Abstract
Following inhalation and deposition in the alveolar region at sufficient dose, biopersistent (nano)materials generally provoke pulmonary inflammation. Alveolar macrophages (AMs) are mediators of pulmonary immune responses and were broadly categorized in pro-inflammatory M1 and anti-inflammatory M2 macrophages. This study aimed at identifying AM phenotype as M1 or M2 upon short-term inhalation exposure to different (nano)materials followed by a postexposure period. Phenotyping of AM was retrospectively performed using immunohistochemistry. M1 (CD68+iNOS+) and M2 (CD68+CD206+ and CD68+ArgI+) AMs were characterized in formalin-fixed, paraffin-embedded lung tissue of rats exposed for 6 hours/day for 5 days to air, 100 mg/m3 nano-TiO2, 25 mg/m3 nano-CeO2, 32 mg/m3 multiwalled carbon nanotubes, or 100 mg/m3 micron-sized quartz. During acute inflammation, relative numbers of M1 AMs were markedly increased, whereas relative numbers of M2 were generally decreased compared to control. Following an exposure-free period, changes in iNOS or CD206 expression correlated with persistence, regression, or progression of inflammation, suggesting a role of M1/M2 AMs in the pathogenesis of pulmonary inflammation. However, no clear correlation of AM subpopulations with qualitatively distinct histopathological findings caused by different (nano)materials was found. A more detailed understanding of the processes underlaying these morphological changes is needed to identify biomarkers for different histopathological outcomes.
Keywords
Introduction
Nanomaterials are widely used in industrial applications. During the production and processing of nanomaterials in the workplace, particulate matter might be released to the air. 1,2 Thus, inhalation is a major route of occupational exposure to nanomaterials. In vivo inhalation studies in laboratory animals suggest a potential hazard of biopersistent nanomaterials to the lungs, commonly initiated by a pulmonary inflammatory response. 3 –6
Early human epidemiological data and rat inhalation studies showed that different poorly soluble dusts at sufficient dose caused pulmonary inflammation upon inhalation exposure, 7 –9 although these materials were of very low inherent toxicity in oral and dermal studies. More recent inhalation toxicity studies in rats showed distinct differences in morphological changes of lungs caused by different poorly soluble nanomaterials. 3,5,10 –14 The inflammatory potency as well as the quality of the inflammation of these biopersistent particles differ largely from each other.
For the present examinations, nano-TiO2, nano-CeO2, multiwalled carbon nanotubes (mwCNT), and micron-sized quartz were chosen as model substances because data from short-term as well as long-term rat inhalation toxicity studies are available for all 4 materials. 3 –5,15 These substances are considered poorly soluble and highly biopersistent. Quartz and mwCNT are known to exhibit specific toxicity due to their surface reactivity and shape, respectively. In the past, TiO2 and CeO2 were considered as “poorly soluble particles with low inherent toxicity”; however, it was shown that both TiO2 and CeO2 nanoparticles provoke distinct pulmonary toxicity in rat lungs upon inhalation exposure. In a 90-day inhalation study, 16 TiO2 caused only minimal to mild lesions comprising particle-laden macrophage accumulation, aggregations in subpleural region, and in centriacinar region at an aerosol concentration of 2 mg/m3. These lesions were associated with minimal hypertrophy and hyperplasia of alveolar epithelial type II cells. Most of these lesions regressed postexposure (13-52 weeks). In comparison, subacute (28 days) and subchronic (90 days) inhalation exposure to CeO2 elicited pulmonary inflammation at very low dust aerosol concentrations (down to 0.1 mg/m3). The CeO2-induced inflammatory reaction persisted during a postexposure period of 13 weeks and progressed during this time to granulomatous inflammation. 10,12 The initial pulmonary inflammation was characterized by an increased number of AMs loaded with amber-colored small particles, diagnosed as histiocytosis, alveolar—with particles. The granulomatous inflammation was characterized by septal/interstitial conglomerates of particle-laden macrophages intermingled with mixed inflammatory cells (lymphocytes, plasma cells). Also, biopersistent nanomaterials with high aspect ratio, such as mwCNT, showed high inflammogenic potential following inhalation exposure. 14 Subchronic exposure to 0.1, 0.5, or 2.5 mg/m3 mwCNT caused dose-dependent initial granulomatous inflammation, characterized by conglomerated macrophages, lymphocytes, and plasma cells, even at the lowest aerosol concentration tested. The inflammation was composed of AMs and polymorphonuclear neutrophils and was located in the centriacinar region. In addition, intraseptal granulomas were observed. 14 With prolonged exposure duration (up to 2 years in rats), long-term effects caused by nanomaterials were observed to persist or progress to fibrosis or possibly lung tumor formation. 3 –5,15
Compared to subchronic inhalation studies, short-term inhalation studies (STIS; 5-day exposure) provide important information on early key elements of pulmonary inflammation caused by nanomaterials. Combining information from quantitative analysis of bronchoalveolar lavage fluid (BALF) with histopathological findings and with information on the location, persistence, regression or progression of effects, STIS are well suitable for the hazard assessment and identification of inflammogenic potency of inhaled nanomaterials. 6,17,18 The STIS proved to be useful in prioritizing 6,19 and grouping nanomaterials. 20 Archived formalin-fixed paraffin-embedded (FFPE) lung tissues of formerly performed STIS were chosen for the present examinations. Rats were inhalation exposed to 100 mg/m3 nano-TiO2, 25 mg/m3 nano-CeO2, 32 mg/m3 mwCNT, or 100 mg/m3 micron-sized quartz. The studies were published previously. 10,13,14 Study design and main findings are summarized in Table 1. In brief, the inhalation exposure to 100 mg/m3 TiO2 caused pulmonary inflammation composed of minimal to moderate diffuse, alveolar histiocytosis, and minimal multifocal infiltration with neutrophils. Importantly, the inflammation declined 14 days after the end of exposure. 13 Exposure to 25 mg/m3 CeO2 resulted in pulmonary inflammation composed of minimal to slight alveolar histiocytosis. Here, inflammation persisted following a 21-day postexposure period. Short-term inhalation exposure to 32 mg/m3 mwCNT led to granulomatous inflammation in lungs of exposed animals. Histopathological findings were not reversible, and granulomatous inflammation persisted during a 21-day post-exposure period. 14 In comparison, 100 mg/m3 quartz leads to a very strong inflammatory response with minimal to moderate diffuse, alveolar histiocytosis in lungs of exposed animals immediately following 5-day exposure. Fourteen days postexposure, increases in inflammatory parameters were not reversible; on the contrary, histological findings of a diffuse inflammatory response, composed of AMs, neutrophils, and cell debris, increased in severity. 13
Short-Term Inhalation Studies (STIS) in Rats.a

Abbreviations: BALF, bronchoalveolar lavage fluid; DMEM/FCS, Dulbecco modified Eagle medium/fetal calf serum; mw-CNT, multiwalled carbon nanotubes; STIS, short-term inhalation studies.
a Summary of Substance Characterization, Study Design, and Main Findings of TiO2 and Quartz STIS, CeO2 STIS, and mwCNT STIS.
Obviously, AMs have a crucial role in pulmonary clearance as well as in orchestrating pulmonary immune responses. 22 Data published during the past 3 decades suggest that these various activities are mediated by distinct subpopulations of macrophages, which are induced by signals they encounter in their local tissue microenvironment. 23 –35 In a rather simplistic view, these subpopulations can be divided into 2 major distinct macrophage phenotypes, which have been categorized broadly as pro-inflammatory/cytotoxic M1 macrophages and anti-inflammatory/wound repair M2 macrophages. 36 –38 Increasing evidence suggests that nanomaterials are capable of activating macrophages to the M1 phenotype, leading to the expression of pro-inflammatory mediators and recruitment of inflammatory cells. Ultimately, nanomaterials may have an impact on the M1/M2 balance, which might change immune defense properties. 39,40 Furthermore, overactivation, characterized by prolonged or excessive release of mediators, of M1 and/or M2 macrophages can contribute to tissue injury and disease progression, such as the development of fibrosis. 22,41
The present study is a retrospective investigation, and the histological specimens examined were derived from available FFPE lungs of the STIS mentioned earlier. 10,13,14 Since AMs represent the immediate, immunocompetent main contact with particulate matter in the alveolar region, the main focus was on the possibility and feasibility to characterize the different phenotypes (M1 or M2) of AMs in existing FFPE lung tissue, which is comparable to the material coming from toxicity studies for regulatory purposes and to elucidate whether early AM polarization can be correlated with the qualitatively different histopathological outcomes of different poorly soluble (nano)materials. Using an immunohistochemical approach, the expression of general (CD68) and specific (iNOS, CD206, ArgI) macrophage markers was detected by double immunolabeling. In addition, we quantitatively analyzed the macrophage subpopulations subsequently to the termination of exposure and following an exposure-free period. We hypothesize that initial macrophage polarization is dependent on intrinsic material properties and affects the different morphological changes observed.
Materials and Methods
Test Substances
TiO2 P25 (CAS No. 13463-67-7) was commercially obtained from the producer (Degussa AG, Frankfurt am Main, Germany) and described in detail in study by van Ravenzwaay et al. 13 CeO2 NM-212 was received from the producer (Umicore, Hanau, Germany) and was extensively characterized in study by Keller et al. 10 The mwCNTs were provided by Nanocyl S.A. (Sambreville, Belgium) under the product name Nanocyl NC 7000. 14 Quartz DQ12 was commercially acquired from the producer (Dörentrup Quarz GmbH, Westfalen, Germany) as depicted in study by van Ravenzwaay et al. 13 For more detailed information on material characterization please refer to Table 1 and study by Wohlleben et al. 21
Short-Term Inhalation Studies
Short-term inhalation studies were performed as described previously. 6,17,18 In brief, Wistar rats were inhalation exposed for 6 hours per day on 5 consecutive days to 100 mg/m3 TiO2, 25 mg/m3 CeO2 NM-212, 32 mg/m3 mwCNT, or 100 mg/m3 quartz aerosols. Concurrent control groups were exposed to conditioned air only. Please note that 3 independent studies were performed. One study for CeO2 exposure (female rats, 5 animals per group), a second one for mwCNT exposure (male rats, 3 animals per group), and a third one for TiO2 or quartz exposure (male rats, 6 animals per group). For details of study design see Table 1. Further, selected tissues, that is, mesenteric lymph nodes, liver, and spleen, were collected as positive control tissues for immunohistochemistry from untreated animals. Tissue selection was based on the expression patterns of the proteins of interest known from the literature. Tissues were collected immediately following killing, lungs were inflated with 10% neutral-buffered formalin (NBF), and all organs were placed in NBF at room temperature. Fixation was followed by standard histotechnical processing and paraffin embedding (Leica ASP300 S; Leica Biosystems, Nussloch, Germany). Subsequently, tissues were sectioned (2-3 µm, Microm HM 355 S; Thermo Scientific), mounted onto slides, and hematoxylin-eosin (HE) stained for standard diagnostic evaluation by a well-experienced and board-certified veterinary pathologist. Histopathological analyses were performed in a blinded manner and according to the International Harmonization of Nomenclature and Diagnostic Criteria. 42 For further details please refer to studies by Keller et al, van Ravenzwaay et al, and Ma-Hock et al. 10,13,14
Immunohistochemistry
For immunohistochemical investigations, tissue sections of the left lung lobe (lobus pulmo sinister), mesenteric lymph nodes, liver, and spleen were mounted onto silane-coated slides (Q Path adhesive slide, Q Path). From each left lung lobe, several sections were prepared. One was used for HE routine staining in the original study (see above); 3 for iNOS-CD68, ArgI-CD68, or CD206-CD68 double immunostaining; and 3 more as negative control for each immunostaining for the present retrospective study. The sections for immunohistochemistry were prepared right before staining. Mesenteric lymph node as well as liver and spleen tissue served as positive control tissue for CD68 immunostaining, and liver tissue served also as positive control tissue for iNOS (Kupffer cells), ArgI (hepatocytes), or CD206 (liver sinusoidal endothelial cells) staining. 43 –49
First, tissue sections were deparaffinized and rehydrated, followed by heat-mediated antigen retrieval, and quenching of endogenous peroxidase activity using 6% hydrogen peroxide. Subsequently, slides were subjected to immunohistochemical staining. Primary polyclonal rabbit anti-iNOS (1:100, ab15323; Abcam), polyclonal rabbit anti-ArgI (1:600, LS-B4660; LifeSpan BioSciences), polyclonal rabbit anti-CD206 (1:5000, ab64693; Abcam) antibody, or serum/IgG negative control (universal negative control serum; Biocare Medical) were applied. As secondary antibody for the first staining antirabbit conjugated to horse radish peroxidase (CytoChem-Plus HRP Polymer Kit; Zytomed Systems) was used according to the manufacturer. As first chromogen served 3,3′-Diaminobenzidine (Zytomed Systems). Subsequently, the same slides were incubated again with a primary antibody, monoclonal mouse anti-CD68 (1:300, MCA341; Abd Serotec), or serum/IgG negative control. Afterward, biotinylated anti-mouse immunoglobulins were applied followed by streptavidin linked to alkaline phosphatase (AP) incubation (link label IHC detection system; BioGenex) according to the manufacturer. As second chromogen served Permanent AP Red (Zytomed Systems).
