Abstract
The goal of this in vitro study was to investigate the effect of lipid emulsion on apoptosis induced by a toxic dose of bupivacaine (BPV) in H9c2 rat cardiomyoblast cell lines. The effect of lipid emulsion on the decreased cell viability and count induced by BPV or mepivacaine (MPV) in the H9c2 cells was assessed using an 3-(4,5-dimethylthiazole-2-yl)-2,5-diphenyl tetrazolium bromide assay or a cell count assay. The effect of BPV or lipid emulsion combined with BPV on cleaved caspase 3, caspase 8, and Bax in H9c2 cells was investigated using Western blotting. A terminal deoxynucleotidyl transferase dUTP2′-deoxyuridine 5′-triphosphate nick end-labeling (TUNEL) assay was performed to detect apoptosis of H9c2 cells treated with BPV alone or lipid emulsion combined with BPV. The magnitude of lipid emulsion-mediated attenuation of decreased cell viability induced by BPV was higher than that of lipid emulsion-mediated attenuation of decreased cell viability induced by MPV. Lipid emulsion attenuated the increases in cleaved caspase 3, caspase 8 and Bax induced by BPV. Lipid emulsion attenuated the increases in TUNEL-positive cells induced by BPV. These results suggest that lipid emulsion attenuates a toxic dose of BPV-induced apoptosis via inhibition of the extrinsic and intrinsic apoptotic pathways. The protective effect of lipid emulsion may be partially associated with the relatively high lipid solubility of BPV.
Introduction
Lipid emulsion as a nonspecific antidote has been used to treat systemic toxicity induced by local anesthetics and other drugs without an antidote. 1,2 In an in vitro experiment, lipid emulsion reverses the toxic dose of local anesthetic-induced vasodilation of isolated rat aorta in a lipid solubility-dependent manner. 3,4 In an in vivo experiment, pretreatment or posttreatment with lipid emulsion is effective for recovery from cardiac arrest caused by a toxic dose of bupivacaine (BPV) with high lipid solubility, but it has no effect on cardiac arrest caused by a toxic dose of mepivacaine (MPV) with low lipid solubility. 5,6 These previous reports support the lipid sink theory, which states that the magnitude of lipid emulsion-mediated extraction of local anesthetics from tissue is dependent on the lipophilicity of the local anesthetics. 1,3 –6
Local anesthetic induces decreased cell viability via enhanced apoptosis in cancer and normal cells. 7,8 A toxic dose of BPV decreases cell viability and induces apoptosis in human chondrocytes. 9 BPV induces apoptosis in H9c2 cells. 10 The local anesthetic cocaine induces apoptosis in fetal myocardial cells. 11 BPV produces a myotoxic effect, leading to myonecrosis. 12 A toxic dose of BPV with high lipid solubility causes cardiac toxicity due to inhibition of sodium channels. 13 However, the effect of lipid emulsion on a toxic dose of BPV-induced cytotoxicity in myocardial cells remains unknown. Therefore, the goal of this in vitro study was to investigate the effect of lipid emulsion on apoptosis induced by a toxic dose of BPV in H9c2 rat cardiomyoblast cell lines and to examine the associated cellular mechanism. Based on previous reports supporting the lipid sink theory, we tested the hypothesis that lipid emulsion would attenuate the decreased cell viability induced by toxic doses of BPV and MPV via the reduction of apoptosis in a lipid solubility-dependent manner. 1 –10
Methods
All experimental procedures and protocols were approved by the Institutional Animal Care and Use Committee at Gyeongsang National University, Korea. All experimental procedures were performed in accordance with the Guide for the Care and Use of Laboratory Animals prepared by the National Academy of Sciences.
