Abstract
Objective:
This study was conducted to assess the ability of the sequential organ failure assessment (SOFA) and acute physiology and chronic health evaluation (APACHE) II scoring systems, as well as the simplified acute physiology score (SAPS) II method to predict group mortality in intensive care unit (ICU) patients who were poisoned with paraquat. This will assist physicians with risk stratification.
Material and methods:
The medical records of 244 paraquat-poisoned patients admitted to the ICU from January 2010 to April 2015 were examined retrospectively. The SOFA, APACHE II, and SAPS II scores were calculated based on initial laboratory data in the emergency department and during the first 24 h of ICU admission. The probability of death was calculated for each patient based on the SOFA score, APACHE II score, and SAPS II. The ability of the SOFA score, APACHE II score, and SAPS II method to predict group mortality was assessed using a receiver operating characteristic (ROC) curve and calibration analyses.
Results:
A total of 219 patients (mean age, 63 years) were enrolled. Sensitivities, specificities, and accuracies were 58.5%, 86.1%, and 64.0% for the SOFA, respectively; 75.1%, 86.1%, and 77.6% for the APACHE II scoring systems, respectively; and 76.1%, 79.1%, and 76.7% for the SAPS II, respectively. The areas under the curve in the ROC curve analysis for the SOFA score, APACHE II scoring system, and SAPS II were 0.716, 0.850, and 0.835, respectively.
Conclusion:
The SOFA, APACHE II, and SAPS II had different capabilities to discriminate and estimate early in-hospital mortality of paraquat-poisoned patients. Our results show that although the SOFA and SAPS II are easier and more quickly calculated than APACHE II, the APACHE II is superior for predicting mortality. We recommend use of the APACHE II for outcome predictions and risk stratification in paraquat-poisoned patients in the ICU.
Introduction
Paraquat (
Numerous treatment strategies for managing PQ poisoning have been proposed, including extracorporeal elimination, immunosuppressants, and antioxidants, but mortality rate remains high even in centers that employ these treatments. 8 –10
A number of laboratory tests and markers have been proposed to assist in risk stratification of PQ-intoxicated patients. 11,12 Plasma PQ concentration is the most reliable predictor of death. 13 Unfortunately, PQ assays are not widely available, particularly in the developing world. Another important predictor of death is the amount of PQ consumed. 14 However, estimates on the amount ingested are often unobtainable or unreliable in many intoxicated patients. In addition, vomiting after ingestion and gastrointestinal absorption of PQ by food can reduce the actual amount of PQ absorbed. 9
Ingesting PQ remains a widely employed means of suicide with a high mortality rate, despite developments in intensive care. 1 Although new advances in treating PQ-poisoned patients in the intensive care unit (ICU) have resulted in increased survival, such measures prolong ICU stays and increase hospital expenses. Informing patients of the severity of the illness at the time of ICU admission helps determine whether expensive treatment can be continued and avoids unnecessary procedures.
Use of scoring systems specifically developed to assess patients at the time of ICU admission has reduced many problems and facilitated treatment planning. The sequential organ failure assessment (SOFA) and acute physiology and chronic health evaluation II (APACHE II) scoring systems, as well as the simplified acute physiology score II (SAPS II) are three tools widely used by most ICUs to predict clinical outcomes. These scoring systems have been evaluated and validated in many centers and can be adjusted according to need. 15 –18
The purpose of the this study was to evaluate the performance of the SOFA, APACHE II, and SAPS II scoring systems for predicting illness severity and mortality of patients poisoned with PQ who were admitted to the ICU.
Materials and methods
Study design
This is a retrospective cross-sectional study conducted from January 2010 to April 2015. 19
Study setting
This study is conducted at a regional emergency center affiliated with an academic university hospital in Changwon, Republic of Korea.
Data collection
The medical records of PQ-poisoned patients admitted to the emergency center were examined carefully. Acute PQ poisoning was defined based on history of exposure, characteristic clinical features, and a urine PQ test. Patients with any of the following conditions were excluded: (1) uncertain exposure history; (2) combined drug exposure; (3) organophosphorus exposure for >24 h prior to presentation; (4) discharge against medical advice and transfer to another hospital; (5) pre-hospital cardiac arrest; (6) <15 years of age; or (7) missing data.
