Abstract
Bixin is a natural red pigment extracted from annatto. Although it is widely used as a coloring agent in food, there are few studies about the effect of this carotenoid on DNA. This study aimed to investigate the effects of bixin on cytotoxicity and genotoxicity induced by doxorubicin in HL60 cells. At concentrations above 0.3 μg/mL, bixin demonstrated cytotoxic effects in HL60 cells. Furthermore, this carotenoid was neither mutagenic nor genotoxic to HL60 cells and reduced the DNA damage induced by doxorubicin. Bixin and doxorubicin showed no apoptotic effect in HL60 cells, but the simultaneous combined treatments showed an increase in the percentage of apoptotic cells. In conclusion, our results showed that bixin modulates the cytotoxicity of doxorubicin via induction of apoptosis. The results of this study provide more knowledge about the toxic effects of anticancer treatments and how the natural compounds can be useful on these therapeutic approaches.
Introduction
Bixin is a natural red dye found in the seed of annatto tree (Bixa Orellana L.), which has generated a growing interest in the scientific community since it is the only natural source of this carotenoid pigment. 1 –3 According to Giuliano et al. 4 Latin America produces about 60% of annatto, followed by Africa (27%) and Asia (12%). The red color of the annatto seed is directly related to the percentage of the soluble fraction of annatto extract bixin (C25H30O4), which is approximately 80%. 5 Bixin is used as a natural colorant in various formulations in food industry, such as in cheese, butter, sausage, and ice cream. 4,5
Annatto has several bioactive properties, such as antioxidant and antiproliferative activities. 6 –8 Compared to other natural carotenoids, bixin is one of the most effective scavengers of the singlet oxygen molecule and may contribute to the protection of cells and tissues against the deleterious effects of free radicals. 5,9,10 Bixin inhibited the proliferation and decreased the viability of tumor cells in dose- and time-dependent manners, induced cell apoptosis, and interfered with cell cycle progression. 11
To evaluate these effects of bixin in combination with an antitumor agent for inhibition or potentiation of chemotherapeutic effect, analyzes were performed with doxorubicin. Doxorubicin is an anthracycline antibiotic highly effective against several cancers. Different mechanisms have been proposed for its antitumor effects, including intercalation in the double helix of DNA with subsequent inhibition of DNA synthesis, DNA alkylation and formation of cross-links, inhibition of topoisomerase II, and activation of apoptotic signaling pathways, being able to produce reactive oxygen species blocking replication of genetic material. 12 –16
In our study, promyelocytic leukemia cell line HL60 was used to evaluate the cytotoxicity and DNA damage of bixin as well as their protective effects against doxorubicin-induced DNA damage.
Materials and methods
Sample preparation and reagents
Dry annatto seeds were collected in the city of Ribeirão Preto, São Paulo State, Brazil. Bixin was extracted from annatto seeds after being washed with ethyl acetate, hexane, and methanol. The solvents were completely evaporated in a roto-evaporator (Büchi Labortechnik, Flawil, Switzerland), and crystallization was performed with dichloromethane/ethanol (1:4). Bixin crystals were formed with 93–95% purity (determined by high-performance liquid chromatography–photodiode array analysis) and were stored under refrigeration and protected from light until use. 17 All solutions containing bixin were prepared immediately prior to use.
Bixin was dissolved in dimethyl sulfoxide (DMSO, Sigma-Aldrich, St Louis, Missouri, USA) at a final concentration of 0.4%. Doxorubicin (DXR) was purchased from Laboratório Bergamo (Taboão da Serra, São Paulo, Brazil). Dulbecco’s modified Eagle’s medium (DMEM), fetal bovine serum (FBS), and the penicillin–streptomycin mix were purchased from Gibco (Carlsbad, California, USA). Normal melting point and low-melting point agaroses were obtained from Invitrogen (Carlsbad, California, USA). All other chemicals were of analytical grade with the highest purity available.
