Abstract
The purpose of this study was to evaluate the toxicity potential of cyanuric acid (CYA) and a combination of melamine (MEL) and CYA in broilers. A total of 1200 male COBB 500 broilers were randomly allocated into 1 of 10 treatment groups by a 5 × 2 factorial design in a 42-d experiment. The dietary treatments were as follows: T1 to T5: basal diets with 0, 10, 20, 33.3, and 50 mg CYA per kg diet; T6 to T10: basal diet with CYA regimens similar to T1 to T5 but with 100 mg MEL per kg diet. There were 6 replication pens with 20 chicks per pen. No differences were observed in alanine transaminase (ALT) and aspartate aminotransferase (AST) activities. But on d 22, uric acid (UA) and creatinine (Crea) concentrations were significantly greater when birds were fed CYA at 33.3 mg/kg with MEL 100 mg/kg, and Crea concentration was also higher when birds were fed CYA at 50 mg/kg. No crystals were found in kidneys but dilated renal tubules and small blood vessel expansion were found in kidneys of birds fed CYA at 50 mg/kg and CYA at 33.3 mg/kg with MEL 100 mg/kg. The apoptosis rate (AR) of kidneys of all birds fed CYA and MEL contaminated diets were higher than the control group. These results indicated that the dietary addition of CYA and MEL could induce kidney damage, and the effects were harmful when the ratio of CYA/MEL was 1:3.
Introduction
In March 2007, large renal failures of pets broke out in North America because of ingesting pet food contaminated with melamine (MEL) and its related contaminants such as cyanuric acid (CYA). 1 Later, in 2008, adulteration of milk products with MEL caused renal failures and even deaths in infants in China. After these incidents, MEL and CYA received considerable public attention.
MEL (1,3,5-triazine-2,4,6-triamine) is an intermediate widely used in the production of amino resins and plastics. MEL is also used in fertilizers and is a metabolite of the pesticide cyromazine in plants and animals.2–4 CYA (1,3,5-triazine-2,4,6-(1H,3H,5H)-trion), a structural analogue of MEL, is also a by-product in the production of plastics and used as a water disinfectant in swimming pools.5 –7 However, CYA is an Food and Drug Administration (FDA)-accepted nitrogen supplement for ruminants. Both CYA and MEL have a low acute toxicity. The oral median lethal doses (LD50) are 3161 mg/kg body weight (BW) and 7700 mg/kg BW in male and female rats, respectively.8–10 Nevertheless, a combination of MEL and CYA can form insoluble crystals that damage renal tubules and cause renal failures. 11
Neither CYA nor MEL is an approved feed or feed additive for human and monogastric animals, 12 but they were illegally added to feed or food falsely to elevate its crude protein content which was determined by the Kjeldahl method. Of course, CYA and MEL can migrate from the environment into feed/food such as plastic wrap or by the degradation of cyromazine. 13 Therefore, it is essential to characterize the toxicity of MEL and CYA.
In previous studies, Ledoux et al. 14 reported young chicks fed with diets containinged MEL revealed histopathologic lesions in kidney and increased mortality increased. In addition, Puschner et al. 15 presumed MEL and CYA in combination is responsible for renal failure in cats. So far no evidence could be found to evaluate the toxicity of CYA alone and the combination of CYA and MEL at different ratio. Therefore, the purpose of this study was to assess the toxicity of CYA with or without MEL to provide information for feed industry.
Materials and methods
Chemicals and reagents
MEL (purity, ≥99.5%) and CYA (purity, ≥98%) were purchased from Sinopharm Chemical Reagent Co., Ltd, Beijing, China.
Birds—feeds and management
This study was approved by the Animal Welfare Committee of Sichuan Agriculture University. A total of 1200 1-d-old male COBB 500 broilers with an average initial BW of 46.25 ± 0.02 g per chick were randomly allotted to 10 treatment groups with 6 replicates each (20 birds per cage) in a completely randomized block design experiment involving a 5 × 2 factorial arrangement of treatments (5 levels of CYA × 2 levels of MEL). All the birds were fed corn-bean basal diets (Table 1 ) providing the same ingredients. Both CYA and MEL were given in addition to the basal diet. Basal diets were formulated according to the requirements recommended by the National Research Council (NRC; 1994). Birds of T1 to T5 were fed basal diets with 0, 10, 20, 33.3, and 50 mg CYA per kg diet. Birds of T6 to T10 were fed CYA regimens similar to T1 to T5 but with 100 mg MEL per kg diet. The diets were offered in crumbles. Feed and water were provided ad libitum. All the birds were housed in stainless steel pens (100 × 200 cm) with fiberglass feeders under standard conditions of temperature, humidity, and ventilation. The experiment lasted 6 weeks, consisting of a starter phase from d 1 to 21 and a finisher phase from d 22 to 42.
