Abstract
The objective of this study was to investigate whether melamine (MEL) is biotransformed into cyanuric acid (CYA) in male broiler chickens. The effects of the dietary addition of MEL and CYA alone and in combination on the growth performance and mortality of the chickens were also investigated. The chickens were divided into six experimental groups and were fed diets with an addition of 50 or 100 mg/kg MEL or 50 or 100 mg/kg CYA of diet, with the contaminants added separately or in combination. The control group was fed a diet without MEL or CYA. At the end of the experiment (day 40), samples of liver, kidney and breast and thigh muscles were collected from 12 birds per treatment group and were analysed for the presence of MEL and CYA. This study showed that MEL is biotransformed into CYA in broilers. Higher concentrations of MEL or CYA in the diet increased the presence of their residues in tissues, and the CYA residue concentration was several times higher in the respective tissues than the MEL residue concentration. The dietary addition of MEL and CYA did not affect the mortality rate, feed conversion ratio or body weight of the chickens.
Introduction
Melamine (2,4,6-triamino-1,3,5-triazine; MEL) is widely used to make plastics, laminates and coating. Moreover, MEL is present as a trace contaminant in nitrogen supplements used in animal feeds (e.g. urea) and can also be found as a metabolite and degradation product of the pesticide and veterinary drug cyromazine. 1 Cyanuric acid (2,4,6-trihydroxy-1,3,5-triazine; CYA), a structural analogue of MEL, is also a by-product of the production of plastics and is used as water disinfectant in pools. 2
In 2007 and 2008, the illegal adulteration of animal feed and raw milk used for infant formula (often to mask insufficient protein content) with either MEL alone or MEL-containing CYA resulted in the illness and death of human infants and companion animals due to the nephrotoxicity associated with the accumulation of MEL-uric acid or MEL-CYA crystals in the kidneys. 3,4
Risk assessment of MEL and CYA in poultry was already conducted on laying hens, broiler chickens, ducks and Japanese quails. 5 –8 Previous poultry research has shown that both MEL and CYA are absorbed from the diet and subsequently can be found in poultry tissues, blood and eggs. In previous study, Brand et al. reported that dietary MEL was toxic to broiler chickens at concentrations higher than 1000 mg/kg of feed. 9 Ding et al. found that dietary CYA alone or in combination with MEL had adverse effects on the performance and health of broiler chickens at considerably lower concentrations. 10
Our previous studies showed that MEL was biotransformed into CYA within the bodies of laying hens, 11–12 which had not been previously shown in poultry. 8,13 Therefore, the objective of this study was to investigate whether MEL is also biotransformed into CYA in broiler chickens. An integral part of the study was to assess the effects of the dietary addition of MEL and CYA alone and in combination on the growth performance and mortality of broiler chickens and to examine the resultant residue distribution in various tissues.
Materials and methods
Birds and experimental design
A total of 252, 1-day-old male broiler chickens (Ross 308) were used in the experiment. Broiler chickens were randomly placed in seven groups (one control and six experimental groups; 36 birds/group). Chicks were weighed, and birds from each group were housed in four floor pens (9 birds/pen) covered with wood shavings. Broiler chickens were housed in the accredited experimental stable of the Department of Animal Nutrition and the Department of Animal Husbandry and Animal Hygiene, University of Veterinary and Pharmaceutical Sciences Brno (UVPS Brno), Brno, Czech Republic, under controlled housing conditions that fully complied with the standards used for fattening of Ross 308 broilers. The experimental procedures were approved by the Animal Welfare Committee of UVPS Brno (no. 1/2011/2220/FVHE).
