Abstract
Loxosceles gaucho spider venom induces in vitro platelet activation and marked thrombocytopenia in rabbits. Herein, we investigated the involvement of platelets in the development of the dermonecrosis induced by L. gaucho venom, using thrombocytopenic rabbits as a model. L. gaucho venom evoked a drop in platelet and neutrophil counts 4 h after venom injection. Ecchymotic areas at the site of venom inoculation were noticed as soon as 4 h in thrombocytopenic animals but not in animals with initial normal platelet counts. After 5 days, areas of scars in thrombocytopenic animals were also larger, evidencing the marked development of lesions in the condition of thrombocytopenia. Histologically, local hemorrhage, collagen fiber disorganization, and edema were more severe in thrombocytopenic animals. Leukocyte infiltration, predominantly due to polymorphonuclears, was observed in the presence or not of thrombocytopenia. Thrombus formation was demonstrated by immunohistochemistry at the microvasculature, and it occurred even under marked thrombocytopenia. Taken together, platelets have an important role in minimizing not only the hemorrhagic phenomena but also the inflammatory and wound-healing processes, suggesting that cutaneous loxoscelism may be aggravated under thrombocytopenic conditions.
Introduction
Accidents inflicted by spider species belonging to the genus Loxosceles may result in severe clinical pictures, with marked local and systemic symptoms. Dermal regions exposed to the venom may exhibit skin complications and progress to dermonecrosis, the hallmark of cutaneous loxoscelism. Currently, these accidents are reported in tropical, subtropical, and even in temperate zones of the globe. 1 In spite of their wide distribution, the pathophysiology of loxoscelism is not completely understood and therapeutic interventions are not standardized yet and are not always effective for treating cutaneous or visceral loxoscelism. 2
In whole South America, the most common species of medical importance are Loxosceles laeta, Loxosceles intermedia, and Loxosceles gaucho. The last one is the main species responsible for accidents in São Paulo State, Brazil. 3 Its venom contains different types of toxins, including phospholipases D, inhibitor cystine knot peptides, and astacin-like metalloproteases. 4 Although loxoscelism may occasionally cause severe systemic manifestations, such as disseminated intravascular coagulation, intravascular hemolysis, and acute renal failure, the dermonecrotic injury is the most common clinical manifestation and also the hallmark of Loxosceles envenomation. The local lesion begins with edema and hemorrhage during the first hours, followed by endothelial injury, polymorphonuclear accumulation, and a severe and disrupted picture of inflammation associated with local thrombosis. Finally, the tissue damage can progress to an ulcerative necrotic skin lesion, whose healing can be difficult and delayed. 5,6
Platelets are crucial for the hemostatic machinery and its regulation. However, other pathophysiologic roles for platelets have been revealed in other conditions so that a growing field of research has addressed their participation in painful, neoplastic, immune, and inflammatory phenomena. 7 –9 In inflammation, platelets act as a natural reservoir of a wide variety of important substances, such as vasoactive amines, purinergic nucleotides, leukotrienes, and cytokines, besides expressing important adhesion molecules and other crucial proteins for immunity and inflammation. 10,11
Decreased platelet count and dermonecrosis resulting from Loxosceles bites have been reported in humans. 12,13 We verified previously that rabbits injected intradermally (i.d.) with Loxosceles gaucho venom develop severe early thrombocytopenia. Interestingly, this platelet consumption was not related with disseminated intravascular coagulation and/or intravascular hemolysis, 14 two important systemic phenomena in the pathogenesis of the visceral loxoscelism. We also confirmed in vitro that this venom and its main component, phospholipase D, promote aggregation, adhesion, and activation of both human and rabbit platelets. 15 In spite of our previous investigations, the role of platelets in the Loxosceles venom-induced dermonecrosis has been poorly investigated. Although thrombi and fibrin formation has been demonstrated to occur in the lesion area, no study has evaluated the participation of platelets in the pathogenesis of the dermonecrotic lesion.
Platelets exert an unique and intertwined link among coagulation, inflammation, and immunity. Currently, it is known that these non-nucleated cells are deeply involved, by distinct manners, in the inflammatory and immunological response. 16,17 Platelets have been even suggested to be potential targets for anti-inflammatory therapy. 18,19 Thus, in the current work, we studied the participation of platelets in the local injury induced by L. gaucho venom in rabbits, and the effect of systemic platelet depletion on the dermonecrotic lesion development.
