Abstract
Thrombocytopenia affects patients undergoing liver transplantation. Intraoperative platelet transfusion has been shown to independently influence survival after liver transplantation at 1 and 5 years. We examined the impact of thrombocytopenia and intraoperative platelet transfusion on short-term graft and overall survival after orthotopic liver transplantation (OLT). A total of 399 patients undergoing first OLT were studied. Graft and overall survival in patients with different degrees of thrombocytopenia and with or without intraoperative platelet transfusion were described. The degree of thrombocytopenia prior to OLT did not affect graft or overall survival after transplant. However, graft survival in patients receiving platelets was significantly reduced at 1 year (P = .023) but not at 90 days (P = .093). Overall survival was significantly reduced at both 90 days (P = .040) and 1 year (P = .037) in patients receiving platelets. We conclude that a consistently lower graft and overall survival were observed in patients receiving intraoperative platelet transfusion.
Introduction
Thrombocytopenia, defined by a platelet count of less than 150 × 109/L, has been reported to occur in upto 76% of patients with cirrhosis. 1,2 Mild thrombocytopenia (platelet count > 75 × 109/L to <150 × 109/L) is of little clinical significance, while moderate thrombocytopenia (50 × 109/L to 75 × 109/L) observed in approximately 13% of patients with cirrhosis may affect treatment decisions. 3 Severe thrombocytopenia (<50 × 109/L) significantly increases the risk of bleeding during invasive procedure and can be associated with significant morbidity and mortality. 4,5 Despite a low platelet count, stable patients with cirrhosis seldom have defects in primary hemostasis that predispose them to bleeding. 5
In patients with cirrhosis, thrombocytopenia occurs because of reduced synthesis of thrombopoietin by the cirrhotic liver and hypersplenism as a consequence of portal hypertension. Thrombopoietin, an essential cytokine produced in the liver, kidney, and skeletal muscles, has been shown to regulate the amount, growth, and size of megakaryocyte ploid. 6 Reduced thrombopoietin messenger RNA expression and decreased reticulated platelet count were found in patients with advanced liver cirrhosis. 7,8 This is further supported by data after liver transplantation where serum thrombopoietin levels were shown to increase significantly. 9,10 Similarly, hypersplenism, characterized by splenomegaly and a significant reduction in 1 or more cellular elements of the blood in the presence of normocellular or hypercellular bone marrow, 11 is common in patients with cirrhosis.
After liver transplantation, the platelet count falls further in almost all patients and reaches a nadir at days 4 to 5. 12,13 The putative mechanisms include increased platelet consumption due to thrombin generation, 13 sequestration of platelets in the reperfused liver graft, 14,15 or a combination of these processes. Curiously, the percentage of fall in platelet count was found to be an independent predictor of survival after liver transplantation, and it was thought that this may be caused by poor graft function. 12 Thrombopoietin concentrations increased significantly after liver transplantation and peaked at days 4 to 6. 9,10,16,17 This is followed by increases in reticulated platelets by day 4 and normalization of peripheral platelet counts within 2 weeks after orthotopic liver transplantation (OLT). 9,10 A previous study has demonstrated that intraoperative platelet transfusion, among other factors, is an independent risk factor for 1- and 5-year survival after OLT. 18 Subsequent report noted that platelet transfusion led to increased 1-year mortality from acute lung injury rather than a direct impact on graft survival per se. 19
In this study, we examined the impact of thrombocytopenia, platelet transfusion, and other factors thought to influence graft and overall survival at 90 days and 1 year after liver transplantation. We postulated that the effect of platelets transfusion on outcome must be transient if not immediate, and its effect on survival may not extend beyond a year after liver transplantation.
Methods
Data Collection
We analyzed 399 patients who had undergone first OLT from 2000 to 2009 retrospectively. During this period, 492 liver transplant operations were performed in our center, but we excluded 62 patients who had retransplantation and 5 with multiple organ transplantation. Another 26 patients were excluded because of missing data. Data were retrieved from the United Kingdom and Ireland Transplant Registry and our center’s hematology laboratory database. Missing data accounted for 5.3% of the data set for intraoperative platelet transfusion.
Graft survival was defined as time from OLT to death or retransplantation, while overall survival was time from transplantation to death. We classified the indications for liver transplantation into the following groups: autoimmune or biliary cirrhosis, alcohol-related cirrhosis, viral cirrhosis, metabolic disease, acute liver failure, and miscellaneous. The OLT for hemochromatosis, α 1 antitrypsin deficiency, Wilson disease, and nonalcoholic steatohepatitis-related cirrhosis were included under metabolic disease. In the autoimmune or biliary cirrhosis category, we included autoimmune hepatitis, primary biliary cirrhosis, primary sclerosing cholangitis (PSC), and cholangiocarcinoma related to PSC (Mayo protocol). The OLT performed for hepatocellular carcinoma was classified under their respective causes of liver cirrhosis. The Model of End-stage Liver Disease (MELD) scoring system is a reliable measure of estimating risk of death in patients with end-stage cirrhosis and is used as a disease severity index to determine organ allocation priorities. 20
Statistiscal Analysis
Statistical analysis was performed using SPSS 17 and GraphPad Prism Version 5.0b. Categorical variables were presented as numbers with percentages while continuous variables as median and interquartile range, as the continuous data were not normally distributed. Categorical variables were compared using the Pearson chi-square test. Comparison of continuous variables was performed using the Mann-Whitney U test or Kruskall Wallis test, where appropriate. We used the Kaplan-Meier method to describe graft and overall patient survival. Statistical significance between the different groups was tested with the Mantel-Cox log rank test. The average length of follow-up was 55 months. Univariate analysis was performed on each variable to determine its effect on graft and overall survival. All variables with a P value of <.10 in the univariate analysis were included in a multivariate survival analysis, using the Cox proportional hazards model. Hazard ratios and 95% confidence intervals are presented. A P value of <.05 was considered statistically significant.
