Abstract
Sodium nitroprusside (SNP) is an antihypertensive drug with proven toxic effects attributed mainly to the production of nitric oxide (NO). Polyunsaturated fatty acids (PUFAs) are widely regarded as functional foods and have been shown to ameliorate the harmful effects of many toxicants. This study examined whether feeding of fish oil (FO)/flaxseed oil (FXO) would have any protective effect against SNP-induced hepatotoxicity and cell death. Male Wistar rats were fed either on normal diet or with 15% FO/FXO for 15 days, following which SNP (1.5 mg/kg body weight) was administered intraperitoneally for 7 days. Animals were killed after treatment, and livers were collected for further analysis. We observed that SNP significantly elevated tissue nitrite levels and lipid peroxidation (LPO) with concomitant perturbation in antioxidant defense systems accompanied with dysregulated glucose metabolism and pronounced cellular death. FO/FXO supplementation to SNP-treated rats caused reversal of tissue injury/cell death and markedly decreased LPO and improved antioxidant defense systems. FO/FXO appear to protect against SNP-induced hepatotoxicity by improving energy metabolism and antioxidant defense mechanism.
Introduction
“We are what we eat.” This adage represents the force behind research efforts in diet manipulation as preventive measures and treatments of various diseased states leading to the concept of “functional foods.” The relation between diet and disease is the subject of great controversy and the last 50 years have been characterized by the understanding of the impact of nutrition and dietary patterns on health.
1
Burr and Burr firstly suggested that unsaturated fatty acids may play a bioeffector role in the growth and development of young humans and animals.
2
Thus, the generic term “essential fatty acid” was used for them. Ingestion of polyunsaturated fatty acids (PUFAs) leads to their distribution to virtually every cell in the body with effects on membrane composition and function, cellular signaling, metabolism, and regulation of gene expression.
3
Dietary supplementation with ω-3 fatty acids (fish oil) has been shown to augment the antioxidant defense system and enhance the resistance to free radical attacks.
4
Their role in regulating the metabolic functions of liver,
5
amelioration of cyclosporine and gentamicin-induced nephrotoxicity, immunoglobulin A nephropathy,
6
and industrial pollutants
7
have also been elucidated. We have previously shown that PUFA protects against sodium nitroprusside (SNP),
Although data are very limited, it has been shown that long-term use of nitrates as donors of nitric oxide (NO) has the potential to induce many pathophysiological conditions. Like all nitrates, SNP works by relaxing blood vessels by quickly breaking down into NO, which is a potent vasodilator. The conversion of SNP to NO produces cyanide as a by-product, which is handled by the liver. SNP has been documented to induce genotoxicity as assessed by measurement of micronucleated lymphocytes.
11
In previous laboratory studies, SNP has been shown to cause DNA strand breaks.
12
SNP generates reactive oxygen species (ROS) during the redox cycling of nitroprusside,
13
and it is converted to a number of products, such as nitrite, NO, iron, cyanide, and oxygen-free radicals including superoxide radical and hydroxyl radical.
13
Peroxynitrite is a potent oxidizing agent formed from the reaction of NO and superoxide radicals, and it plays a critical role in the induction of inflammatory reaction and apoptosis, besides being associated with tumor promotion and/or progression.
14,15
The fact that NO is capable of triggering apoptosis is consistent with its ability to induce DNA damage, the inhibition of DNA synthesis, and cell cycle arrest.
16,17
Recent studies showed the influence of SNP on the processes in mitochondria, particularly on the respiratory chain, on cell function, and even cell death. Nanomolar concentrations of NO immediately, specifically, and reversibly inhibit cytochrome oxidase in competition with oxygen, while higher concentrations of NO and its derivatives (peroxynitrite, nitrogen dioxide, or
Considering the potential clinical benefits of FO and FXO, the present study was undertaken to examine the effect of prior administration of FO and FXO on SNP-induced hepatotoxicity, carbohydrate metabolism, cellular death, and antioxidant parameters. We hypothesize that on account of their intrinsic antioxidant effects, FO and FXO would ameliorate SNP-induced hepatotoxicity. The results obtained indicate that dietary supplementation with FO and FXO markedly protected against SNP-induced hepatotoxicity parameters.