Cell line
The human promyelocytic leukemia cell line (HL60) was obtained from American Type Culture Collection (ATCC, Rockville, Maryland, USA) and cultured in DMEM containing 10% heat-inactivated FBS and 1% antibiotic mix (penicillin/streptomycin). The cells were maintained at 37°C in a humidified atmosphere of 5% carbon dioxide (CO2) and 95% air in an incubator (Forma Series II, Thermo Electron Corporation, Beverly, Massachusetts, USA). In all experiments, the cells were incubated 24 h before bixin and/or cisplatin treatments.
MTT assay
HL60 cells (1 × 105 cells/well in 96-well plates) were incubated at 37°C for 24 h with different concentrations of bixin (0.01–3.00 µg/mL), DXR (0.01–3.00 µg/mL), or a simultaneous combination of both (bixin + DXR). A negative control containing only cells and a solvent control (cells treated with 0.4% DMSO) were also evaluated. After treatment, the plates were incubated in the 3-[4,5-dimethylthiazol-2-yl]-2,5-diphenyltetrazolium bromide (MTT) solution (final concentration of 0.5 mg/mL) for 3 h. The dark blue formazan crystals, which were formed in intact cells, were dissolved with DMSO, and the absorbance at 570 nm was measured with a microenzyme-linked immunosorbent assay (ELISA) reader (Sigma-Aldrich). The results were expressed as the percentage of MTT reduction relative to the absorbance measured from negative control cells or DMSO. All assays were performed in triplicate.
CBMN-Cyt assay
The in vitro micronucleus test was conducted as described by Fenech
18
with slight modifications. Cells (1 × 106 in a 25-cm2 flask) were incubated 24 h before treatment with DXR (0.01 µg/mL), bixin (0.05, 0.08, or 0.10 µg/mL), or a simultaneous combination of both (bixin + DXR 0.01 µg/mL). The concentrations of bixin and doxorubicin used in the cytokinesis-block micronucleus cytome (CBMN-Cyt) were established based on the results of the MTT assay (concentrations of bixin and doxorubicin with cell viability above 80%). A solvent control (culture of 0.4% DMSO-treated cells) was also evaluated. Cytochalasin B (6.0 µg/mL) was added to arrest cytokinesis after 44 h of growth, and the cells were harvested and placed in a cold hypotonic solution (1% sodium citrate) and fixed three times in cold methanol: acetic acid (3:1) 72 h after initiating the culture. The initial addition of fixative contained 1% formaldehyde, which enhances cytoplasm preservation. Slides were prepared and stained in a 40 µg/mL solution of Acridine orange (Sigma–Aldrich). In three independent experiments, 2000 binucleated cells (BNs) with 1–4 micronuclei (MNs) were scored at 1000× magnification in Zeiss fluorescence microscope. Additionally, the frequency of nucleoplasmic bridges (NPBs) and nuclear buds (NBUDs) were evaluated using the criteria of Fenech.
18
The nuclear division index (NDI) was also calculated to evaluate the altered mitotic activity and/or cytostatic effects according to the following formula
19
:
SCGE comet assay
Comet assays were performed under alkaline conditions according to the methods of Singh et al. 20 and Tice et al. 21 with slight modifications. HL60 cells seeded at 1 × 106 cells in a 25-cm2 flask. To analyze the genotoxic effect, the cells were treated with bixin (0.05, 0.08, 0.10 µg/mL) or DXR (0.05 µg/mL) for 4 h. In order to analyze the antigenotoxic effect, the cells were treated with simultaneous combination of bixin and DXR. A solvent control (culture of 0.4% DMSO-treated cells) was also evaluated. Microscope slides were covered by a thin layer of 1.0% normal melting-point agarose (Invitrogen), which was dissolved in calcium and magnesium free phosphate-buffered saline at 60°C. Upon solidification of the agarose, 10.00 µL of a freshly prepared suspension of treated or control HL60 cells was mixed with 100.00 µL of low-melting–point agarose (0.5%) at 37°C and placed on the microscope slides. The cells were covered with a coverslip and incubated at 4°C for 10 min. After solidification of the low-melting–point agarose, the coverslips were removed, and the slides were incubated in cold lysis solution (2.5 M sodium chloride, 