Abstract
Background:
Exposure to diesel exhaust particles (DEP) has long been associated with increased cardiovascular morbidity and mortality. The development of DEP toxicity seems to be linked to inflammation in which macrophages play a critical role. Macrophages can be polarized into proinflammatory M1 or anti-inflammatory M2 macrophages. The aim of this study was to identify the role of inflammation in DEP-induced toxicity by assessing the effects of DEP on macrophage polarization.
Methods:
Monocyte-derived macrophages (Mϕ) were stimulated with interferon γ and lipopolysaccharide or interleukin (IL)-4 to obtain M1 and M2 subtypes, respectively. To test the polarization capacity of DEP, Mϕ cells were exposed to DEP and compared to Mϕ, M1, and M2. We also studied the effects of DEP on already-polarized M1 or M2. The M1 markers assessed were tumor necrosis factor α (TNF-α) and IL-1β, while the M2 markers were the mannose receptor C type 1 (MRC-1) and transglutaminase 2 (TGM2).
Results:
Western blots revealed a 31 kDa band corresponding to pro-IL-1β, but only in M1-polarized macrophages. In M1, we also observed an upregulation of TNF-α messenger RNA (mRNA) expression. MRC-1 and TGM2 mRNA expression were only significantly enhanced in M2. DEP had no effect on any of the M1/M2 markers assessed. Moreover, DEP were not able to modify the phenotype of already-polarized M1 or M2.
Conclusion:
Mϕ incubation with DEP did not have any effect on macrophage polarization, at least on the markers assessed in this study, namely, TNF-α/IL-1β for M1, and MRC-1/TGM2 for M2. Hence, these data argue against an important role of inflammation in DEP-induced vascular toxicity.
Introduction
A large number of epidemiological and clinical studies have associated inhalation of ambient particulate matter (PM) to increased cardiovascular morbidity and mortality. 1 –3 Indeed, urban air pollution does not only contribute to the development and exacerbation of respiratory diseases like asthma, chronic obstructive pulmonary disease, and lung cancer but also to the progression of atherosclerosis and to the development of myocardial infarction. 3 These adverse effects are more strongly linked to the fine and ultrafine particulate fractions, that is, to PM with an aerodynamic diameter of less than 2.5 μm (PM2.5) because they have the capacity to penetrate deep into the lungs and may even pass directly into the circulatory system. 1,2 A major part of this PM2.5 fraction is constituted by diesel exhaust particles (DEP).
The precise pathway through which DEP influence cardiovascular risk has not yet been fully elucidated, but one mechanism thought to be involved in this toxicity is the production of an oxidative stress and an inflammation in the lung with a subsequent secondary systemic inflammatory state leading to cardiovascular dysfunctions. 4,5 Inflammation is therefore considered to be a key step in the development of the negative health effects induced by DEP. 6 Many studies have focused on the role of inflammation in the toxicity induced by DEP, but the results are very often contradictory. While some authors observe, either in the bronchoalveolar fluid of mice exposed to PM10 7 or in human alveolar or monocyte-derived macrophages (Mϕ) exposed to DEP, 8 –12 an increase in proinflammatory cytokines like tumor necrosis factor α (TNF-α), interleukin (IL)-1β, IL-6, and IL-8, 7 –10 others find no alteration 11,12 of those markers. 6
When inflammation occurs, monocytes will be released from the bone marrow to the blood and can be recruited to the tissue at any point of their maturation process to give rise to specific tissue macrophages. 13 Macrophages form the first line of defense against pathogens and play a critical role in the initiation, maintenance, and resolution of inflammation. 13,14
Mϕ can develop into different phenotypes according to the nature of their environment. The classically activated macrophages, called M1, arise in response to the T helper type 1 (Th1) cytokine interferon γ (IFN-γ) and lipopolysaccharide (LPS). They have bactericidal and tumoricidal activity, generate reactive oxygen species and nitric oxide, promote Th1 responses, and produce high proinflammatory cytokines like TNF-α, IL-1β, IL-6, and IL-12. 13 –16 On the other hand, the alternative activated macrophages, called M2, generated by the T helper type 2 (Th2) cytokines IL-4 or IL-13, play a role in tissue repair, tissue remodeling, matrix deposition and healing, and they promote Th2 responses. 13,15,16 They express high levels of scavenger mannose and galactose receptors, produce high levels of IL-10 and IL-1 receptor antagonist, and overexpress transglutaminase 2 (TGM2) which has recently been determined as an M2 marker. 14,15,17,18 Macrophage polarization has been studied thoroughly in certain diseases like bacterial infection, cancer, and asthma, 16,18 –20 but very little research has focused on how DEP can modulate macrophage polarization.