Materials and methods
Chemicals
For the purpose of this study, the following chemicals were used: fish oil (FO; Menhaden, Sigma Chemical Co., St Louis, Missouri, USA), flaxseed oil (FXO; Omega Nutrition, Vancouver, Canada), SNP (Sigma Chemical Co.), Griess reagent (Sigma Chemical Co.). William’s medium E, antibiotic–antimycotic solution, and fetal bovine serum were obtained from Life Technologies (Paisley, UK). All other chemicals used were of analytical grade and were purchased either from Sigma Chemical Co. or Sisco Research Laboratory (Mumbai, Maharashtra, India).
Diet
A nutritionally adequate laboratory pellet diet was obtained from Aashirwad Industries, Chandigarh, India. Pellets were crushed finely and mixed with (a) 15% FO and (b) 15% FXO and stored in airtight containers. Vitamin E as
Experimental design
The animal experiments were conducted according to the guidelines of Committee for Purpose of Control and Supervision of Experiments on Animals (CPCSEA), Ministry of Environment and Forests, Government of India (S.O.2778(E), [10/11/2010]). Adult male Wistar rats (8 rats/group) weighing 150–180 g fed with standard rat chow (Aashirwad Industries) and water ad libitum were conditioned for 1 week before the start of the experiment. Rats were divided into four groups. They were fed on a normal diet (control and SNP) or diet-containing 15% FO (FOSNP) or 15% FXO (FXOSNP). After 15 days, SNP was administered intraperitoneally (1.5 mg/kg body weight/day) for 7 days to all groups except control. Control animals received an equivalent volume of normal saline. The rats were killed 24 h after the last injection under light ether anesthesia. Blood samples were collected, and liver was harvested and processed for the preparation of homogenates and hepatocyte culture as described below.
Preparation of primary hepatocytes and cell culture
Hepatocytes were isolated by in situ collagenase perfusion of liver from animals at the end of experiments as described previously. Livers were perfused first with an oxygenated solution I (10 mM 4-(2-hydroxyethyl)-1-piperazineethanesulfonic acid (HEPES), 6.7 mM potassium chloride (KCl), 145 mM sodium chloride (NaCl), and 2.4 mM ethylenediaminetetraacetic acid), pH 7.4 at 37°C, and then with solution II (100 mM HEPES, 6.7 mM KCl, 67 mM NaCl, 10 g/l albumin, 4.8 mM calcium chloride, and 0.05% collagenase A), pH 7.4 at 37°C. Thereafter, tissue was gently minced and cell suspension filtered through a nylon mesh. Hepatocytes were centrifuged and washed three times at 50 g for 5 min in William’s medium E, pH 7.4, supplemented with 1 μM insulin, 0.6 μM hydrocortisone, 15 mM HEPES, 100 U/ml penicillin, 100 μg/ml streptomycin, 0.25 μg/ml amphotericin, 2 mM glutamine, and 26 mM sodium bicarbonate. Hepatocytes (150,000 cells/cm2) were plated in a tissue culture plates coated with collagen type I (Iwaki, Gyouda, Japan) and cultured in supplemented William’s medium E, pH 7.4, containing 5% fetal bovine serum. After 2 h, culture medium was removed and replaced by fresh supplemented medium without fetal bovine serum, and the culture was maintained for 24 h without treatment. After stabilization of cell culture, hepatocytes were analyzed for annexin and propidium iodide (PI) binding or 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT) assay as described subsequently.
Serum chemistries
Blood samples were collected at the time of killing the animals, and serum was separated by centrifuging at 1008 relative centrifugal force (RCF) and stored at −20°C until further use. Serum glutamate oxaloacetate transaminase (SGOT), serum glutamate pyruvate transaminase (SGPT), and glucose levels were determined directly in serum samples using commercially available kits from Span diagnostics, Mumbai, Maharashtra, India.
Preparation of liver homogenate
The liver samples kept in ice-cold tris(hydroxymethyl)aminomethane (Tris)-buffered saline were homogenized in a glass Teflon homogenizer in 10 mM Tris-hydrochloric acid (HCl) buffer (pH 7.5) to get 10% (w/v) homogenate, followed by high-speed homogenization in an Ultra Turrex Kunkel homogenizer. Aliquots of liver homogenates were divided into three parts. The first part of the homogenate was centrifuged at 1792 RCF, for 10 min at 4°C, and the supernatant was saved for assaying the enzymes of carbohydrate metabolism and liver marker enzymes, second part was centrifuged at 2800 RCF for 15 min at 4°C and the supernatant was used for assay of free radical scavenging enzymes, and the third part was used for the estimation of total thiol (–SH) and lipid peroxidation (LPO).