Cell culture
The rat embryonic ventricular myocardial cell line (H9c2) was cultured in Dulbecco’s modified Eagle’s medium (HyClone; GE Healthcare, Salt Lake City, Utah, USA) supplemented with 10% fetal bovine serum (Gibco; Life technologies, Grand Island, New York, USA), 2 mM
Cell viability
Cell viability was determined colorimetrically using a 3-(4,5-dimethylthiazole-2-yl)-2,5-diphenyl tetrazolium bromide (MTT) assay as previously described. 14 Cells at the exponential phase were seeded at 105 cells/well in 24-well plates. H9c2 cells were treated with a local anesthetic (3 × 10−4 M BPV or 5.1 × 10−3 M MPV) alone for 24 h, SMOFlipid® emulsion (LE; 0.062% or 0.124%) alone for 25 h, and local anesthetic (3 × 10−4 M BPV or 5.1 × 10−3 M MPV) for 24 h after pretreatment with LE (0.062 or 0.124%) for 1 h. Then, 500 µl of 0.1 mg/ml MTT solution was added to each well (0.05 mg/well) and incubated for 3 h at 37°C in the dark. The supernatants were aspirated, the formazan crystals in each well were dissolved in 200 µl of dimethyl sulfoxide for 30 min at 37°C, and absorbance at 570 nm was read on a VersaMax® microplate reader (Molecular Devices, Sunnyvale, California, USA).
Cell count assay
H9c2 cells were seeded at a density of 105 cells/well into 24-well plates, incubated for 24 h, and synchronized to quiescence by serum starvation for 12 h. H9c2 cells were pretreated with 3 × 10−4 M BPV alone for 24 h and 3 × 10−4 M BPV for 24 h after pretreatment with LE (0.062 or 0.124%) for 1 h. The cells were trypsinized, and cell numbers were determined using an automated cell counter, Countess II FL (Life Technologies, Bothel, Washington, USA), according to the manufacturer’s instructions.
Western blot
The effect of LE (0.062% and 0.124%) on the cleaved caspase 3, cleaved caspase 8, and Bax induced by BPV (3 × 10−4 M) was examined in H9c2 cells using Western blotting as described previously. 15 Cells were lysed in PRO-PREP protein extract solution (iNtRON Biotechnology, Houston, Texas, USA) to obtain total cell lysates, and the lysates were centrifuged at 100,000g for 20 min at 4°C. Protein concentrations were determined using the Bradford method. 16 For preparation of sample loading, equal volumes of 2× sodium dodecyl sulfate sample buffer (0.1 mol/L Tris-hydrochloride, 20% glycerol, 4% sodium dodecyl sulfate, and 0.01% bromophenol blue) and supernatant fractions from the lysates were mixed. Proteins (30 µg) were separated by 10% sodium dodecyl sulfate-polyacrylamide gel electrophoresis for 90 min at 110 V. The separated proteins were transferred onto polyvinylidene difluoride membranes for 2 h at 20 mA using SD Semi-dry Transfer Cells (Bio-Rad Laboratories, Hercules, California, USA). After blocking the membranes using 5% nonfat milk in Tris-buffered saline (pH 7.0), the membranes were incubated overnight at 4°C with specific antibodies at a dilution of 1:500 in 5% skim milk in Tris-buffered saline containing Tween-20. The bound antibody was detected with a horseradish peroxidase-conjugated secondary antibody. Membranes were washed and developed using the Western Blotting Luminol Reagent system (iNtRON Biotechnology) and autoradiography.
TUNEL assay
A terminal deoxynucleotidyl transferase 2′-deoxyuridine 5′-triphosphate nick end-labeling (TUNEL) assay was performed to detect apoptosis of H9c2 cells treated with BPV (3 × 10−4 M) alone or LE (0.062% or 0.124%) combined with BPV (3 × 10−4 M) as previously described. 14 Immunohistochemical procedures for detecting apoptotic H9c2 cells were performed using a TUNEL kit (Roche Applied Science, Indianapolis, Indiana, USA), according to the manufacturer’s instructions. Fluorescence staining was viewed with a fluorescence microscope (Olympus Optical Co. Ltd Fluoview 500, Tokyo, Japan). TUNEL-positive cells were imaged and counted.
