Abstract
Mylotarg (Gemtuzumab ozogamicin [GO]), an antibody drug conjugate comprising a CD33-directed antibody linked to calicheamicin, is approved for use in certain acute myeloid leukemia patients. Following reports of prolonged thrombocytopenia and hemorrhagic events in a subset of patients, a detailed series of in vitro and ex vivo studies was performed at the request of regulators, both to look at the effects of GO on platelet production and to determine whether treatment with GO was likely to affect platelet aggregation under a variety of conditions. Treatment with GO resulted in cellular cytotoxicity and/or decreased differentiation during human megakaryocyte development. However, GO did not impair platelet aggregation under the experimental conditions evaluated. Ultimately, the effect of GO on megakaryocyte development observed in our studies was determined to have no impact on the risk-benefit assessment in the intended patient population, as thrombocytopenia is a known side effect of GO, and monitoring of platelet counts in patients is already strongly recommended.
Introduction
Drug-related platelet effects, including thrombocytopenia and decreased platelet function, are commonly reported adverse reactions in patients undergoing cancer treatment.1-3 Gemtuzumab ozogamicin (GO; Mylotarg), an antibody-drug conjugate (ADC) consisting of a calicheamicin derivative linked to a humanized IgG4 directed against CD33, is approved for use in patients with newly diagnosed or refractory CD33-positive acute myeloid leukemia (AML).4-6 Protracted thrombocytopenia unaccompanied by other cytopenias has been reported in some GO patients, with various proposed mechanisms that include an immune-mediated process, decreased thrombopoietin secretion due to sinusoidal obstruction syndrome (SOS), or impaired platelet production due to targeting of CD-33-expressing hematopoietic precursors.4,7-10 Following reports of ≥grade III thrombocytopenia and bleeding in patients treated with GO + daunorubicin 5 when compared with daunorubicin alone, a regulatory health agency requested an investigation into the impact of GO treatment specifically on megakaryocytes and platelet production, as well as an evaluation of the effect of GO on platelet function. The request included evaluation of platelet function in patients concurrently treated with anticoagulant therapies and those with concomitant severe renal dysfunction.
Existing nonclinical data from GO investigations in animal species provided little understanding concerning the mechanism of the prolonged platelet decreases reported in patients. However, these studies would not be expected to detect an on-target risk to megakaryopoiesis in humans; GO does not bind the CD33 target antigen in nonclinical species and therefore nonclinical toxicology studies would only facilitate identification of off-target effects. In toxicology studies that evaluated GO in rats and cynomolgus monkeys, platelet decreases were only observed during a single-dose study in rats with concurrent liver injury; no effects on platelets and only minor decreases in bone marrow hematopoietic (primarily erythroid) cellularity were reported in longer duration studies. An investigation into the mechanism of hepatic SOS and thrombocytopenia in cynomolgus monkeys administered antibody-calicheamicin conjugates also used a nonbinding construct to characterize target-independent effects of ADC treatment. 10 In that study, an acute decrease in platelets was observed and was attributed to platelet accumulation within hepatic sinusoids; platelet numbers returned to baseline values after the first 3-week dosing cycle. Subsequent administrations resulted in less severe acute post-dosing effects, but those recoveries were not as robust as the initial one, resulting in a gradual decrease in circulating platelet numbers over the course of the 63-day study. Decreases in serum thrombopoietin were noted and, although there was no effect on megakaryocyte or other hematopoietic cellularity within the bone marrow of study animals, the authors suggested that with ongoing hepatic damage, inadequate stimulation of the bone marrow might develop. The GO-related inhibition of platelet production due to a target-dependent effect in patients has been proposed; however, there are conflicting reports regarding the extent to which CD33 is expressed on megakaryocytic lineage cells and their progenitors, with most concluding that expression exists only in a small minority of these cells.11-14 Under such circumstances, an in vitro megakaryopoiesis assay using CD34+ cells derived from healthy human donors may help to investigate any potential direct effects of GO on platelet production from bone marrow progenitor cells. 15
Platelet function can be assessed by several methods, although logistical factors, such as timing, sample handling, laboratory location relative to sampling, and suspected mechanism of dysfunction can influence platelet integrity and assay selection.16-18 These factors, combined with the specific regulatory requests, made it logistically problematic to obtain platelet samples from diverse, targeted patient populations, and then to submit them promptly for assessment under a uniform and controlled set of experimental conditions. For these reasons, in vitro platelet systems using platelets from healthy human donors were pursued. This approach was supported by the fact that in vitro evaluation of platelet aggregation using anticoagulants that act directly on platelets has been described,19,20 and also that characteristics of uremic plasma have been demonstrated to play a role in diminished platelet function, aggregation included, in patients with impaired renal function, based on functional testing of normal platelets exposed to uremic plasma.21-23 Accordingly, we believe that multiple aspects of this complex request were recapitulated satisfactorily in vitro, using platelets obtained from normal donors at the testing site.