In our previous study, we showed that DEP impair vascular function through the generation of an oxidative stress. 21 In order to further identify the mechanisms involved in DEP toxicity, we wished to explore the effects of DEP on inflammation and, more particularly, on macrophage polarization.
Materials and methods
Chemicals
Bovine serum albumin, dimethylsulfoxyde (DMSO), 3-(4,5-dimethyl-2-thiazolyl)-2,5-diphenyl-2H-tetrazolium bromide (MTT), fetal bovine serum (FBS), LPS from
Cell culture
THP-1 cells, a human acute monocytic leukemia cell line (gift from the Laboratory of Structure and Function of Biological Membranes (ULB), and obtained from HPA cultures (Salisbury, United Kingdom)), were cultured in RPMI-1640 medium supplemented with 10% FBS, 2 mM
Macrophage differentiation and polarization
THP-1 cells were differentiated into Mϕ macrophages by overnight incubation with 100 nM of PMA. 22 The cells were then washed and cultured with PMA-free RPMI medium for 30 h before polarization. They were polarized overnight (18 h of incubation) into proinflammatory M1 macrophages using 10 ng/mL of LPS and 20 ng/mL of IFN-γ 23 ; or into anti-inflammatory M2 macrophages using 15 ng/mL of IL-4. 24,25
DEP characteristics
DEP (SRM 2975, Industrial Forklift), a sample of combustion-derived particles, were purchased from the National Institute of Standards and Technology (NIST, Gaithersburg, MD, USA). The particle size distribution of PBS-suspended DEP was assessed by dynamic light scattering using a Zetasizer nano ZS (Malvern Instruments, Worcestershire, UK). Size distribution (
DEP exposure
DEP suspensions were prepared as previously described.
21
In short, DEP (2 mg/mL) were suspended in PBS, vortexed, and sonicated for 2 min by probe sonication. They were sonicated for 15 min in a bath sonicator just before use. Aqueous DEP extracts were prepared in the same way, except that they were sonicated for 30 min, centrifuged at high speed (20 min at 14,000 ×
To study the polarization capacity of DEP, we exposed Mϕ macrophages overnight (18 h) to either DEP suspensions (10 µg/mL) or aqueous DEP extracts (100 µg/mL). DEP concentrations were chosen according to previous studies. 10,21 We also exposed M1 and M2 macrophages for an additional 24 h with DEP suspensions or DEP extracts.
MTT cell viability test
THP-1 cells were seeded in a 96-well plate at a density of 100,000 cells per well and cultured overnight in RPMI medium containing 100 nM of PMA to achieve their differentiation. The next day, cells were washed and cultured for an additional 24 h in RPMI. Cells were then treated with various concentrations of DEP suspensions or DEP extracts (1, 5, 10, 50, 100, 250, and 500 µg/mL) for 18 h. Cells were thereafter incubated with MTT solution (0.5 mg/mL) for 3.5 h at 37°C in a 5% CO2 atmosphere. Finally, medium was discarded and replaced by DMSO to solubilize formazan crystals. Absorbance was measured at 540 nm.
Western blot analysis
Cells were differentiated and polarized in 12-well plates before being lysed in lysis buffer containing 1% of Triton X-100 and 0.5% of protease inhibitor cocktail. Protein concentration was determined using the bicinchoninic acid assay (Life Technologies, Ghent, Belgium). Extracted proteins were separated on 15% polyacrylamide gels and transferred to polyvinylidene difluoride membranes. Membranes were blocked for 1.5 h with 5% nonfat dry milk in Tris-Buffered Saline Tween 20 at room temperature and then incubated overnight at 4°C with primary antibody directed against IL-1β (1:4000 dilution; Thermo Fisher Scientific). After washing, membranes were incubated with anti-rabbit horseradish peroxidase–conjugated secondary antibody for 1 h at room temperature (1:5000 dilution; Thermo Fisher Scientific). Immunolabeled proteins were visualized using an enhanced chemiluminescent detection system (SuperSignal™ West Femto, Thermo Fisher Scientific).
RNA extraction and quantitative real-time polymerase chain reaction
Total RNA was extracted from polarized and DEP-exposed macrophages using ReliaPrep™ RNA Cell Miniprep System (Promega, Leiden, the Netherlands) according to the manufacturer’s instructions. RNA concentration and purity were determined by optical density measurement using a nanodrop spectrophotometer. Reverse transcription was carried out using iScript reverse transcription supermix for real-time quantitative polymerase chain reaction (RT-qPCR; BioRad, Temse, Belgium).