Results
At least three-quarters of patients had thrombocytopenia prior to liver transplantation (301 of 399), with a median platelet count of 95 (61-149) × 109/L. The main causes of cirrhosis leading to transplant were autoimmune/biliary related, alcohol, and viral hepatitis. Approximately two-thirds of patients received platelet or plasma transfusions during OLT. In our cohort, 164 (41.1%) patients had mild thrombocytopenia with a median platelet count of 102 (86-123) × 109/L, 70 (17.5%) patients had moderate thrombocytopenia, 63 (54-68) ×109/L, and 67 (16.8%) patients had severe thrombocytopenia, 42 (33-46) ×109/L. The major indication for OLT in patients with severe thrombocytopenia was viral cirrhosis, while alcohol-related liver cirrhosis caused predominantly mild to moderate thrombocytopenia. Patients with progressively more severe thrombocytopenia had higher MELD score (P < .01) and required more platelet (P < .001) and plasma transfusion (P < .001) during OLT (Table 1).
Baseline Characteristics of Patients With and Without Thrombocytopenia.a,b
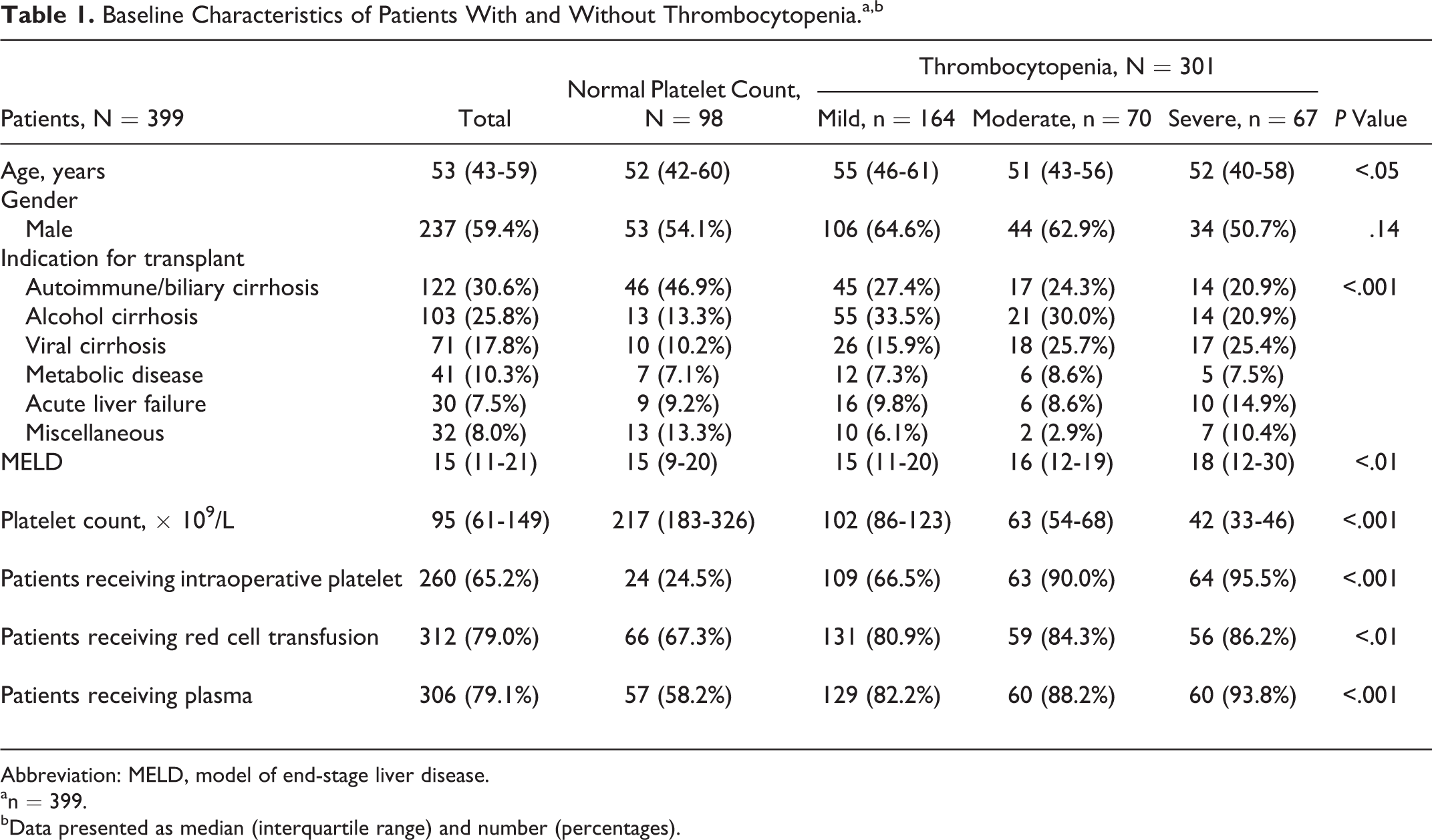
Abbreviation: MELD, model of end-stage liver disease.
an = 399.
bData presented as median (interquartile range) and number (percentages).
When we compared patients with normal platelet count versus the 3 varying groups of thrombocytopenia (mild, moderate, and severe), we did not find any differences in either graft or overall survival at 90 days and 1 year after liver transplantation (Figure 1). However, graft survival was reduced significantly in patients receiving intraoperative platelets at 1-year follow-up (P = .023) but not at 90 days (P = .093; Figure 2). When we examined whether hepatic artery thrombosis was the predominant cause for graft failure, there were no significant differences in the incidence of hepatic artery thrombosis in patients with or without intraoperative platelet transfusion at 90 days (P = .82) and 1 year (P = .28). Overall survival was significantly reduced at both 90 days (P = .040) and 1 year (P = .037) in patients receiving platelet transfusion during OLT (Figure 2). Patients receiving intraoperative platelet transfusion were at increased risk of death due to septicemia (90-day mortality, 2.88% vs 0.39%, P = .033; 1-year mortality, 3.60% vs 0.39%, P = .012; Table 2).