Assay of liver marker enzymes
The activities of alkaline phosphatase (ALP) was determined using
Assay of carbohydrate metabolism enzymes
The activities of the enzymes involving oxidation of NADH or reduction of nicotinamide adenine dinucleotide phosphate were determined spectrophotometrically on Cintra 5 fixed for 340 nm using 3 ml of assay in a 1-cm cuvette at room temperature (28–30°C). The enzyme assays of malate dehydrogenase (MDH), malic enzyme (ME), glucose-6-phosphate dehydrogenase (G6PDH), glucose-6-phosphatase (G6Pase), and fructose-1,6-bisphosphatase (FBPase) activities were studied as described by Khundmiri et al. 28 Hexokinase was estimated by the method of Crane and Sols 29 and the remaining glucose was measured by method of Somogyi, as described by Nelson. 30
Assay of enzymatic and nonenzymatic antioxidants
Superoxide dismutase (SOD) was assayed by the method of Marklund and Marklund. 31 Catalase (CAT) and glutathione peroxidase (GSH-Px) activities were determined by the method of Giri et al. 32 and Flohe and Gunzler, 33 respectively. Total –SH was determined by the method of Sedlak and Lindsay 34 and LPO by the method of Ohkawa et al. 35 Protein concentrations in the homogenates were determined the method of Lowry et al., 36 as modified by Yusufi et al. 37
Determination of tissue nitrite
Tissue nitrite levels were quantified using the Griess assay. 38 Liver homogenates were centrifuged at 5000 RCF for 10 min. Five hundred microliter supernatant was removed, and an equal volume of modified Griess reagent was added, and the samples were incubated in the dark for 20 min. Standard curves were generated using 0–20 μM sodium nitrite. Absorbance was measured in standards and samples at 550 nm. Concentrations of nitrite were determined from the standard curve, which was linear over the entire concentration range tested. Values were expressed as nanomoles per gram tissue.
Flow cytometric analysis
Detection of apoptotic cells was carried out using annexin V/PI staining kit (Invitrogen, Waltham, Massachusetts, USA). Hepatocytes were harvested and washed with cold phosphate-buffered saline twice and resuspended in 200 μl binding buffer at a concentration of 1 × 106 cells/ml. The cells were incubated with 5 μl annexin V–fluorescein isothiocyanate and 1 μl PI in the dark for 15 min. In total, 300 μl binding buffer was then added in each tube prior to being analyzed with an LSRFortessa cell analyzer (BD Biosciences, San Jose, California, USA).
MTT assay
Hepatocytes were seeded at 1 × 104 cells/well in 96-well plates, and after 24 h, MTT (5 mg/ml) was added to each well, and the plate was incubated for 4 h at 37°C. The formazan product was dissolved by adding 100 μl dimethyl sulfoxide to each well. The MTT absorbance value was detected at 590 nm in a microplate reader.
Statistical analyses
Data are expressed as mean ± SEM as indicated for at least five different preparations. Statistical evaluation was conducted by one-way analysis of variance. A probability level of
Results and discussion
FO/FXO ameliorate SNP-induced dysregulation of carbohydrate metabolism
The liver plays a central role in whole-body glucose and lipid homeostasis. In the fasted state, it maintains blood glucose levels by producing glucose via glycogen breakdown/gluconeogenesis. In the postprandial state, the liver stops glucose production simultaneously under the influence of insulin increasing its uptake to replenish glycogen and triglyceride stores. In our study, SNP administration to animals resulted in increased glycolysis as measured by the activity of hexokinase (HK; Figure 1(a)) with a concomitant increase in the specific activity of LDH (Figure 1(b)). As LDH catalyzes the formation of lactate from pyruvate under conditions of hypoxia which is then excreted out of the cells, this points to a shift in the glucose metabolism toward anerobic mode. Under normal physiological conditions, pyruvate is shuttled into the mitochondria via conversion to acetyl coenzyme A, thereby leading to complete oxidation of glucose. We found that on SNP treatment, the activities of key tricarboxylic acid (TCA) cycle enzymes such as MDH and ME were significantly reduced (Figure 1(c) and (d)), thereby pointing again to a shift toward anerobic metabolism. Interestingly, when FO and FXO were given to rats along with SNP treatment, the observed dysregulation of glycolysis and TCA cycle were significantly improved. FO/FXO treatment significantly improved the activities of glycolytic enzymes HK and LDH (Figure 1(a) and (b)) and TCA cycle enzymes MDH and ME (Figure 1(c) and (d)).