Quantitative Immunohistochemistry
Slides were digitalized using a slide scanner (NanoZoomer 2.0HT; Hamamatsu) and ndp.scan software (NanoZoomer Digital Pathology v2.5; Hamamatsu). Evaluation of scanned left lung tissue was performed with ndp.view software (NanoZoomer Digital Pathology version 2.5; Hamamatsu Photonics, Hamamatsu, Japan).
Each left lung was assessed for 4 immunohistochemical markers (CD68 [macrophage marker], iNOS [M1], CD206 [M2], ArgI [M2]), applying 3 double-immunolabeling protocols (CD68-iNOS, CD68-ArgI, or CD68-CD206). Hence, each lung was analyzed 3 times (1 slide for each double-immunostaining). Positively stained macrophages were identified by a red reaction product covering the cytoplasm in case of CD68 immunolabeling, eventually accompanied by a brown reaction product covering the cytoplasm for iNOS, ArgI, or CD206 immunolabeling. Only cells located in the alveolar region and showing typical AM morphology regarding size and shape were included in the analysis. Apoptotic or necrotic cells exhibiting red and/or brown reaction product were excluded from quantification. The number of macrophage cells in lung tissue per high-power field (HPF) differed between the control groups and the treatment groups, the latter being diagnosed with diffuse histiocytosis. Thus, the total lung tissue area analyzed differed between the different exposure groups. For control animals, more HPFs than for treated animals had to be evaluated to reach the number of 100 CD68+ AMs. Overall, a total of 100 CD68+ cells per lung section, scattered throughout the tissue, were counted manually. Considering the objectives of this article, macrophages present at sites of inflammation were preferentially assessed. Counting was truncated once the minimum number of 100 AMs was reached. Out of those 100 CD68+ cells, double-stained AMs were counted. Then, for each lung section, the relationship of single-/and double-labeled AM was calculated.
Statistical Analysis
Group sizes varied from 3 animals per group in the mwCNT study to 5 animals per group in the CeO2 study and 6 animals per group in the TiO2 and quartz study. Percentages of M1 or M2 marker double-positive AMs were calculated from counted CD68+ AMs. Then, data were evaluated using arcsine transformation. The arcsine transformation is indicated when dealing with percentages. It pulls the values close to 0 and 100 apart while compressing the mean values. This enables a more powerful evaluation of substantial changes, for example, from 4% to 8% compared to changes from 50% to 54%. Each dose group was compared to the concurrent control group of the respective study. Because of differences in the study design, the dose groups were not compared to each other. Therefore, an unpaired t test was performed (dose group vs concurrent control). In lungs of control animals, baseline expression of iNOS is approximately 0%. Thus, a 1-sided t test for iNOS data was chosen. Baseline expression of CD206 and ArgI is more variable, and both increase and decrease in CD206 or ArgI expression were assumed. Here, a 2-sided t test was performed. 50 Value of P < .05 was considered significant. Accordingly, labeling * for P ≤ .05 or ** for P ≤ .01 was applied. Data are presented as mean ± standard deviation.