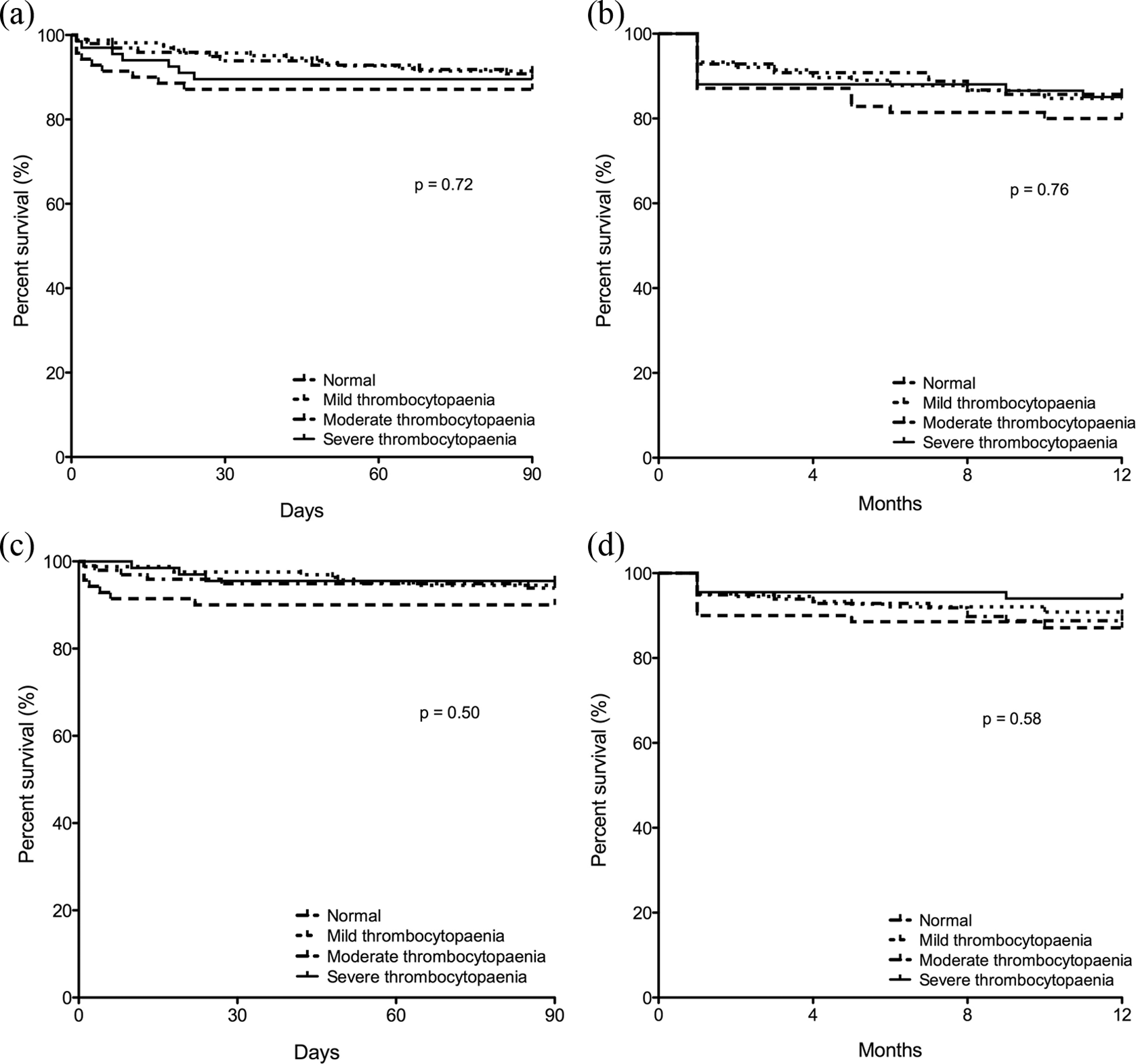
Kaplan-Meier plot of graft (a and b) and overall (c and d) survival in patients with different degrees of thrombocytopenia at 90 days and 1 year after transplantation.