Materials
All drugs were of the highest purity and commercially available. BPV was obtained from Reyon Pharmaceutical Co., Ltd (Seoul, Korea). MPV was donated by Hana Pharmaceutical Co., Ltd (Gyeonggi-do, Korea). MTT solution was obtained from Sigma Chemicals (St Louis, Missouri, USA). LE was obtained from Fresenius Kabi Korea (Seoul, Korea). Anti-cleaved caspase 3 and caspase 8 antibodies were obtained from Cell Signal Technology (Beverly, Massachusetts, USA). The anti-Bax antibody was obtained from Santa Cruz Biotechnology (Santa Cruz, California, USA). The rat embryonic ventricular cardiomyoblast H9c2 cell line was obtained from the American Type Culture Collection (Manassas, Virginia, USA).
Data analysis
The effect of local anesthetic (BPV or MPV) alone, lipid emulsion alone, and combined treatment with lipid emulsion and local anesthetic (BPV or MPV) on the cell viability or cell count of H9c2 cells was analyzed using a one-way analysis of variance (ANOVA) followed by Bonferroni’s posttest. The effect of BPV and MPV on the magnitude of lipid emulsion-mediated attenuation of decreased cell viability was analyzed using an unpaired Student’s t-test. The effect of lipid emulsion on the increased cleaved caspase 3, caspase 8, and Bax induced by BPV was analyzed using a one-way ANOVA followed by Bonferroni’s posttest. The effect of BPV alone or combined treatment with lipid emulsion and BPV on the TUNEL-positive cells was analyzed using a one-way ANOVA followed by Bonferroni’s posttest. p Values less than 0.05 were considered significant.
Results
BPV (3 × 10−4 M) and MPV (5.1 × 10−3 M) decreased cell viability in H9c2 cells (p < 0.001 vs. control; Figure 1). Lipid emulsion (0.062% and 0.124%) attenuated the decreased cell viability induced by BPV (3 × 10−4 M; p < 0.01 vs. 3 × 10−4 M BPV alone; Figure 1(a)), whereas lipid emulsion (0.062% and 0.124%) slightly inhibited the decreased cell viability induced by MPV (5.1 × 10−3 M; p < 0.05 vs. control; Figure 1(b)). Lipid emulsion (0.062% and 0.124%) alone slightly increased cell viability (p < 0.001 vs. control; Figure 2(a)). The magnitude of lipid emulsion-mediated attenuation of BPV (3 × 10−4 M)-induced decreases in cell viability was higher than that of lipid emulsion-mediated attenuation of MPV (5.1 × 10−3 M)-induced decreases in cell viability (p < 0.001; Figure 2(b)). BPV (3 × 10−4 M) decreased the cell count of H9c2 cells (p < 0.001 vs. control; Figure 3), whereas lipid emulsion (0.062% and 0.124%) inhibited the decreased cell count induced by 3 × 10−4 M BPV (p < 0.001 vs. 3 × 10−4 M BPV alone; Figure 3).

The effect of LE on the decreased cell viability induced by BPV (N = 8, (a)) or MPV (N = 10, (b)) in H9c2 cells using an MTT assay. H9c2 cells were treated with 3 × 10−4 M BPV alone or 5.1 × 10−3 M MPV alone for 24 h, and 3 × 10−4 M BPV or 5.1 × 10−3 M MPV for 24 h after pretreatment with LE (0.062 or 0.124%) for 1 h. Data are shown as the means ± SD. (a) *p < 0.001: versus control; †p < 0.001; #p < 0.01: versus 3 × 10−4 M BPV alone. (b) *p < 0.001: versus control; †p < 0.001; #p < 0.05: versus 5.1 × 10−3 M MPV alone. LE: SMOFlipid® emulsion; BPV: bupivacaine; MPV: mepivacaine; MTT: 3-(4,5-dimethylthiazole-2-yl)-2,5-diphenyl tetrazolium bromide; SD: standard deviation.