Results
Immunohistochemistry of Histological Sections of Rat Lungs Exposed to Different (Nano)Materials
In the lungs of control animals, small, spindle-shaped CD68+ AMs were found sparsely scattered throughout the tissue. In some animals, occasionally, single macrophage-like cells were found unstained. This is consistent with previous findings, which showed a high and specific CD68 expression in the vast majority of mature, resident AMs. 51 CD68 immunostaining was restricted to the cytoplasm of macrophage cells, without any background staining. Immunolabeling of iNOS in lung tissue was found cytoplasmatically in CD68+ AMs of treated but not of control animals. Further, untreated animals showed a high abundance of CD68+CD206+ macrophages, whereby specific immunostaining was discernible in the cytoplasm. Positive signal of ArgI was observed in the nuclei of bronchial and alveolar epithelial cells as well as in the nuclei of the cells of the vasculature. Nuclear ArgI signal was also found in AMs; therefore, ArgI+ staining was defined for quantification as brown reaction product in the cytoplasm of CD68+ cells.
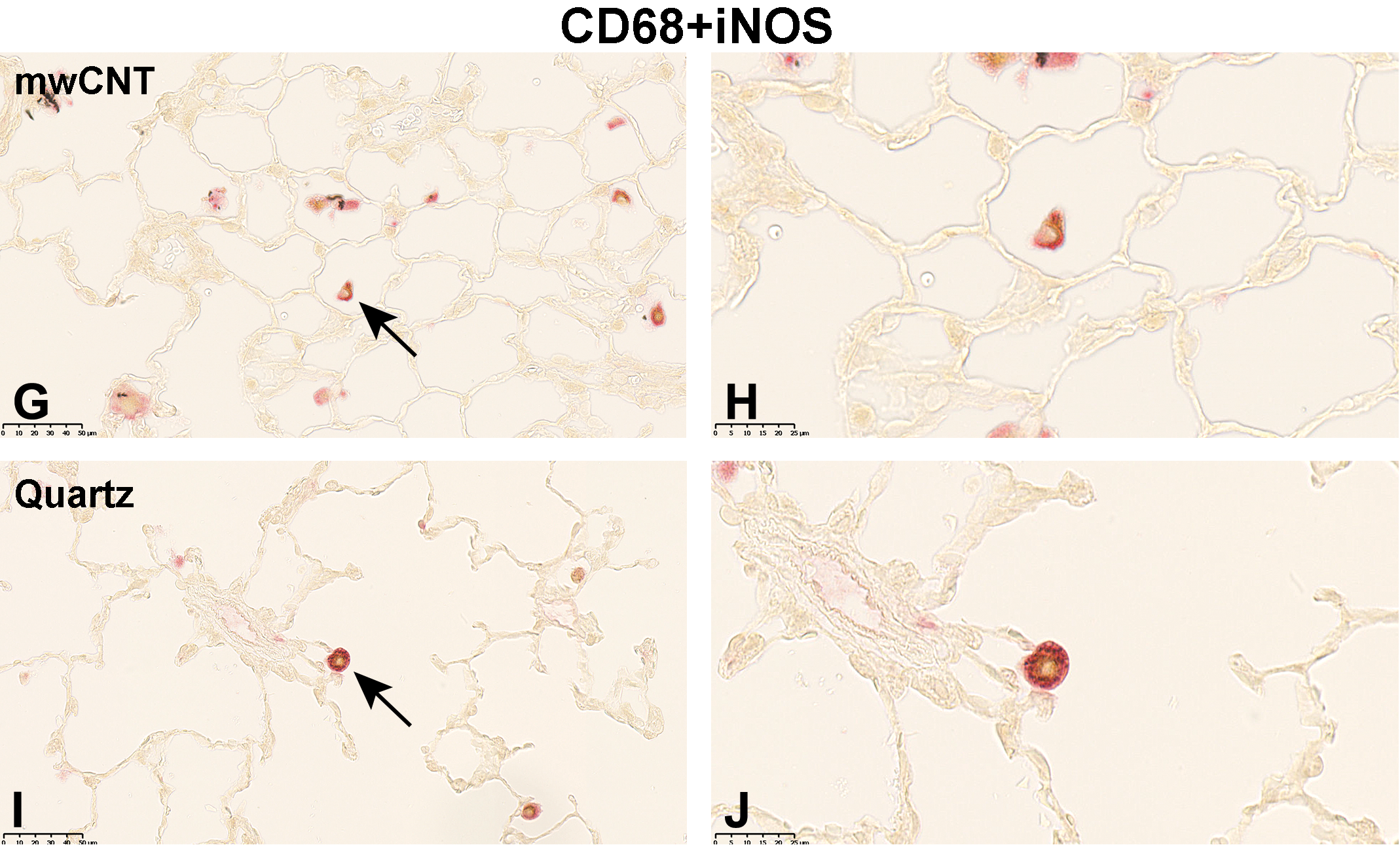
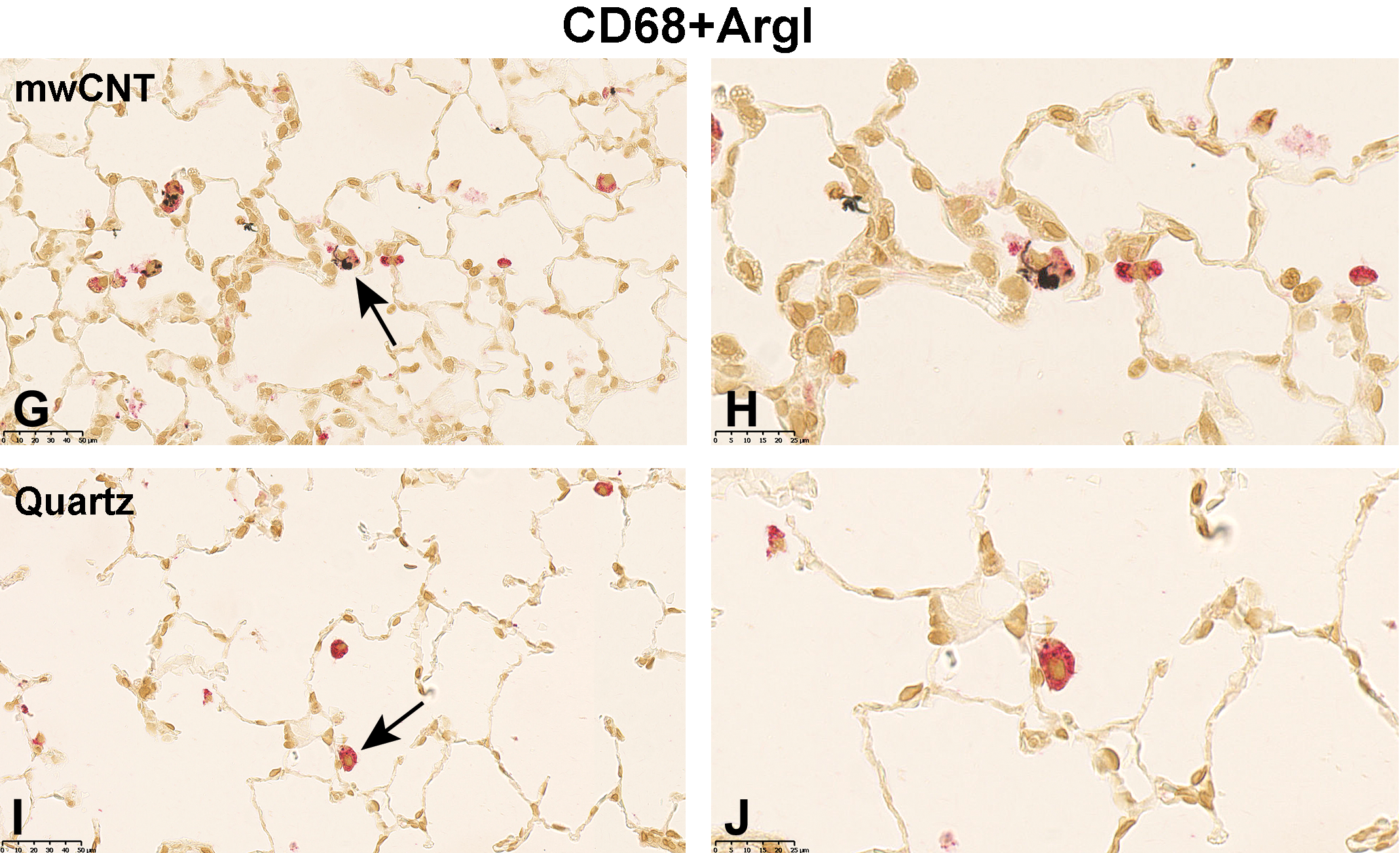
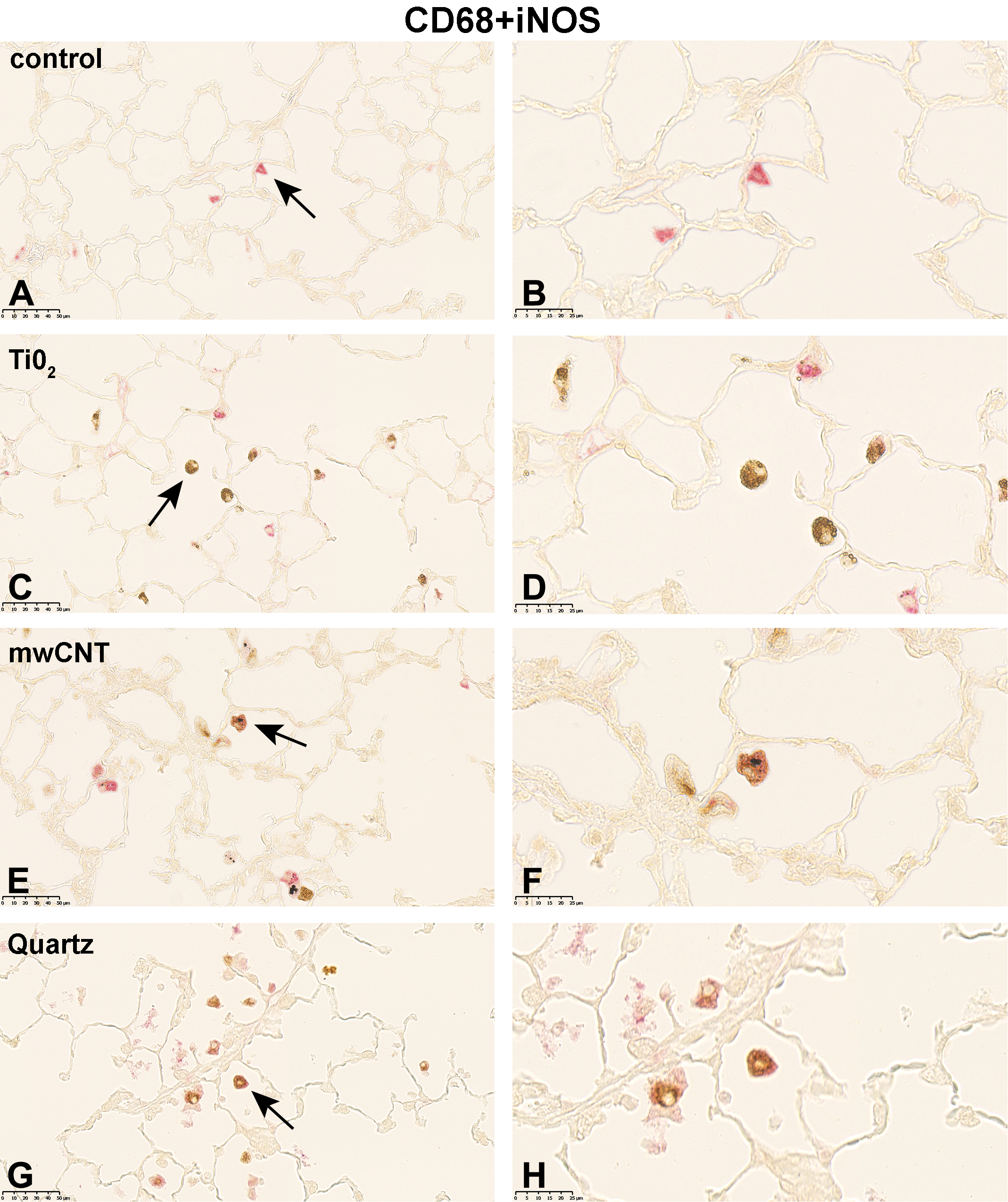
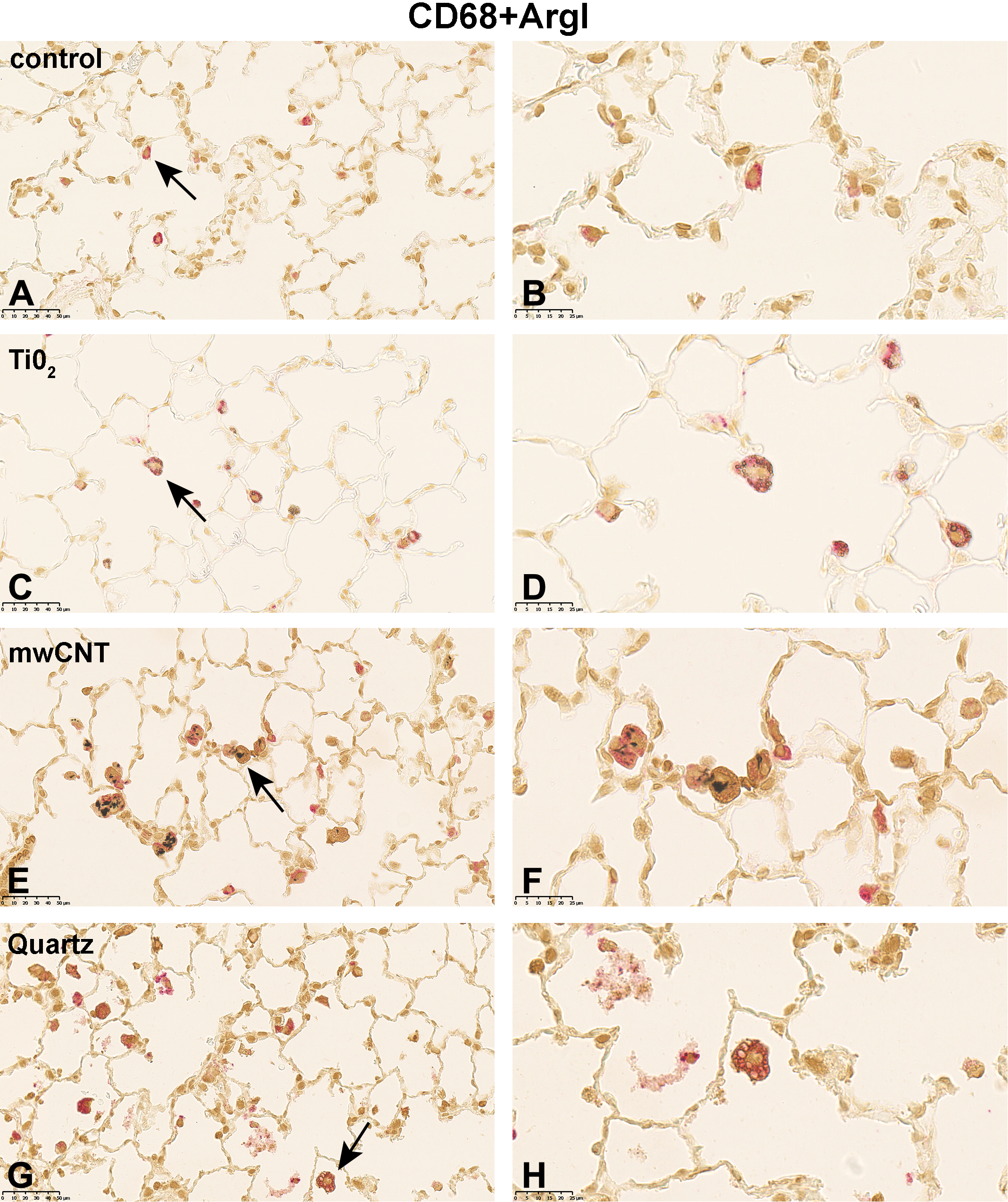
Following 5-day exposure to 100 mg/m3 TiO2, a large proportion of CD68+ AMs was fully loaded with gray, globular material, presumably TiO2 agglomerates. In addition, CD68 staining was accompanied by cytoplasmic iNOS (Figure 1C and D), ArgI (Figure 2C and D) or CD206 (Figure 3C and D) immunolabeling. In the 25 mg/m3 CeO2 treatment group, particulate matter in the cytoplasm of CD68+ AMs was noted, which was accompanied by an increase in cellular size and a more globular cell shape. Additionally, particulate matter and cell debris were found in the alveolar lumen. Immunohistochemical M1 (Figure 1E and 1F) and M2 (Figure 2E and F, and 3E and F) specific double-staining was restricted to the cytoplasm. Subsequent to the 5-day exposure to 32 mg/m3, mwCNT CD68+ AMs were laden with black fibrous structures. Cell debris as well as intact CD68+iNOS+ (Figure 1G and H), CD68+ArgI+ (Figure 2G and H), or CD68+CD206+ (Figure 3G and H) cells could be observed. In comparison, exposure of rats to 100 mg/m3 quartz resulted in considerable amount of cell debris and necrotic cells in the alveolar lumen, which were stained with red reaction product (Figures 1I and J; 2I and J; and 3I and J).