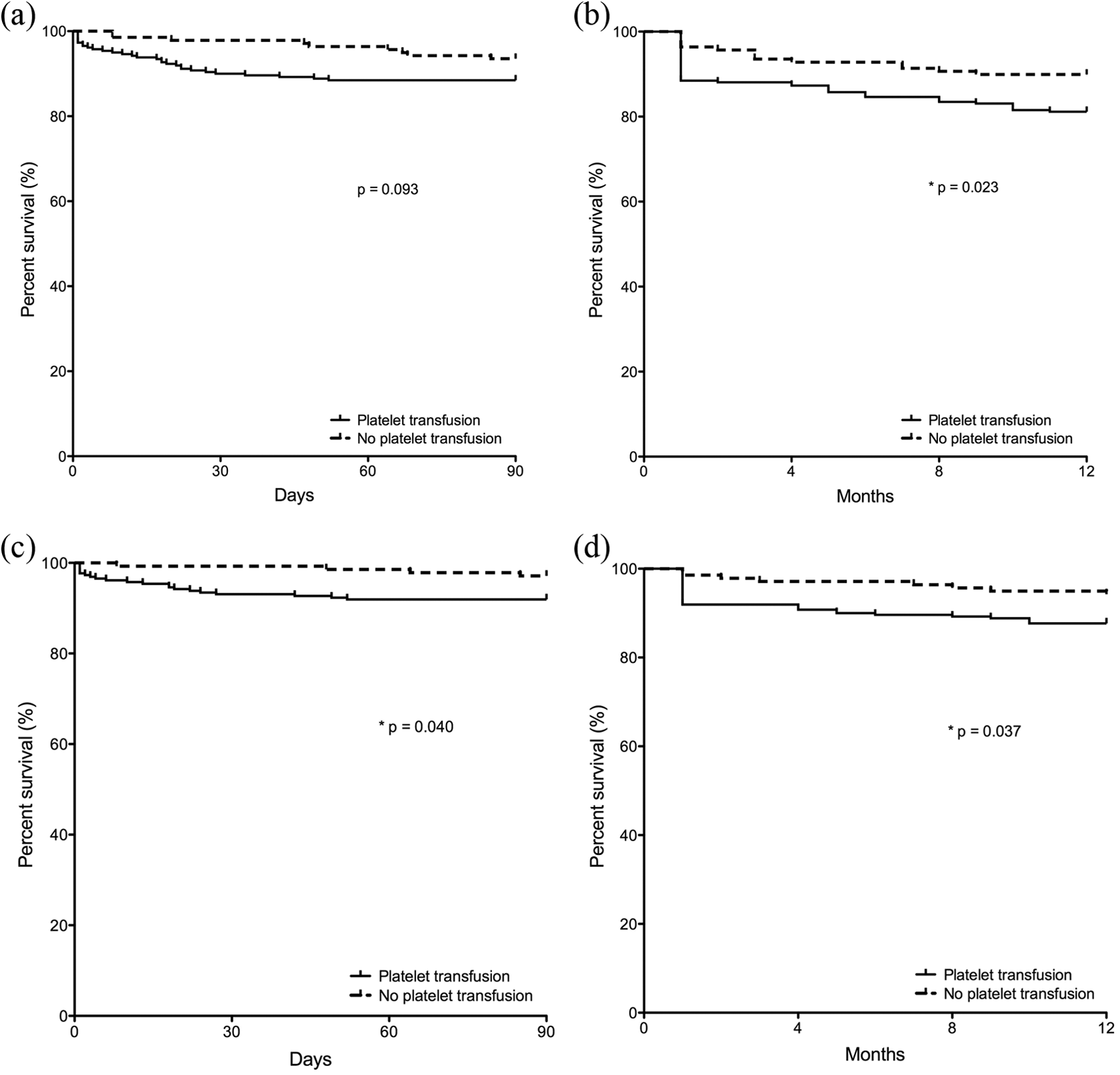
Kaplan-Meier plot of graft (a and b) and overall (c and d) survival in patients with and without intraoperative platelet transfusion at 90 days and 1 year after transplantation.
Causes of Death at 90 Days and 1 Year After Liver Transplantation in Patients Receiving Intraoperative Platelet Transfusion Compared to Those Who Did Not.
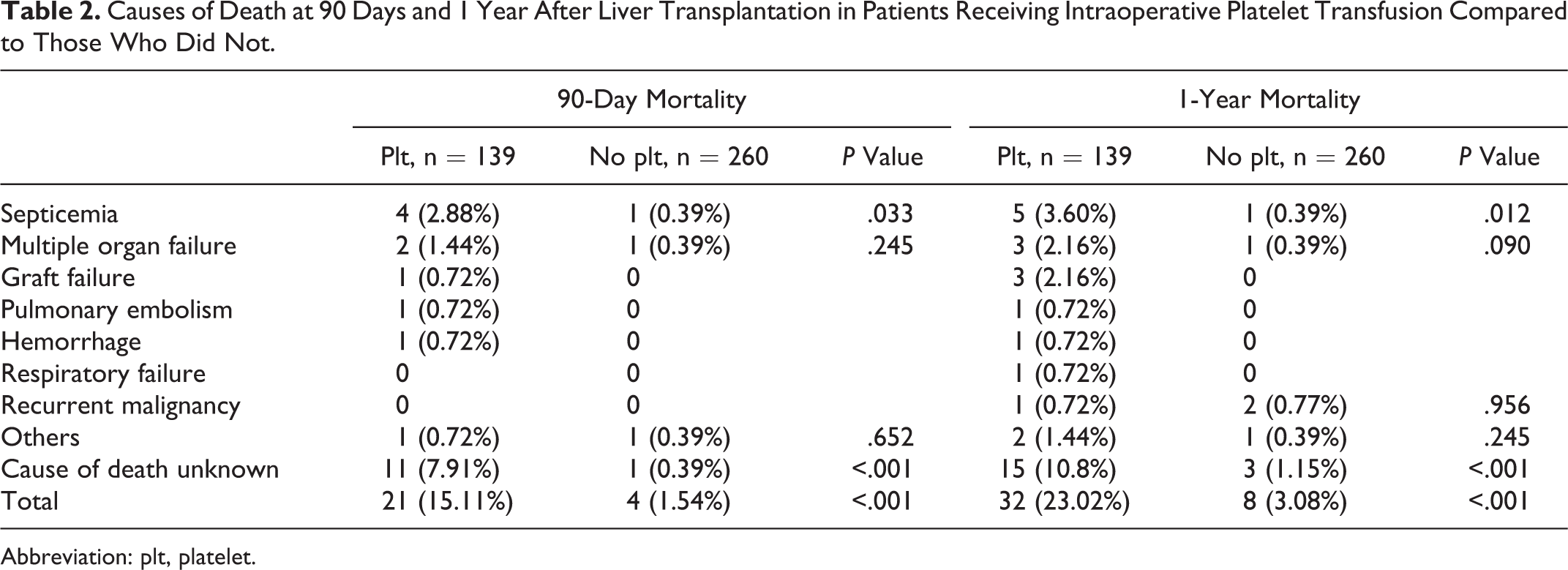
Abbreviation: plt, platelet.