Materials and Methods
Effects of GO on Human Differentiated Megakaryocytes and Platelets
Test article and positive control
Serial dilutions were prepared from the 1 mg/mL GO stock in a vehicle comprising 0.07% sodium phosphate, 1.55% sucrose, 0.58% sodium chloride, and 0.91% dextran (pH of 7.3) to generate stock concentrations of 3, 10, and 30 µg/mL. Furthermore, 5-Florouracil (5-FU), used as a positive control in this assay was diluted in dimethyl sulfoxide (DMSO) to generate working stock concentrations of 0.05 and 0.5 mg/mL, which was further diluted 1:50 volume/volume (v/v) with X-Vivo Medium (Lonza Bioscience, Alpharetta, Georgia).
Cells and treatment
Human CD34+ cells, derived from normal bone marrow from 3 different donors (NorCal Biologics, California) were stored in the gaseous phase of liquid nitrogen. On the day of the experiment, cells were thawed rapidly, contents of each vial were diluted in 10 mL of Iscove’s modified Dulbecco’s medium containing 10% fetal bovine serum (IMDM + 10% FBS) and washed by centrifugation (approximately, 1200 rpm for 10 minutes, room temperature). The supernatant was discarded and the cell pellets were resuspended in IMDM + 10% FBS. A cell count (3% glacial acetic acid) and viability assessment (trypan blue exclusion test) were performed for each bone marrow sample. Cells from three donors were pooled and then plated in X-Vivo medium (Lonza; Lot No. 0000677610) containing 50 ng/mL rhSCF (Peprotec; Lot No. 41534 F1216) and 50 ng/mL rhTpo (Peprotech; Lot No. 101544 A2517) at approximately 20,000 cells per well in 96-well plates in a humidified incubator at 37°C, 5% CO2. Cells were dosed in triplicate with the test articles described above; 10 µL of each test solution was added to 190 µL of cell suspensions in X-Vivo media to achieve the desired, clinically relevant test concentrations of 0.15, 0.5, and 1.5 µg/mL for GO or 0.05, 0.5, and 5 µg/mL for 5-FU. Wells containing no solvent, the test article vehicle, and a DMSO solvent control (for 5-FU) were also included. PBS was added to wells not containing cells to reduce evaporation over the course of the study. Four replicate plates were prepared, one for each time point tested.
Flow cytometry
A cocktail was prepared with anti-CD41-FITC (Beckman Coulter; Cat No. IM0649U, Lot No. 63) and 30 µM DAPI (Biolegend; Lot No. B161925). On study days 3, 7, 10, and 14, the plate was centrifuged and the medium was removed. The wells were then washed by centrifugation with PBS at 1500 rpm for 10 minutes. The cocktail (17.5 µL per well) containing anti-CD41 and DAPI was added and incubated for 30 minutes at 4°C and then washed by centrifugation with PBS+ 2% FBS twice prior to reading on the cytoFLEX (Beckman Coulter). Megakaryocyte differentiation was assessed using CD41 expression (CD41 dim, CD41 mid, and CD41 bright) levels. Samples were evaluated for relative percentage and absolute count of early, mid-, and late-stages of megakaryocytes as well as absolute platelet count at each test concentration and time point in triplicate. The mean ± standard deviation of triplicate measures were reported. A two-tailed standard t test was performed to assess statistical significance.
Effects of GO on Human Platelet Aggregation
Vehicle control
A stock vehicle solution of the previously described GO excipient was prepared. On days of use, the stock solution was diluted in 0.9% NaCl to create a working solution.
Test article
The test article was diluted in water to generate a stock of 1 mg/mL, and then diluted on the day of use in 0.9% sodium chloride (vehicle) to provide a dosing formulation at 7.5 µg/mL.
Anticoagulants
Rivaroxaban (Cayman Chemical; Lot No. 0458274-40): working solution of 30 µg/mL was prepared by diluting a thawed aliquot of 3 mg/mL stock solution 100-fold in DMSO. Dabigatran (Cayman Chemical; Lot No. 0464319-28): working solution of 30 µg/mL was prepared by diluting a thawed aliquot of 0.3 mg/mL stock solution 10-fold in DMSO. Ticagrelor (Cayman Chemical; Lot No. 0494081-14): working solution of 500 µM was prepared by diluting a thawed aliquot of 10 mM stock solution 20-fold in DMSO. Acetylsalicylic Acid (Sigma-Aldrich; Lot No. SLBN2916V): 24 mg of ASA was dissolved in 10 mL of DMSO to create a stock/working solution of 13.3 mM.
Platelet-rich and platelet-poor plasma (PPP) samples
Human whole blood was collected on days of analysis in BD Vacutainer 3.2% sodium citrate tubes from healthy human donors who had fasted for approximately 4 hours. Donors were screened to ensure that they were nonsmokers who had not taken prescription drugs, acetylsalicylic acid, or nonsteroidal anti-inflammatory drugs within the 7 days prior to blood collection. Whole blood samples were mixed gently and placed at ambient condition until centrifugation. The blood samples from each donor were centrifuged at 200 relative centrifugal field (RCF) for 10 minutes at 21°C to obtain platelet-rich plasma (PRP). The remaining blood was re-spun in a centrifuge set at 2400 RCF for 10 minutes at 21°C (break set to the on position) to obtain the PPP. The PRP test sample was analyzed on the ADVIA 120 hematology instrument for platelet count, and then standardized using PPP as a diluent to obtain a final platelet concentration between 200 × 103 cells/µL and 300 × 103 cells/µL. Once the standardized PRP sample was obtained, it was analyzed on the ADVIA 120 hematology instrument for a final confirmation of the platelet count.