For RT-qPCR, primers were designed using the computer Primer3 program for human TNF-α, mannose receptor C type 1 (MRC-1), TGM2, and hypoxanthine guanine phosphoribosyltransferase (HPRT) as housekeeping gene (Table 1).
Primer sequences.

HPRT: hypoxanthine guanine phosphoribosyltransferase; TNF-α: tumor necrosis factor α; MRC-1: mannose receptor C type 1; TGM2: transglutaminase 2.
For each sample, amplification reaction was performed in triplicate using SsoAdvanced™ Universal SYBR® Green Supermix (BioRad, Temse, Belgium), specific primers and diluted template complementary DNA. Signal detection and analysis of results were performed using the BioRad CFX96 Touch™ Real-Time PCR Detection System and the BioRad CFX Manager™ Software. Relative quantification was achieved with the comparative 2−ΔΔ
Statistical analysis
Results were expressed as means ± SEM,
Results
Effect of DEP on cell viability
DEP suspensions (Figure 1(a)) and DEP extracts (Figure 1(b)), at the concentrations used, did not affect viability of Mϕ macrophages, at least after 18 h of exposure (

Cell viability test (
DEP are not able to polarize Mϕ macrophages into M1 macrophages
Expression of IL-1β and TNF-α was used to assess M1 polarization. For IL-1β expression, we performed a Western blot analysis with an antibody that detects IL-1β and pro-IL-1β. The expression of pro-IL-1β (31 kDa) was only detected in M1 macrophages (Figure 2).

Western blot analysis of IL-1β protein expression in Mϕ, M1, M2, and DEP-exposed Mϕ macrophages. Experiments were repeated three times with similar results. DEP: diesel exhaust particle.
The expression of TNF-α, on the other hand, was determined by RT-qPCR. Similar results were observed, namely, an overexpression of relative TNF-α mRNA only in M1 macrophages (Figure 3).

Relative TNF-α mRNA expression in Mϕ, M1, M2, and DEP-exposed Mϕ macrophages. Values represent means ± SEM in arbitrary units. **
These results suggest that DEP are not able to polarize Mϕ macrophages into proinflammatory M1 macrophages.
DEP are not able to polarize Mϕ macrophages into M2 macrophages
For M2 polarization, we assessed the expression of the M2 markers MRC-1 and TGM2 by RT-qPCR. Relative MRC-1 (Figure 4(a)) and TGM2 (Figure 4(b)) mRNA expressions were only significantly increased in M2 macrophages. These results suggest that DEP are not able to polarize Mϕ macrophages into anti-inflammatory M2 macrophages.
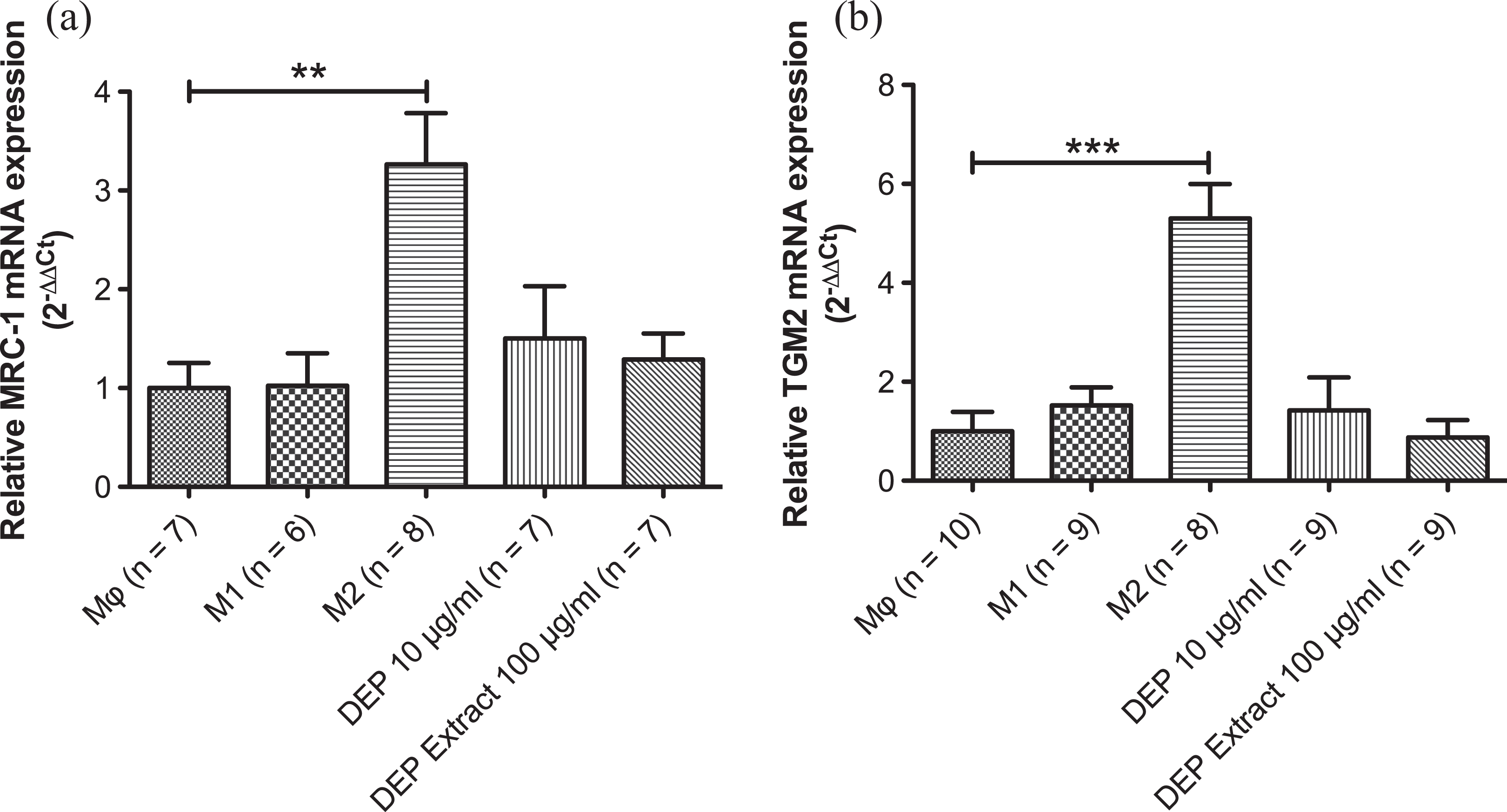
Relative MRC-1 (a) and TGM2 (b) mRNA expression in Mϕ, M1, M2, and DEP-exposed Mϕ macrophages. Values represent means ± SEM in arbitrary units. **
Effect of DEP on already-polarized M1 and M2 macrophages
In additional experiments, we wanted to verify whether DEP could reverse the polarity of M1 or M2 macrophages. Therefore, we incubated M1 or M2 macrophages for an additional 24 h with either DEP suspensions or DEP extracts and determined the expression of M1 and M2 markers through RT-qPCR.