Univariate analyses identified age, MELD score, and indication for OLT as significant risk factors for graft survival in 90 days and intraoperative platelet transfusion and transplant indication for 1-year graft survival (Table 3). When all of these variables were entered into a multivariate Cox regression model, only the indication for OLT was found to be an independent predictor of 90-day and 1-year graft survival. The hazard ratios for different transplant indications are shown in Table 4. Liver transplantation for acute liver failure was significantly associated with graft failure, with a hazard ratio of 16.91 (1.61, 177.16) at 90 days and 5.53 (1.25, 24.39) at 1 year.
Univariate Analyses of Various Factors on Graft Survival at 90 Days and 1 Year.
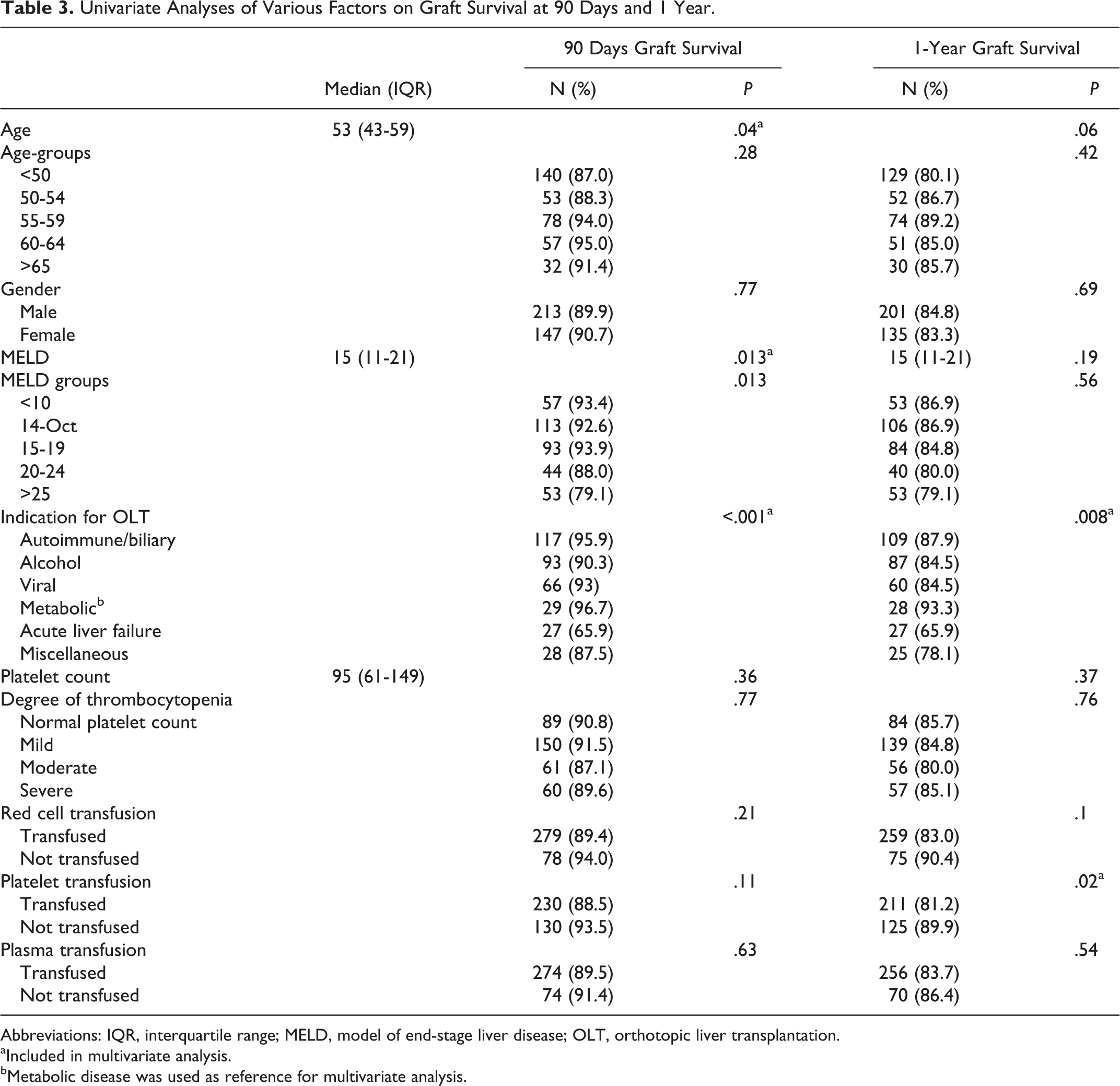
Abbreviations: IQR, interquartile range; MELD, model of end-stage liver disease; OLT, orthotopic liver transplantation.
aIncluded in multivariate analysis.
bMetabolic disease was used as reference for multivariate analysis.
Multivariate Cox Regression Analysis of Graft Survival.