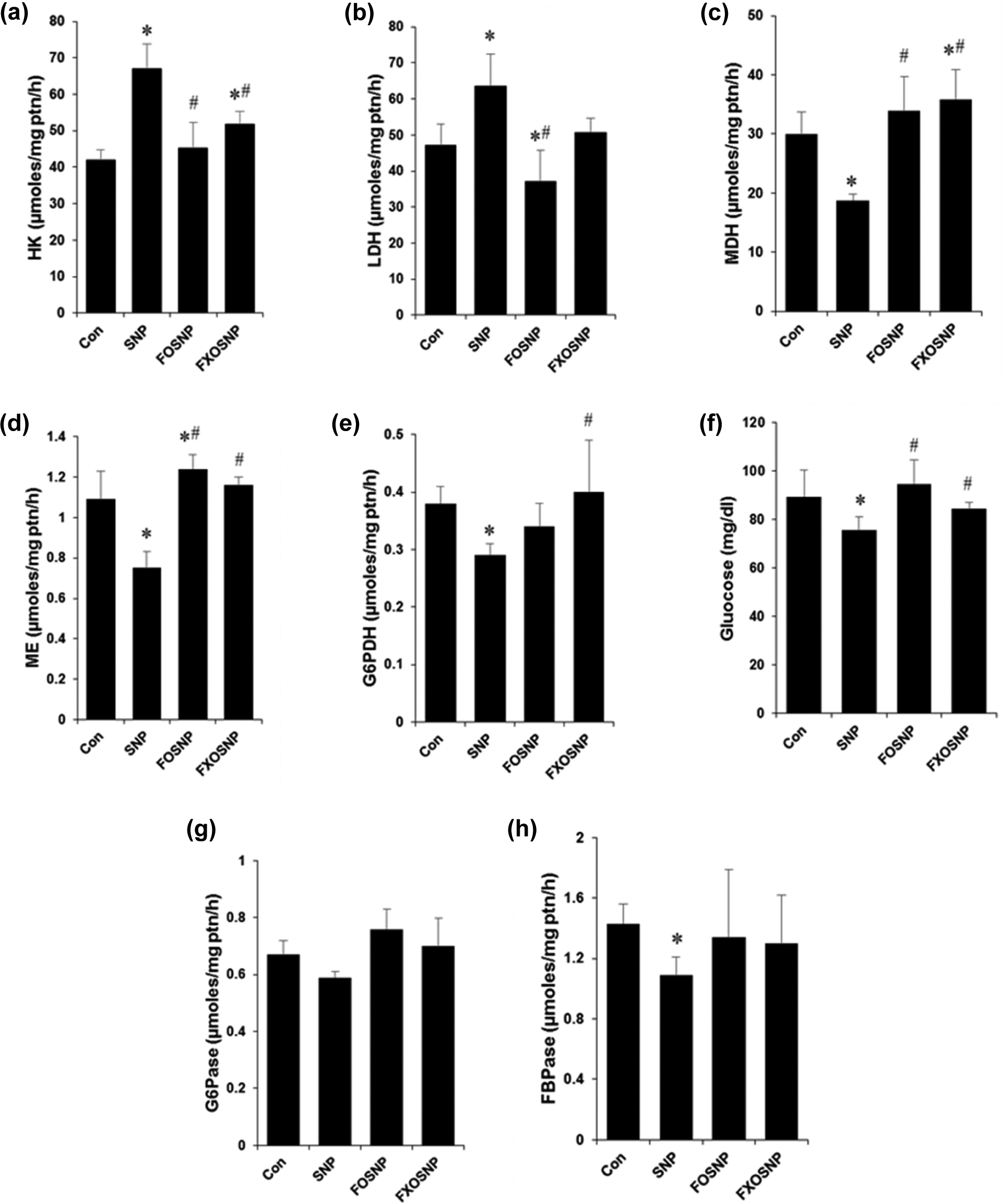
FO/FXO ameliorate SNP-induced dysregulation of carbohydrate metabolism. (a, b) Liver homogenates from treated animals were analyzed for specific activity of gluconeogenic enzymes HK and LDH; (c) glucose was estimated in serum by
G6PDH is the first and rate-limiting enzyme of the pentose phosphate pathway that shunts metabolites from the glycolytic pathway toward the production of nucleotides. It also produces reduced nicotinamide adenine dinucleotide phosphate (NADPH) and helps in maintaining the cellular levels of glutathione, which protects cells from oxidative damage. SNP effected in a significant reduction in the specific activity of G6PDH in liver homogenates of treated rats (Figure 1(e)). FO/FXO when given along with SNP were able to hinder the reduction in G6PDH activity (Figure 1(e)).