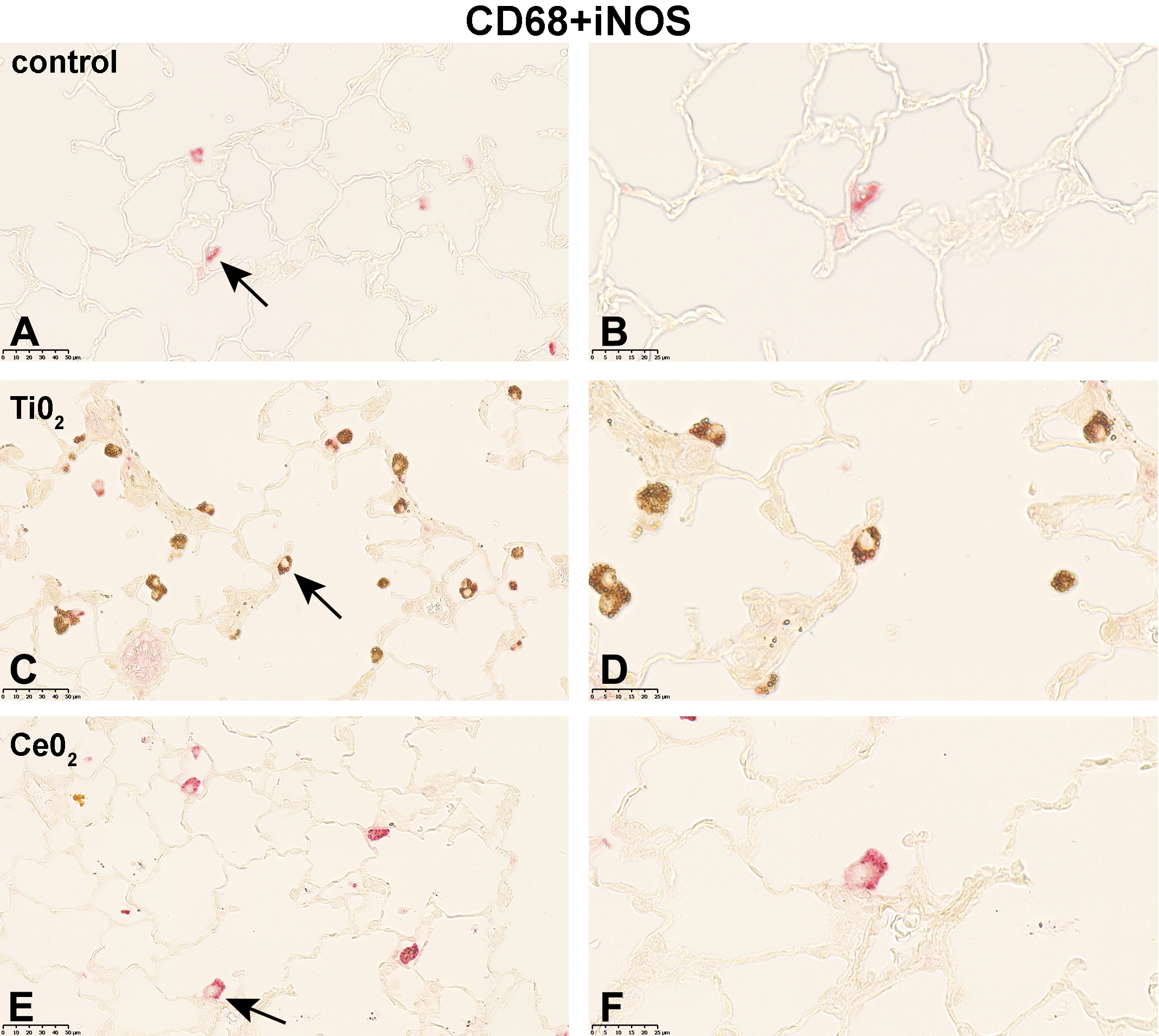
Micrographs of left lung sections of rats exposed to different (nano)materials for 5 days. Expression of iNOS (M1 marker) shortly after the last exposure was visualized by immunohistochemistry. Binding of antibodies was visualized using a red chromogen for the AM marker CD68 and a brown chromogen for the M1 marker iNOS (A, C, E, G, and I). Arrows indicate AMs shown with higher magnification in (B, D, F, H, and J). Representative sections from each treatment group are shown. Controls were exposed to air only. AM indicates alveolar macrophages.
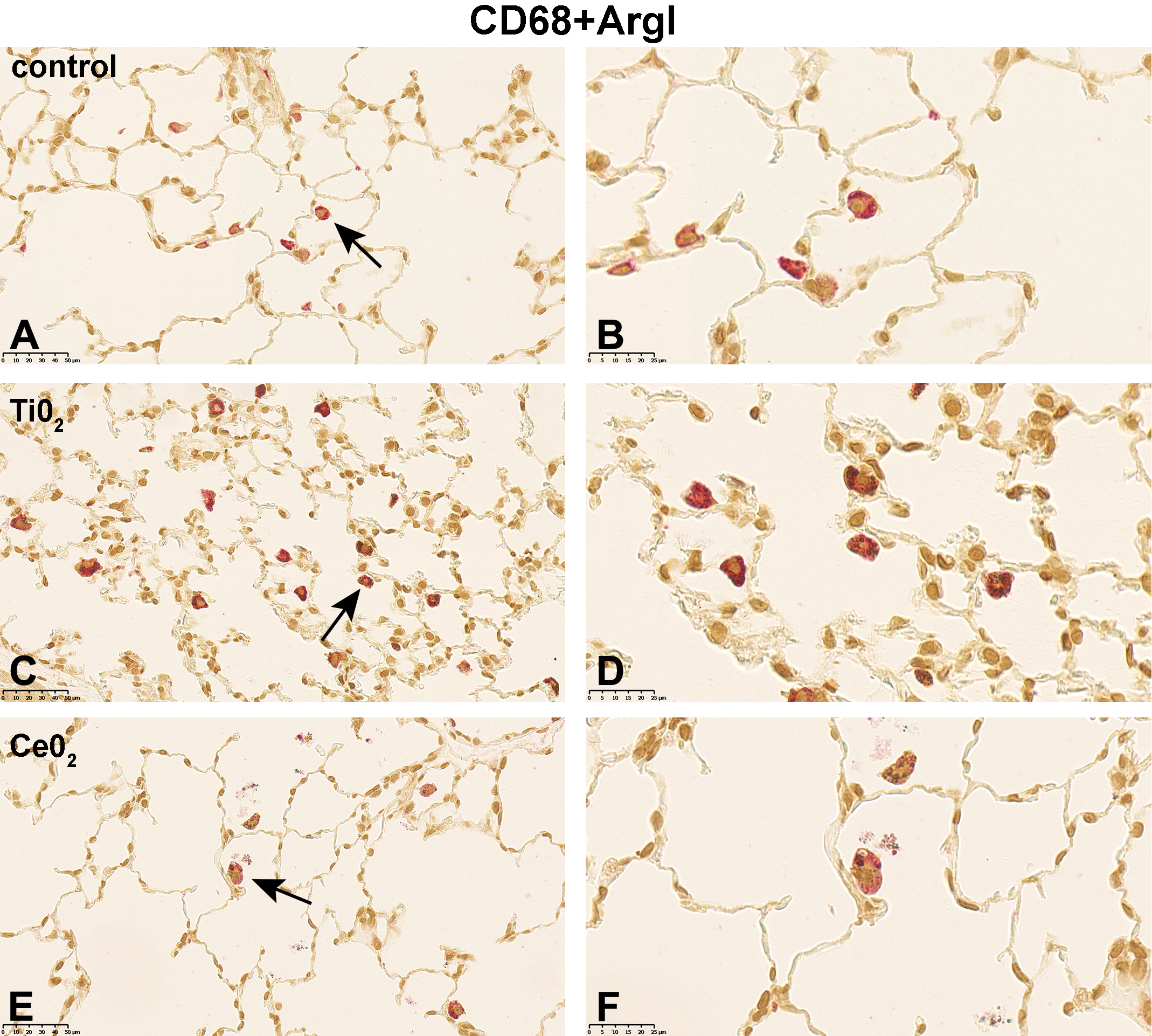
Micrographs of left lung sections of rats exposed to different (nano)materials for 5 days. Expression of ArgI (M2 marker) shortly after the last exposure was visualized by immunohistochemistry. Binding of antibodies was visualized using a red chromogen for the AM marker CD68 and a brown chromogen for the M2 marker ArgI (A, C, E, G, and I). Arrows indicate AMs shown with higher magnification in (B, D, F, H, and J). Representative sections from each treatment group are shown. Controls were exposed to air only. AM indicates alveolar macrophages.
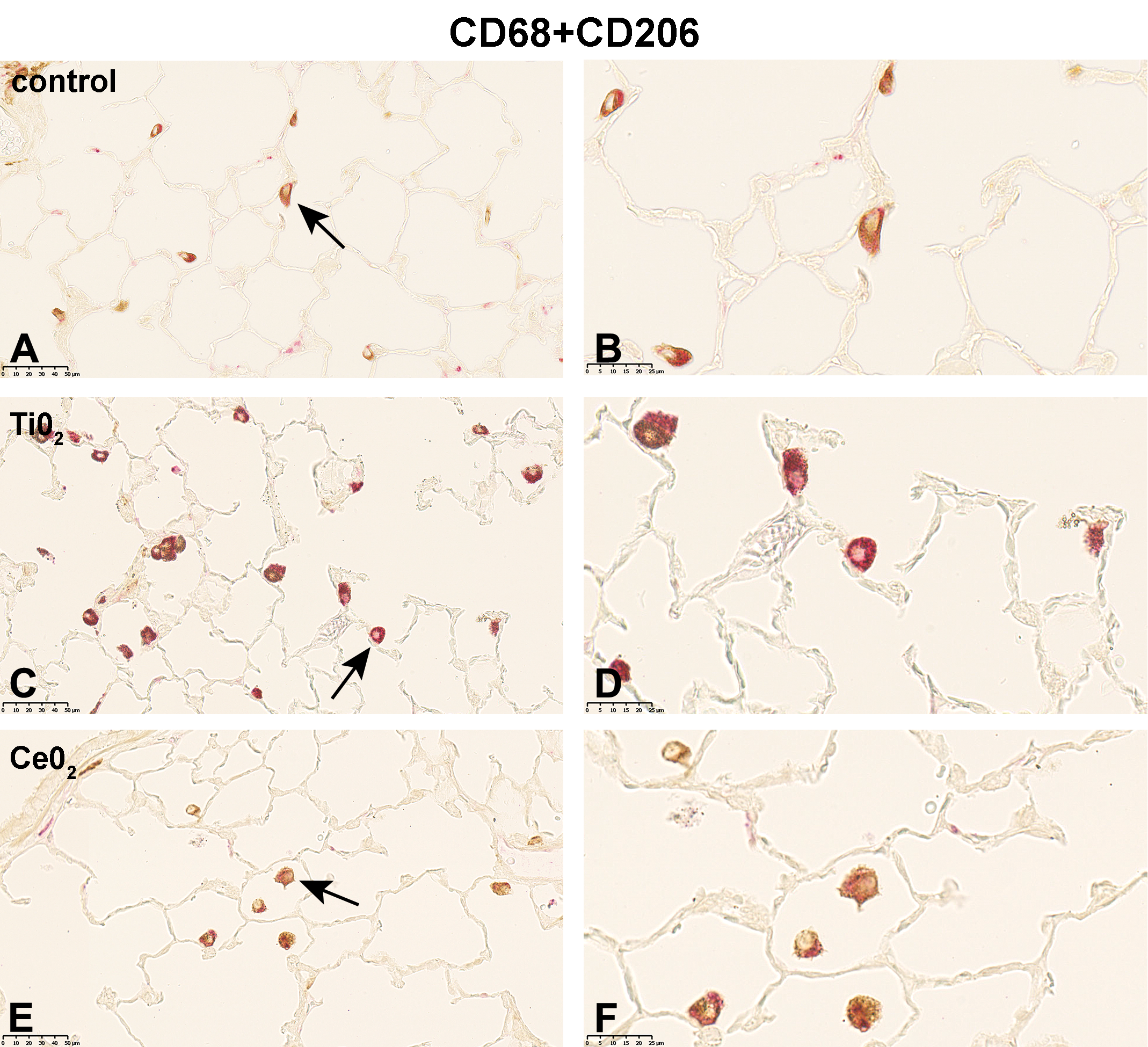
Micrographs of left lung sections of rats exposed to different (nano)materials for 5 days. Expression of CD206 (M2 marker) shortly after the last exposure was visualized by immunohistochemistry. Binding of antibodies was visualized using a red chromogen for the AM marker CD68 and a brown chromogen for the M2 marker CD206 (A, C, E, G, and I). Arrows indicate AMs shown with higher magnification in (B, D, F, H, and J). Representative sections from each treatment group are shown. Controls were exposed to air only. AM indicates alveolar macrophages.
