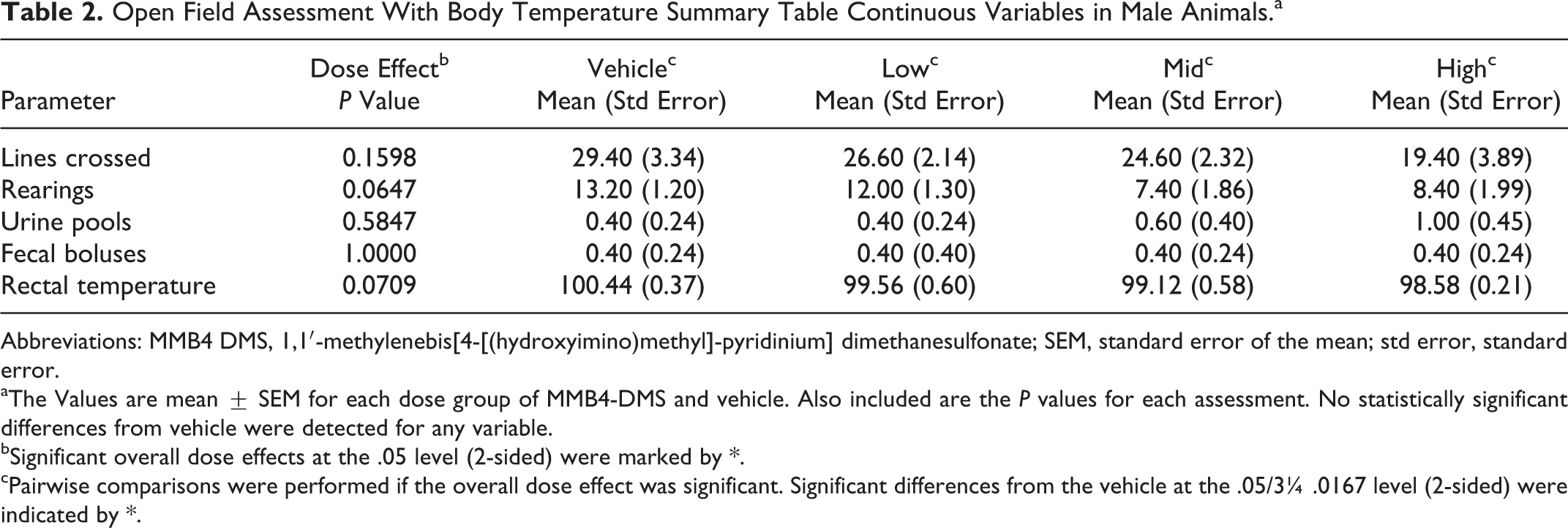
Means and standard errors of the reported measurements for the open-field assessment and body temperature (continuous variables) were calculated for each dose group and are presented in Tables 2 (males) and 3 (females). Based upon the outcome of the 1-way ANOVA applied to these continuous measurements, no significant differences among dose groups were observed in male rats. For the females, statistically significant differences in rectal temperature were observed among the dose groups at the .05 level (Table 3), with the temperatures of animals receiving the 170 and 340 mg/kg dose levels of MMB4 DMS significantly decreased when compared to the vehicle dose.
Open Field Assessment With Body Temperature Summary Table Continuous Variables in Male Animals.a
Abbreviations: MMB4 DMS, 1,1′-methylenebis[4-[(hydroxyimino)methyl]-pyridinium] dimethanesulfonate; SEM, standard error of the mean; std error, standard error.
aThe Values are mean ± SEM for each dose group of MMB4-DMS and vehicle. Also included are the P values for each assessment. No statistically significant differences from vehicle were detected for any variable.
bSignificant overall dose effects at the .05 level (2-sided) were marked by *.
cPairwise comparisons were performed if the overall dose effect was significant. Significant differences from the vehicle at the .05/3¼ .0167 level (2-sided) were indicated by *.