Standardized medical emergency procedures were followed to assess PQ intoxication. Gastric lavage was performed on all subjects observed within 2 h after ingestion. About 100 g of Fuller’s earth in 200 mL of 20% mannitol was administered if intoxication had occurred within 12 h. Hemoperfusion was recommended after confirmed PQ ingestion by a urine test in all patients. All of these procedures were conducted after acquiring permission and informed consent from the patients or their next of kin. The urinary PQ level was measured semi-quantitatively using the dithionite method. 20
Medical records were examined carefully, and two investigators collected the following parameters: age, sex, systolic and mean arterial blood pressure (MAP) (mmHg), heart rate, respiratory rate, body temperature, initial Glasgow coma score (GCS), arterial blood gas analysis (pH, PaO2, and PaCO2), FiO2, laboratory data (white blood cell count (WBC), hematocrit, platelet count, and sodium, potassium, creatinine, albumin, glucose, bilirubin, and C-reactive protein levels), use of mechanical ventilation, seizure, pneumonia, mortality, SOFA score, APACHE II score, and SAPS II. The investigators who collected the data were blinded to the study objectives. They collected the same data and compared it for accuracy.
The outcome parameter was 60-day mortality. The SOFA score, APACHE II score, and SAPS II were calculated as described previously. 16,18,21 Based on the logistic regression predictive model for the scoring systems, the probability of death was calculated and compared between survivors and non-survivors.
Statistical analysis
Data were analyzed using SPSS ver. 21.0 (SPSS Inc., Chicago, Illinois, USA) and MedCalc ver. 12.2 (MedCalc Inc., Mariakerke, Belgium). Data are presented as mean ± standard deviation, median with interquartile range, or frequency. Differences between the two groups were tested using the independent two-sample
Ethics statement
This study protocol was approved by the institutional review board (IRB) of Samsung Changwon Hospital (IRB no. 2015-SCMC-039-00). Informed consent was waived by the board.
Results
Among the 244 patients, 219 from January 2010 to April 2015 were included in the study. Twenty-five patients with the conditions shown in Figure 1 were excluded.

Paraquat-poisoned patients treated between January 2010 and April 2015.
Mean age was 63 years (range, 20–97 years) and 138 (63.0%) patients were male. The cause of poisoning (incidental vs intentional) and ingestion route (oral vs non-oral) were significantly different between survivors and non-survivors (