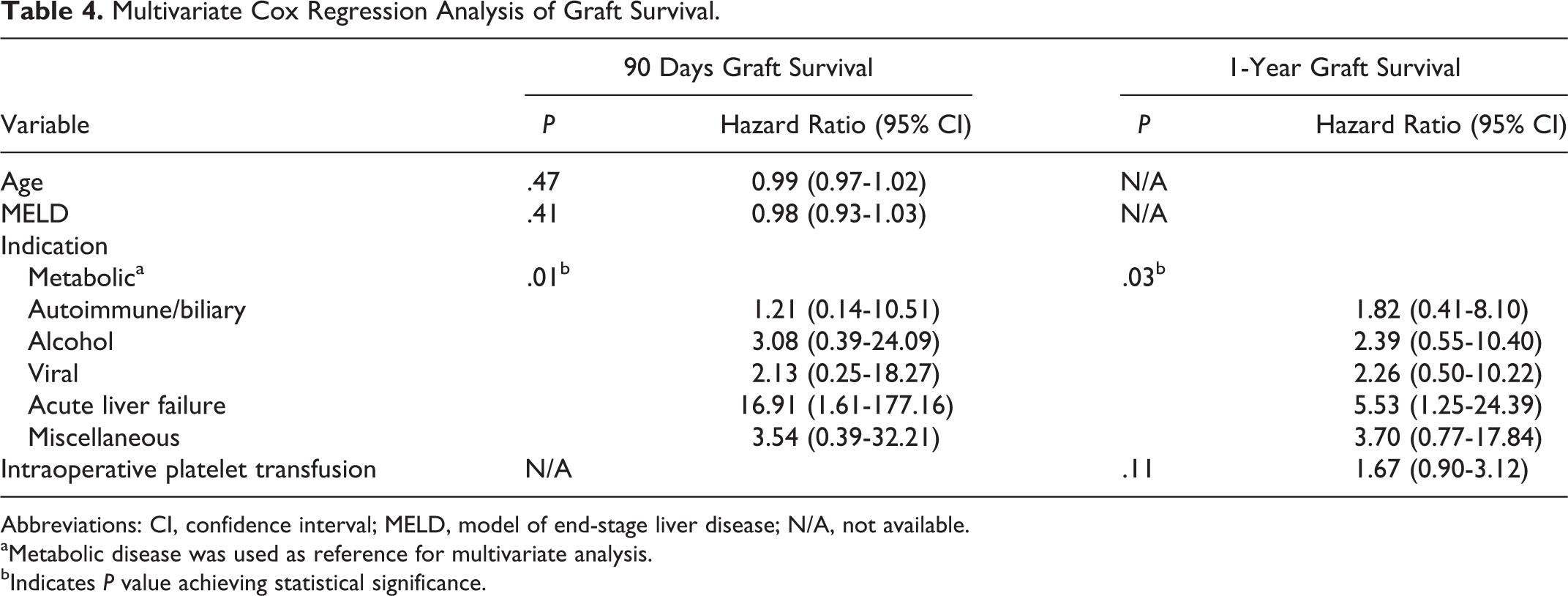
Abbreviations: CI, confidence interval; MELD, model of end-stage liver disease; N/A, not available.
aMetabolic disease was used as reference for multivariate analysis.
bIndicates P value achieving statistical significance.
Similarly, univariate analyses for overall patient survival indicated that the MELD score, indication for OLT, and intraoperative platelet transfusion were significant for 1-year survival, while these factors and intraoperative red cell transfusion were significant for 90-day survival (Table 5). However, multivariate analysis showed that none of these factors were independent predictors of outcome (Table 6).
Univariate Analyses of Various Factors on Overall Patient Survival at 90 Days and 1 Year.
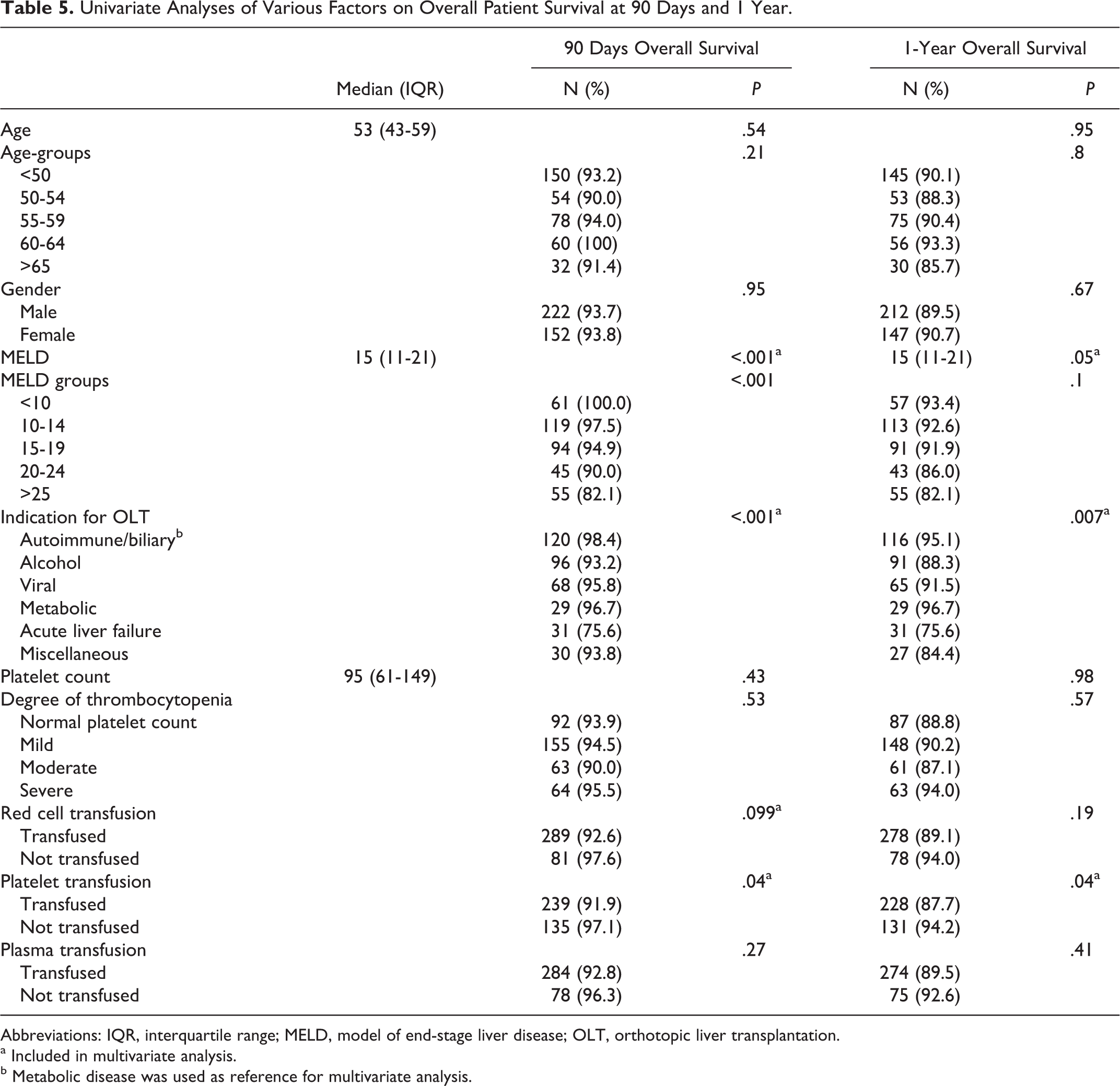
Abbreviations: IQR, interquartile range; MELD, model of end-stage liver disease; OLT, orthotopic liver transplantation.