Materials and methods
Animals and L. gaucho venom
Procedures involving animals were performed in accordance with the Guide for Care and Use of Laboratory Animals (2011) and the International Guiding Principles for Biomedical Research Involving Animals (2012). Protocols for animal experimentation were approved by the Ethical Committee for the Use of Animals at Butantan Institute (process number 129/2003). Adult male New Zealand albino rabbits, obtained from the Animal House of Butantan Institute, and three adult female goats, used for immunization procedures in the farm of the mentioned institution, were maintained under appropriate conditions for each species.
Specimens of L. gaucho were collected from São Paulo State, Brazil. Extraction of spider venom was performed by micropipette aspiration after electrostimulation of the cephalothorax region, as previously described. 20 Venom was pooled and immediately frozen until use.
Production of rabbit antiplatelet serum and IgG purification
Blood collection in rabbits, plasma-rich platelet preparation, platelet washing, platelet lysis, and protein content determination were performed as described previously. 21 Eight milliliters of platelet lysate suspension (400 μg mL−1) were injected i.d. into the medial dorsal region of two goats for production of rabbit antiplatelet serum. For the first immunization, the suspension was mixed with complete Freund adjuvant. Other three immunization boosters were repeated, using incomplete Montanide ISA 50V® (Seppic, Paris, France) as adjuvant. Immunoglobulin G (IgG) antibody titers were checked monthly in the goat serum by enzyme-linked immunosorbent assay (ELISA). Another goat was immunized only with the vehicle for obtaining control and inert IgG. One month after the fourth immunization, 200 mL of blood was withdrawn from each goat, and the serum was processed as described previously for obtaining purified antiplatelet IgG. 22 The protein content of IgG solutions was determined by the bicinchoninic acid assay. 23
Experimental groups and blood collection
In preliminary experiments, purified IgG obtained was injected in rabbits to determine the best doses to induce thrombocytopenia. IgG administration (doses of 30 mg kg−1, body weight
Complete blood cell counts were determined in a Serono-Baker hematological counter, model 9020 + AX. Differential leukocyte count was carried out on blood films stained with panchromatic stain.
Superficial and histological analysis of the local lesion
After euthanasia, superficial areas of the lesion (edema, erythema, ecchymosis, and/or necrosis) were drawn in transparent paper and measured by the software KS300 (Zeiss, Munich, Germany). Skin samples corresponding to the necrotic site and its adjacent areas were immediately fixed in 2% paraformaldehyde solution in phosphate-buffered saline, pH 7.4. Following 18 h of fixation, samples were transferred to 70% of ethanol and processed routinely for paraffin inclusion.
Histological sections were stained by Fraser–Lendrum’s method to allow a general analysis of the lesion as well as a better visualization of platelets and fibrin. The presence of platelets and their deposition in the lesion were studied by immunohistochemical staining using chicken immunoglobulin Y antibodies for labeling the rabbit platelet GPIIb-IIIa, as described previously. 24
Phagocytosis of opsonized zymosan by rabbit polymorphonuclears
Phagocytosis assays were performed in order to investigate the function and activity of circulatory polymorphonuclears after envenomation. After 4, 24, or 120 h of venom injection, leukocytes were isolated as described previously. 25 Phagocytosis of opsonized zymosan was performed with adaptations described elsewhere. 26 Briefly, heparinized blood (15 U mL−1) was mixed with Histopaque (Sigma-Aldrich, St Louis, Missouri, USA) and then centrifuged at 400g for 30 min at room temperature. Polymorphonuclears were separated in the bottom of centrifugation tube and then submitted to hypotonic hemolysis. Cells were washed, counted, and made up to a concentration of 4 × 105 mL−1. Then, they were incubated with opsonized zymosan particles (1 × 106 mL−1) for 40 min, and slides were prepared in a cytological centrifuge (20g, 100 s). After staining with Rosenfeld method, the number of phagocytosed zymosan particles by polymorphonuclears was counted. Scores were established considering the count of particles found within 200 polymorphonuclears.
Von Willebrand Factor, prothrombin time and activated partial prothrombin time assays
Circulating levels of von Willebrand factor (vWF) were determined in order to evaluate whether the combination of different treatments (venom, saline, antiplatelet IgG, or control IgG) would induce endothelial damage. A methodology previously described elsewhere 24 was employed using a commercial antibody (DiaSorin, Stillwater, Minnesota, USA) that recognizes both human and rabbit vWF by a sandwich-ELISA assay.