Demographic and laboratory findings between survivors and non-survivors.

MAP: mean arterial pressure; GCS: Glasgow Coma Scale; WBC: white blood cell count; RR: respiratory rate; CRP: C-reactive protein (mg/dL); APACHE II: acute physiology and chronic health evaluation II; SAPS II: simplified acute physiology score II; SOFA: sequential organ failure assessment.
Increased lactate, amylase, glucose, and decreased MAP and HCO3 were associated with a significantly higher risk of death after controlling for cause of poisoning, ingestion route, age, MAP, HCO3, lactate, creatinine, potassium, AST, glucose, pH, GCS score, PaO2, amylase, and lipase (Table 2).
Multiple logistic regression analysis of clinical parameters predicting mortality after paraquat ingestion.a
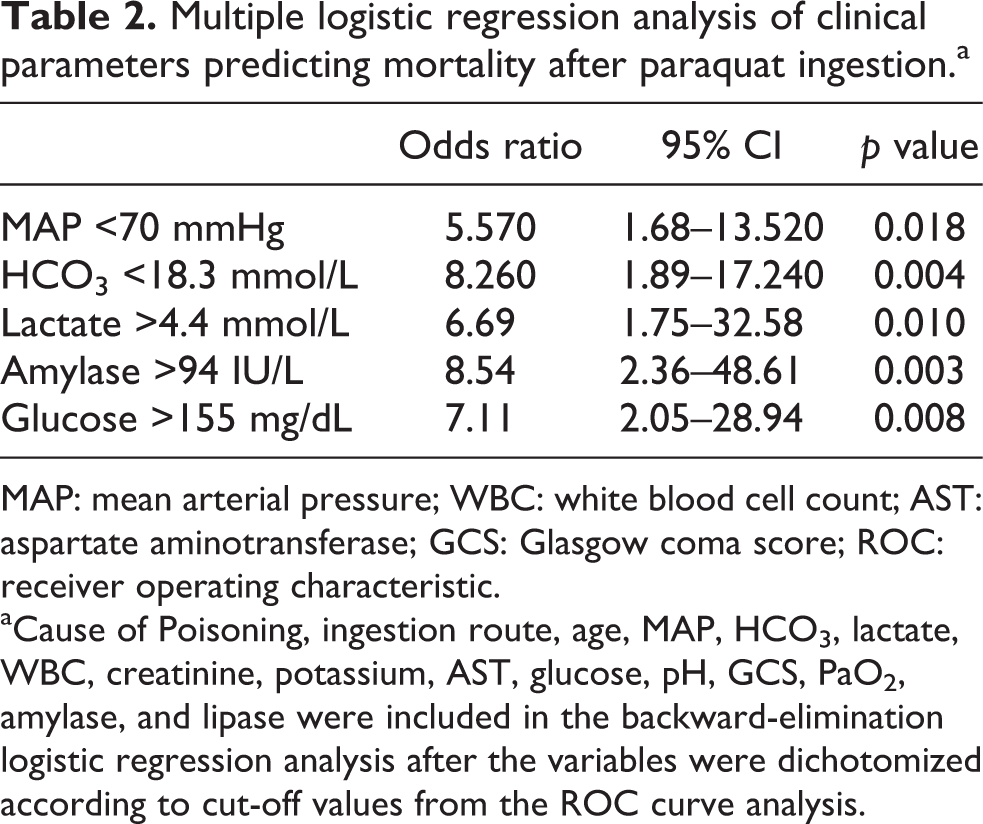
MAP: mean arterial pressure; WBC: white blood cell count; AST: aspartate aminotransferase; GCS: Glasgow coma score; ROC: receiver operating characteristic.
aCause of Poisoning, ingestion route, age, MAP, HCO3, lactate, WBC, creatinine, potassium, AST, glucose, pH, GCS, PaO2, amylase, and lipase were included in the backward-elimination logistic regression analysis after the variables were dichotomized according to cut-off values from the ROC curve analysis.
All scoring systems were significantly different between survivors and non-survivors. That is, the APACHE II scores were 15.3 ± 8.4 in the non-survivor group and 5.7 ± 3.9 in the survivor group (
The area under the ROC curve was calculated to evaluate the predictive value of the scoring systems. The APACHE II scoring system had an area of 0.850 and a cut-off value of 9, the SAPS II score had an area of 0.835 and a cut-off value of 32, and the SOFA score had an area of 0.716 and a cut-off value of 3 (Figure 2).

Receiver operating curves for predicting death according the SOFA, APACHE II, and SAPS II scoring systems. The areas under the curve and 95% confidence intervals for these indicators were 0.716 (0.642–0.790) for the SOFA, 0.850 (0.795–0.905) for the APACHE II, and 0.835 (0.775–0.894) for the SAPS II. SOFA: sequential organ failure assessment; APACHE II: acute physiology and chronic health evaluation II; SAPS: simplified acute physiology score.
The probabilities of death in the SOFA, APACHE II, and SAPS II model are shown in Table 3 for the survivor and non-survivor groups. The overall method was significantly associated between survivors and non-survivors with a good probability of death.
Mean and standard deviation for the SOFA, APACHE II, and SAPS II values.
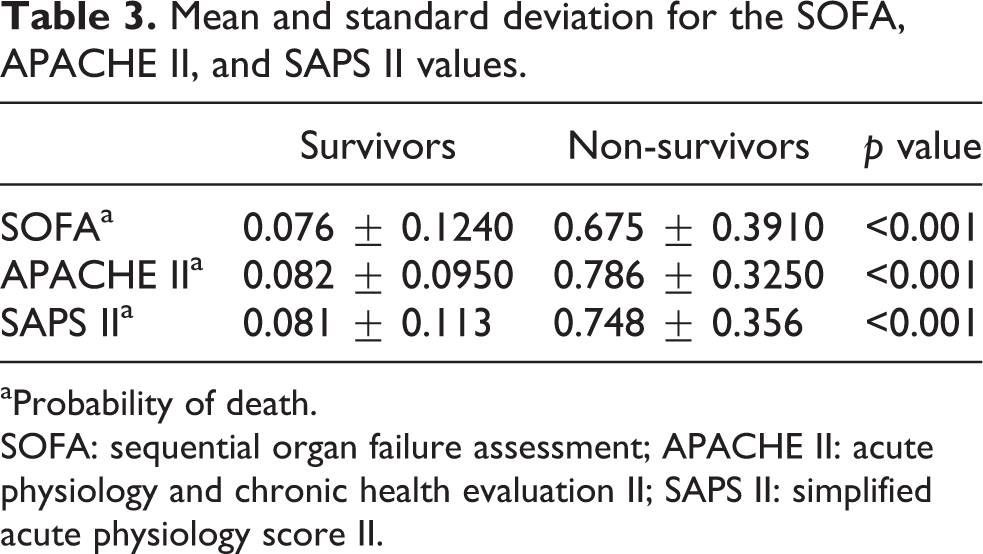
aProbability of death.