100 mM ethylenediaminetetraacetic acid (EDTA), 10 mM Tris (pH 10), 1% Triton X-100, and 10% DMSO) at 4°C overnight. After lysis, the slides were placed in a horizontal electrophoresis unit containing alkaline electrophoretic solution (300 mM sodium hydroxide and 1 mM EDTA, pH > 13) for 20 min at 4°C with an electric field strength of 25 V (0.78 V/cm) and a current of 300 mA, which allows DNA to unwind, exposing alkali-labile sites. The slides were then washed with cold neutralization buffer (0.4 M Tris-hydrochloric acid, pH 7.5) for 15 min and fixed in ethanol for 2 min. After drying at room temperature, the slides were stained with 30 µL ethidium bromide (20.00 µg/mL) and immediately analyzed using a fluorescence microscope (Axiostar plus, Zeiss, Germany) equipped with a 515–560 nm excitation filter, a 590 nm barrier filter, and an integrated digital camera (Axiocan, Zeiss). One hundred randomly nucleoids (50 nucleoids from each replicate slide) were evaluated per treatment using CometScore software (Tri-Tek Corporation, Sumerduck, Virginia, USA). Three independent experiments were performed. DNA damage was quantified by the percentage of DNA in the comet tail (% DNA).
Apoptosis index analysis by epifluorescence microscopy
After the cells (1 × 106) were exposed to bixin (0.05, 1.00, or 2.50 μg/mL), doxorubicin (0.50 μg/mL), or a simultaneous combination of both for 24 h, a morphological evaluation was performed in order to discriminate between the living cells and the necrotic or apoptotic ones. In this experiment, treatment with camptothecin (CPT), used as a positive control for apoptosis, was included. A solution with the mixture of the non-permeant dye propidium iodide (PI 5.00 μg/mL), fluorescein diacetate (FDA – 15.00 μg/mL), and Hoechst 33342 (2.00 μg/mL) was prepared in phosphate-buffered salt solution, and then 2.00 μL of the fluorescent mix was added to the cells (100.00 μL final volume). After 5 min incubation at 37°C in 5% CO2 atmosphere, the cell suspension was washed and resuspended in 1.00 mL of the medium.
22
Samples were dropped onto slides for observation using a fluorescent microscope (Zeiss AxioStar Plus) with an ultraviolet light filter. Three independent experiments were performed, and 500 cells were assessed for treatment according to the following morphological and staining
22
: (a) normal cells: blue nucleus, spherical, and intact—green cytoplasm; (b) apoptotic cells: blue nucleus with apoptotic corpuscles—green cytoplasm; and (c) necrotic cells: red and spherical nucleus—red cytoplasm. The apoptotic index was calculated according to the following formula
22
:
Statistical analysis
The results presented are expressed as the mean ± standard deviation of three independent experiments (N = 3). The data were subjected to multiple variance analysis (analysis of variance) and the post hoc Tukey test, using GraphPad Prism 2.01 software. A value of p < 0.05 was considered statistically significant for all the parameters evaluated.
Results
Evaluation of bixin and DXR cytotoxicity in HL60 cell line
The results of the cytotoxic effects of bixin (0.01–3.00 µg/mL) and DXR (0.01–3.00 µg/mL) are presented in Figure 1. Treatments between 0.01 and 0.10 µg/mL of bixin have shown cell viability above 80%. Treatments from 0.30 µg/mL of bixin presented significant cytotoxicity compared with the control (p < 0.05). DXR had a higher toxicity in HL60 cells. Only at very low concentrations (0.01–0.05 µg/mL) DXR showed cell viability above 80%. In treatments between 0.08 and 3.0 μg/mL, the viability of this chemotherapy ranged from 36% to 75%.

Evaluation of cytotoxicity of BXN and DXR on HL60 cells using MTT method. Cell cultures were treated with BXN (0.01–3.00 μg/mL) or DXR (0.01–3.00 μg/mL) for 24 h. C = negative control (for DXR) and DMSO solvent control (for BXN). The experiments were performed in triplicate, and the bars represent mean ± SD of three independent experiments. *p < 0.05: statistically different from C. BXN: bixin; DXR: doxorubicin; DMSO: dimethyl sulfoxide; MTT: 3-[4,5-dimethylthiazol-2-yl]-2,5-diphenyltetrazolium bromide.