(a) Effect of LE alone on the viability of H9c2 cells. H9c2 cells were treated with LE (0.062% and 0.124%) alone for 25 h. Data are shown as means ± SD (N = 8 in each group). *p < 0.001: versus control. (b) The effect of BPV (N = 8) and MPV (N = 10) on the magnitude of LE-mediated attenuation of decreased cell viability in H9c2 cells. H9c2 cells were treated with 3 × 10−4 M BPV alone or 5.1 × 10−3 M MPV alone for 24 h, and 3 × 10−4 M BPV or 5.1 × 10−3 M MPV for 24 h after pretreatment with LE (0.062% or 0.124%) for 1 h. Data are shown as the means ± SD. The magnitude of the LE-mediated attenuation of decreased cell viability induced by local anesthetics is expressed as the difference in cell viability between LE plus local anesthetics (BPV or MPV) and local anesthetics (BPV or MPV) alone. *p < 0.001: versus 5.1 × 10−3 M MPV. LE: SMOFlipid® emulsion; SD: standard deviation; BPV: bupivacaine; MPV: mepivacaine.

The effect of BPV alone or combined treatment with LE and BPV on the count of H9c2 cells using a cell count assay as described in the Methods section. H9c2 cells were treated with 3 × 10−4 M BPV alone for 24 h or 3 × 10−4 M BPV for 24 h after pretreatment with LE (0.062% or 0.124%) for 1 h. Data are shown as the means ± SD (N = 8 in each group). *p < 0.001: versus control; †p < 0.001: versus 3 × 10−4 M BPV alone; BPV: bupivacaine; LE: SMOFlipid® emulsion; SD: standard deviation.
BPV (3 × 10−4 M) increased cleaved caspase 3 expression (p < 0.001 vs. control; Figure 4), whereas lipid emulsion (0.062% and 0.124%) attenuated the increased cleaved caspase 3 induced by 3 × 10−4 M BPV (p < 0.001 vs. 3 × 10−4 M BPV alone; Figure 4). BPV (3 × 10−4 M) increased the expression of cleaved caspase 8 