Ingredients and chemical composition of the basal diet (%)

a Each kilogram of diet contains 25,000 IU of vitamin A (VA), 5000 IU of VD3, 12.5 IU of VE, 2.5 mg of VK3, 1.0 mg of VB1, 8.0 mg of VB2, 3.0 mg of VB6, 15 μg of VB12, 250 μg of biotin, 17.5 mg of nicacid, 12.5 mg of calcium pantothenate, 80 mg of Fe, 20 mg of Cu, 60 mg of Mn, 80 mg of Zn, 0.3 mg of Se, and 0.35 mg of I. AP: Available phosphorus, CP: Crude protein, Lys: Lysine, ME: Metabolizable energy, Met: Methionine, SAA: Sulphur amino acid.
Sample collection
On d 22 and 43, two birds from each cage were randomly selected and weighed. Blood samples were collected from the wing vein into a sterile syringe and centrifuged at 3000
Determination of biochemical serum markers
Sera were defrosted at 4°C; and immediately after defrosting the sera werethen used to determine the activities of aspartate aminotransferase (AST), and alanine transaminase (ALT), and the concentrations of creatinine (Crea), and uric acid (UA) by automatic biochemical analyzer, as soon as possible after defrost. The test kits were purchased from Jiancheng Bio. Co., Nanjing, China.
Organ size
The relative weight (% BW) of livers and kidneys were calculated according to the following formula:
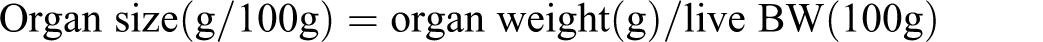
Histopathologic measurements of livers and kidneys
Sections of liver and kidney tissues fixed in formalin were embedded in paraffin and stained with hematoxylin–eosin for light microscopy. All biopsies were examined independently and in a blinded fashion by four investigators. For each specimen, five histologic features were evaluated. For each feature, the investigator indicated the degree of involvement by placing a mark on a continuous 0–2 scale. On this scale, 0 represented normal or no involvement; 1 represented no serious pathological changes; and 2 represented obvious or serious pathological changes. For each individual histologic parameter, a mean score was computed from the separate scores of the 4 investigators to evaluate the degree of pathological changes. The individual features of the liver that were evaluated included (1) cell tumefaction; (2) fatty degeneration; (3) cell granular degeneration; (4) cell hemorrhage; (5) vacuolus degeneration; and the evaluated individual features of the kidney included (1) dilated renal tubules; (2) small blood vessel expansion; (3) acute, interstitial, mononuclear cell infiltration; (4) interstitial hemorrhage; (5) epithelial cell expansion.
Determination of AR
Sections in PBS solution were made into cell homogenates through physical method and passed through a 0.05-mm screen. After centrifugation at 500–800 rpm, the homogenates were washed 2 times using PBS solution (pH = 7.0–7.4) and the cell concentrations were adjusted to 1 × 10 6 cells/ml. After all the preparations have been completed, 100 μl of cell suspension was dropped into the flow tube and 5 μl annexin V-fluorescein isothiocyanate (FITC) and 5 μl polytrans isoprene (PI) were infused. The tube was made to stand for 15 min at room temperature away from light after homogenizing. Then 400 μl of binding buffer was added, homogenized, and determined by flow cytometry. The AR of kidney was analyzed using CellQuest (Becton, Dickinson and Company, NJ, USA). All these work must be done within 6 h.
Statistical analysis
Data were expressed as mean ± SD and were analyzed by one-way analysis of variance (ANOVA) using Statistical Package for Social Sciences 13.0 (SPSS). Data were also analyzed as a 5 × 2 factorial arrangement (grade levels of CYA; grade levels of MEL) using the general linear model (GLM) procedure of SPSS. The model included the effects of CYA, MEL, and the two-way interaction. Each replicate was an experimental unit. Differences were considered statistically significant at
Results
Analyses of CYA and MEL values in experimental diets
The determined CYA and MEL levels in the finisher experimental diets are summarized in Table 2 . The recovered amounts of MEL ranged from 86% to 92% and CYA ranged from 53% to 81%, which are in line with expectations when mixing and assay errors are considered.