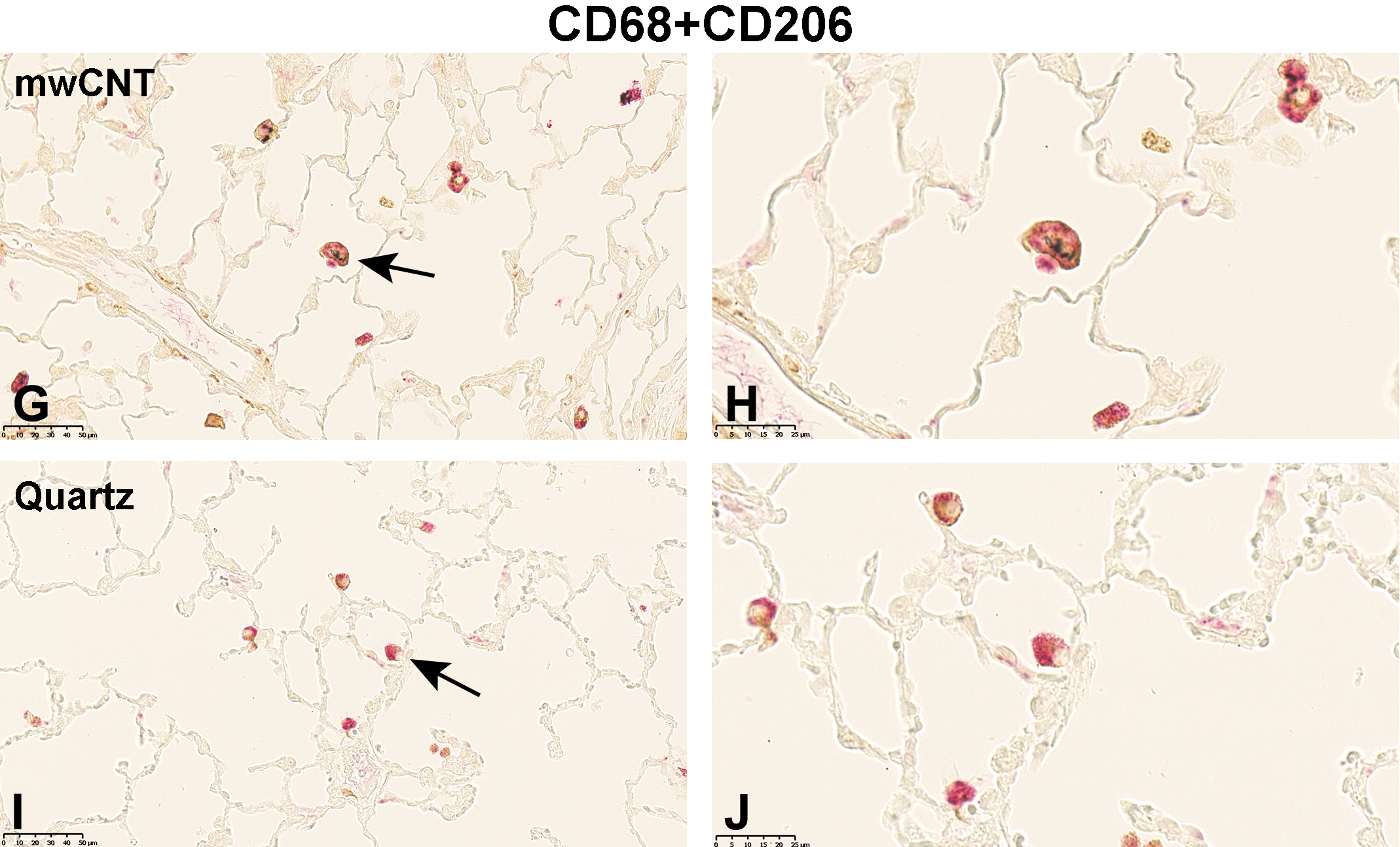
At the end of a 14-day postexposure period, following exposure to TiO2, the distribution of M1-specific immunostaining of cells changed. Only AMs that obviously internalized gray TiO2 particles were CD68+ and iNOS+ (Figure 4C and 4D). Alveolar macrophages not obviously loaded with particulate matter were CD68+ only. For the M2-specific double immunolabeling of AMs, such a separation was not noticeable. Here, CD68+ArgI+ (Figure 5C and D) or CD68+CD206+ (Figure 6C and D) immunostaining of AMs was not obviously associated with the cellular content. Twenty-one days postinhalation exposure to 32 mg/m3 mwCNT and in contrast to the TiO2 exposure group, the M1-specific immunolabeling of AMs did not show an apparent association with the staining and the cellular content (Figure 4E and F). Interestingly, the intracellular localization of the CD206 immunostaining translocated in a few cells from the cytoplasm to the cell membrane (Figure 6E and F). In rats treated with 100 mg/m3 quartz, the 14-day postexposure period resulted in AMs that were increased in size, more globular in shape, and more vacuolated. The pronounced accumulation of cell debris found in the alveolar space, which was stained with red reaction product, was very prominent. Notably, the staining pattern of CD206 changed (Figure 6G and H). So far, CD206 labeling was found cytoplasmatically; here, the immunolabeling was concentrated at the cellular surface for most of the cells, similar but more pronounced than in the mwCNT group.
Micrographs of left lung sections of rats exposed to different (nano)materials following an exposure-free period (14 days in case of TiO2 and quartz, 21 days in case of mwCNT). Expression of iNOS (M1 marker) was visualized by immunohistochemistry. Binding of antibodies was visualized using a red chromogen for the AM marker CD68 and a brown chromogen for the M1 marker iNOS (A, C, E, and G). Arrows indicate AMs shown with higher magnification in (B, D, F, and H). Representative sections from each treatment group are shown. Controls were exposed to air only. AM indicates alveolar macrophages; mwCNT, multiwalled carbon nanotubes.
Micrographs of left lung sections of rats exposed to different (nano)materials following an exposure-free period (14 days in case of TiO2 and quartz, 21 days in case of mwCNT). Expression of ArgI (M2 marker) was visualized by immunohistochemistry. Binding of antibodies was visualized using a red chromogen for the AM marker CD68 and a brown chromogen for the M2 marker ArgI (A, C, E, and G). Arrows indicate AMs shown with higher magnification in (B, D, F, and H). Representative sections from each treatment group are shown. Controls were exposed to air only. AM indicates alveolar macrophages; mwCNT, multiwalled carbon nanotubes.
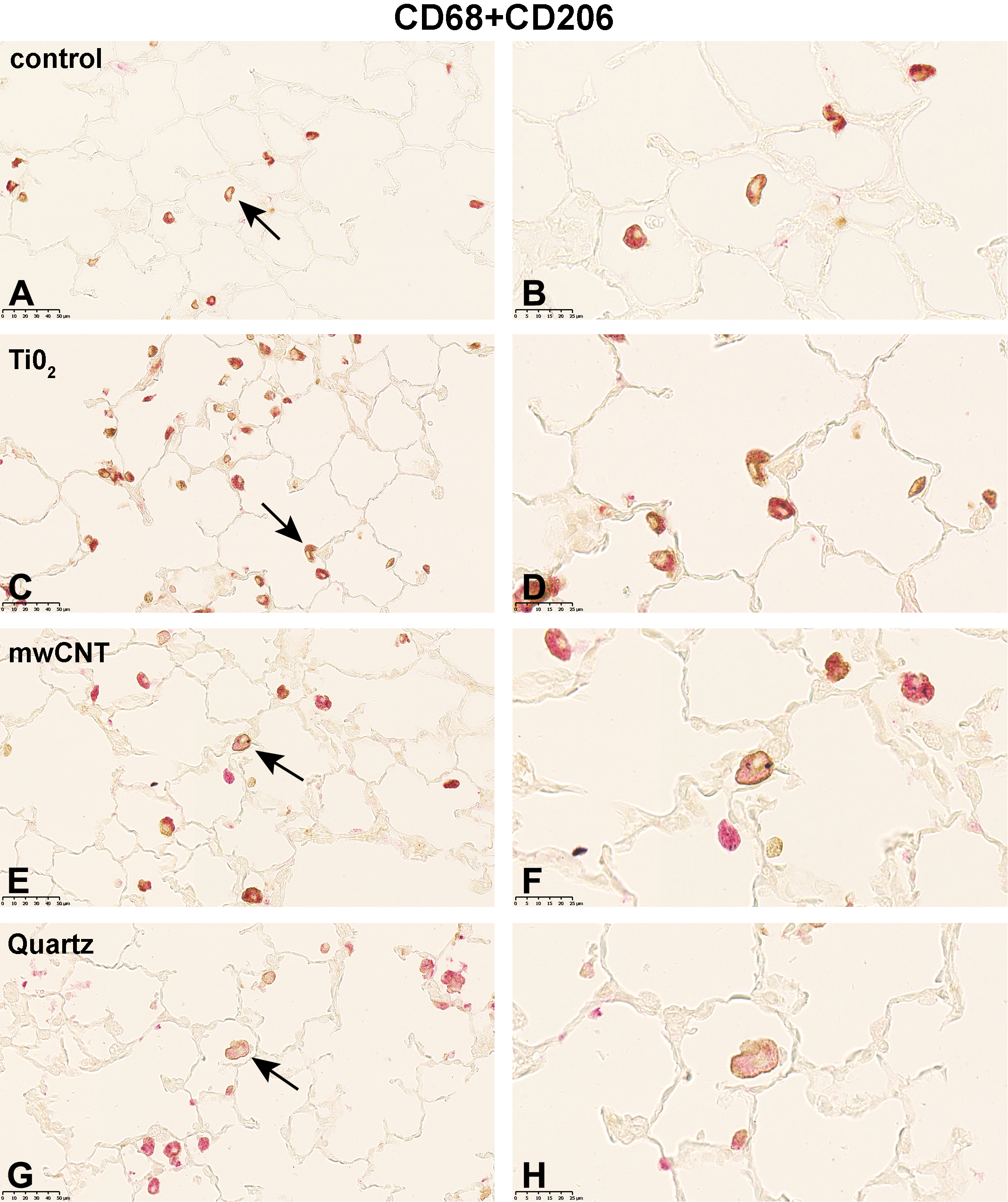
Micrographs of left lung sections of rats exposed to different (nano)materials following an exposure-free period (14 days in case of TiO2 and quartz, 21 days in case of mwCNT). Expression of CD206 (M2 marker) was visualized by immunohistochemistry. Binding of antibodies was visualized using a red chromogen for the AM marker CD68 and a brown chromogen for the M2 marker CD206 (A, C, E, and G). Arrows indicate AMs shown with higher magnification in (B, D, F, and H). Representative sections from each treatment group are shown. Controls were exposed to air only. AM indicates alveolar macrophages; mwCNT, multiwalled carbon nanotubes.