In order to evaluate whether any of the treatments promoted alterations in the coagulation system, prothrombin time (PT) and activated partial prothrombin time (APPT) were measured following the instructions of the manufacturer (Dade-Behring, Deerfield, Illinois, USA).
Statistical analysis
Two-way analysis of variance, followed by Tukey test, was used to analyze data and compare the results among the different groups. Statistical analyses were carried out on STATA™ software version 5.0 and SigmaStat software version 2.03 (SSPS, Chicago, Illinois, USA). Differences with p < 0.05 were considered statistically significant.
Results
Purified antiplatelet IgG (30 mg kg−1) induced expressive thrombocytopenia in rabbits. Complete blood cell counts at 1, 2, 4, 6, 24, 48, and 72 h showed that this dose did not affect the white and red series (data not shown). The thrombocytopenic status was sustained for at least 24 h after antiplatelet IgG administration in Thr-Ctr group.
As shown in Figure 1, L. gaucho venom progressively decreased platelet and leukocyte (neutrophil) counts in N-Exp group (animal with initial normal platelet count) until 24 h. Erythrocyte counts were not modified under any treatment. Platelet and leukocyte counts returned to normal after 120 h of venom injection.

Values of platelet, erythrocyte, leukocyte, and neutrophil counts in rabbit peripheral blood at 4, 24, and 120 h after i.d. injection of L. gaucho venom (5 μg). Data are expressed as mean ± SEM (n = 5). *p < 0.05 versus corresponding control at time zero.
Figure 2 depicts that phagocytosis index of polymorphonuclears isolated from blood circulation were not affected by antiplatelet IgG, Loxosceles venom, nor the association of both treatments. In addition, APTT and PT, and vWF levels were unaltered by all treatments employed in the present study, evidencing that no important alteration in hemostasis occurred.

Phagocytosis index by polymorphonuclear leukocytes, PT, APTT, and plasma vWF levels in rabbits submitted to different protocols of treatment. Data are expressed as mean ± SEM (n = 5). PT: prothrombin time; APTT: activated partial thromboplastin time; vWF: von Willebrand factor.
Macroscopic measurements of the cutaneous lesion after venom injection are shown in Figure 3. Small areas of ecchymosis were evidenced after 4 h in the Thr-Exp group; however, any kind of hemorrhagic lesion was observed in the N-Exp group. After 120 h, the extension of the necrotic scar was also significantly larger in the Thr-Exp group than that produced in the normal platelet count (N-Exp) group.

Extension of local reaction (edema, erythema, and ecchymosis), and necrotic scar in the skin of Thr-Exp (thrombocytopenic) or N-Exp (with initial normal platelet count) rabbits after 4, 24, and 120 h of L. gaucho venom (5 μg) i.d. injection. Superficial areas of lesions were measured, analyzed, and correlated to the different treatments. Data are expressed as mean ± SEM (n = 5). *p < 0.05 versus N-Exp group.
Histopathological analysis (Figure 4) evidenced a more severe progression of the dermonecrotic lesion in thrombocytopenic animals than in those with initial normal platelet counts. Hemorrhage and edema were more intense in the Thr-Exp group (Figure 4B) than in the N-Exp group at 4 h (Figure 4A). Similarly, at 24 h, disrupted fibrin deposition was more evident and severe in thrombocytopenic animals. Fibrin deposition (fibrinoid areas) was a more expressive picture in the Thr-Exp group (Figure 4D) than in the N-Exp group (Figure 4C). Finally, after 120 h, the region of scar formation was deeper and more extensive in the Thr-Exp group (Figure 4F) than in the N-Exp group (Figure 4E). In thrombocytopenic animals, a more extensive area of granulomatous tissue was visualized under the necrotic skin, indicating the presence of leukocytes and more intense tissue repair.

Histological sections of rabbit skin showing hemorrhage, edema, and necrotic scar induced by L. gaucho venom (5 μg) in thrombocytopenic and normal rabbits. Hemorrhage was visualized by staining of erythrocytes in orange, and edema by clear areas without staining, mainly in the subjunctive space above muscular fibers; after 4 h, hemorrhage and edema were more intense in animals of the Thr-Exp group (B) than in those of the N-Exp group (A). Fibrinoid areas (red staining) showing fibrin deposition were more noticed in the Thr-Exp (D) than in the N-Exp (C) group at 24 h. The magnitude and depth of necrotic scars, visualized in nuances of yellow and orange, were more pronounced in the Thr-Exp group (F) than in the N-Exp group at 120 h (E). Animals of the Thr-Exp group, after the fifth day of envenomation, also presented outstanding zones of granulomatous tissue, seen in purple and located below and left of the scar region (F). G: Histological section of control skin (animal of N-Ctr group), showing the normal aspect of collagen fibers, stained in blue. Staining by Fraser–Lendrum's method. Bar scale: 500 μm.