A two-phase feeding programme (starter and finisher) was used. The starter and finisher diets were offered from days 1 to 15 and 16 to 40, respectively. The composition of the basal diet is outlined in Table 1. The chickens in the control (C) group were fed basal diets without MEL or CYA. For the experimental groups, MEL (M2659; Sigma-Aldrich, Saint-Quentin Fallavier, France) and CYA (185809; Sigma-Aldrich) alone and in combination were added to the basal diets. The M50 and M100 groups were given the basal diet with the addition of 50 or 100 mg/kg MEL of feed, respectively; the K50 and K100 groups were given the basal diet with the addition of 50 or 100 mg/kg CYA of feed, respectively; the MK50 group was given the basal diet supplemented with 50 mg/kg MEL and 50 mg/kg CYA; and the MK100 group was given the basal diet supplemented with 100 mg/kg MEL and 100 mg/kg CYA. The experimental period lasted for 40 days. Feed and water were supplied ad libitum.
Ingredient and nutrient composition of the basal diets.

NaCl: sodium chloride; Ca: calcium; P: phosphorus; Mg: magnesium’; FTU: phytase unit; BGU: β-glucanase unit; EXU: endoxylanase unit.
aIn 1 kg of feed: 8000 IU of vitamin A, 2500 IU of vitamin D3, 50 mg of α-tocopherol, 1.5 mg of vitamin K3, 4 mg of vitamin B1, 6.5 mg of vitamin B2, 3 mg of vitamin B6, 0.015 mg of vitamin B12, 0.15 mg of biotin, 2.5 mg of folic acid, 30 mg of nicotinic acid, 12.5 mg of calcium pantothenate, 250 mg of betaine, 17 mg of butylhydroxytoluene, 6 mg of propyl gallate, 2.7 mg of ethoxyquin, 50 mg of ferrous sulphate monohydrate, 80 mg of manganese oxide, 80 mg of zinc oxide, 8.7 mg of copper sulphate, 1 mg of potassium iodide, 0.15 mg of sodium selenite, 0.25 mg of cobalt sulphate, 250 FTU of phytase, 120 BGU of glucanase, 5500 EXU of xylanase.
Birds were observed twice daily for any signs of illness and behavioural changes. On days 11, 20, 29 and 40, the broiler chickens were weighed and feed intake on a pen basis was recorded. Mortality was recorded daily, and it is expressed as the percentage ratio of dead birds from the total number of initially used broilers in a group. The average body weight (BW) gain and feed intake adjusted for mortality were used to calculate the feed conversion ratio (FCR; feed:gain).
Sample collection and analyses
At the end of the experiment (day 40), 12 birds/group (3 birds/pen) were randomly selected for MEL and CYA tissue residue analysis. The chickens were stunned and exsanguinated by cutting the jugular vein. The MEL and CYA levels were measured in samples of the liver, kidney and breast and thigh muscles. Tissue samples were homogenized, dried in an oven at 105°C and ground. The samples were stored in a freezer until sample preparation for gas chromatography–tandem mass spectrometry (GC-MS/MS) analysis. The detection limit of the assays was 0.2 mg/kg of MEL and 0.2 mg/kg of CYA of dry matter tissue. Determination of the MEL and CYA levels by GC-MS/MS was performed in an accredited laboratory (State Veterinary and Food Institute Dolný Kubín, Slovak Republic).
Statistical analyses
Statistical analyses were performed using the STATISTICA CZ version 10 software (StatSoft, Inc. Tulsa, Oklahoma, USA). One-way analysis of variance was used to determine the differences in both MEL and CYA residue levels in the tissues, as well as BW and FCR differences between groups of broilers. The differences for these parameters were tested according to the following statistical model:
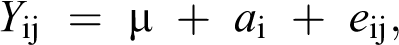
where Y ij is variance associated with parameter a, µ is the overall mean, a i is a treatment effect and e ij is the error term. The averages for pens were treated as the experimental units, and differences between treatments were analysed for a significance using Tukey’s test. The data are presented as an average value and accompanied by the standard error of the mean. A χ 2 test with the Yates correction was used to compare the differences in mortality rate among groups of broilers. Differences were considered significant at p < 0.05.