Uremic PPP samples
Human whole blood was collected from 5 human donors with glomerular filtration rates of less than 45 (≥stage 3b renal failure) and processed as described above to obtain uremic platelet-poor plasma (UPPP). Donors were otherwise healthy and non-dialyzed, had not used acetylsalicylic acid within 4 hours of sampling, and were not taking anticoagulant compounds with known direct effects on platelet function. Samples were stored at –20°C. The UPPP samples were subsequently used to standardize normal volunteer PRP samples as described above, creating platelet-rich uremic plasma (PRUP) to obtain a final platelet concentration between 200 × 103 cells/µL and 300 × 103 cells/µL.
Optical aggregometry
Optical platelet aggregation was performed using a Chrono-Log Aggregometer Model 700. Platelet-poor plasma (PPP) was used as a reference sample for 100% light transmission, whereas the standardized (Std) PRP test sample was used as a reference sample for 0% light transmission. Samples were incubated for 15 minutes at 37°C prior to being loaded in duplicate, and then aggregometry assessment was performed according to manufacturer’s instructions, using the following platelet aggregation agonists: adenosine diphosphate (ADP; Chrono-Log Corporation; Lot Nos. 3458/3460) at 10 µM, collagen (Chrono-Log Corporation; Lot No. 3456) at 2 µg/mL, arachidonic acid (AA; Chrono-Log Corporation; Lot No. 4209) at 1 mM, ristocetin (Chrono-Log Corporation; Lot No. 1304-23) at 1.25 mg/mL, and thrombin receptor activator peptide 6 (TRAP-6; Sigma-Aldrich; Lot No. 035M4761V) at 10 µM in samples from two donors, and 16 µM in a sample from one donor. Readings from the aggregometer were transferred to the Chrono-Log AGGRO/LINK 8 software, which generated an aggregation curve and calculated the maximal amplitude of the aggregation curve in percent, plus the area under the curve (AUC) in percent/minute. Descriptive statistical analysis of mean and standard deviation of the amplitude of the aggregation curve and AUC were subsequently performed using Microsoft Excel software.
Results
A GO-Related Effect Was Observed on Early and Mid-Stage Megakaryopoiesis
To evaluate the effect of GO on platelet production, megakaryocytes and platelets were derived from CD34+ human bone marrow cells in the presence of GO at clinically relevant concentrations of 0.15, 0.5, and 1.5 µg/mL in the following 3, 7, 10, and 14 days. Treatment of CD34+ cells with GO resulted in decreases in total CD41+ megakaryocyte numbers at all concentrations, with a decrease observed in the CD41 mid cells on day 3 at the highest concentration, and additional decreases at all concentrations of GO in the CD41 dim and mid subsets on day 7, followed by decreases in all CD41 subsets at all concentrations of GO on days 10 and 14 (Figure 1). Decreases in mature platelet numbers were also observed on day 14 at all concentrations of GO. Although a GO-related impact on early and mid-stage megakaryocyte development was observed, it was unclear whether the impact on late-stage megakaryocyte and platelet development was directly related to GO treatment or was the result of a test article-related decrease in megakaryocyte survival.