and Bax (cleaved caspase 8 and Bax: p < 0.001 vs. control; Figure 5(a) and (b)), whereas lipid emulsion (0.124%) attenuated the increased expression of cleaved caspase-8 and Bax induced by 3 × 10−4 M BPV (cleaved caspase 8 and Bax: p < 0.001 vs. 3 × 10−4 M BPV alone; Figure 5(a) and (b)).
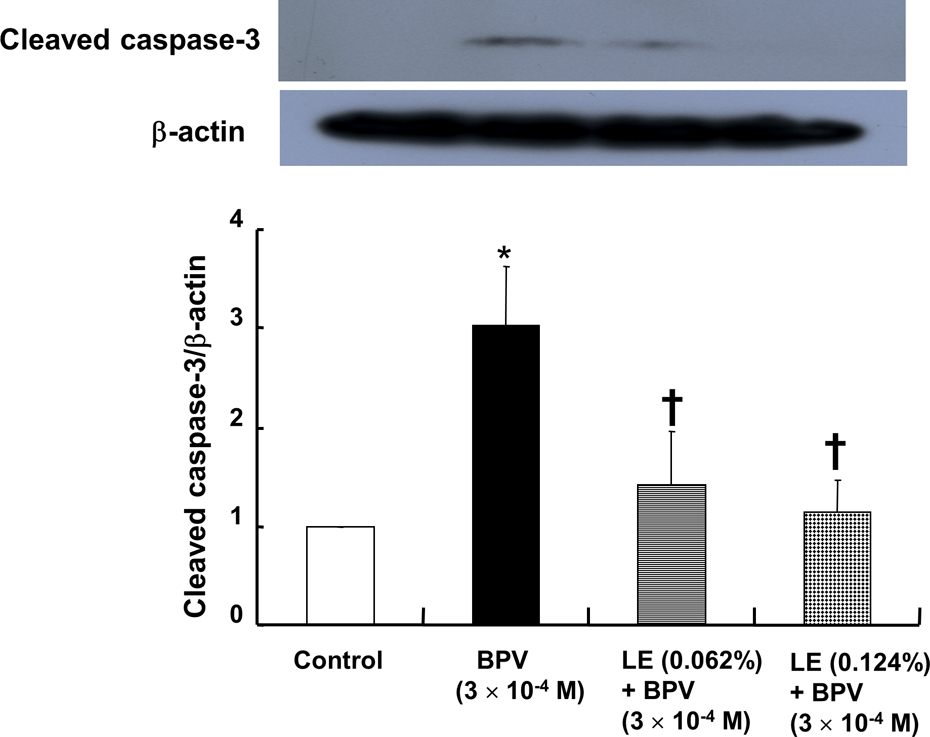
The effect of BPV alone or combined treatment with LE and BPV on cleaved caspase 3 expression in H9c2 cells. H9c2 cells were treated with 3 × 10−4 M BPV alone for 24 h or 3 × 10−4 M BPV for 24 h after pretreatment with LE (0.062% or 0.124%) for 1 h. Expression of cleaved caspase 3 was examined by Western blot analysis as described in the Methods section. Data are shown as the means ± SD (N = 4). N indicates the number of independent experiments. *p < 0.001: versus control; †p < 0.001: versus 3 × 10−4 M BPV alone; BPV: bupivacaine; LE: SMOFlipid® emulsion; SD: standard deviation.

The effect of BPV alone or combined treatment with LE and BPV on the expression of cleaved caspase 8 (N = 5, (a)) and Bax (N = 6, (b)) in H9c2 cells. H9c2 cells were treated with 3 × 10−4 M BPV alone for 6 h or 3 × 10−4 M BPV for 6 h after pretreatment with LE (0.124%) for 1 h for the detection of caspase 8 expression. H9c2 cells were treated with 3 × 10−4 M BPV alone for 12 h or 3 × 10−4 M BPV for 12 h after pretreatment with LE (0.124%) for 1 h for the detection of Bax expression. Expression levels of cleaved caspase 8 and Bax were examined by Western blot analysis as described in the Methods section. Data are shown as the means ± SD. N indicates the number of independent experiments. *p < 0.001: versus control; †p < 0.001: versus 3 × 10−4 M BPV alone; BPV: bupivacaine; LE: SMOFlipid® emulsion; SD: standard deviation.