Values of CYA and MEL in the experimental diets

CYA: cyanuric acid, MEL: melamine, ND: not detectable.
Biochemical serum markers
No differences were observed in ALT and AST activities (Table 3
). However, on d 22, the concentration of UA and Crea increased when birds were fed diets containing CYA and MEL (Table 4
). Crea concentrations were significantly higher when birds were fed CYA at the concentration of 50 mg/kg;, moreover, UA and Crea concentrations were significantly greater when birds were fed CYA at 33.3 mg/kg with MEL 100 mg/kg (the ratio of CYA/MEL is 1:/3;
Activities of alanine transaminase (ALT) and aspartate aminotransferase (AST)a

CYA: cyanuric acid, MEL: melamine.
a The data are expressed as mean ± SD (
Concentrations of uric acid (UA) and creatinine (Crea)a

CYA: cyanuric acid, MEL: melamine.
a The data are expressed as mean ± SD (
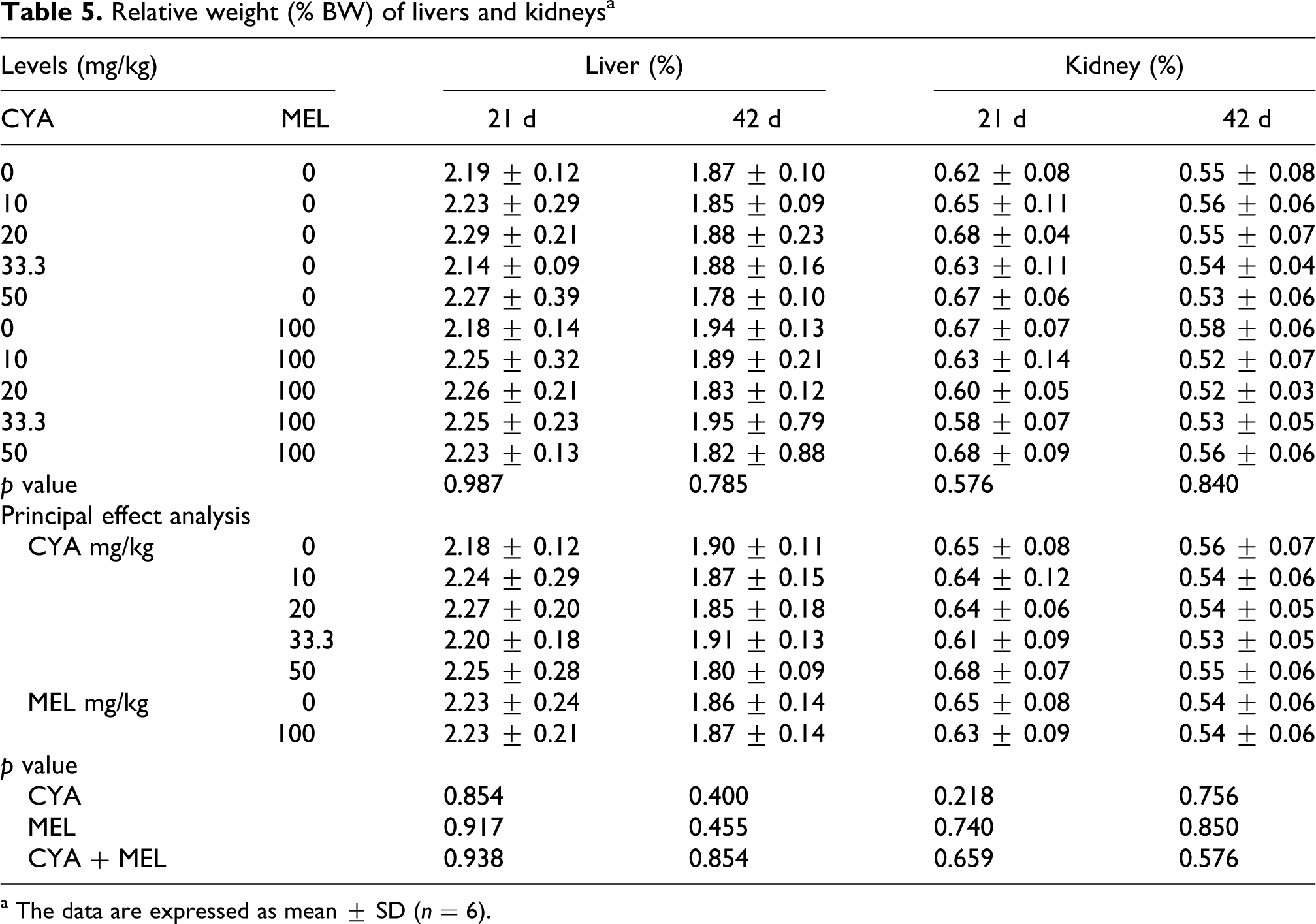
Organ size
Relative weight (% BW) of livers and kidneys of broilers are presented in Table 5
. No differences were observed in liver and kidney weight (
Relative weight (% BW) of livers and kidneysa
a The data are expressed as mean ± SD (
Histopathologic findings