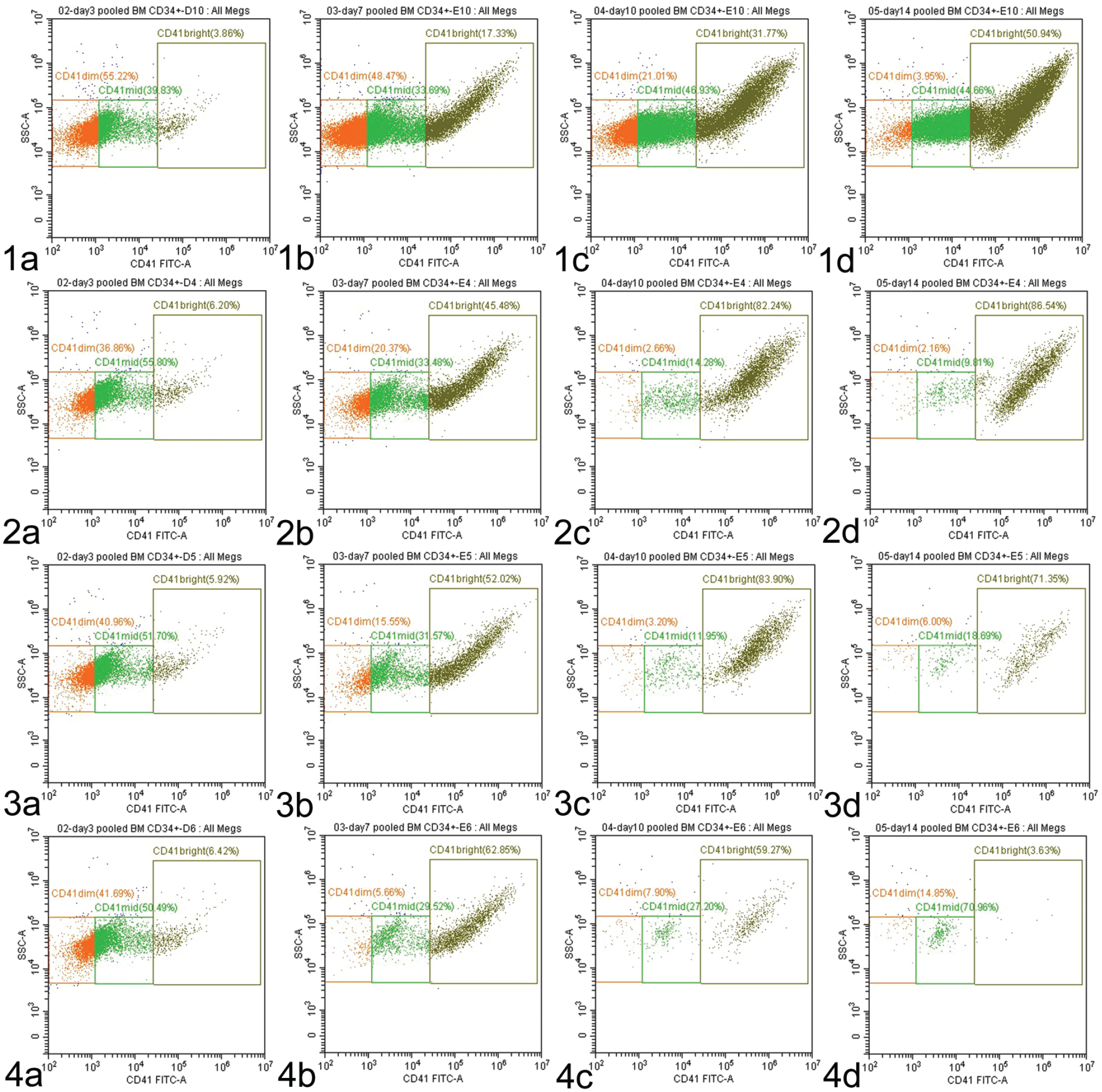
Representative flow plots of CD41+ megakaryocytes (MK) in the presence and absence of GO. As they mature, megakaryocytes normally acquire brighter CD41 expression and increasing side scatter (SSC); CD41 brightness is divided into CD41 dim (orange), CD41 mid (bright green), and CD41 bright (dark green) expressing cells. Row 1 depicts vehicle-treated cells with increasing CD41 brightness and side scatter on day 3 (1a), day 7 (1b), day 10 (1c), and day 14 (1d). Row 2 shows cells treated with 0.15 mg/mL GO, with no differences from control on Day 3 (2a) followed by decreases in CD41 dim and mid cells on day 7 (2b), and CD41 dim, mid, and bright cells on days 10 (2c) and 14 (2d). Row 3 shows cells treated with 0.5 mg/mL GO with no differences from control on day 3 (3a) decreases in CD41 dim and mid cells on day 7 (3b), and CD41 dim, mid, and bright cells on days 10 (3c) and 14 (3d). Row 4 shows cells treated with 1.5 mg/mL GO, with decreases in CD41 mid cells on day 3 (4a), decreases in CD41 dim and mid cells on day 7 (4b), and CD41 dim, mid, and bright cells on days 10 (4c) and 14 (4d). BM indicates bone marrow; FITC, fluorescein isothiocyanate, GO, gemtuzumab ozogamicin.
Treatment With GO Did Not Affect Platelet Aggregation
A series of experiments were conducted to determine the impact of GO on platelet aggregation. First, we evaluated the effects of a range of clinically relevant GO concentrations on platelet aggregation as measured by light transmittance aggregometry (LTA), with platelets derived from healthy human donors, both with GO alone and when concurrently treated with commonly prescribed anticoagulants which act directly on platelets. Next, we evaluated the effects of GO on platelet aggregation with platelets from healthy human donors that were perfused with plasma from uremic patient donors. Results below are displayed as AUC.
No GO-related effects were observed on platelet aggregation in PRP samples from healthy human donors at clinically relevant concentrations of 0.15, 0.5, and 1.5 µg/mL when evaluated either alone or in the presence of various platelet aggregation agonists (ristocetin, collagen, ADP, AA, or TRAP-6 (Figure 2). In addition, when PRP samples from healthy human donors were co-treated with 0.5 µg/mL GO and an anticoagulant (rivaroxaban, dabigatran, ticagrelor or acetylsalicylic acid) in the presence of platelet aggregation agonists, there was no GO-related exacerbation of expected effects of the individual anticoagulants (Figure 3).