Analysis of sequential histological sections stained by Fraser–Lendrum’s method (Figure 5A and C) and by immunolabeling of platelets (Figure 5B and D) confirmed the distinctive patterns of thrombus formation induced by L. gaucho venom at the site of venom injection. Some large venom-induced thrombi had expressive presence of platelets (Figure 5C), which were confirmed by immunolabeling (Figure 5D). But even in animals of the Thr-Ctr group, with very low platelet counts, thrombi formation was confirmed by histological analysis. Intense fibrin formation, red cells, and leukocytes were observed in large thrombi found in vascular bed in the Thr-Exp group at 24 h (Figure 5A). A sequential section of Figure 5A, shown in Figure 5B, was immunolabeled for platelets, and confirmed a marked thrombus formation, even in the condition of very low platelet count in circulation. In general, vascular damage was easily noted. The absence of an endothelial cell layer on extensive areas of the vessel wall, the presence of leukocytes, and thrombus formation – under the presence or absence of platelets – are important elements indicating local hemostatic disturbances and inflammatory reaction.
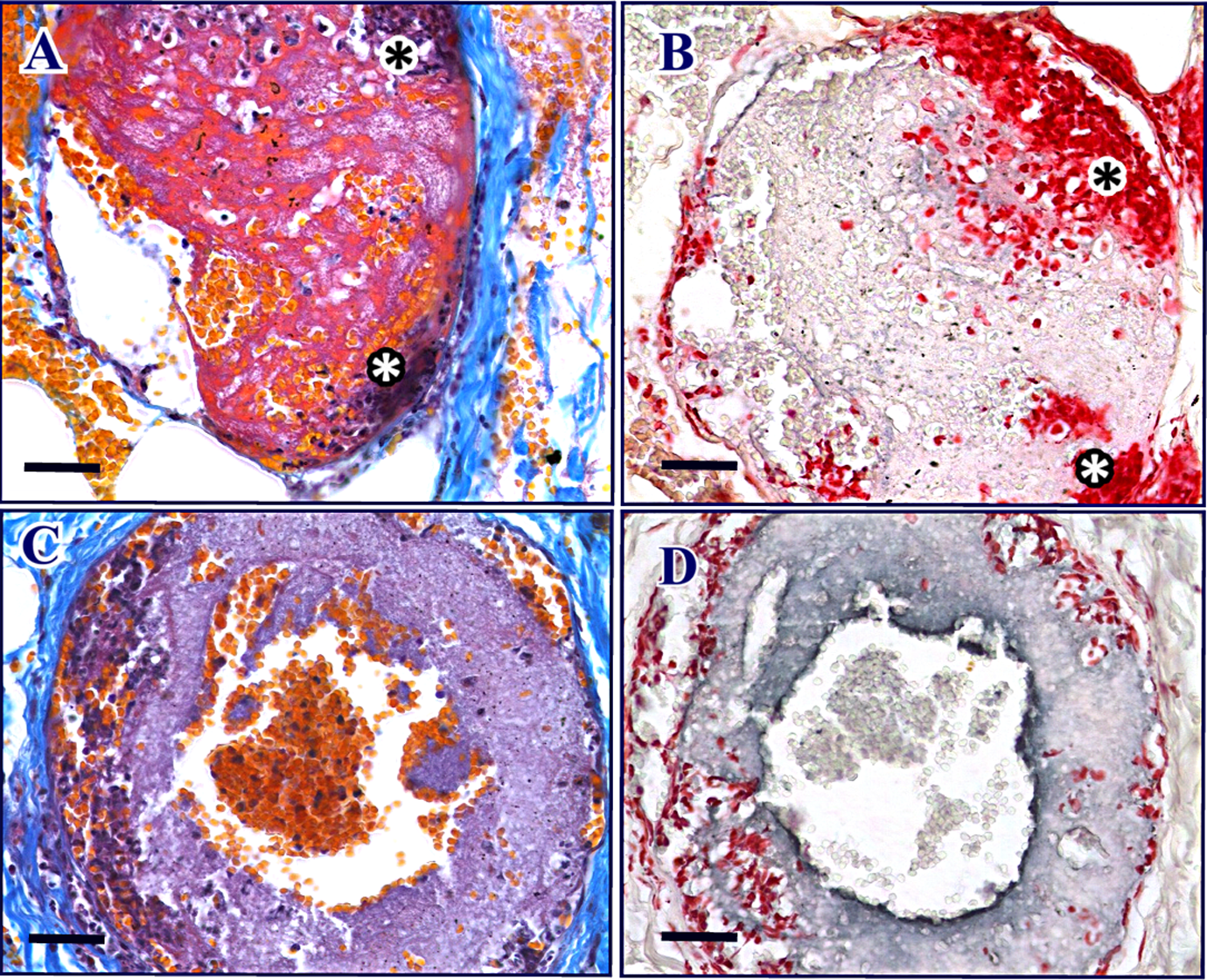
Dermal tissue thrombi induced by L. gaucho venom (5 μg) in either thrombocytopenic (A and B) or normal (C and D) rabbits visualized in sequential histological sections. (A) Large thrombus was observed in vascular bed, which presented erythrocytes – stained in orange – and leukocytes with dark-stained nuclei, within an intense fibrin formation (red) in animals of the Thr-Exp group after 24 h of venom administration. Sections were stained by platelet–fibrin staining method. This thrombus obstructed almost completely the vessel lumen. In some points, there is expressive agglomeration of polymorphonuclears (marked with asterisks) near the vessel wall. (B) Sequential sections of A were submitted to immunolabeling of platelets and confirmed the poor presence of platelets within the thrombus. In A, leukocyte nuclei are stained in dark blue, while in B, the same nuclei are stained in intense red. In both A and B, there is no evidence of consistent presence of platelets. (C) Massive radial deposition of platelets, presenting a purple