Results
MEL residue was detected in all evaluated tissues of the 40-day-old broilers from the M50, M100, MK50 and MK100 groups (Table 2). Within these experimental groups, the MEL residue concentration decreased as follows: kidney > breast muscle > thigh muscle > liver. Higher MEL levels in the feed (100 mg/kg MEL vs. 50 mg/kg MEL) resulted in a highly significant increase in the MEL residue level in all of the broiler tissues that were investigated, even when the chickens were simultaneously fed CYA (MK50 and MK100 groups). Furthermore, the level of MEL residue in each of the tissues was similar in the M50 and MK50 groups as well as in the M100 and MK100 groups. MEL was not detected in any tissues from either the broiler chickens fed with CYA only contaminated diet (K50 and K100 groups) or those in the C group.
Concentration of MEL (mg/kg of dry weight) in tissues of broiler chickens fed with a particular diet.a

MEL: melamine; CYA: cyanuric acid; C: control, M50: 50 mg/kg MEL of feed, M100: 100 mg/kg MEL of feed, K50: 50 mg/kg CYA of feed, K100: 100 mg/kg CYA of feed, MK50: 50 mg/kg MEL + 50 mg/kg CYA of feed, MK100: 100 mg/kg MEL + 100 mg/kg CYA of feed; ND: not detected.
aMeans within a column with different superscript letters (b, c, and d) differ (p < 0.01).
CYA residue was detected in all of the examined broiler tissues from the M50, M100, K50, K100, MK50 and MK100 groups (Table 3). CYA residue concentrations in the tissues decreased as follows: kidney > breast muscle > thigh muscle > liver. Higher CYA levels in the feed (K50 vs. K100 group) increased the residue levels of CYA in all of the studied tissues (p < 0.01). The CYA residue was also found in the studied tissues when the feed was contaminated with MEL alone (M50 and M100 groups), and the higher dose of dietary MEL highly significantly increased CYA residue levels in the broiler tissues studied. Furthermore, the levels of CYA residue in broiler tissue samples from the M100 and K50 groups were similar. When diets were simultaneously contaminated with MEL and CYA (MK50 and MK100 groups), the levels of CYA residue in the liver and the breast and thigh muscles did not increase significantly. Indeed, the CYA residue concentrations in these tissues fell within the range observed in the M50 and M100 groups. The level of CYA residue in the kidneys was highest in the MK100 group (105.0 mg/kg) and was significantly higher than that in the MK50 group (28.1 mg/kg). CYA residue was not detected in any of the tissues studied from the C group.