Quantitative Evaluation of the Phenotype of Immunolabeled AMs
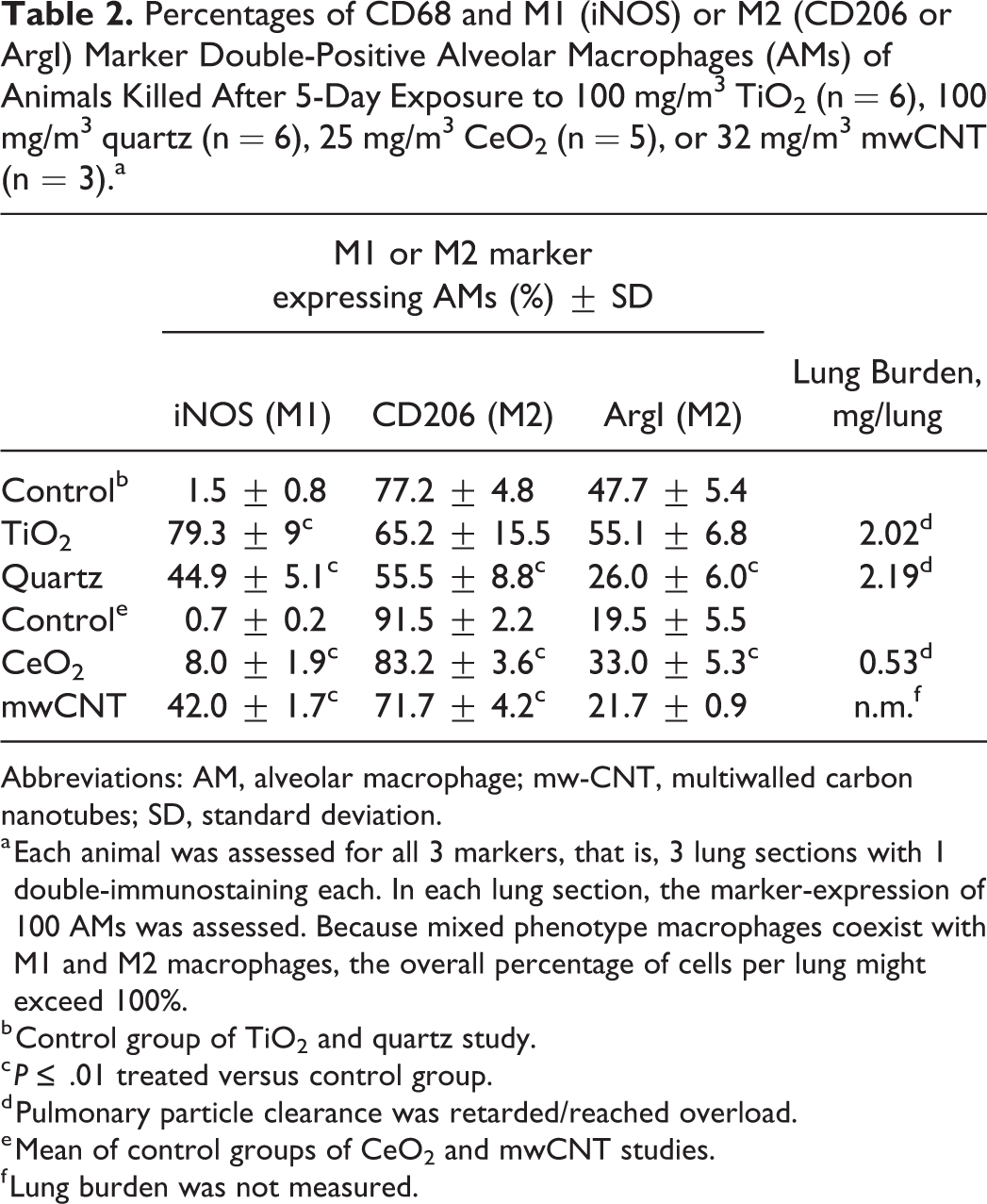
To more objectively characterize the occurrence of polarized AMs, we quantified immunohistochemically labeled AMs positive for the general AM marker CD68 and M1 (iNOS) or M2 (CD206 and ArgI) macrophage markers in lungs of (nano)material-exposed animals. In all concurrent control groups following 5-day exposure, the baseline expression of the M1 marker iNOS was low, that is, between 0% and 2% of counted cells. The exposure to TiO2, however, increased the expression of iNOS in AMs to 79%. In contrast, CeO2 exposure led to a small but significant increase in iNOS expression to 8%. In animals exposed to mwCNT, iNOS expression levels were significantly elevated to 42%. And quartz caused a significant increase up to 45% (Table 2). Further, we evaluated the expression of M2 macrophage markers after the 5-day exposure period. The percentages of CD68+CD206+ macrophages generally decreased following exposure to nanomaterials or quartz. A reduction of CD206 frequency was observed in TiO2-treated animals (65%) compared to control animals (77%), however, not significant. A slight but significant decrease was observed in CeO2- (control: 93%, CeO2: 83%) and mwCNT- (control: 90%, mwCNT: 72%) exposed animals. And a marked decrease was observed following exposure to quartz (control: 77%, quartz: 56%; Table 2). Quantitative analysis of ArgI expression revealed variable values of the control groups of the 3 different inhalation studies (Tables 2 and 3). One possible explanation is that the animals of the TiO2 and quartz study were subjected to surgery (implantation of BrdU-minipumps). This might have affected the immunological state of the animals. Compared to the concurrent control group of the TiO2 and quartz study, ArgI expression in TiO2-exposed animals was not altered (control: 48%, TiO2: 55%), whereas it significantly decreased in quartz-exposed animals to 26%. CeO2 exposure led to a significant increase in the percentage of ArgI+ cells (33%) compared to the control (17%). The exposure to mwCNT, however, did not alter the expression of ArgI in treated (22%) compared to control (22%) animals (Table 2).
Percentages of CD68 and M1 (iNOS) or M2 (CD206 or ArgI) Marker Double-Positive Alveolar Macrophages (AMs) of Animals Killed After 5-Day Exposure to 100 mg/m3 TiO2 (n = 6), 100 mg/m3 quartz (n = 6), 25 mg/m3 CeO2 (n = 5), or 32 mg/m3 mwCNT (n = 3).a
Abbreviations: AM, alveolar macrophage; mw-CNT, multiwalled carbon nanotubes; SD, standard deviation.
a Each animal was assessed for all 3 markers, that is, 3 lung sections with 1 double-immunostaining each. In each lung section, the marker-expression of 100 AMs was assessed. Because mixed phenotype macrophages coexist with M1 and M2 macrophages, the overall percentage of cells per lung might exceed 100%.
b Control group of TiO2 and quartz study.
c P ≤ .01 treated versus control group.
d Pulmonary particle clearance was retarded/reached overload.
e Mean of control groups of CeO2 and mwCNT studies.
f Lung burden was not measured.
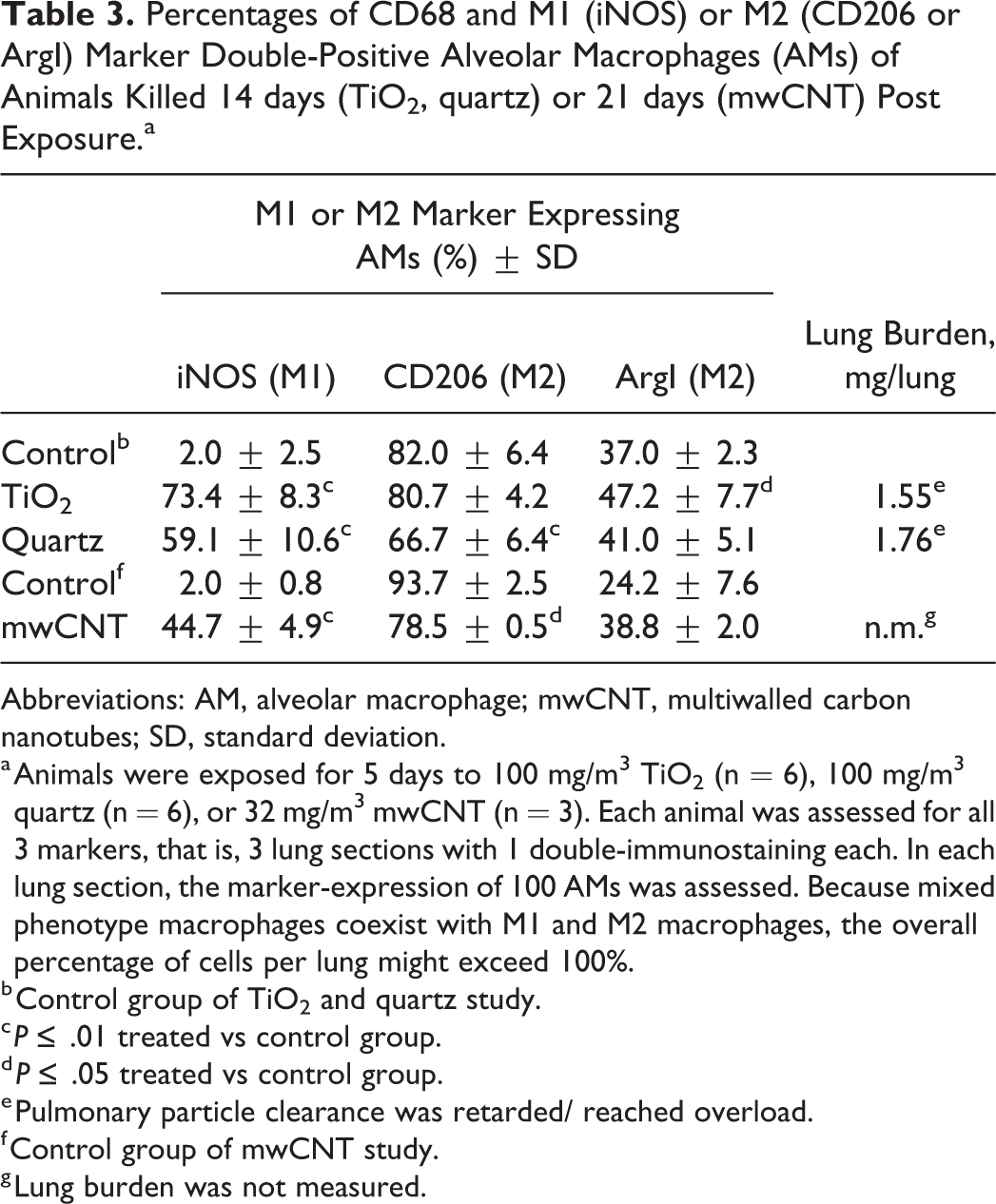
Percentages of CD68 and M1 (iNOS) or M2 (CD206 or ArgI) Marker Double-Positive Alveolar Macrophages (AMs) of Animals Killed 14 days (TiO2, quartz) or 21 days (mwCNT) Post Exposure.a
Abbreviations: AM, alveolar macrophage; mwCNT, multiwalled carbon nanotubes; SD, standard deviation.