As discussed above, one of the major functions of the liver is to provide glucose to the body in times of glucose deprivation. The liver carries out this function by activating the process of gluconeogenesis, which involves converting metabolites from the TCA cycle and glycolysis to glucose and is then exported to the bloodstream. In our study, we observed that SNP-treated rats show a decrease in blood glucose levels (Figure 1(f)) along with reduced activities of key gluconeogenic enzymes G6Pase and FBPase (Figure 1(g) and (h)). Importantly, FO and FXO supplementation to SNP-treated rats resulted in improvement of gluconeogenic enzyme activities (Figure 1(g) and (h)) and normalization of blood glucose levels (Figure 1(f)).
Overall, our results indicate that SNP treatment to rats resulted in severe dysregulation of glucose homeostasis and decreased liver function. We observed a shift of metabolism to anerobic glycolysis concomitant with TCA cycle inhibition, an effect that is normally a hallmark of cancer cell as stated in the “Warburg effect”. Similar to cancer cells using vast amount of glucose to compensate for the inactive TCA cycle, we also observed a decrease in blood glucose levels on SNP treatment. This may result due to the ability of NO and other NO-derived free radicals to irreversibly damage mitochondria and inhibit the respiratory chain as described earlier. 18 The reduction in blood glucose could be due to increased glycolysis or decreased gluconeogenesis, the exact mechanism warrants further studies. These protective effects exhibited by FO and FXO can be attributed to the fact that both FO and FXO alone were able to effectively enhance the activities of enzymes, 39,40 resulting in overall improvement of carbohydrate metabolism in renal tissues similar to those reported in rat liver. 3 ω-3 PUFA have been shown to coordinately regulate the expression of several enzymes involved in carbohydrate and lipid metabolism. 41
FO/FXO improve SNP-mediated perturbation of antioxidant defense mechanisms
SNP is an NO donor compound, which is not only an important bioregulatory agent but also an endogenous cytotoxin, mutagen, and/or carcinogen. Excessive production/accumulation of NO always has the ability to result in the formation of other reactive intermediates of nitrogen. Reactive nitrogen species (RNS) produce cellular injury and necrosis via several mechanisms including peroxidation of membrane lipids, proteins, and DNA (Dawson and Dawson, 1996). A major cellular defense against free radicals is provided by SOD and CAT, which together convert superoxide radicals first to hydrogen peroxide and then to molecular oxygen and water. Other enzymes, for example, GSH-Px use —SH-reducing power of GSH to reduce oxidized lipids and protein targets of RNS. Our results show that SNP administration to control rats caused severe damage to the liver most likely by RNS/ROS generation as apparent by increased tissue accumulation of nitrite (Figure 2(a)) and altered activities of the above-mentioned antioxidant enzymes and total –SH content that resulted in a profound increase in LPO (Figure 2(b) to (f)). As consistent with previous studies, CAT and GSH-Px were inhibited by SNP-induced NO production 42,43 (Figure 2(e) and (f)). The decrease in SOD activity may be due to unavailability of its substrate that forms peroxynitrite after reaction with NO (Figure 2(c)). The ω-3 fatty acids possess antioxidant-enhancing activity, thereby raising the efficiency of antioxidant defense system 39,40 and therefore the feeding of FO/FXO diet to SNP-treated rats prevented SNP-induced augmentation of LPO and suppression of antioxidant enzyme activities. FO/FXO diet improved the activities of antioxidant enzymes SOD, CAT, and GSH-Px with a concomitant increase in the tissue levels of reduced GSH. This beneficial effect of FO/FXO oils may be attributed to the antioxidant properties of PUFAs.