BPV (3 × 10−4 M) increased TUNEL-positive H9c2 cells (p < 0.001 vs. control; Figure 6), whereas lipid emulsion (0.062% and 0.124%) attenuated the increased TUNEL-positive cells induced by 3 × 10−4 M BPV (p < 0.001 vs. 3 × 10−4 M BPV alone; Figure 6).
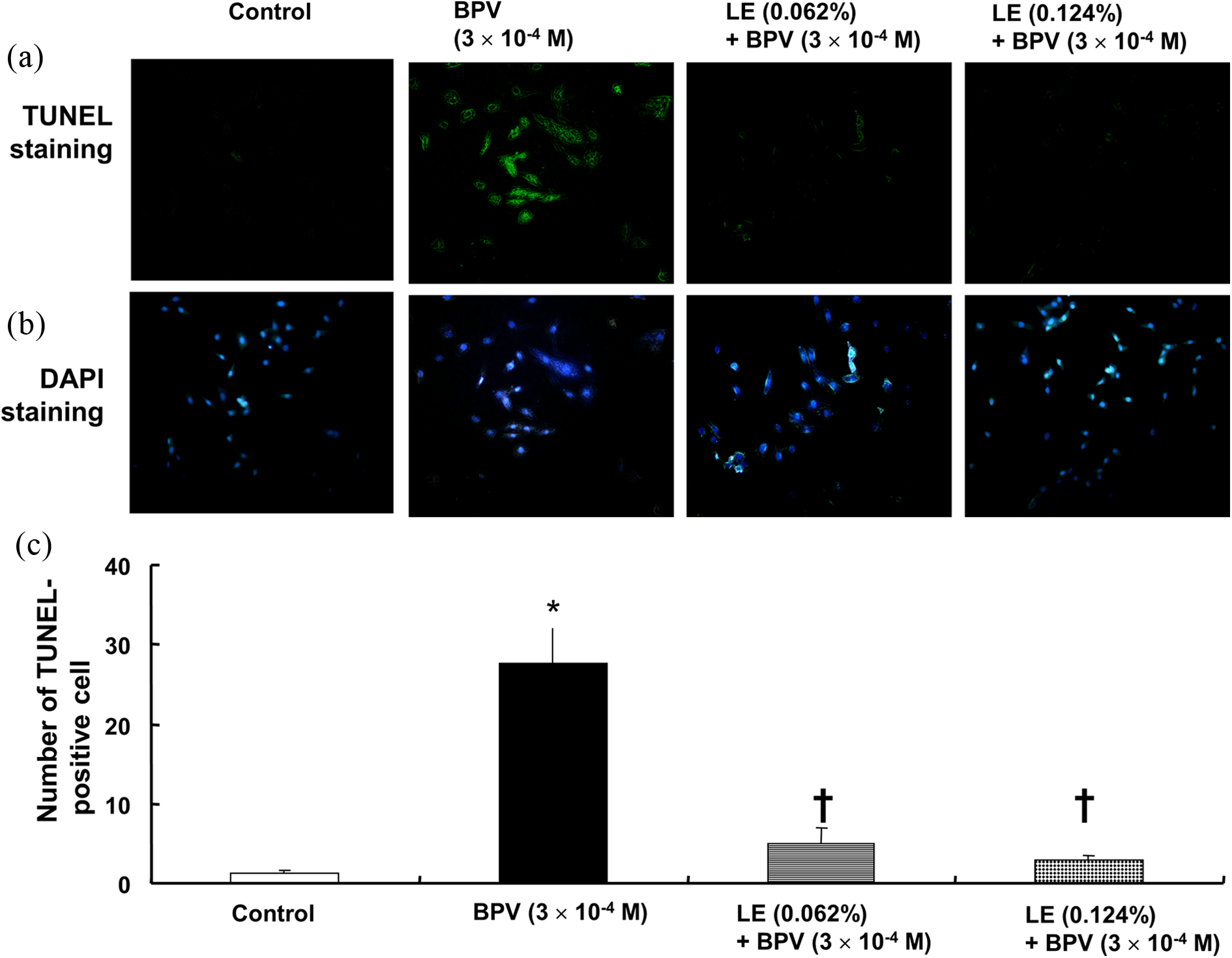
(a) TUNEL staining of H9c2 cells treated with BPV alone or with LE combined with BPV. H9c2 cells were treated with 3 × 10−4 M BPV alone for 24 h or 3 × 10−4 M BPV for 24 h after pretreatment with LE (0.062% or 0.124%) for 1 h. (b) Representative micrographs of TUNEL staining of H9c2 cells are shown. (c) Number of TUNEL-positive H9c2 cells in the control, BPV alone, and combined treatment with LE and BPV. Data are shown as the means ± SD (N = 3 in each group). *p < 0.001: versus control; †p < 0.001: versus 3 × 10−4 M BPV alone; TUNEL: terminal deoxynucleotidyl transferase 2′-deoxyuridine 5′-triphosphate nick end-labeling; BPV: bupivacaine; LE: SMOFlipid® emulsion; SD: standard deviation.
Discussion
This study suggests that lipid emulsion attenuates apoptosis induced by toxic doses of BPV via the inhibition of the extrinsic and intrinsic apoptotic pathways in H9c2 cells, which appears to be partially associated with a relatively high lipid solubility of BPV. The major findings of this in vitro study are as follows: (1) The magnitude of the lipid emulsion-mediated attenuation of decreased cell viability induced by BPV was higher than that of decreased cell viability induced by MPV; (2) lipid emulsion attenuated the increased expression of cleaved caspase 3 and caspase 8 and Bax induced by BPV; and (3) lipid emulsion inhibited the increased number of TUNEL-positive cells induced by BPV.