a Animals were exposed for 5 days to 100 mg/m3 TiO2 (n = 6), 100 mg/m3 quartz (n = 6), or 32 mg/m3 mwCNT (n = 3). Each animal was assessed for all 3 markers, that is, 3 lung sections with 1 double-immunostaining each. In each lung section, the marker-expression of 100 AMs was assessed. Because mixed phenotype macrophages coexist with M1 and M2 macrophages, the overall percentage of cells per lung might exceed 100%.
b Control group of TiO2 and quartz study.
c P ≤ .01 treated vs control group.
d P ≤ .05 treated vs control group.
e Pulmonary particle clearance was retarded/ reached overload.
f Control group of mwCNT study.
g Lung burden was not measured.
After an exposure-free period of 14 days, the percentage of iNOS-positive cells in animals exposed to TiO2 remained elevated at 73.4% but decreased compared to values 1 day postexposure. Similarly, the increased iNOS expression values in animals exposed to mwCNT did not change following a postexposure period of 21 (45%) days. In contrast, animals exposed to quartz exhibited even higher percentage of iNOS+-positive cells, significantly increased to 59% compared to the control and the 5-day exposure group (Table 3). Further, CD206 expression in the TiO2 exposure group recovered to 81% (control: 82%), whereas it remained significantly decreased in mwCNT (79%, control: 94%) and quartz (67%, control: 82%) exposure groups (Table 3). The careful evaluation of the ArgI expression levels of the postexposure groups revealed a general increase. In TiO2-treated animals, ArgI expression remained elevated at 47% compared to 37% of the concurrent controls. For the mwCNT- and quartz-treated groups, a progressive increase in ArgI expression levels to 39% and 41% could be observed 14 and 21 days postexposure, respectively (Table 3).
Discussion
The present investigation was conducted to assess whether possible differences in early AM polarization upon short-term (5 days) inhalation exposure correlates with variable histopathological findings of poorly soluble (nano)materials, especially regarding the possible onset of granulomatous inflammation. Using an immunohistochemical approach, we identified AM subpopulations, which appeared in lung tissue following exposure to poorly soluble (nano)materials. Further, we quantitatively analyzed the initial macrophage polarization shortly after 5-day exposure and the polarization state seen after 14 or 21 days postexposure. TiO2, CeO2, mwCNT, and quartz served as model substances, representing thoroughly studied materials with high biopersistence and qualitatively different histopathological effects (Tables 1 and 4). However, the available FFPE specimen did not allow for mechanistic investigations or high-throughput quantification as quantitative real-time polymerase chain reaction or flow cytometry of nonparaffinized tissue would have allowed, respectively. However, immunohistochemistry allowed specific assessment of polarized AMs in the alveolar lumen in situ, without the need for sophisticated immunophenotyping of pulmonary immune cells. In addition, it was repeatedly described that bronchoalveolar lavage techniques are not capable of detaching sessile macrophages out of the lungs. 55,56 Furthermore, using an immunohistochemical approach restricted the current investigations to a simplistic classification of macrophages exhibiting merely a pro-inflammatory M1 or anti-inflammatory M2 phenotype. Future studies, using for instance flow cytometry, could provide more detailed phenotyping of macrophages. For more information on cytokine expression, please refer to the BALF analysis of the respective study publication. Measured lung burdens are summarized in Table 4. Similarly, assessing biokinetics was not the motivation of the current study; therefore, please refer to the respective publications. Furthermore, there is an emerging concept that AMs and interstitial macrophages within rodent lungs are differentially recruited 57 following exposure to particles 58 and that they differ in their cytokine secretion profiles. 57,59 Investigations in macaques 60 revealed that both cell types, AMs as well as interstitial macrophages, are CD68+. Thus, differentiation between resident AMs and recruited interstitial macrophages in the alveolar lumen is not possible with the current study design. However, this was not the focus of the present study.
Summary of the Test Results Obtained in the Present Study for Alveolar Macrophage Polarization (%) and Obtained in Published Inhalation Studies, Which Measured Lung Burden and Examined Histological Lung Sections After Short-Term or Subchronic Exposure to Different (Nano)Materials.
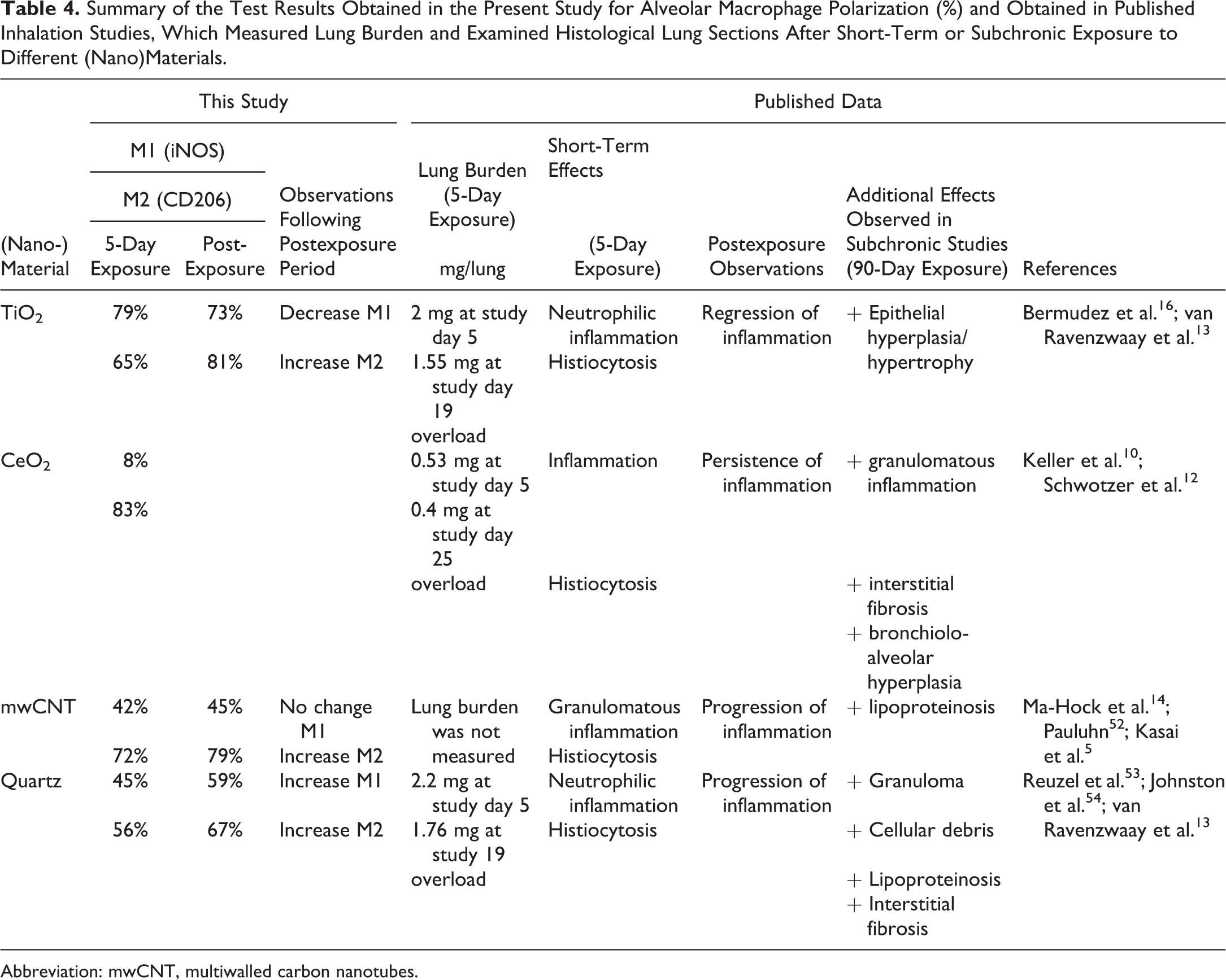
Abbreviation: mwCNT, multiwalled carbon nanotubes.
Immunohistochemical Method for Labeling AMs in Lung Sections Using Macrophage Markers
Our immunohistochemical observations of the expression of CD68, iNOS, ArgI, and CD206 are in line with previous studies. CD68, often handled as the pan’ macrophage marker, was discussed not to be a suitable marker for macrophages in general but rather a marker indicative of phagocytosis. 61,62 However, we found high expression of CD68 in AMs and only rarely single cells with macrophage-like appearance throughout all study groups not labeled for CD68, resembling the findings of Zaynagetdinov et al. 51 Therefore, we considered immunolabeling of cells with anti-CD68 antibody as a suitable tool to identify AMs.
iNOS serves commonly as marker of pro-inflammatory macrophages (M1). 37 It becomes early induced in the frame of a pro-inflammatory immune response. Like other investigators, we observed no expression of iNOS in control animals but strong iNOS induction in AMs of treated animals. 63,64 Thus, we found iNOS a specific and suitable immunohistochemical marker for the identification of M1 macrophages.
Further, and in accordance with previous observations, 43,65 we detected the enzyme ArgI (M2 marker), which was initially described as a cytoplasmic enzyme, 66 in the cytoplasm and nuclei of AMs and within the nuclei of other cell types of histological lung sections. Available data demonstrate a colocalization of arginase with heterochromatin, suggesting an important role for arginase, or its family, in nuclear events related to cell cycle progression. 67 In addition, ArgI immunohistochemical labeling was previously observed in rat bronchial and alveolar epithelial cells, 68 and it was shown that ArgI plays an important role in physiological blood vessel function when expressed in endothelial and smooth muscle cells. 69,70 Considering the available data, our observations in the present study are in accordance with previously published results, and thus, the use of cytoplasmic ArgI immunolabeling for M2 identification using the current staining protocol seemed well justified.