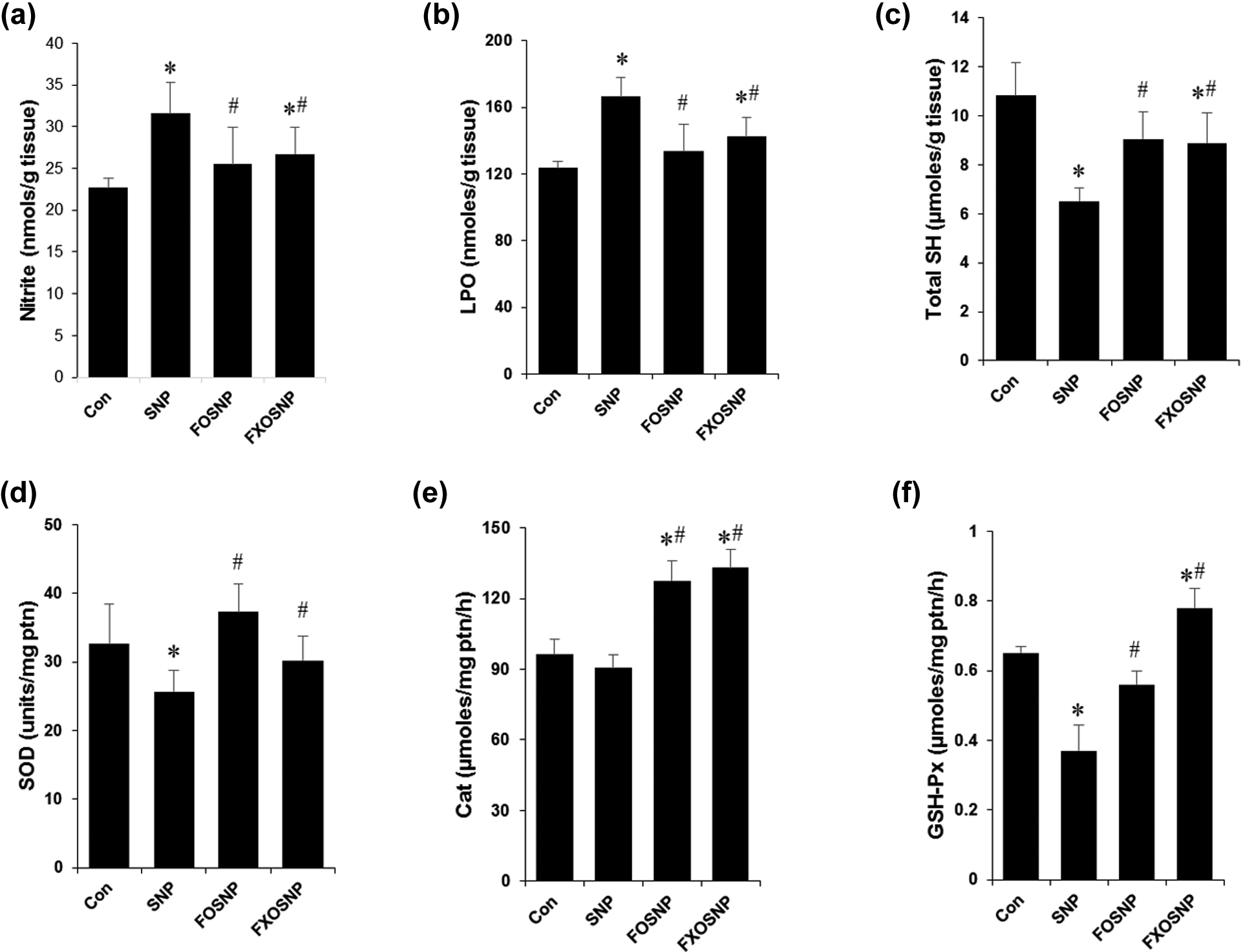
PUFAs prevent SNP-induced perturbation in the antioxidant defense mechanisms. (a) Nitrite levels were estimated in liver homogenates using the Griess reagent according to manufacturer’s protocol and the values were normalized by the amount of tissue in grams; (b) LPO was quantified in crude liver homogenate as described in Materials and methods and the values were normalized by the amount of tissue in gram; (c) Reduced GSH was estimated in rat liver homogenates and values were normalized by the amount of tissue in gram; (d) to (f) activities of antioxidant enzymes SOD, CAT, and GSH-Px was quantified as described in materials and methods and values were normalized by the amount of protein. Values are mean ± SEM of five to seven animals. *
FO and FXO protect against SNP-induced liver injury and cell death
Apart from maintaining whole-body glucose and lipid homeostasis, one equally important function of the liver is to detoxify the body of harmful chemicals and drugs. We have already shown that SNP is potentially toxic to the kidney structure and function, 9 therefore here we investigated the potential harmful effects of SNP on the liver. We found that after 7 days of SNP treatment, there was profound liver damage as evident from the significant increase in SGOT and SGPT in the serum and ALP in the liver homogenates (Figure 3(a) to (c)). The release of these enzymes usually indicates tissue injury and necrosis to the liver, which may be due to the generation of NO and other reactive intermediates. Interestingly FO and FXO supplementation was able to provide a protective effect to the rat liver as evident by the significant improvement in serum levels of SGOT and SGPT (Figure 3(a) and (b)).
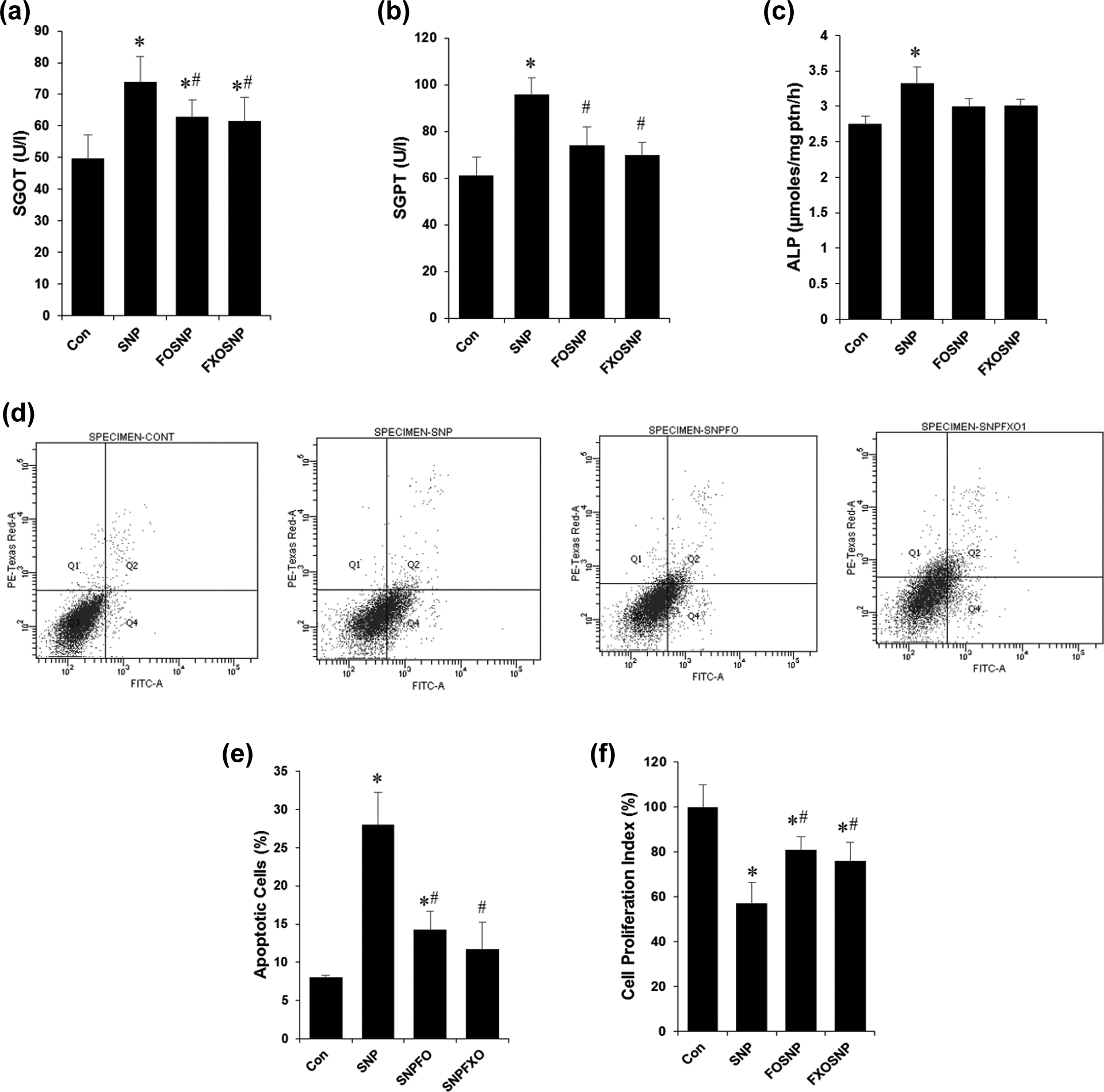