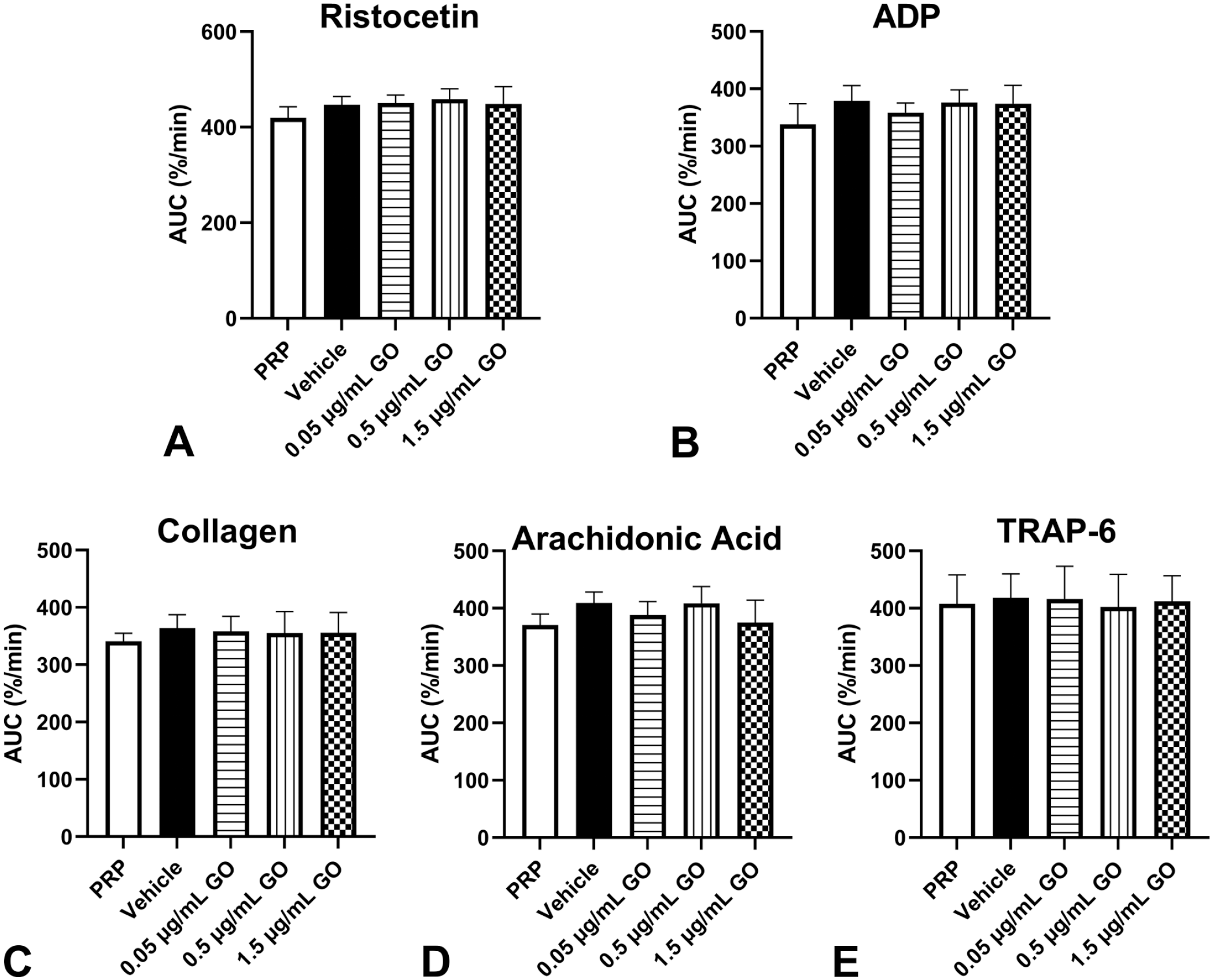
Mean GO-related effects on platelet aggregation in platelet-rich plasma samples from healthy human donors. Platelet-rich plasma derived from 2 male and 1 female human donors were incubated with increasing concentrations of GO (0.05, 0.5, 1.5 µg/mL) and stimulated, all set up in duplicate, with agonists (A) ristocetin (1.25 mg/mL), (B) ADP (10 µM), (C) collagen (2 µg/mL), (D) arachidonic acid (1 mM), or (E) TRAP-6 (10 µM). Optical aggregometry was performed and the results are expressed as the mean ± SD of the area under the curve (percent/minute). No GO-related effects on platelet aggregation were observed. TRAP-6 results only include n = 2 as one of the donor results had a very low response and was inconsistent with the other samples tested. ADP indicates adenosine diphosphate; AUC, area under the curve; GO, gemtuzumab ozogamicin; PRP, platelet-rich plasma; TRAP, thrombin receptor activator peptide.