Discussion
Platelets are one of the circulating target cells for Loxosceles venoms. 14,15,27,28 However, their participation at the site of the bite has never been investigated. On that account, in this study, we investigated the role of platelets in the local dermonecrotic lesion induced by L. gaucho venom in rabbits, which is an experimental animal model that reproduces the pattern of skin injury observed in human victims. 29 Herein, we addressed the involvement of platelet participation in the dermal lesion induced by Loxosceles venom, and their importance for local fibrin deposition and thrombus formation. We showed that hemorrhage, edema, ecchymosis, and scar formation induced by Loxosceles venom were more severe during platelet depletion.
The cell profile in peripheral blood of rabbits inoculated with L. gaucho venom in the present study was comparable to that reported previously. 15 However, the extent of thrombocytopenia, leukopenia, and neutropenia was less severe in the current investigation since an inferior amount of venom was used here. Our current protocol was effective to induce intense and long lasting platelet depletion for at least 24 h, period of time which allows dermonecrotic lesion to reach its maximum size in rabbits. 29
Endothelial cell injury induced in situ by Loxosceles venom has been unequivocally shown, by demonstrating the attack of endothelial cell membrane, 30 interleukin-8 and monocyte chemoattractant protein-1 release, E-selectin expression, and cleavage of thrombomodulin and protein C receptor in cell surface. 31 –33 Morphologically, L. intermedia venom induce an increase in filopodia projections, cell retraction, disorganization of actin microfilaments, anoikis, and cell detachment. 34,35 These mechanisms help to understand our observations using L. gaucho venom, which produced denuded areas in the inner vessel wall, with marked absence of endothelial cells. Furthermore, large thrombi were observed in direct contact with the subendothelial matrix – easily visualized as collagen fibers – evidencing the in situ activation of blood coagulation, even under the condition of thrombocytopenia.