Pretreatment with BPV (0.25% and 0.5%) for 72 h in human chondrocytes reduces cell viability. 9 BPV (10−3 M) treatment for 18 h decreases cell viability in cultured cortical astrocytes. 17 Similar to previous reports, BPV (3 × 10−4 M) that exceeds a clinically relevant serum concentration of BPV causing systemic toxicity in an in vivo model inhibited viability of H9c2 cells. 9,17,18 In addition, a toxic dose of MPV (5.1 × 10−3 M) that is higher than the plasma concentration (1.27 × 10−4 M) of MPV used to produce hypotension in an in vivo study attenuated cell viability of H9c2 cells in the current study. 19 However, the magnitude of the lipid emulsion-mediated attenuation of decreased cell viability was higher in H9c2 cells treated with BPV (3 × 10−4 M) than in H9c2 cells treated with MPV (5.1 × 10−3 M; Figure 2(b)). As lipid emulsion (0.062% and 0.124%) alone slightly enhanced cell viability (Figure 2(a)), this slight lipid emulsion-mediated inhibition of decreased cell viability induced by MPV seems to be due to the slightly increased cell viability induced by the lipid emulsion itself. In the previous studies, using both in vivo and in vitro models, treatment with lipid emulsion enhances recovery from toxic dose BPV-induced cardiac arrest and reverses severe vasodilation induced by a toxic dose of BPV and levobupivacaine, whereas treatment with lipid emulsion has no effect on either cardiac arrest or vasodilation induced by a toxic dose of MPV. 3 –6 These previous reports support the lipid sink theory that a lipid emulsion-mediated scavenging effect of a toxic dose of local anesthetic from tissue is dependent on the lipid solubility of the local anesthetic. 2 –6 In agreement with previous reports, as the lipid solubility of BPV is higher than that of MPV (n-octanol/buffer partition coefficient: BPV [346] vs. MPV [21]), the enhanced lipid emulsion-mediated attenuation of the decreased cell viability induced by BPV (3 × 10−4 M) compared with the MPV (5.1 × 10− 3 M)-induced decreased cell viability found in the current study seems to be associated with the relatively high lipid solubility of BPV. 2 –6,20 In addition, the cytoprotection provided by the lipid emulsion is associated with an inhibition of mitochondrial permeability transition pore opening via the phosphorylation of glycogen synthase kinase-3β through a pathway involving phosphoinositide 3-kinase, Akt, and extracellular signal-regulated kinase. 21 Further study regarding the role of the pathway involving phosphoinositide 3-kinase on the lipid emulsion-mediated attenuation of apoptotic cell death induced by toxic doses of local anesthetics is needed. It has been reported that lipid solubility of local anesthetics is moderately correlated with the potency of local anesthetic-induced cytotoxicity in human T-lymphoma cells. 22 BPV (3 × 10− 4 M) and MPV (5.1 × 10− 3 M) reduced cell viability of H9c2 cells to 48 ± 8% and 39 ± 2%, respectively. Taking into consideration this result, a limitation of this study is the fact that we did not use equipotent toxic doses of BPV and MPV that decrease cell viability of H9c2 cells to 50%.