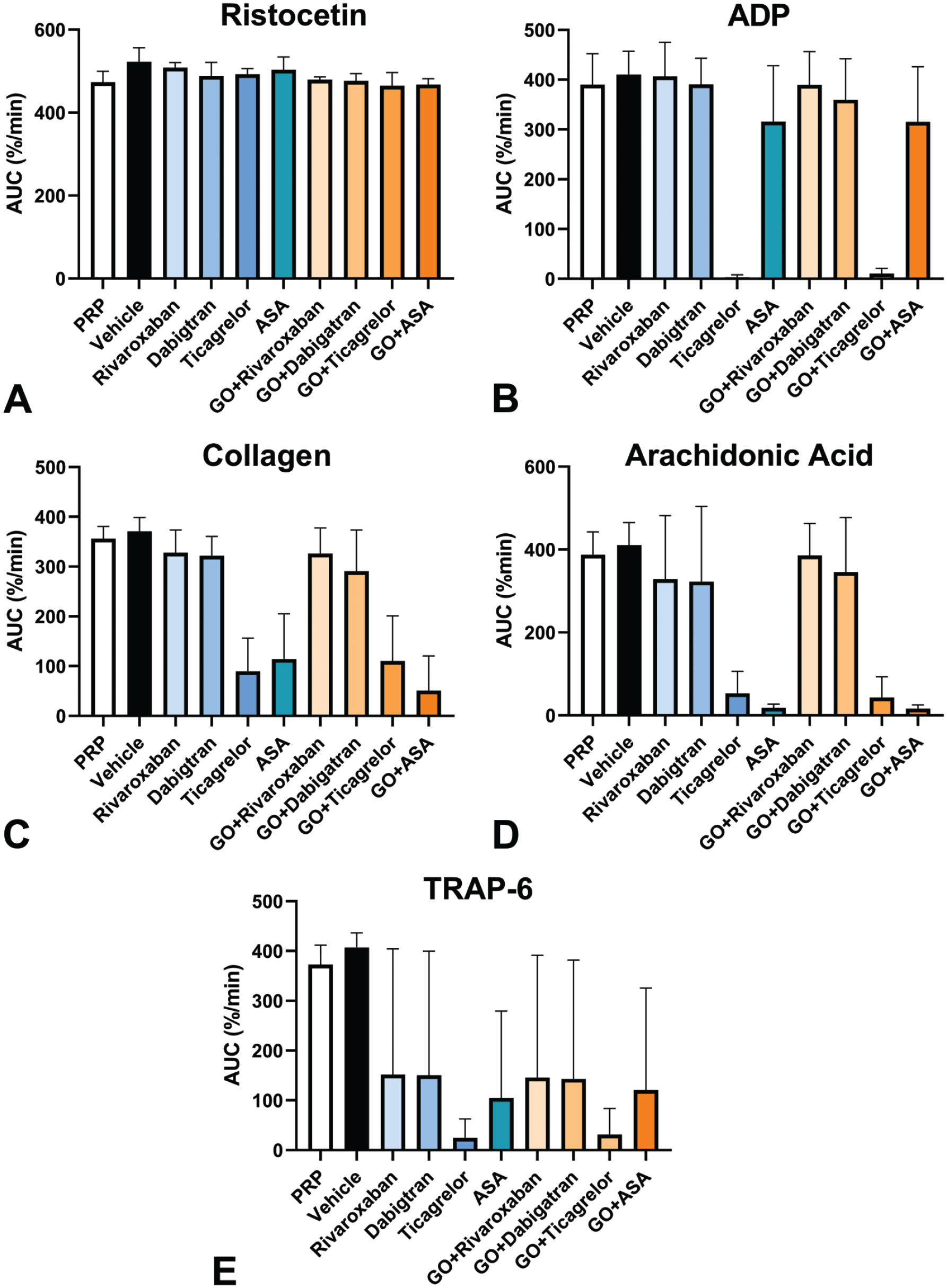
Mean effects on platelet aggregation in platelet-rich plasma samples from healthy human donors treated with anticoagulants with and without 0.5 µg/mL GO. Platelet-rich plasma derived from 2 male and 1 female human donors were incubated with or without anticoagulants (Rivaroxaban [300 ng/mL], Dabigatran [300 ng/mL], Ticagrelor [0.5 µM] or Acetylsalicylic Acid (ASA) [133 µM]) and stimulated, all set up in duplicate, with agonists (A) ristocetin (1.25 mg/mL), (B) ADP (10 µM), (C) collagen (2 µg/mL), (D) arachidonic acid (1 mM), or (E) TRAP-6 (10 µM). Optical aggregometry was performed and the results are expressed as the mean ± SD of the area under the curve (percent/minute). No GO-related exacerbation of the expected individual effects of the anticoagulants were seen. ADP indicates adenosine diphosphate; AUC, area under the curve; GO, gemtuzumab ozogamicin; PRP, platelet-rich plasma; TRAP, thrombin receptor activator peptide.