The cytotoxic effect of the combination of bixin and DXR in HL60 cells was also evaluated at the same concentrations used in the apoptosis index analysis in order to compare the results of bixin cytotoxicity with the induction of apoptosis. Figure 2 shows the results of different treatments of HL60 cells, which were treated with DXR (0.50 μg/mL), associated to three concentrations of bixin (0.05, 1.00, or 2.50 μg/mL). All combinations of bixin and DXR decreased (p < 0.05) cell viability by about 15% when compared with doxorubicin treatment (0.50 μg/mL).
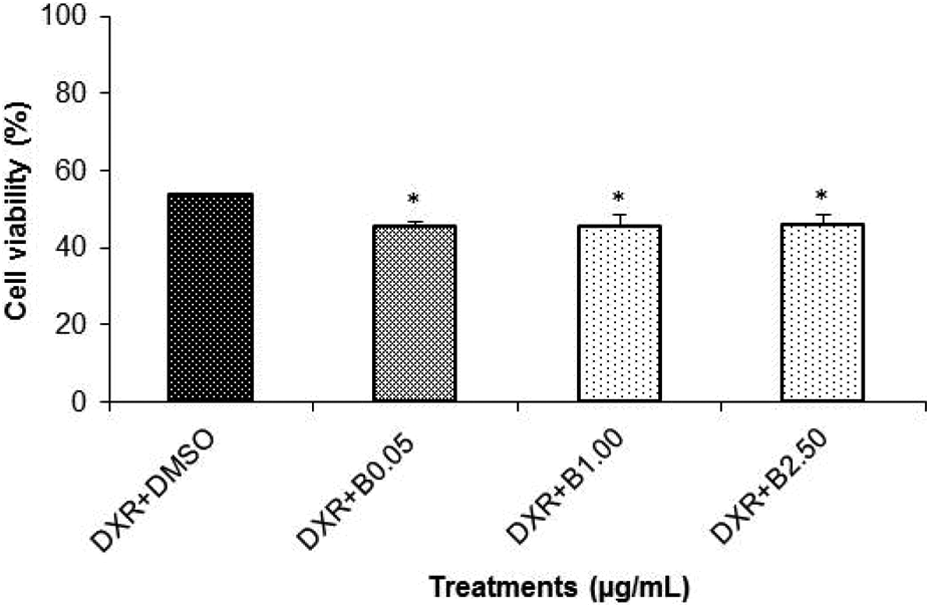
Effect of combined treatment of B with DXR on HL60 cells using MTT method. Cell cultures were treated with 0.50 μg/mL of DXR and 0.05, 1.00, or 2.50 μg/mL of B for 24 h. The experiments were performed in triplicate, and the bars represent the mean ± SD of three independent experiments. *p < 0.05: statistically different from the DXR + DMSO. B: bixin; DXR: doxorubicin; DMSO: dimethyl sulfoxide; MTT: 3-[4,5-dimethylthiazol-2-yl]-2,5-diphenyltetrazolium bromide.
Evaluation of bixin and DXR mutagenicity in HL60 cell line by the CBMN-Cyt assay. In the CBMN-Cyt assay, the percentage of the BNs should be around 30%, indicating a single division cycle after the addition of cytochalasin B. Only 0.01 μg/mL of DXR treatment showed sufficient number of BNs (35%), hence, this concentration was used as inducing DNA damage. Higher concentrations of bixin or DXR were not evaluated in CBMN-Cyt assay since these concentrations were cytotoxic to HL60 cells and thus we did not obtain the minimum number of BNs (30%).
The CBMN-Cyt results (Table 1) showed no significant difference in MNs frequency of cells treated with three different concentrations of bixin (0.05, 0.08, or 0.10 μg/mL) when compared with the DMSO control (p > 0.05). The treatment with DXR showed significant increase (p < 0.05) of MNs frequency, indicating its mutagenicity and that the assay was effective. Two concentrations of bixin (0.05 and 0.08 μg/mL) significantly decreased (p < 0.05) the frequency of MNs induced by DXR.