Concentration of CYA (mg/kg of dry weight) in tissues of broiler chickens fed with a particular diet.a
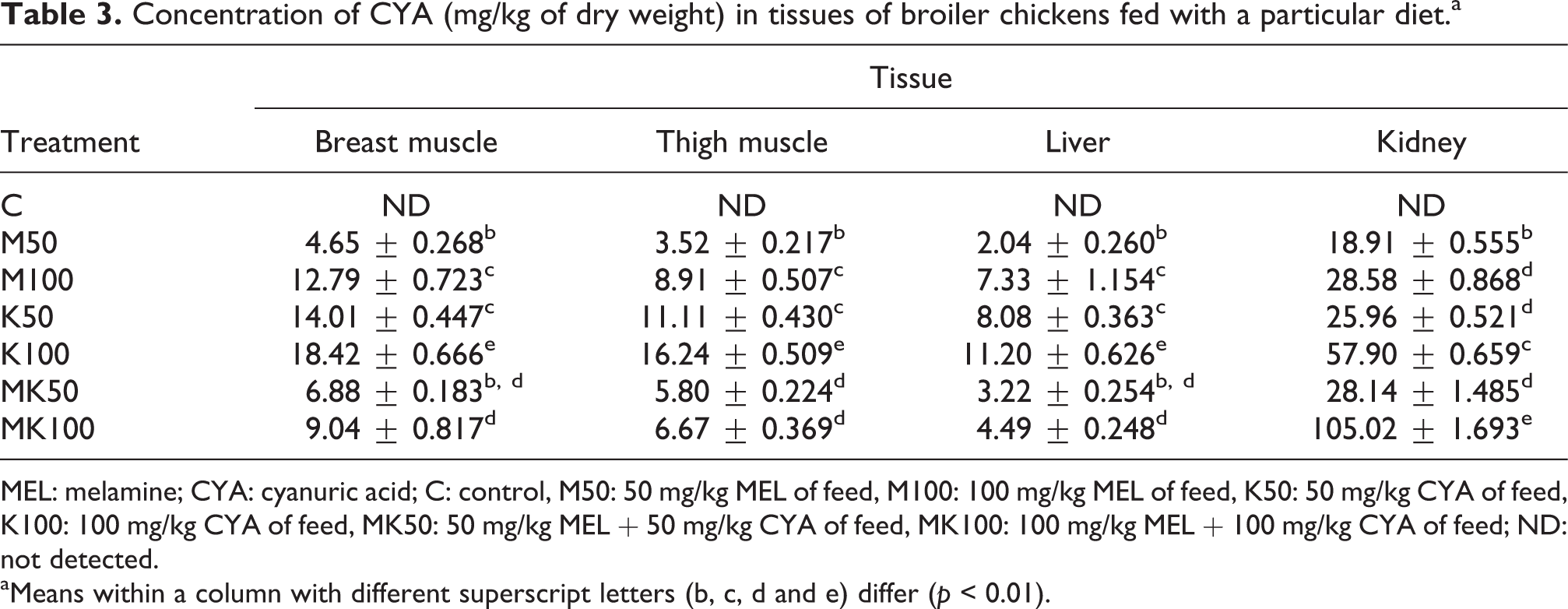
MEL: melamine; CYA: cyanuric acid; C: control, M50: 50 mg/kg MEL of feed, M100: 100 mg/kg MEL of feed, K50: 50 mg/kg CYA of feed, K100: 100 mg/kg CYA of feed, MK50: 50 mg/kg MEL + 50 mg/kg CYA of feed, MK100: 100 mg/kg MEL + 100 mg/kg CYA of feed; ND: not detected.
aMeans within a column with different superscript letters (b, c, d and e) differ (p < 0.01).
The mean BWs of the broiler chickens in each group are listed in Table 4. At 11, 20, 29 and 40 days of age, the contamination of the diet did not significantly affect BW (p > 0.05).
BW (g) of broiler chickens over the course of the experiment.

MEL: melamine; CYA: cyanuric acid; C: control, M50: 50 mg/kg MEL of feed, M100: 100 mg/kg MEL of feed, K50: 50 mg/kg CYA of feed, K100: 100 mg/kg CYA of feed, MK50: 50 mg/kg MEL + 50 mg/kg CYA of feed, MK100: 100 mg/kg MEL + 100 mg/kg CYA of feed; NS: non-significant; BW: body weight.
The average values of calculated FCR in all the groups of broilers during the experimental period are listed in Table 5. No clear trend in the level of FCR values was observed among the respective periods. For the whole period of fattening, the lowest FCR value was found in the C group (1.57) and the highest value was found in the K100 group (1.67); higher values were also observed in the M100 and MK50 groups (1.66). The mortality rate for the period of 40 days was not significantly different (p > 0.05) among the investigated groups of broilers (Table 5). The mortality ranged from 0% (K50) to 11.1% (K100) for the experimental groups and a rather high mortality was also found in the C group.
FCR (feed/gain) and mortality (1 to 40 days) of broiler chickens over the course of the experiment.a

FCR: feed conversion ratio; C: control, M50: 50 mg of melamine.kg−1 of feed, M100: 100 mg of melamine.kg−1 of feed, K50: 50 mg of cyanuric acid.kg−1 of feed, K100: 100 mg of cyanuric acid.kg−1 of feed, MK50: 50 mg of melamine + 50 mg of cyanuric acid.kg−1 of feed, MK100: 100 mg of melamine + 100 mg of cyanuric acid.kg−1 of feed; NS: non-significant.
aMeans within a row with different superscript letters (b and c) differ (p < 0.05).