No crystals were found in the kidneys of the birds, but the livers and kidneys were damaged to various degrees because of the addition of CYA and MEL compared to the control group (Figures 1 and 2). No serious pathological changes were observed in the liver of birds fed diets supplemented with 10 or 20 mg of CYA with or without 100 mg of MEL/kg of feed and 33.3 mg of CYA/kg of feed (Figure 1A-D, G-H
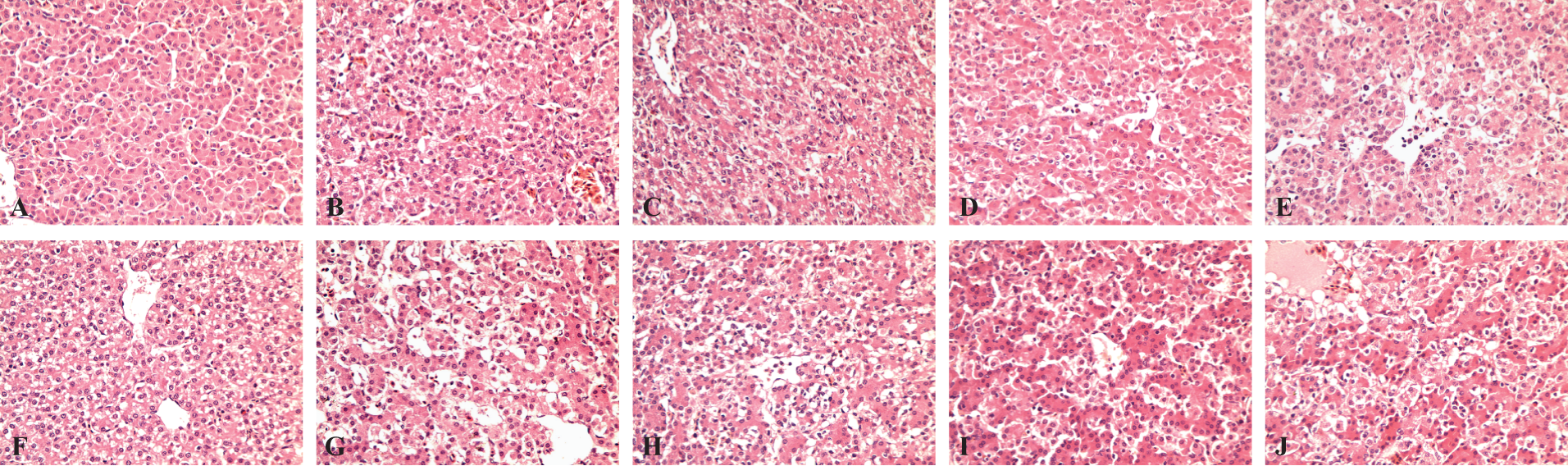
Pathological changes in livers from broilers that received cyanuric acid (CYA) with or without melamine (MEL) for 42 d (hematoxylin–eosin [HE] staining, original magnification, ×400). (A) Control; (B) 10 mg of CYA/kg of diet; (C) 20 mg of CYA/kg of diet; (D) 33.3 mg of CYA/kg of diet; (E) 50 mg of CYA/kg of diet; (F) 100 mg of MEL/kg of diet (G) 10 mg of CYA with 100 mg of MEL/kg of diet; (H) 20 mg of CYA with 100 mg of MEL/kg of diet; (I) 33.3 mg of CYA with 100 mg of MEL/kg of diet; and (J) 50 mg of CYA with 100 mg of MEL/kg of diet.

Pathological changes in kidneys from broilers that received cyanuric acid (CYA) with or without melamine (MEL) for 42 d (hematoxylin–eosin [HE] staining, original magnification, ×400). (A) Control; (B) 10 mg of CYA/kg of diet; (C) 20 mg of CYA/kg of diet; (D) 33.3 mg of CYA/kg of diet; (E) 50 mg of CYA/kg of diet; (F) 100 mg of MEL/kg of diet; (G) 10 mg of CYA with 100 mg of MEL/kg of diet; (H) 20 mg of CYA with 100 mg of MEL/kg of diet; (I) 33.3 mg of CYA with 100 mg of MEL/kg of diet; and (J) 50 mg of CYA with 100 mg of MEL/kg of diet.