FO/FXO protect against SNP-induced liver injury and cell death. (a) and (b) Serum levels of liver marker enzymes SGOT and SGPT were estimated using commercially available kits; (c) specific activity of ALP was estimated in liver homogenates and values normalized by the amount of protein; (d) flow cytometric analysis of primary hepatocytes isolated from liver of treated animals; (e) graphical representation of data from (d); (f) cell viability of primary hepatocytes isolated from liver of treated animals was estimated using the MTT assay. Values are mean ± SEM of five to seven animals. *
The potential of SNP to induce apoptosis and cell cycle arrest directly from NO liberation has been well established in previous studies. In our study, we found that indeed SNP is able to promote cell death via apoptosis as indicated by the significant increase in annexin V-positive hepatocytes (Figure 3(d) and (e)). This was accompanied by a significant decrease in cellular proliferation as measured by the formation of formazan end product in the MTT assay (Figure 3(f)). The MTT assay is also a measurement of metabolically active and viable cells, as the dye is reduced in the mitochondria and this strengthens our previous results, which shows an altered metabolic state upon SNP treatment (Figure 1(a) to (h)). When FO/FXO supplementation was extended to SNP-treated rats, the observed hepatotoxic effects were significantly diminished on account of the intrinsic biochemical and antioxidant properties of FO/FXO (Figure 3(d) to (f)). We observed a significant decrease in the percent of apoptotic cells (Figure 3(d) and (e)) and a significant improvement in cell viability (Figure 3(f))
Conclusion