Local anesthetics, including BPV, induce decreased cell viability via apoptosis in neuron, myocyte, and H9c2 cells. 7,10 In addition, local anesthetics, including BPV, tetracaine, and lidocaine, attenuate cancer cell proliferation via apoptosis. 8 The cellular signaling pathways responsible for apoptosis are divided into the intrinsic and extrinsic pathways that are associated with mitochondrial stress and the death receptor, respectively. 22 BPV decreases procaspase 3 in human chondrocytes and induces apoptosis in H9c2 cells. 9,10 Similar to previous reports, BPV (3 × 10−4 M) increased cleaved caspase 3, which is a common downstream signal molecule of the intrinsic and extrinsic apoptotic pathways. 7,9,10,23 BPV (0.5%) reduces procaspase 8 expression in human chondrocytes. 9 Lidocaine causes apoptosis via the intrinsic pathway in human Jurkat T-lymphoma cells. 24 The local anesthetic cocaine produces apoptosis via the release of mitochondria cytochrome c. 11 These previous reports suggest that local anesthetic-induced apoptosis seems to be mediated by the intrinsic apoptotic pathway associated with the mitochondrial pathway. 9,11,24 However, in the current study, as BPV (3 × 10−4 M) induced the increased expression of both the extrinsic pathway proapoptotic protein cleaved caspase 8 and the intrinsic pathway proapoptotic protein Bax in H9c2 cells, BPV-induced apoptosis appears to be associated with both the extrinsic and intrinsic pathways. 23 As lipid emulsion inhibited the increased cleaved caspase 8 and Bax induced by BPV in H9c2 cells, lipid emulsion-mediated inhibition of BPV-induced apoptosis appears to be associated with attenuation of both the extrinsic and intrinsic apoptotic pathways. It has been reported that local anesthetic-induced apoptosis is mediated by mitogen-activated protein kinase, reactive oxygen species, and cytochrome c. 11,17,25 In addition, BPV induces apoptosis via the activation of inducible nitric oxide synthase in H9c2 cells. 10 Thus, further study regarding the signaling pathway upstream of toxic dose BPV-induced apoptosis in H9c2 cells is needed to elucidate the detailed signaling pathway. A TUNEL assay was performed to confirm the lipid emulsion-mediated attenuation of apoptosis induced by a toxic dose of BPV. A previous study reported that BPV induces TUNEL-positive H9c2 cells. 10 Consistent with the previous report and reinforced by the BPV (3 × 10−4 M)-induced cleaved caspase 3 expression observed in the current study, BPV (3 × 10−4 M) increased TUNEL-positive cells. 10 However, lipid emulsion decreased the increased number of TUNEL-positive cells induced by BPV (3 × 10−4 M), suggesting that lipid emulsion attenuates BPV (3 × 10−4 M)-induced apoptosis in H9c2 cells.
The proposed mechanisms responsible for lipid emulsion-mediated recovery from a BPV-induced cardiovascular collapse include lipid sink, inotropic effect, fatty acid supply, and drug redistribution. 2,26 Several case reports demonstrated that treatment with lipid emulsion, including Intralipid® or Lipofundin® medium-chain triglyceride and long-chain triglyceride (MCT/LCT), is effective in treating local anesthetic systemic toxicity induced by levobupivacaine, BPV, lidocaine, and ropivacaine. 27 –31 Although we used H9c2 cells derived from a rat heart in the current experiment, it has been reported that H9c2 cells show properties of skeletal muscle. 32 Even with this limitation, taken together with previous reports and current results, these results suggest that lipid emulsion not only provides a beneficial effect for recovery from cardiovascular collapse induced by a toxic dose of local anesthetics, but it also seems to reduce BPV-induced cytotoxicity of myocardial cells. 32 SMOFlipid® is composed of 61% long-chain triglyceride and 39% medium-chain triglyceride. 33 Further studies regarding which type of lipid emulsion, including Intralipid® (100% long-chain triglyceride) and Lipofundin® MCT/LCT (50% long-chain triglyceride and 50% medium-chain triglyceride), is more effective in reducing BPV-induced apoptosis remain to be conducted. 34
Taken together, these results suggest that lipid emulsion attenuates apoptosis induced by a toxic dose of BPV via attenuation of the extrinsic and intrinsic apoptotic pathways in H9c2 cells. This lipid emulsion-mediated inhibition of BPV-induced apoptosis appears to be partially associated with a relatively high lipophilicity of BPV compared with MPV.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was supported by Basic Science Research Program through the National Research Foundation of Korea (NRF) funded by the Ministry of Education (2013R1A1A2057459).