Discussion
In our study, the contamination of feed with MEL led to the deposition of MEL residue in all studied broiler chicken tissue, and this result was found in both individual contamination and simultaneous contamination with MEL and CYA. The administration of 100 mg/kg MEL vs. 50 mg/kg MEL of feed, alone or in combination with CYA, led to a highly significant increase in the MEL residue level in the studied tissues, which is in accordance with the findings of previous studies. 10,14 However, in our study, the same dose of MEL in the diet (100 mg/kg MEL) resulted in a markedly higher MEL residue level than was detected by Lü et al. 14 In their experiment, a dose of 500 mg/kg MEL of diet was necessary to reach a comparable level of MEL residue in the tissue. In our study, the concentrations of MEL residue in the tissues decreased in the following order: kidney > breast muscle > thigh muscle > liver. These results are consistent with the findings published in broilers that were fed an MEL-contaminated diet for 42 days. 14 However, in younger broilers (21- to 28-day-old), the MEL residue concentration in the tissues decreased in a different order: kidney > liver > breast muscle. 9,14 Previous poultry research has shown that MEL metabolism can also be affected by the length of the MEL exposure period. 14,15 Lü et al. and Valat et al. found that longer MEL exposure period in broiler chickens and laying hens resulted in decreased MEL residue level in liver tissue in comparison with muscle tissue, which is in accordance with our study. We suggest that long-term MEL exposure period may subsequently lead to faster depletion of MEL in liver tissue with the aim of its faster excretion from chicken body.
CYA residue was detected in all broiler tissues studied from all experimental groups, even in birds fed with MEL-only contaminated diets. These results show that MEL is biotransformed into CYA in broilers and is in accordance with the previous studies looking at laying hens fed with MEL-contaminated diet. 11,12 In contrast, Dong et al. and Zhang et al. found that MEL was not metabolized into CYA in laying hens and Japanese quails. 8,13 Dorne et al. state that CYA may be produced by microbial degradation of MEL in the gastrointestinal tract 16 ; however, this process occurs mainly in ruminants and has not yet been described in detail in poultry. Moreover, Zheng et al. found that in rats, MEL was biotransformed by intestinal microorganisms (namely by Klebsiella terrigena) into CYA which subsequently became the main component of kidney stones. 17 Our results indicate that the biotransformation of MEL into CYA may primarily take place in the liver. According to Dorne et al. in most animal species, MEL and CYA are eliminated by renal excretion, and more rapid elimination was observed for CYA than that for MEL. 16 Our results were in accordance with these findings, and we observed that the concentration of CYA residue in the kidneys was several times higher than the concentrations of MEL residue in the kidneys, which also suggests the intensive biotransformation of MEL into CYA in broilers. When chickens were given simultaneous contamination (the MK100 group), the concentration of CYA residue was 25 times higher than that of the MEL residue. The levels of CYA residue in the tissues of broilers from all experimental groups decreased in a manner similar to those of MEL residue: kidney > breast muscle > thigh muscle > liver. In general, the tissue CYA residue levels were similar between the groups of broilers fed 100 mg/kg MEL and those fed 50 mg/kg CYA of feed. When the diet was contaminated with CYA only, the higher dose (100 mg) caused a highly significant increase in the residue levels of CYA in all tissues. This effect was not observed in liver, thigh and breast muscles when chickens were fed diets contaminated with both CYA and MEL. The levels of CYA residue in tissues from these broilers were relatively low, and the CYA residue concentrations fell within the range found for the groups of chickens fed MEL only contaminated diets. However, when the diet was contaminated with a combination of CYA and MEL, the residue level of CYA in kidneys increased almost 4 times in the MK100 group (105 mg/kg) compared with the MK50 group (28 mg/kg). Moreover, the level of CYA residue in the kidneys of broilers from the MK100 group was almost double that of the broilers fed the diet contaminated with 100 mg/kg CYA of feed (K100).