aIncluded in multivariate analysis.
bMetabolic disease was used as reference for multivariate analysis.
Multivariate Cox Regression Analysis of Overall Patient Survival.
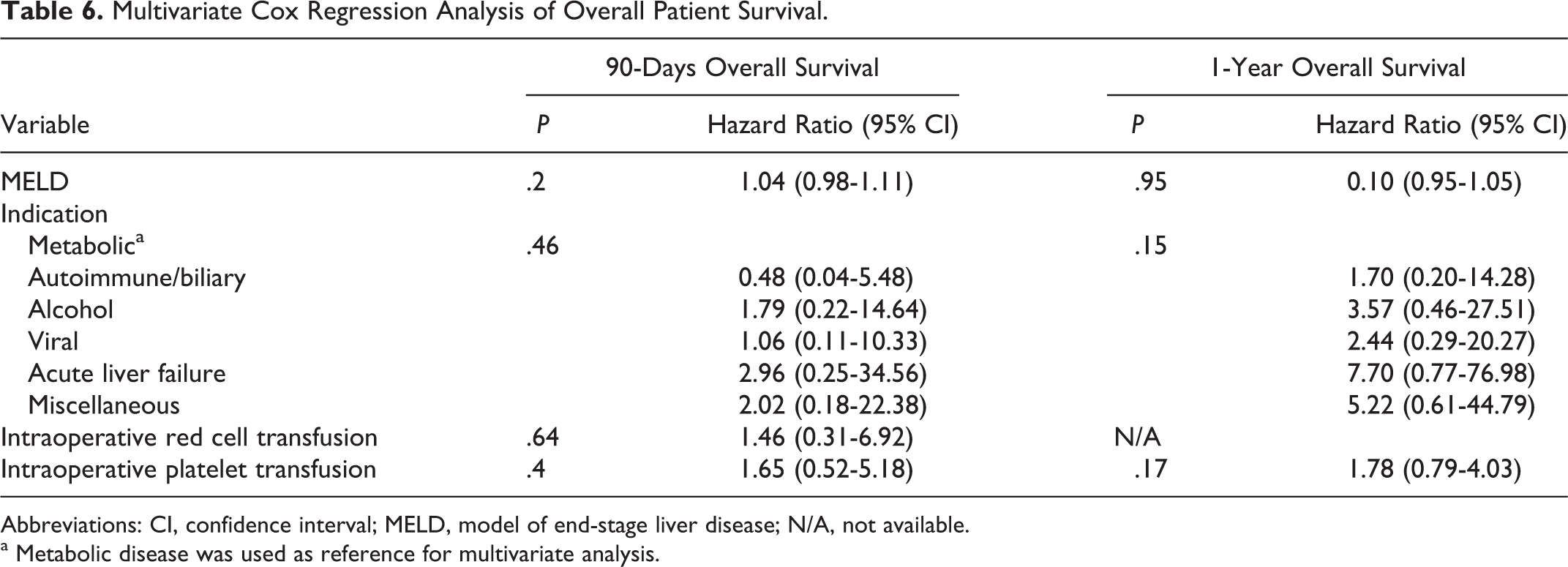
Abbreviations: CI, confidence interval; MELD, model of end-stage liver disease; N/A, not available.
aMetabolic disease was used as reference for multivariate analysis.
Discussion
Thrombocytopenia as a result of reduced synthesis of thrombopoietin and hypersplenism is a common problem in patients with cirrhosis. The prevalence of thrombocytopenia in end-stage liver cirrhosis prior to transplantation is similar to previous reports. 1 The slight differences in the proportion of mild (41.1%), moderate (17.5%), and severe (16.8%) thrombocytopenia, compared to previous reported studies, reflect the liver transplant cohort of patients included in this study rather than the actual population with cirrhosis. Similarly, the indications for liver transplantation in Ireland are not identical to other centers. As described earlier, the most common indication for OLT in our center during the study period is autoimmune and biliary-related cirrhosis (30.6%), when compared to centers in North America where the leading indications for liver transplant is hepatitis C virus-related cirrhosis. 21
Although patients with increasing severity of thrombocytopenia were deemed sicker with higher MELD score (P < .01) and required more blood products (P < .001) during OLT, there were no significant differences in graft and overall survival at 90 days and 1 year for patients with normal platelet counts or varying degrees of thrombocytopenia before transplantation. When we examined the effect of intraoperative platelet transfusion on outcome after OLT, we found that overall patient survival was significantly lower in patients receiving platelet transfusions at both 90 days and 1 year. In contrast, graft survival was reduced significantly in patients receiving intraoperative platelet transfusion at 1-year follow-up but not at 90 days (P = .093). These findings would suggest that intraoperative platelet transfusion, rather than thrombocytopenia per se, is associated with a poor outcome after liver transplantation. In our multivariate analysis, only the indication for liver transplantation was an independent predictor for graft loss at 90 days and 1 year. Although patients receiving intraoperative platelet transfusion have a significantly reduced survival at both 90 days and 1 year, we did not find platelet transfusion to be an independent risk factor for overall survival in our multivariate analysis.