SOFA: sequential organ failure assessment; APACHE II: acute physiology and chronic health evaluation II; SAPS II: simplified acute physiology score II.
The predictive power of the mortality estimates for the overall study population according to the SOFA, APACHE II, and SAPS II is shown in Table 4. Sensitivity of the SOFA was lower than that of the APACHE II (
Sensitivities, specificities, and accuracy rates of the SOFA, APACHE II, and SAPS II scoring systems for predicting mortality.
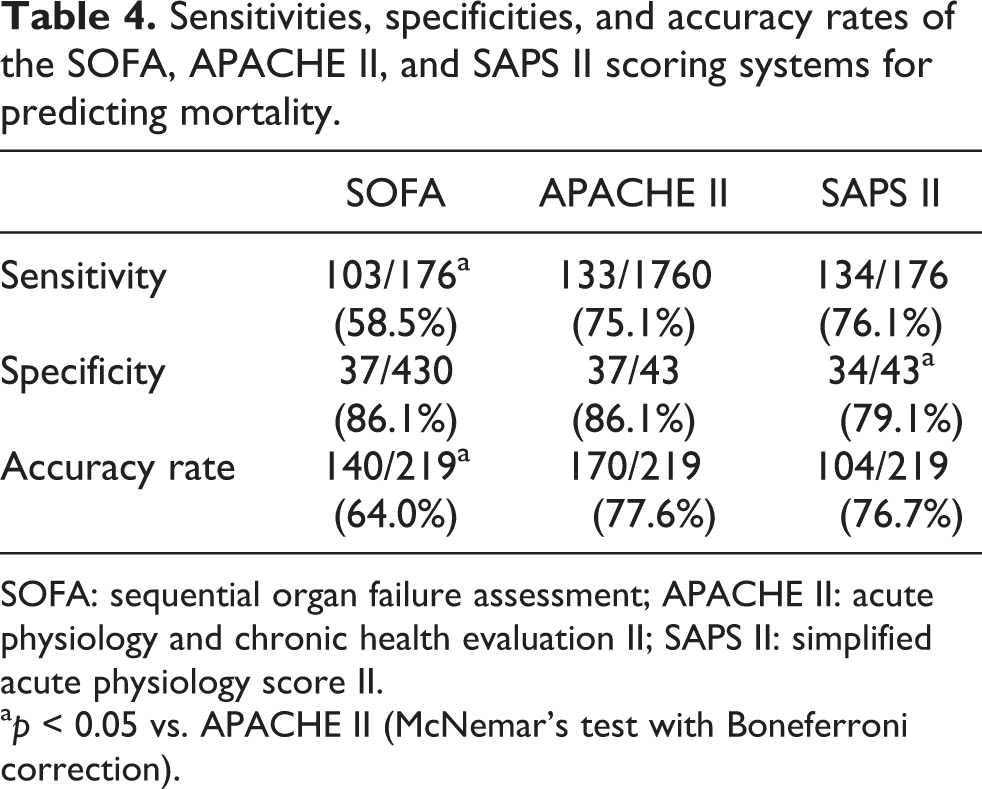
SOFA: sequential organ failure assessment; APACHE II: acute physiology and chronic health evaluation II; SAPS II: simplified acute physiology score II.
a
The SMR values for the SOFA, APACHE II, and SAPS II were 0.97 (95% confidence interval, 0.18–1.53), 0.86 (0.36–1.68), and 4.39 (1.73–13.27), respectively. The
Discussion
Several pathways have been identified as causative factors for the critical toxicity of PQ in humans. First, the lungs are vulnerable to PQ intoxication. Second, reactive oxygen species (ROS)-mediated cell signal transduction causes inflammation. Third, PQ causes mitochondrial damage. Finally, PQ releases the free form of iron from ferritin, aggravating ROS production. 24 Patients with severe PQ poisoning usually die due to multi-organ failure and cardiogenic shock with 1–4 days. If the clinical course is prolonged, pulmonary fibrosis can develop, which is often fatal within several weeks. 9
PQ poisoning severity differed depending on the compound ingested, the quantity, the way it was ingested, and when treatment was initiated. Mortality can decrease in PQ-poisoned patients if they are treated appropriately. Other methods have been reported to determine the severity and prognosis of PQ poisoning.