Open Field Assessment With Body Temperature Summary Table Continuous Variables in Female Animals.a
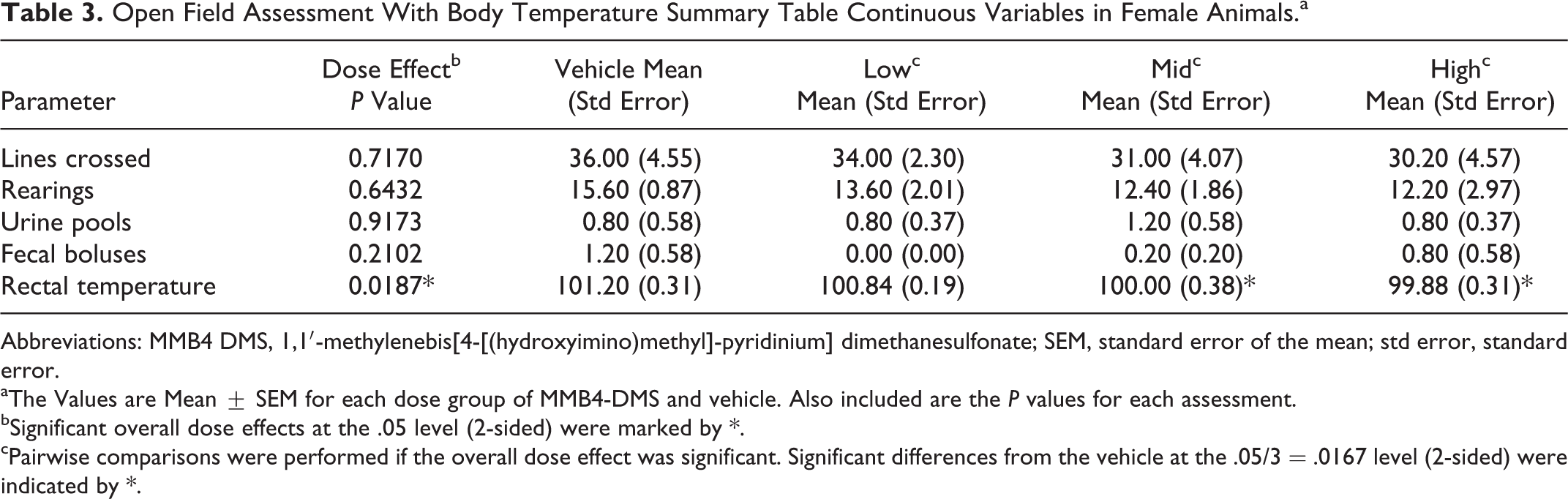
Abbreviations: MMB4 DMS, 1,1′-methylenebis[4-[(hydroxyimino)methyl]-pyridinium] dimethanesulfonate; SEM, standard error of the mean; std error, standard error.
aThe Values are Mean ± SEM for each dose group of MMB4-DMS and vehicle. Also included are the P values for each assessment.
bSignificant overall dose effects at the .05 level (2-sided) were marked by *.
cPairwise comparisons were performed if the overall dose effect was significant. Significant differences from the vehicle at the .05/3 = .0167 level (2-sided) were indicated by *.
Discussion
Toxicity caused by nerve agents can result in respiratory arrest within 2 to 3 minutes and circulatory arrest only a few minutes after, if left untreated. 11 Thus, there is dire need for a more effective AChE reactivator that also causes minimal cardiovascular stress/injury. HI-6 (asoxime) and MMB4 have been used as treatments for OP poisoning in many countries around the world, but 2-PAM is currently the only licensed oxime for the treatment of nerve agent poisoning in the United States. 2,3 Atropine and 2 of the bispyridinium oximes, HI-6 and HLö 7 (1-[[[4-(aminocarbonyl)pyridinio]methoxy]methyl]-2,4-bis [(hydroxyimino)methyl]pyridinium), are in consideration to replace 2-PAM. These compounds were previously evaluated in the guinea pig. Unfortunately, the results suggested that while antidote treatment rapidly restored heart rate and mean arterial pressure, as well as improved the respiratory function, these combinations exhibited the potential to induce lethal arrhythmias. 11
The MMB4 may prove to be a better reactivator candidate than 2-PAM 5 for some nerve agents but scant information with regard to cardiovascular, pulmonary, and CNS effects or safety of this compound was found in the current literature. Because of the lack of data in cardiovascular, pulmonary, and CNS effects of MMB4 DMS, in the present study, we evaluated the cardiopulmonary and CNS responses to an intramuscular injection of MMB4 DMS with a goal of extrapolating the data to find a better treatment of nerve agent poisoning in humans.
Cardiovascular/Pulmonary Effects
This study contains data that were collected while the animals were periodically restrained for pulmonary function evaluation as well as when in their home cages unrestrained. The comparisons used in this study were made to the appropriate values, for example, in-sling test article data compared to in-sling vehicle data. However, the anticipated differences between in-cage and in-sling data can have an effect upon the values obtained while restrained as compared to data collected while in cages and thus the interpretation of the data. To minimize such effects, the animals utilized in this study were selected and progressively trained over a period of 18 days to accept both the sling restraint and the head dome apparatus used for collecting high-quality pulmonary function data. Following this training paradigm, the differences between the in-sling, and in-cage baseline values were compared to assess the effect of restraint upon the data being collected. For the present study, the differences between in-sling and in-cage mean arterial pressures were 5.9 and 5.1 mm Hg for males and females, respectively. The differences between in-sling and in-cage for heart rates were −3.0 and −5.3 bpm for males and females, respectively. It is unlikely that these relatively small differences between the in-cage and in-sling restrained blood pressure and heart rate data would markedly affect the interpretation of the hemodynamic data.