CBMN-Cyt assay of HL60 cells treated with B (0.05, 0.08, or 0.10 µg/mL), DXR (0.01 µg/mL), and the respective combinations.a
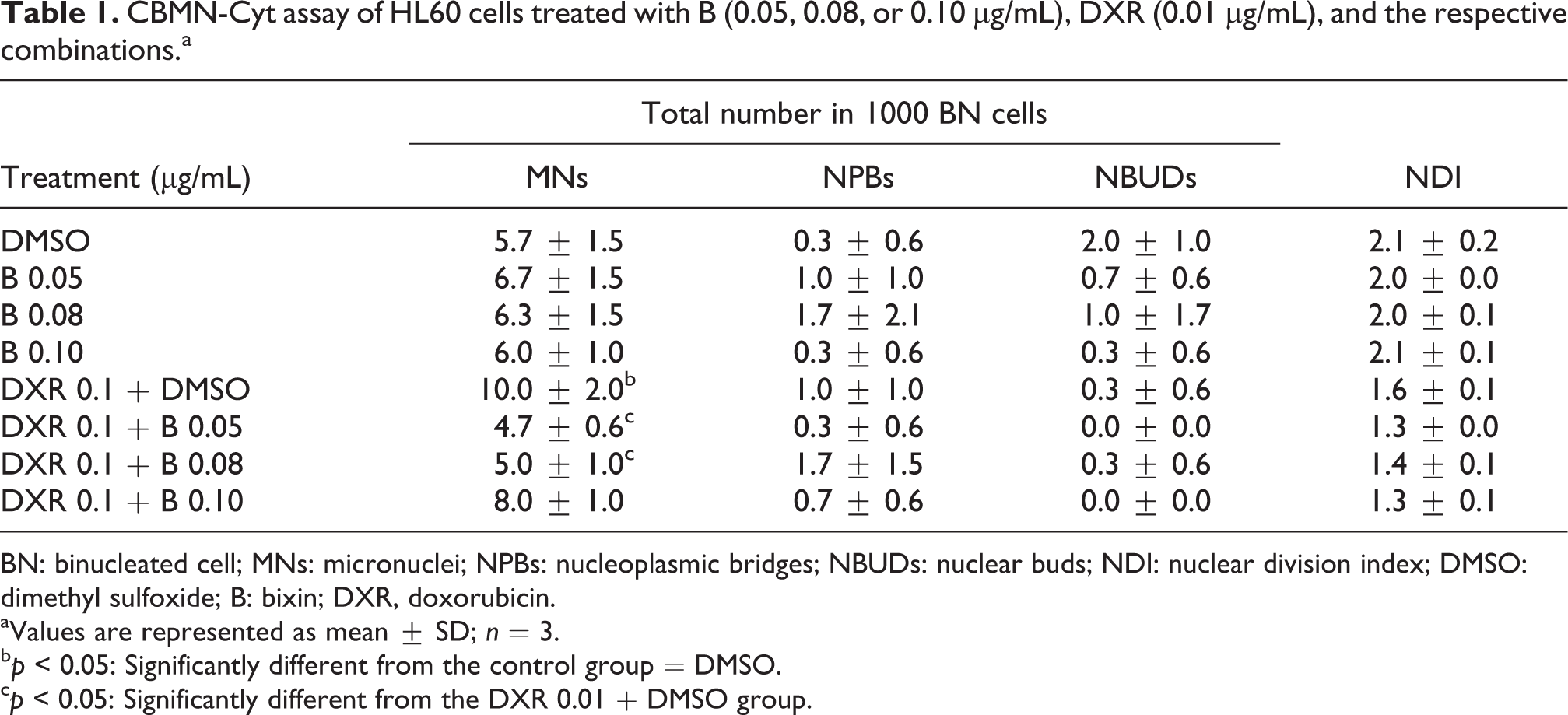
BN: binucleated cell; MNs: micronuclei; NPBs: nucleoplasmic bridges; NBUDs: nuclear buds; NDI: nuclear division index; DMSO: dimethyl sulfoxide; B: bixin; DXR, doxorubicin.
aValues are represented as mean ± SD; n = 3.
bp < 0.05: Significantly different from the control group = DMSO.
cp < 0.05: Significantly different from the DXR 0.01 + DMSO group.