AR of kidneys
Higher AR was observed in kidneys collected from birds fed CYA and MEL contaminated feed on d 22 in our study (Table 6
). The AR of kidneys tended to increase when broilers were fed diets contaminated with CYA and MEL. The AR of kidneys significantly increased when birds were fed CYA at 50 mg/kg (
The apoptosis rate (AR) of kidneysa

a The data are expressed as mean ± SD (
Discussion
In previous studies, the toxicities of CYA and MEL mainly affected kidneys thereby and inducing renal damage, 16–20 while serum hepatic enzyme concentrations have been shown to be normal in pets from outbreaks of renal failure associated with MEL and CYA in 2004 and 2007. 1 However, Neerman et al. 21 found that glutamic pyruvic transaminase ([GPT] AST) activity increased in mice after injection with MEL dendrimer at 40 mg/kg BW. In a clinical setting, liver or kidney injury is often detected using a battery of blood tests. In our study, ALT and AST activities were chosen to represent the enzymes that reflect the hepatic health and no differences were observed. Reasons for this difference may be that the supplemental levels CYA and MEL supplemental levels were not high enough to induce obvious liver damage in our study, and toxicities of CYA and MEL may be different in various animal species. 22 Puschner et al. 15 reported that both MEL and CYA would harm the kidney. Brown et al. 1 also measured the serum Crea and blood urea nitrogen (BUN) concentrations that can be presumed to reflect renal health and found both Crea and BUN concentrations were beyond the normal range. However, the major urinary nitrogenous components of birds and mammals are different, and UA is the end product in nitrogen metabolism in birds, 23 ; so UA rather than BUN was expected to reflect the renal health in our study. It was possible that not only the level of CYA and MEL addition but also the ratio of CYA and MEL would play a role in renal damage, and the toxicity of CYA is higher than MEL. According to previous studies, the toxicity of combinations of CYA and MEL combined was higher than CYA or MEL alone. Moreover, Xu et al. 24 found that CYA could combine with MEL at a ratio of 1:/3. Our study showed that all these may explain the harmful effects of UA and Crea concentration was the greatest when CYA/MEL ratio was 1:3 in our study. Both CYA as the main effect and the interaction of CYA and MEL significantly increased the Crea concentration, which may demonstrate this possibility.
Organ size can partially reflect organ’s health. 25 But it was possible that the dosage of CYA and MEL used in our study was not enough to induce severe damage to livers and kidneys.
In order to ensure crystal preservation, all samples were embedded in paraffin for 24 h and the histologic features were evaluated as soon as possible. In previous studies, high supplemental levels of CYA and MEL could cause kidney and liver damage but low levels were not serious in both the liver and kidney. 26,27 Moreover, MEL-CYA crystals were found in some cases.1, 11,15 Our study used much lower levels of CYA and MEL (CYA ≤ 50 mg, MEL ≤ 100 mg/kg of diet) compared to other studies (CYA ≥ 2000 mg/kg, MEL ≥ 5000 mg/kg of diet), 15 and this may be the reason for not causing serious liver and kidney damage and for lack of MEL-CYA crystal formation.
Apoptosis is a regular destruction of a cell. It is the result of an underlying conserved, endogenous cell death program to make the cell number homeostatic. But a balance between cell proliferation and cell death is essential for the normal functions.28–30 In our study, higher AR of the kidney was observed when birds were fed diets containing CYA and MEL; and based on this, we concluded that addition of both CYA and MEL could damage the kidneys to a certain degree but low levels of CYA and MEL could only affect few sensitive indexes such as the AR.
In conclusion, we presumed that CYA and MEL would harm the kidney of broilers to a certain degree. UA and Crea concentrations and the AR of kidney are sensitive to nephrotoxicity caused by CYA and MEL. Moreover, not only the concentrations of CYA and MEL but also the ratio of CYA and MEL may influence the toxicities. In our study, the harmful effects were the greatest when the CYA/MEL ratio was 1:3.
Footnotes
Acknowledgment and Funding
This trial was financially supported by the project of feed safety in the China, Ministry of Agriculture, and the specific research supporting program for academic sustentation research team in Sichuan Agricultural University.