The results showed that DEP were not able to modify the phenotype of M1 or M2 macrophages. Indeed, TNF-α mRNA was only overexpressed in M1 macrophages and M1 subsequently incubated with DEP (Figure 5(a) and (c)) while TGM2 mRNA expression was only significantly enhanced in M2 macrophages and M2 subsequently incubated with DEP (Figure 5(b) and (d)).

Relative TNF-α (a and c) and TGM2 (b and d) mRNA expression in Mϕ, M1, M2, and M1 macrophages subsequently incubated with DEP (a and b) or in Mϕ, M1, M2, and M2 macrophages subsequently incubated with DEP (c and d). Values represent means ± SEM in arbitrary units. *
Discussion
It is now widely accepted that DEP exposure increases cardiovascular morbidity and mortality. One of the mechanisms thought to be involved in the toxicity of DEP is the development of an inflammatory state in the lung with the release of circulating factors capable of inducing a systemic inflammation. 2,4 The role of inflammation in DEP toxicity remains controversial and very few studies have focused on the effects of DEP on macrophage polarization.
In many aspects, ingredients and characteristics, DEP are similar to cigarette smoke. 27 Although cigarette smoke seems to direct the immune system to a Th2 profile, 28 it is unclear whether DEP favors Th1 or Th2 response. DEP exposure has been consistently linked to inflammation (Th1 profile) indicating that DEP would most likely polarize macrophages into an M1 state. 6,27,29 However, exposure to PM has also been associated with pulmonary diseases like asthma and pulmonary fibrosis (Th2 response) 18,20,30 and decreases the phagocytic function mostly attributable to the M1 subtype, suggesting a conversion into an M2 profile. 14,31
In our study, we exposed Mϕ cells to DEP in order to see whether DEP were able to induce their polarization into M1 or M2 macrophages. M1 macrophages are detectable by their ability to release the proinflammatory cytokines TNF-α and IL-1β. 15 We observed no alteration of those two M1 markers after DEP exposure, either for the whole DEP suspension or for the aqueous extract of DEP. Kocbach et al. showed similar results, as they noted no modifications of the mRNA expressions of TNF-α and IL-1β after exposure of THP-1 cells to DEP. 32 They also showed no alteration of IL-10 and IL-4 (M2 markers) mRNA expression after DEP exposure, which is in line with our results, that is, no change of our M2 markers MRC-1 and TGM2 in DEP-exposed Mϕ macrophages. On the contrary, DEP were shown to suppress LPS-induced TNF-α and IL-8 release in alveolar macrophages, 11,31,33 and the production of IFN-γ, IL-2, and IL-10 in LPS-stimulated splenic mononuclear cells, 34 which may provide evidence that DEP are involved in Th2 response.