Many donor- and host-related as well as peri- and postoperative factors may contribute to survival after liver transplantation. A negative impact of intraoperative platelet transfusion on survival after liver transplantation was initially described in 2003. 22 This study showed that intraoperative transfusion of 20 or more units of platelets was associated with poor outcome after liver transplantation. 22 A more recent exhaustive study on exploring the risk factors for survival after liver transplant reported that intraoperative transfusion of platelets and red blood cells is associated with poor outcome at 1 year and 5 years. 18 Interestingly, this result was not replicated in other liver transplant centers. 23 Moreover, available literature on the impact of platelet transfusions on surgical outcomes remained inconsistent. 24,25 In patients with acute gastrointestinal bleeding, a restrictive transfusion strategy (blood transfusion only when hemoglobin falls below 7 g/dL) significantly improved outcome when compared to a liberal strategy (transfused when hemoglobin is below 9 g/dL), particularly in patients with Child A or B cirrhosis. 26
Patients with compensated liver cirrhosis have complex hemostatic changes that coexist in equilibrium. 27 –29 These complex hemostatic changes may be thrown into disarray during liver transplantation. Patients with cirrhosis have defects in primary hemostasis, abnormal clotting due to impaired production of pro- and anticoagulant factors, low-grade coagulation activation, and hyperfibrinolysis. 5 Alterations in both platelet count and function may occur during liver transplantation. A reduction in platelet count could be due to hemodilution, immunologic reactions or sequestration of platelets in the liver graft after reperfusion. 15 Studies have shown that platelet count decreases by 30% to 50% after graft reperfusion because of platelet entrapment in the liver graft. 30 –32 Similarly, platelet function is thought to decline throughout the course of liver transplantation. 30 –32 The 2 proposed mechanisms for this include the activation of platelets after graft reperfusion 15,31 and the development of a hyperfibrinolytic state due to the release of tissue-type plasminogen activator. 33,34
In liver transplantation, platelets are not only essential for primary hemostasis after surgery but have been implicated in the pathogenesis of inflammation, ischemia–reperfusion injury, angiogenesis, tissue repair and regeneration, and even allograft rejection. 15,32,35,36 Hence, platelets are now thought of as both “friend and foe” of liver transplantation. 37 Platelets initiate clot formation at sites of damaged endothelium allowing injured vessels to cease bleeding. Moreover, platelets are now thought to mediate tissue repair in ischemia–reperfusion injury through the release of serotonin. 36,38 Serotonin stimulates hepatocyte mitosis in vitro, while in vivo, platelet-derived serotonin had been shown to enhance liver regeneration. 36,39
Conversely, platelet transfusions have been associated with poor outcome after liver transplantation. 18 Although platelets are involved in the pathogenesis of postoperative thrombosis, large retrospective studies and our data have not shown that intraoperative transfusion was associated with a significant increase in incidence of hepatic artery thrombosis. 40,41 Platelets have been shown to contribute to ischemia–reperfusion injury initially after liver transplantation. 35 Platelets interact with activated endothelium in the liver graft, induced by cold ischemia during organ retrieval. This interaction is mediated by adhesion molecules highly expressed on activated platelets and endothelium, such as selectins and integrins. 37 Immediately after graft reperfusion during liver transplantation, platelets induce apoptosis of sinusoidal endothelial cells. 32,42 Platelets have also been shown to mediate the immune response by augmenting the recruitment of CD4 T cells to hepatic sinusoids 43 and may contribute to allograft rejection through glutamate receptor signaling. 44
Platelets for transfusion are prepared from either whole blood (buffy coat-based or platelet-rich plasma) or via apheresis. Platelet concentrates prepared by apheresis only requires 1 donor, while 4 to 8 donors may be needed for platelets derived from whole blood. In Ireland, approximately 80% of platelets are derived from apheresis versus 20% from 4 whole blood donations through the buffy coat process at present. The proportion of pooled to apheresis platelets was higher in the year 2000 compared to 2009. (J. Fitzgerald, e-mail and discussion, October 12, 2012). Apheresis platelets have been linked with a lower rate of infection complications, tranfusion reactions, and alloimmunization. 45 Universal leukodepletion of blood components was implemented in Ireland since 1999 to reduce the risk of variant Creutzfeldt-Jakob disease. In this study, we were not able to examine the impact of different platelet preparations on outcome. We suspect that the majority of patients who received platelet transfusions probably received a combination of apheresis and pooled platelets, making the data difficult to interpret.
In summary, we conclude that thrombocytopenia is a major problem in patients with cirrhosis undergoing liver transplantation, with up to a third of these patients classified as having moderate to severe thrombocytopenia. Interestingly, the degree of thrombocytopenia prior to liver transplantation does not affect graft or overall survival after transplant. We observed a consistently lower graft and overall survival after OLT in patients with platelet transfusions; whether this is directly related to the effect of intraoperative platelet transfusion is not clear.
Footnotes
Acknowledgments
We wish to recognize the dedicated contribution of our liver transplant team.
Authors’ Note
Jun Liong Chin designed the study, collected and analyzed the data, and prepared the manuscript; Syafiah Hanis Hisamuddin was involved in study design, collected and analyzed the data, and reviewed the manuscript; Aoife O’Sullivan was involved in study design, collected the data, and reviewed the manuscript; Grace Chan was involved in study design and assisted with statistical analysis and manuscript preparation; and P. Aiden McCormick provided the concept, reviewed the data, and the manuscript. This article was previously presented at the Oral presentation in the National Autumn Meeting of the Irish Society of Gastroenterology, November 2011, Dublin.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