In this study, we compared the performance of the SOFA, APACHE II, and SAPS II scores of patients poisoned with PQ in the ICU to predict mortality.
The APACHE II uses a point score based on initial values of 12 routine physiological measurements, patient age, and medical history to provide a general disease severity measure. This system stratifies a wide variety of patients according to prognosis because of the strong and consistent relationship between acute physiological dysfunction and the risk of death due to acute illness. 25 –27 The APACHE II system is a useful index for evaluating poisoning severity due to multiple organ system involvement. 18 Apparently, the APACHE II system was chosen as a practical tool in predicting the prognosis of acute PQ-poisoned patients. Our study showed that APACHE II system had more superior discriminative power (AUC = 0.850) than the SOFA score (AUC = 0.716) and SAPS II system (AUC = 0.835). Therefore, we guess that the APACHE II system may be a practical tool in predicting the prognosis of acute PQ-poisoned patients.
The SOFA score was developed during a consensus conference organized by the European Society of Intensive Care and Emergency Medicine. 16 The SOFA score calculates a summary value for the degree of dysfunction of six sets of organs (respiratory, coagulation, liver, cardiovascular, central nervous system, and renal). Four levels of dysfunction are identified in the SOFA score for each organ system. Organ dysfunction is associated with high rates of ICU morbidity and mortality, and, as such, treatment for these disorders accounts for a high proportion of the ICU budget. 28,29 Despite the fact that the SOFA score is easier to calculate for PQ-poisoned patients, its utility to predict mortality in our population was not as good as that of the APACHE II and SAPS II scoring systems.
The SAPS II was designed to measure disease severity in patients <15 years of age admitted to the ICU. 21 A point score is calculated based on 12 routine physiological measurements during the first 24 h, health status information, and information obtained at admission. This calculation method results in a predicted mortality. Although the SAPS II was useful for predicting outcomes of PQ-poisoned patients in our study, it is relatively complex and it was less valuable for this use than the APACHE II scoring system.
Our study found that the APACHE II score estimates mortality risks with satisfactory accuracy in patients PQ-poisoned. The assessment of illness severity with the APACHE II score had been demonstrated to be reliable in ICUs. As a result from overcrowding, our EDs sometimes also function as ICUs, handling monitoring needs, and providing adequate treatment for ill patients at a lower cost. This may explain why the APACHE II score was predictive of mortality in our database with satisfactory discriminative power and function.
Fortunately, we could confirm that mortality of PQ-poisoned patients was predicted precisely using any of the three scoring systems upon ICU admission. Although all of these scores can be used as an objective parameter to identify PQ-poisoned patients being monitored in the ICU, the APACHE II score had the best overall performance compared with the other two scoring systems. The APACHE II score also had strong function and discrimination power. This score was accurate and useful for stratifying risk among PQ-poisoned patients presenting to the ICU.
Our conclusions are limited by the single-center retrospective nature of the study, and the results may lack wider applicability due to missing data and the small sample size. In addition, we could not determine the effect of the time difference between patient arrival to the emergency department and the time they ingested the poison because it was a retrospective study. Last, we did not measure plasma PQ levels, which may have more accurately reflected the severity of PQ exposure. Therefore, a multi-center or randomized trial should be conducted and plasma PQ levels determined to avoid these limitations and confirm our results.
Conclusions
We evaluated performance of the SOFA, APACHE II, and SAPS II scores for predicting mortality from PQ poisoning in patients treated in the ICU. These three predictive scoring systems provided potentially valuable prognostic information to evaluate the severity and mortality of PQ-poisoned patients. The APACHE II score is somewhat complex, but was more significantly associated with mortality and had stronger discriminative power for predicting mortality compared to those of the SOFA and SAPS II. We recommend that the APACHE II score be used to predict outcomes and stratify risk among PQ-poisoned patients treated in the ICU.
Footnotes
Authors’ Note
The authors alone are responsible for the content and writing of the article.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