When platelets from healthy human donors were perfused with plasma from uremic patient donors, no GO-related effects were observed on platelet aggregation with four PRUP samples co-treated with GO and platelet agonists (Figure 4). Lower platelet aggregation was observed with one human uremic donor’s platelet-rich uremic plasma (PRUP) sample when co-treated with GO and platelet agonists ADP or TRAP-6. However, there were no GO-related effects on platelet aggregation in the PRUP samples from the same uremic donor co-treated with GO and the remaining agonists (collagen, AA, or ristocetin). More detailed inquiry into the medical background of the plasma donor with the affected PRUP revealed that this patient was treated with the beta blocker carvedilol, a compound that has been specifically associated with decreases in platelet function.24-26
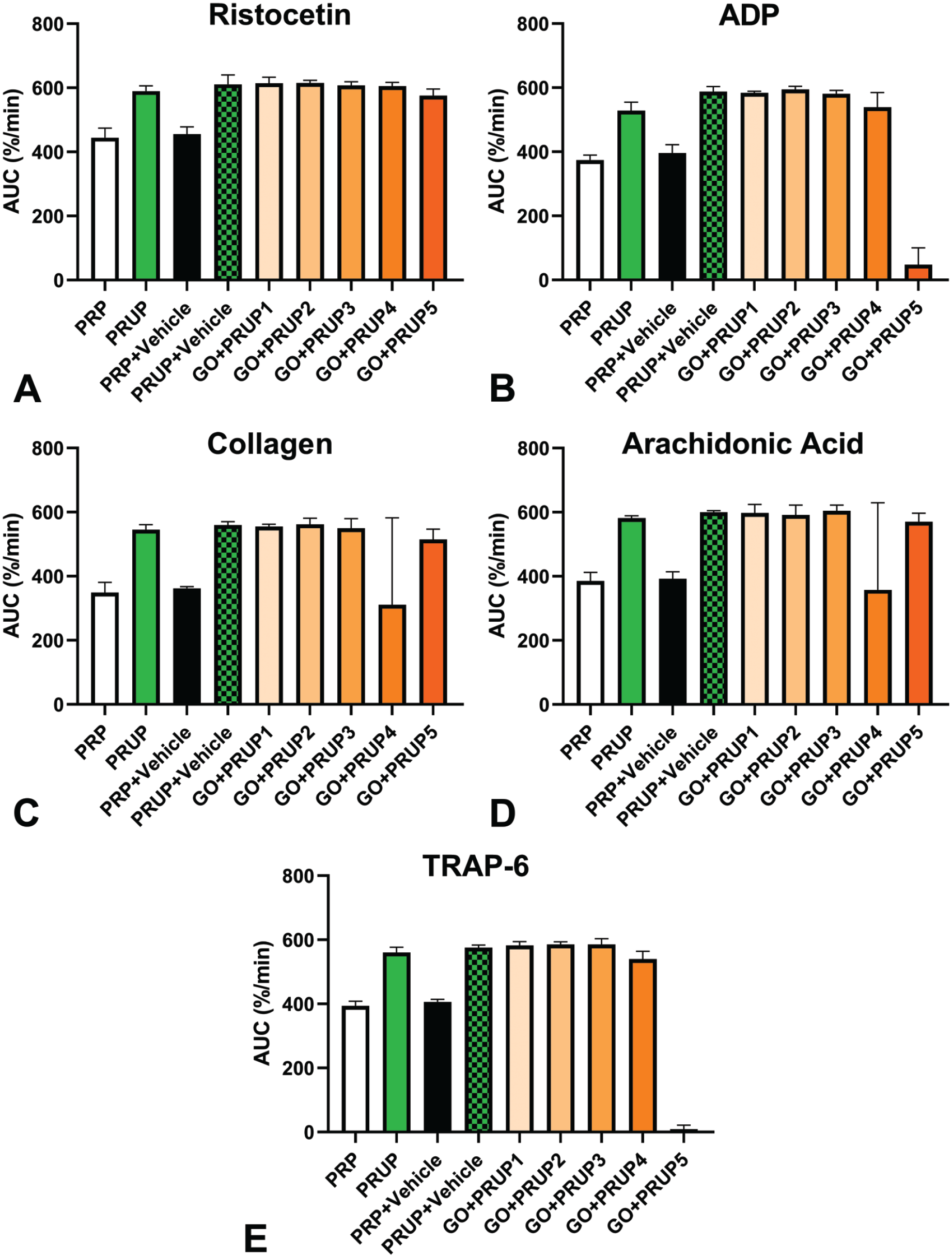
Mean effects of 0.5 µg/mL GO on platelet aggregation in platelet-rich plasma samples perfused with plasma from uremic patient donors. Platelet-rich plasma samples derived from 2 female and 1 male human donors were perfused with platelet-poor plasma from 5 different renal-impaired donors and then treated with GO and stimulated with agonists, all set up in duplicate, (A) ristocetin (1.25 mg/mL), (B) ADP (10 µM), (C) collagen (2 µg/mL), (D) arachidonic acid (1 mM) or (E) TRAP-6 (10 µM). No GO-related effects were observed in four of the five human uremic samples. For the individual with decreases in platelet aggregation after stimulation with ADP or TRAP-6, those decreases were attributed to a concurrent medication. AUC indicates area under the curve; GO, gemtuzumab ozogamicin; PRP, platelet-rich plasma; PRUP, platelet-rich uremic plasma; TRAP, thrombin receptor activator peptide.