Severe leukocyte infiltration, with prominent neutrophil presence, is undoubtedly a classical signal of tissue inflammation in cutaneous loxoscelism. 36,37 Many studies in the last decades have pointed out that neutrophil migration and its presence in inflammatory processes are influenced by platelet activation and correlated events, such as the expression of adhesion molecules – P-selectin, E-selectin, CD40, and intercellular adhesion molecule-1 – and the liberation of a great spectrum of inflammatory mediators after platelet degranulation. 19 Thus, platelets have been shown to increase leukocyte recruitment in the inflammatory response. 38 Herein, focusing on platelet influence in the local inflammatory response induced by L. gaucho venom, we evidenced that neutrophil infiltration and the consequent vasculitis is not diminished by the condition of systemic platelet depletion. The more rapid and intense inflammatory phenomena in thrombocytopenic animals suggest that platelets do not play a pivotal role in exacerbating neutrophil recruitment in cutaneous loxoscelism. Histopathological analysis confirmed that expressive neutrophil infiltration occurs even under platelet depletion. Our findings are in agreement with other reports showing that thrombocytopenia did not interfere with neutrophil migration neither in a model of tendon injury 39 nor in a model of skin lesion. 40
Local hemorrhage, which appeared macroscopically as ecchymotic areas, was visualized as early as 4 h at the site of venom inoculation in thrombocytopenic animals (Thr-Exp group). The absence of local hemorrhage at the same time interval in envenomed animals with normal initial platelet counts (N-Exp group) demonstrates the importance of platelets in delaying and minimizing the local hemorrhage induced by venom. Results from PT and APTT showed that the coagulation system was functioning normally, ruling out the possibility of systemic alterations in blood coagulation. Interestingly, and in agreement with our observations, some studies have provided evidences that the condition of thrombocytopenia alone is not enough to explain hemorrhagic states. For instance, the isolated condition of low platelet counts was not demonstrated to induce bleeding, and the occurrence of hemorrhage was associated with the presence of inflammation. 41 In line with these observations, our results showed a more severe picture of venom-induced hemorrhage under the condition of platelet depletion. In addition, edema and scar formation were more severe in thrombocytopenic animals. These findings suggest that platelets are important to minimize many of the injurious events whose progression culminates in dermonecrotic loxoscelism. Since Loxosceles venoms induce per se a marked local inflammatory reaction, 42,43 hemorrhagic phenomena may be intensified by platelet depletion. Furthermore, it is worth noting the high presence of sphingomyelin on platelet membrane, 44 the main molecular target for Loxosceles phospholipase D components. The lack of natural substrate for phospholipase D on platelet surface during thrombocytopenia is a condition that may exacerbate the activity of this enzyme in other target cells. Thus, a possible role in the dermal protection may be lost, under the condition of platelet depletion, in the local of Loxosceles injury.
Loxosceles venom-induced dermonecrosis has its pathogenesis intimately related to disrupted inflammation and local activation of blood coagulation. 45 New mediators have been demonstrated as potential participants in the dermonecrotic process, such as lysophosphatidic acid (LPA). Although we have evidenced that Loxosceles venom-induced platelet activation is not directly dependent on LPA, 15 receptors for this molecule exposed in other cell types could broaden the spectrum of biological effects, such as the leukocyte attraction and the consequent inflammatory response. The LPA pathway was recently demonstrated to mediate the liberation of cytokines by fibroblasts exposed to Loxosceles venom, 46 what reinforces the participation of LPA in the inflammation induced by Loxosceles venom.
Local hemorrhage and dermonecrosis might result from the association of local inflammation and unregulated activation of hemostasis since both phenomena were observed at the site of venom action. 14 Leukocyte infiltration, hemorrhage, and extensive formation of thrombi induced by Loxosceles venoms are compatible with fibrinoid necrosis. 37 This picture was observed herein in both conditions, with and without platelet depletion. Fibrin deposition and thrombus formation occurred at the site of venom injection in a platelet-independent manner, demonstrating that local activation of blood coagulation induced by Loxosceles venom minimally depends on circulating platelets. In fact, Loxosceles venoms attack not only endothelial cells, as mentioned earlier, but also extracellular matrix components, such as heparin sulfate proteoglycan, fibrinogen, entactin, and fibronectin. 13 Thus, all these injurious actions may explain, at least in part, the triggering events of coagulation disorders seen in the local loxoscelism.
In conclusion, thrombocytopenic animals developed a more severe picture in the initial development, progression, and resolution of the dermonecrotic lesion induced by Loxosceles venom. Thus, our results suggest that platelets have a protective role during the development of the dermonecrotic lesion induced by this spider venom.
Footnotes
Acknowledgment
Authors would like to thank Dr Hisako G Higashi and Ronaldo A Ferreira for their assistance with the antibody production in goats.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Fundacão de Amparo à Pesquisa do Estado de São Paulo (FAPESP, Brazil; grant 03/00841-0), Agencia Nacional de Promoción Científica y Tecnológica (Argentina; PICT-2010-1908), and Fundação Butantan (Brazil).
Author’s Note
The present research was part of the PhD thesis of FLT, accomplished at the Department of Pathology, Faculty of Veterinary Medicine and Zootechny, University of São Paulo, Brazil.