Furthermore, as shown in Table 1, the low NPBs and NBUDs frequencies were not significantly different from the DMSO control. The CBMN-Cyt assay also evaluates the NDI, which was similar for all control and experimental groups.
Evaluation of DNA damage in HL60 cell line by the comet assay (SCGE)
The results of the genotoxic evaluation of bixin and DXR are presented in Figure 3. The extent of DNA damage was evaluated by tail intensity (% DNA in the tail) parameter. Treatments with bixin (0.05, 0.08, or 0.10 μg/mL) showed no significant difference (p > 0.05) in %DNA compared with the DMSO control group, while treatment with DXR showed significant increase (p < 0.05) of DNA damage. The treatments of bixin + DXR did not show any statistical difference (p > 0.05) in the %DNA compared to DXR group.

Evaluation of DNA damage by comet assay in HL60 cells treated with B (0.05, 0.08, or 0.10 μg/mL), DXR (0.05 μg/mL), and their combinations. Three independent experiments were performed and, in each experiment, 100 nucleoids were analyzed per treatment. Values are represented as mean ± SD; *p < 0.05: significantly different from the DMSO group. Photomicrographs of ethidium bromide-stained comet assay with a nucleoid intact and different damage intensities. Original magnification ×40; staining with ethidium bromide. DMSO: dimethyl sulfoxide; DXR: doxorubicin; B: bixin.
Apoptosis index analysis by epifluorescence microscopy
Figure 4 shows morphological analysis with three different dyes (PI, FDA, and Hoechst) in the HL60 cell line. This assay was able to detect apoptotic, necrotic, and also viable cells. Figure 5 shows the results from the analysis of the apoptotic index. CPT at 10 μM was used as positive control for apoptosis.

Analysis of apoptotic nucleus by fluorescence microscopy in HL60 cell line. (a) The arrow number 1 indicates normal living cells showing green fluorescence in the cytoplasm and integrates nucleus (Hoechst); arrow number 2 indicates dead cells population, either necrotic or apoptotic, each cell appearing with the DNA stained a red color (PI) as a consequence of the loss of active membrane control of permeability; (b) arrow number 3 indicates an apoptotic cell population, showing a green cytoplasm (FDA) and spotted nuclear bodies in blue. A representative figure from each group at ×40 magnification is shown here. Microphotographs were shown as representative results from three independent experiments. Details of procedures are described in Materials and Methods section. FDA: fluorescein diacetate; PI: propidium iodide.
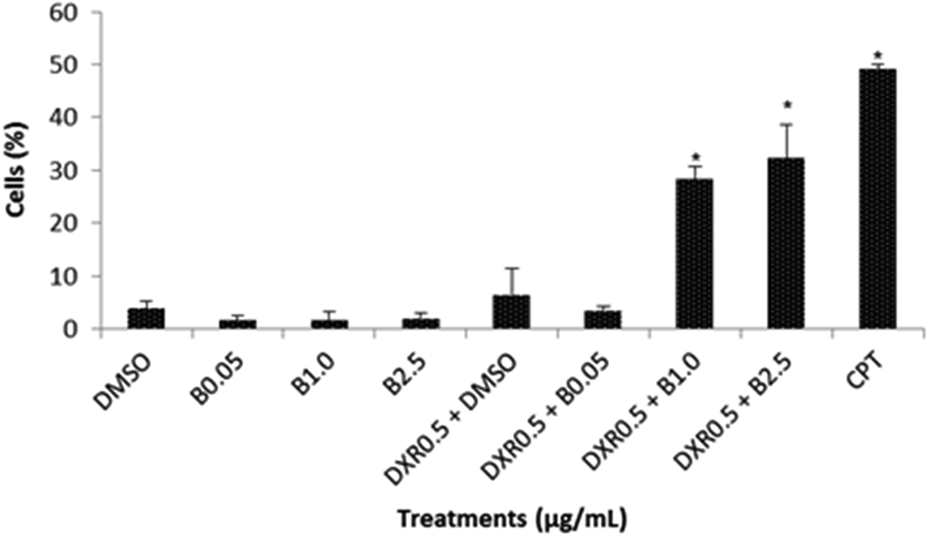
Apoptotic index in HL60 cells exposed to B (0.05; 1.00, or 2.50 μg/mL) and DXR (0.50 μg/mL). DMSO is negative control, and CPT is the positive control (10 μM/mL). Values are represented as mean ± SD for three independent experiments, where each measurement was performed in triplicate. *p < 0.05: compared negative control and DXR + DMSO. B: bixin; DXR: doxorubicin; DMSO: dimethyl sulfoxide; CPT: camptothecin.