However, Jaguin et al. showed that DEP treatment increased the secretion of the M2 marker IL-10 in human blood monocytes-derived Mϕ. 35 Furthermore, they observed modifications of the mRNA expression of M1 and M2 markers in DEP-exposed macrophages, in particular, an induction of the M1 markers TNF-α and IL-8 in DEP-exposed M1, as well as a downregulation of the M2 markers MRC-1 and nitric oxide synthase 3 in DEP-exposed M2, while mRNA levels of IL-10 in this subtype remained unchanged. This indicates that DEP are able to alter the expression of some M1 and M2 markers. Yet those results were observed in Mϕ macrophages exposed to DEP in the presence of IFN-γ or IL-4, while we exposed the Mϕ macrophages solely to DEP (in the absence of the polarizing factors). The differences in our results suggest that the presence of the polarizing factors is essential for DEP-induced polarization. The lack of effect of DEP alone was also reported by Thevenot et al. who observed that PM exposure of mice along with alcohol intake led to enhanced M2 polarization, while inhalation of PM alone was not able to alter the expression of the M2 markers assessed. 30 This suggests that the environment can alter the way macrophages respond to DEP, while DEP alone are unable to do so.
Another explanation for the disparity in the results of those studies is that only a few phenotypical markers are assessed per macrophage subset, which may not be enough to completely rule out a direct effect of DEP. It is well known that a large variety of macrophage phenotypes exists and that M1 and M2 are only the two extreme limits of this spectrum. 13
A second part of the current study consisted in assessing the response of already-polarized M1 and M2 macrophages to DEP exposure, because there is strong evidence that macrophages can switch from one phenotype into another in response to different conditions. 14 We therefore incubated M1 and M2 macrophages with DEP and measured the expression of the M1 and M2 markers TNF-α and TGM2, respectively. However, after an exposure time of 24 h, DEP were unable to reverse the polarity of M1 or M2 macrophages. This conclusion has also been formulated by Jaguin et al., namely, that DEP are unable to switch polarization profiles of macrophages from one phenotype to another. 35
Finally, dissimilarities in how macrophages react to PM have been reported to be related to the characteristics of the PM. 27 However, these results are very difficult to compare as very few authors give a clear description of the particles used. Nevertheless, Becker et al. assessed the effects of particulate size on cytokine production in alveolar macrophages and observed that inflammatory mediators were preferentially induced by coarse PM rather than ultrafine PM like DEP. 31 In line with the PM characteristic hypothesis, Schwarze et al. reviewed in vitro studies of pro- and anti-inflammatory responses of lung cells to various kinds of DEP, focusing particularly on the proinflammatory effects of DEP. 6 They concluded that the inflammatory effects of DEP are attributable to the soluble organic fraction of DEP. This could explain why we did not see increased proinflammatory cytokine release in our study given that we used aqueous DEP extracts or suspensions while Jaguin et al., who used the dichloromethane extract, did observe some modifications. 35 Hence, these data show that in addition to DEP concentration, DEP components and the environment can alter macrophage polarization, 27,36,37 but the complexity of DEP characteristics makes it very difficult to identify the exact DEP compounds or peculiarities responsible for the pro- or anti-inflammatory effects of DEP, making it necessary to conduct more systemic research in this field.
Conclusion
In conclusion, our data demonstrate that DEP alone are not able to polarize Mϕ macrophages either into proinflammatory M1 macrophages or into anti-inflammatory M2 macrophages. Indeed, they were not able to modify the expression of the M1 and M2 markers used in this study.
Moreover, DEP alone did not modify the phenotype of M1- or M2-polarized macrophages arguing against an important role of inflammation in DEP-induced vascular toxicity.
Footnotes
Acknowledgements
The authors gratefully acknowledge Dr Nathalie Wauthoz (Laboratory of Pharmaceutics and Biopharmaceutics, Faculty of Pharmacy, Université Libre de Bruxelles) for the determination of particle size distribution, and Prof. Michel Vandenbranden (Laboratory of Structure and Function of Biological Membranes, Université Libre de Bruxelles) for the gift of the THP-1 cells.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by a grant from the “Fonds pour la Chirurgie Cardiaque”.