In our previous studies, we have found that MEL is biotransformed into CYA in laying hens. 11,12 In laying hens, dietary addition of MEL (100 mg/kg MEL) for 35-day period resulted in decreasing MEL residue concentration in tissues in the following order: kidney > breast muscle > liver > thigh muscle. MEL residue levels were 2 to 3 times higher in the tissues of laying hens than in broilers (M100) in this study. Detected residues of CYA in laying hens were found only in liver (7 mg/kg) and kidneys (6.3 mg/kg). However, in laying hens, the CYA residue was also depleted into eggs. Its highest level was found in eggs at the end of the experiment (14.1 mg/kg). In this study, the same dose of MEL in the diet for broiler chickens (M100) resulted in the markedly higher CYA residue level in kidneys in comparison with laying hens in our previous studies. It seems that broiler chickens show higher ability to biotransform MEL into CYA than laying hens.
We found that the dietary doses of MEL and CYA that we used did not affect the growth intensity of broiler chickens during the experimental period, similar to findings reported by previous studies. 10,14 The decrease of growth intensity may be expected if the chickens are fed higher doses of MEL; Brand et al. observed a decrease of growth performance in broilers fed with 1000 mg/kg MEL of feed. 9 We observed higher calculated FCR values for the 40-day period in all experimental groups of broilers in our study compared with the control group (1.57), and the highest FCR values (p > 0.05) were found in the K100, M100 and MK50 groups (1.66–1.67). We assume that the metabolization of nonprotein nitrogen (i.e. MEL and CYA) in broilers could be related to their increased energy requirement, which could lead to increased feed intake.
The dietary addition of MEL and CYA in our experiment did not significantly increase the mortality rate of broiler chickens. The highest mortality was found in broilers fed 100 mg/kg CYA of feed (11.1%); however, a relatively high mortality rate was also found in the control group (8.3%). Our results are in agreement with Lü et al. who found that the administration of up to 1000 mg/kg MEL of feed had no effect on broiler mortality. 14 However, Ding et al. found that MEL and CYA in the diet damaged the kidneys and livers of broiler chickens and that higher doses resulted in even more serious lesions. 10 Moreover, Wang et al. found that the dietary addition of MEL and CYA induces kidney damage in chickens, with the most harmful effects occurring when the CYA/MEL ratio was 1:3. 7 In their study, when broilers received 100 mg/kg MEL and 33.3–50 mg/kg CYA of feed, dilated renal tubes and small blood vessel expansion, but no crystals, were found in the kidneys.
This study has shown that MEL is biotransformed into CYA in broiler chickens. The MEL and CYA residue levels in all evaluated tissues increased with increasing doses of MEL and CYA in the feed (p < 0.01), and the CYA residue levels were several times higher than MEL residue levels. Furthermore, the levels of CYA residue in broiler tissue samples from the M100 and K50 groups were similar. The MEL and CYA residue levels decreased in tissues in the following order: kidney > breast muscle > thigh muscle > liver. The level of CYA residue in the kidneys was highest in the MK100 group (105.0 mg/kg) and was significantly higher (p < 0.01) than that in the MK50 group (28.1 mg/kg). At 11, 20, 29 and 40 day of age, the dietary addition of MEL and CYA did not affect the body weight of broiler chickens (p > 0.05). The lowest calculated FCR value for the 40-day period was found in chickens from the C group (1.57) and the highest value (p > 0.05) was found in the K100 group (1.67). In addition, the mortality rate for the period of 40 days was not different among the investigated groups of broilers (p > 0.05).
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by the Ministry of Education, Youth and Sport of the Czech Republic [grant number MSM6215712402].