The present study indicated dose-related increases in blood pressure and heart rate for both the sexes, generally of greater magnitude and statistically significant for females. Pressures and rates were higher, as compared to vehicle, beginning shortly after dosing, reaching maximum values from 5 to 16 hours (male high dose) after dosing and from 10 minutes to 16 hours (female high dose) after dosing.
The MMB4 DMS-related increases in blood pressure and heart rate were observed in both the sexes at all the dose levels but were more significant and more prolonged with increased MMB4 DMS concentration especially in the females, who appeared to be more sensitive to these test article-related blood pressure alterations.
Worek et al demonstrated that guinea pigs developed reduced heart rate (sinus bradycardia), complete atrioventricular block, followed by indioventricular rhythm after nerve agent poisoning (tabun, sarin, soman, or VX), and subsequent antidote treatment (atropine or atropine and Hi 6 or HLö 7). 11 The authors noted that no ventricular tachyarrhythmias were detected prior to death with nerve agent alone. Atropine or atropine plus oxime restored sinus rhythm in guinea pigs with respiratory function greater than 50% of baseline; however, guinea pigs with respiratory function below 50% of baseline, in some cases, produced ventricular tachyarrhythmias within 1 minute of treatment. The lethal arrhythmias induced by Worek et al 11 can therefore be attributed to the atropine-containing antidote combinations used to treat nerve agent intoxication. In the present study, there were no changes in ECG rhythm or morphology attributable to MMB4 DMS or reductions in respiratory function.
The QTc prolongation indicates a delay in ventricular repolarization of the myocardium. The QTc prolongation in both the sexes appears to be MMB4 DMS-related, with females being more sensitive (effects occurring at a lower dose) and exhibiting greater prolongation than their male counterparts at any given dose level compared to the vehicle group. It appears that MMB4 DMS has an effect on the 3 evaluated parameters of pulmonary data, that is, respiratory rate, tidal volume, and minute volume, with greater effects observed in female dogs. All 3 parameters were modestly increased in the male test subjects when compared to the vehicle in large part because of a single male outlier (no statistically significant differences). However, it is clear that the females did respond adversely to the drug, with 3 of the 4 females exhibiting dramatic increase in respiratory rate in the mid- and high-dose groups. Although no significant alterations to tidal volume were apparent in the female test subjects, an evident increase in the high-dose group minute volume does appear to be MMB4 related. Although the potential for MMB4 DMS to affect the pulmonary parameters of only a single male animal out of 4 was considered low and the respiratory parameters for males did not respond similarly to the females, it appears that MMB4 DMS affected the respiratory system of the dogs in this study.
Neurobehavioral Effects
The only toxicologically significant change observed was decreased rectal temperature reported in female rats treated with 170 or 340 mg/kg doses of MMB4 DMS, as compared to controls. Although it was determined that MMB4 DMS slightly increased body temperature in dogs, the higher doses of MMB4 DMS in the rats appeared to lower body temperature. Other than species differences to regulate body temperature, the authors are unsure of the potential mechanism of the body temperature discrepancies observed in response to MMB4-DMS in these studies.
Conclusion
The MMB4 DMS administered at doses of 20, 50, and 100 mg/kg by intramuscular injection to male and female dogs produced moderate increases in blood pressure and heart rate, slight reductions in PR interval, slight prolongations of the QTc interval, and moderate increases in respiratory function (respiratory rate). These aberrations were most noticeable at the 100 mg/kg concentration. There were no alterations in ECG rhythm or morphology associated with MMB4 DMS. The MMB4 DMS administered at 60, 170, and 340 mg/kg to both male and female rats induced no acute neurobehavioral effects.
Footnotes
Acknowledgments
The authors wish to thank Dr. Vincent Brown for the editorial assistance.
Authors’ Note
The opinions and assertions contained herein are the private views of the authors and are not to be construed as official or reflecting true views of the Department of the Army or the Department of Defense.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was funded by the United States Army; Chemical Biological Medical Systems (CBMS)/Medical Identification and Treatment Systems (MITS) under contract SP0700-00-D-D3180, Delivery Order 0599 (Task 771), and Delivery Order 0600 (Task 789).