As reported earlier, the mannose receptor CD206 is highly expressed by AMs under homeostatic conditions. 51,71 In addition, it was early recognized as an important marker for alternative activation of macrophages (M2). 72 Accordingly, we found specific and abundant immunolabeling for CD206 of AMs of control animals and used CD206 as a marker for M2 macrophage identification. The combination of cell membrane and cytoplasmic immunostaining derives from the fact that CD206 continuously recycles between the surface membrane of macrophages and intracellular phagolysosomal compartments. 73,74 Therefore, cytoplasmic occurrence is repeatedly seen in immunohistochemical investigations. 62,64
Polarization of AM Correlates With Histopathological Findings and the Postexposure Pro- or Regression
As previously reported, 5-day inhalation exposure to the (nano)materials examined caused pulmonary inflammation that was diagnosed by light microscopic examination of HE-stained slides and changes in respective BALF parameters. 10,13,14 Histological findings were consistent with the original publications (Table 1). In accordance with the ongoing inflammatory process, our examination using immunohistochemistry followed by quantitative evaluation revealed relative increase in the expression of the M1 marker iNOS in CD68+ AMs, which was accompanied by a decrease in the M2 marker CD206. This is consistent with the pro-inflammatory characteristics of M1 and anti-inflammatory characteristics of M2 macrophages. The most significant relative increase of M1 macrophages was observed in TiO2-exposed animals, while the decrease of M2 macrophages was most prominent in quartz-exposed animals. Although TiO2 (nano)particles were described as poorly soluble and of low toxicity, several studies suggested that TiO2 might induce cellular oxidative stress and ultimately lead to the expression of pro-inflammatory mediators, including pro-inflammatory enzymes such as iNOS. 75 –77 Noteworthy, CeO2 elicited pulmonary inflammation similar to the other materials investigated, but iNOS expression levels were only slightly increased compared to the others. Notably, inflammation caused by 25 mg/m3 CeO2 dust aerosol concentration progressed to a granulomatous inflammation following 28-day exposure plus 34 days postexposure. 10 Given the fact that CeO2-induced inflammatory changes that included granulomatous inflammation following a post-exposure period, more detailed investigations might unravel possible differences in the initiation and progression of effects.
Following an exposure-free period, light microscopic examination of HE-stained lung sections and BALF analysis of the original publications revealed that biological effects elicited by the investigated (nano)materials changed differently over time (regression, persistency or progression of effects induced by TiO2, mwCNT or quartz, respectively). 10,13,14 Correspondingly, differences were seen in temporal patterns of AM subsets depending on the nanomaterial. In general, our quantitative analysis revealed that the occurrence of M1 and M2 macrophages correlated with the persistence, progression, or regression of pulmonary inflammation. For instance, CD68+iNOS+ M1 macrophage percentages decreased, whereas CD68+CD206+ M2 macrophage percentages recovered in case of resolution of inflammation. This statement is based on the observations made for TiO2, where a decline of inflammatory parameters in BALF and a disappearance of granulocytic infiltration in lung tissue indicated a regression of the inflammatory response. It remains to be elucidated whether this applies also to other materials showing transient inflammatory responses in rat lungs. In case of persistent pulmonary inflammation, increased CD68+iNOS+ (M1) as well as decreased CD68+CD206+ (M2) cell numbers did not substantially change over time, although exposure was terminated. This was observed for animals exposed to 32 mg/m3 mwCNT, where granulomatous inflammation persisted 21 days postexposure. Furthermore, progressive inflammation was correlated with additional relative increase of M1 macrophages after an exposure-free period. For example, quartz histopathological findings increased in severity during the postexposure period, which was paralleled with a significant relative increase of CD68+iNOS+ AMs when compared to the control and the exposure group analyzed immediately following the last exposure.
Interestingly, only in TiO2-exposed animals of the postexposure group, iNOS expression was restricted to AMs harboring grey, globular material, assumed to be TiO2 agglomerates. CD68+ AMs not obviously laden with particulate matter did not show iNOS+ immunolabeling in the postexposure group. One explanation for this might be that TiO2 particles themselves induced intracellular signaling, which gave rise to iNOS expression, either through binding to respective effector proteins or through their surface reactivity, which might have disturbed chemical cell homeostasis. Another explanation is that the local pulmonary microenvironment changed during the postexposure period from a pro- to an anti-inflammatory one. Monocytes, which entered the lung at a late time point, where engulfment of TiO2 particles from the alveolar surface was already accomplished and resolution of inflammation was already ongoing, did not encounter M1 inducing signals.
In addition, we observed a change in localization of the immunolabeling of CD68+CD206+ AMs for 2 postexposure groups. Intracellular localization of CD206 changed from a diffuse (cell membrane and cytoplasm) to a cell membrane–restricted manner, to some extent in the mwCNT postexposure group (Figure 6E and F) and to a greater extend in the quartz postexposure group (Figure 6G and H). The reason for and relevance of this observation is unclear. However, the importance of intact intracellular trafficking of the phagocytic receptor CD206 becomes particularly apparent when considering its role in the resolution of inflammation. 33,78,79
In addition to CD206, ArgI is a well-known and important marker for M2 polarization of macrophages. 27,80 Several studies point to the crucial role of the arginine metabolism in immune responses and the herewith associated regulation of iNOS and ArgI expression. 81 –84 Because M2 macrophages exert a wide spectrum of immunological functions, which led to subdividing them into different subtypes, 85 we chose 2 markers for M2 identification, each indicative for the different processes macrophages are involved in. In the present study, ArgI expression was found to be variable between the control groups of the 3 different inhalation studies. One possible explanation is that the animals of the TiO2 and quartz study were subjected to surgery. For the purpose of assessing the proliferation rate of bronchial cells, small BrdU-containing minipumps were implanted beneath the skin of the experimental animals on the second day of exposure. The implantation of the BrdU-minipumps might have affected the immunological state of the animals. Thus, careful data analysis should be conducted, and values of treatment groups should be compared to the concurrent control group of the respective study only. When comparing the ArgI values of the treatment groups with the respective concurrent control group, CD68+ArgI+ macrophages were significantly upregulated in CeO2-exposed animals and significantly downregulated in the quartz exposure group. This observation might trigger speculations, considering that ArgI is suspected to be involved in granuloma formation and healing processes, 82,86,87 and CeO2 causes granulomatous inflammation and quartz progressive inflammation and tissue destruction. However, although granulomatous inflammation was also observed in mwCNT-exposed animals, ArgI expression was not changed. Nevertheless, we consider our data as valid because several studies reported contradictory results on the role of ArgI in tissue repair processes, fibrosis, and granuloma formation. 81 –83,88 –90 Thus, the exact role of ArgI in pulmonary inflammation and fibrosis is still not fully resolved.
An increasing body of data demonstrates that nanomaterials differentially influence macrophage polarization. TiO2 as well as quartz particles were repeatedly found to differently influence M1- and M2-specific protein expression. 39,77,91 –94 Further, there is first evidence that CeO2 and mwCNT might affect macrophage polarization as well. 90,95 –99 However, several aspects should be considered when investigating macrophage polarization and biological effects of inhaled nanomaterials in vivo. Obviously, the dose is a central element in eliciting toxicity, in addition, duration of exposure, but also dose rate should be considered. All these factors will differently influence the spatiotemporal dynamics of M1 and M2 polarization. For instance, Kumar et al found a dose-dependent shift in macrophage polarization. 92 Others found a time-dependent sequential switch in the appearance of macrophage subpopulations. 64,90,96,100 Interestingly, first evidence points out that the crucial balance of M1 and M2 immune reactions is not necessarily sequential, but there are various pathways, which are early stimulated at the same time and which seem to be independent from each other, each leading to a different aspect of the observed pathophysiology. 101 –103 Unraveling these pathways will add to our understanding of the pathogenesis of morphological changes observed, the exact contribution of AMs, and might identify powerful, specific biomarkers, which potentially allow for the prediction of long-term outcome following nanomaterial exposure. 77,104,105
In conclusion, the overall appearance of pro-inflammatory M1 and anti-inflammatory M2 macrophages correlated with histopathological findings indicative for pulmonary inflammation upon short-term (5 days) inhalation exposure at the end of the exposure as well as after a postexposure period (14 or 21 days). Initial pulmonary inflammation correlated with a strong upregulation of M1 and a downregulation of M2 AMs, and sequential changes in macrophage polarization correlated with changes in histopathological findings postexposure. However, AM polarization was not indicative for the different quality of histopathological findings observed with different (nano)materials at later time points in longer term studies. Between the 4 investigated (nano)materials, no substantially different expression pattern of M1 or M2 AMs at an early time point (5 day exposure) or 14/21 days postexposure was seen, which could explain the different histopathological patterns (especially granulomatous inflammation) observed at later time points in longer term studies. A distinct induction of AM polarization might indicate different underlying initiating events, which in turn might explain the various histopathological outcomes after long-term exposure to different inhaled (nano)materials. Unraveling the underlying pathways might identify biomarkers, which could allow for the prediction of the quality of histopathological outcome following long-term exposure. For this, a more in-depth understanding of the underlying processes of nanomaterial-induced histopathological changes and the role of AMs in the pathophysiology is needed. Investigations on biokinetics and cytokine-mediated signaling will add to this understanding. Additional or more specific early markers of long-term inhalation outcomes will promote further development of scientifically sound adverse outcome pathways for inhalation hazard of biopersistent particulate matter.
Supplemental Material
TPX896347_Supplement_Figure_1a-e - Appearance of Alveolar Macrophage Subpopulations in Correlation With Histopathological Effects in Short-Term Inhalation Studies With Biopersistent (Nano)Materials
TPX896347_Supplement_Figure_1a-e for Appearance of Alveolar Macrophage Subpopulations in Correlation With Histopathological Effects in Short-Term Inhalation Studies With Biopersistent (Nano)Materials by Johanna Koltermann-Jülly, Lan Ma-Hock, Sibylle Gröters and Robert Landsiedel in Toxicologic Pathology
Supplemental Material
TPX896347_Supplement_References - Appearance of Alveolar Macrophage Subpopulations in Correlation With Histopathological Effects in Short-Term Inhalation Studies With Biopersistent (Nano)Materials
TPX896347_Supplement_References for Appearance of Alveolar Macrophage Subpopulations in Correlation With Histopathological Effects in Short-Term Inhalation Studies With Biopersistent (Nano)Materials by Johanna Koltermann-Jülly, Lan Ma-Hock, Sibylle Gröters and Robert Landsiedel in Toxicologic Pathology
Footnotes
Acknowledgments
The authors gratefully thank Claus-Michael Lehr for helpful discussion and Heike Marxfeld for valuable discussion on IHC. They particularly thank Thomas Tatarewicz and Hans-Robert Hofmann for excellent laboratory support on IHC, and Martina Dammann for valuable help with data transformation.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Johanna Koltermann-Jülly (J.K.-J.), Lan Ma-Hock (L.M.-H.), Sibylle Gröters (S.G.), and Robert Landsiedel (R.L.) are employees of BASF SE, a company producing nanomaterials.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