Bixin and DXR showed no apoptotic effect (p > 0.05) in HL60 cells compared with DMSO control group (p > 0.05). However, the combined treatments of bixin (1.00 or 2.50 μg/mL) and DXR showed an increase in the percentage of apoptotic cells (p < 0.05).
Discussion
Tibodeau et al. 23 observed cytotoxic effects of bixin (20–400 μM) in a variety of cancer cell lines such as A549 (lung), U2OS (osteosarcoma), PC3 (prostate), HCT-116 (colon), MCF7 (breast), DRO (anaplastic thyroid), and BHP5-16 (papillary thyroid). It is interesting to note that carotenoids are most commonly cited due to their antioxidant properties, inhibiting effects of other toxic substances, and protecting cells and tissues from deleterious effects; on the other hand, some studies have reported prooxidant effects, which could lead to its cytotoxicity. 24 –28 These apparently conflicting studies indicate the possibility that carotenoid properties prooxidants versus antioxidants may be substance and/or dosage specific. 23
The cytotoxicity of DXR was evaluated by Lüpertz et al. 29 in colon cancer cells (Hct-116), and 24 h after incubation, a dose-dependent effect with 55–70% of viable cells using treatment that ranged from 0.30 μg/mL to 5.50 μg/mL of DXR was observed. Quan et al. 30 evaluated the cytotoxic effect of DXR on tumor cell line GBC-SD (gallbladder carcinoma), a type of tumor known as insensitive to chemotherapy, and noted that the cell viability was significantly inhibited by high concentrations of DXR, for which the half maximal inhibitory concentration value was 15.00 μg/mL.
Within this context, in the present investigation, HL60 cells, which are a human promyelocytic leukemia cell line, were used to evaluate the cytotoxicity of different concentrations of bixin and DXR. This was carried out from the MTT assay (a tetrazolium salt reduction assay) that provides sensitive measurements of the normal metabolic status of cells, particularly that of the mitochondria, where measurements reflect early cellular redox changes. 31
Bixin concentrations between 0.30 and 3.00 μg/mL showed less than 80% cell viability (between 40% and 77%). Although these concentrations are low, bixin showed to be cytotoxic. This could indicate a potential anticancer activity, and other studies have shown cytotoxicity of bixin in different tumor cells using higher concentrations. 23,25
We investigated the cytotoxic activity of DXR in HL60 cells in order to find a concentration that modulates the response to cellular damage. Thus, it was observed that HL60 cells are very sensitive to treatment with DXR, whereas in the lower concentration (0.01 μg/mL), the cell viability was 83.4%.
It is known that the clinical use of DXR may be limited with the occurrence of drug resistance to tumor cells and toxicity to normal cells. Therefore, concomitant use of high doses of bixin could improve the clinical usefulness of DXR. It is of great interest to elucidate the mechanism of a specific compound in combination with an antitumor agent for inhibition or potentiation of chemotherapeutic effect in tumor cells. At concentrations above 0.30 μg/mL, bixin was cytotoxic to HL60 cells, and when it was administered with DXR, it increased its cytotoxicity by about 15%.