Discussion
As a modality, ADCs intend to increase the therapeutic index of potent cytotoxic drugs by selectively delivering them to a specified tumor cell population. This approach often relies upon the specificity and/or relative overexpression of the target antigen, followed by efficient internalization of the molecule, and then cleavage of the linker to release the cytotoxic agent.27,28 Although any component of the molecule can contribute to ADC-related toxicity, it is most commonly thought to be mediated by the cytotoxic agent (also known as the “payload.”) Nonclinical testing of ADCs warrants careful consideration of target molecule expression to aid in selection of a cross-reactive species; however, target-dependent toxicities may be driven by factors other than the presence and pattern of target expression, and target-independent toxicities are also a major source of safety liability in the clinic.29-31
In the case of GO, nonclinical in vivo studies were only considered relevant for detection of target-independent toxicities due to lack of cross-species reactivity with the target antigen. Therefore, an in vitro investigation on human cells was performed to determine whether GO influenced human megakaryopoiesis and platelet production. Our results showed an impact on early and mid-stage megakaryocyte development from pooled CD34+ bone marrow cells although it was unclear whether the subsequently observed effect on late-stage megakaryocyte and platelet development was directly related to GO treatment or the result of decreased megakaryocyte survival. Although as previously noted, many reports indicate either absent or minimal CD33 expression on megakaryocytic cells,11-14 occasional reports of higher expression on megakaryocytes and megakaryocytic precursor cells do exist. 32 It is also possible that a contributor to the impact on megakaryopoiesis is an off-target effect not identified in the nonclinical studies in the species and/or at the doses evaluated. Target-independent effects specifically affecting megakaryocytes have been described for other ADCs, including both trastuzumab emtansine and AGS-16C3F.33-35 Either an on-target or off-target effect could be exacerbated by diminished thrombopoietin levels, which have been reported to accompany SOS, a known side effect of treatment with calicheamicin ADCs.31,36 Finally, it has also been demonstrated that in some AML patients, MPL (the thrombopoietin scavenging receptor) expression on neoplastic blast cells is notable and a key predictor of thrombocytopenia; in such patients, lower than normal levels of thrombopoietin (and therefore lower platelet numbers) would be expected to exist prior to starting treatment with GO.
We also evaluated the effects of GO on platelets from normal donors alone or in the presence of known platelet aggregate agonists (ADP, collagen, AA, ristocetin, or TRAP-6). In addition, regulators requested an evaluation of the effects of GO in patients with severe renal dysfunction as this population frequently exhibits altered hemostasis.37-39 However, performing controlled in vitro investigations on platelet function using samples from specific patient populations was logistically challenging. Light transmittance aggregometry, which is widely used for assessment of platelet function, must be performed on PRP samples prepared immediately after blood sampling, and most standardization documents recommend that that blood samples be exposed to minimal agitation and vibration to minimize artifactual platelet activation and that testing take place within 4 hours.17,40 To address the first part of the request, platelets from normal donors were either treated with anticoagulants that have been demonstrated to affect platelet function in vitro 19,20 or were perfused with plasma from renal-impaired donors in the presence of the previously described platelet aggregation agonists as characteristics of uremic plasma have been demonstrated to play a role in diminished platelet function, based on functional testing of normal platelets exposed to uremic plasma.21-23 Samples were then treated with GO in the presence of the same aggregation agonists. No GO-related effects were observed on platelet aggregation in four of five human uremic donor PRUP samples treated with GO. However, an apparent lesser degree of platelet aggregation was observed in one renal-impaired donor’s uremic plasma sample using platelets from all three normal platelet donors when co-treated with GO and aggregation agonists ADP or TRAP-6. To address whether this effect was related to GO treatment, more detailed inquiry into the medical background of the plasma donor was pursued. It was subsequently discovered that this renal-impaired donor was being treated with the beta blocker carvedilol. Beta blockers, and particularly nonselective lipophilic beta blockers, such as carvedilol, are known to significantly reduce platelet aggregation, and carvedilol’s anti-aggregatory effects on platelets have specifically been reported when ADP and thrombin were used as platelet aggregation agonists.24-26 Although no GO-related effects on aggregation were observed on normal human platelets either alone, when exposed to common anticoagulants, or when perfused with uremic plasma, it remains possible that patients with functionally impaired platelets could exhibit an enhanced tendency toward hemorrhage, especially if they are thrombocytopenic. However, platelet function is known to be altered in a subset of AML patients as a result of diminished activation capacity inherent to the disease, and platelet functional assessment of AML patients has recently been shown to be a better predictor of propensity toward bleeding than platelet counts, even when platelet numbers are extremely low.41,42
In conclusion, these studies revealed that treatment with GO resulted in cellular cytotoxicity and/or decreased differentiation during human megakaryocyte development, but that GO did not impair platelet aggregation under the experimental conditions evaluated. Ultimately, the effect of GO on megakaryocyte development observed in our studies was determined to have no impact on the risk-benefit assessment in the intended patient population as thrombocytopenia is a known side effect of GO and monitoring of platelet counts in patients is already strongly recommended.