Although widely used as coloring and antioxidant in food, there are few studies on the effect of the carotenoid bixin in DNA. Alves de Lima et al. 32 found that the annatto showed no mutagenic properties in vivo using bone marrow cells of male Swiss mice. In our previous study, we demonstrated the absence of genotoxic effects of bixin in PC12 cells. 9 In the present study, the evaluated concentrations of bixin presented neither mutagenic (Table 1) nor genotoxic effects (Figure 3) on HL60 cells; moreover, in combined treatment with DXR, it reduced the MNs frequency (p < 0.05) compared with DXR treatment.
By associating the CBMN-Cyt to the comet assay after bixin + doxorubicin combined treatment, these results suggest the damage mechanism involved. The CBMN-Cyt detects both aneugenic and clastogenic effects, while comet assay is often considered more sensitive and detects DNA single-strain breaks. 33 As the value of NPBs is positively correlated with the frequency of MN, induction of MN in the absence of NPBs may be indicative of an aneugenic agent. Then, these results could suggest an aneugenic process that resulted in permanent changes in the DNA structure.
Apoptosis is a form of regulated and controlled cell death that plays an important role in the control and development of the organism. During this period, the normal cells undergo differentiation, and depending on the biological processes that are involved, they may or may not be directed to apoptosis. In contrast, tumor cells fail to complete differentiation or reacquire the ability to resume the differentiation program and reenter the cell cycle. Thus, tumor cells stop undergoing apoptosis and are able to survive in an unfavorable environment under conditions in which oxygen and growth factors are limited. 34 Apoptosis is characterized by severe morphological and biochemical changes, such as nuclear condensation and cleavage of chromatin fragments or oligonucleossomais. 35
According to Tibodeau et al., 23 due to the lipophilic nature of bixin, this could potentially integrate and damage cell membranes, which potentially induce necrosis rather than apoptosis. In our study, we found that bixin showed low induction of apoptosis after 24 h of treatment (Figure 5). However, we also found that, in the concentrations of 1.00 and 2.50 μg/mL, bixin was cytotoxic on the MTT assay. This may be due to damage to cell membranes and may indicate cell death by necrosis.
Regarding DXR, the results have shown low induction of apoptosis. However, in the same concentration (0.5 μg/mL), DXR was cytotoxic by MTT assay (Figure 1), demonstrating that necrotic cell death may have occurred. Lupertz et al. 29 reported that DXR is able to induce apoptosis cell death only in particular conditions of doses and treatments. In addition to apoptosis, cells can be efficiently eliminated after DNA damage due to necrosis, mitotic catastrophe, autophagy, as well as premature senescence, which is characterized by an irreversible arrest of cell division. 36 –38 Thus, it is noted that other types of death, in addition to apoptosis, can be induced after treatment with DXR.
The combined treatments of bixin (1.00 or 2.50 μg/mL) and DXR showed an increase in the percentage of the apoptotic cells. This could indicate that the combination of DXR and bixin, in appropriate concentrations, modulates the action of DXR via induction of apoptosis. In a recent study, Tibodeau et al. 23 found that a myeloma cell line, resistant to doxorubicin, was not considered resistant to bixin, and further, this carotenoid showed selectivity for tumor cells when assessed also in normal cells from bone marrow CD138.
In conclusion, HL60 cells showed high sensitivity to the cytotoxicity of DXR, which caused DNA damage, demonstrated in the comet assay and MN test. Bixin was cytotoxic to HL60 cells; however, this carotenoid demonstrated neither mutagenic nor genotoxic effects, it presented protective effect on DXR-DNA–induced damage, and when it was associated with DXR, it increased the apoptotic index. The results of this study provide more knowledge about the toxic effects of anticancer treatments and how natural compounds can be useful on these therapeutic approaches.
Footnotes
Acknowledgements
The authors would like to thank Regislaine Valeria Burin, PhD, and Joana D’Arc Castania Darin (FCFRP-USP) for technical assistance.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was supported by CNPq, National Council for Scientific and Technological Development, Brazil.