Several approaches utilizing different mechanisms have been attempted to reduce chemical- and drug-induced nephrotoxicity and other adverse effects. In the past few years, much interest has been focused on the role of naturally occurring dietary substances for the control and prevention of various chronic diseases that resulted in ω-3 fatty acid-rich food products from animal and plant sources being marketed as a health supplement. This puts so much pressure on the scientific community to unravel the potential beneficial effects and possible side effects of these supplements. As can be seen from the results, feeding of FO and FXO enriched in ω-3 fatty acids enhanced resistance to SNP-mediated hepatotoxicity and cell death and perturbation in antioxidant defense systems. Since SNP is an NO donor, most of these effects can be attributed to NO-induced RNOS formation. Our results thus support the rationale that ω-3 fatty acids-enriched FO and FXO may be an effective dietary supplementation in the management of SNP-induced nephrotoxicity and other pathologies in which antioxidant defense mechanism are decelerated and may provide a cushion for a prolonged therapeutic option against SNP-mediated hepatotoxicity and other adverse alterations without harmful side effects. Further, we can also say that there is not much significant difference between the protective effect of FO versus FXO, and therefore, vegetarians around the world who cannot consume FO due to dietary or religious beliefs can get the same benefits using FXO.
Footnotes
Acknowledgment
MWK and SP acknowledge the support provided by Indian Council of Medical Research (ICMR), India, via Junior Research Fellowship (JRF) and Senior Research Fellowship (SRF), and SAK acknowledges the support provided by Council of Scientific and Industrial Research (CSIR), India.
Conflict of interest
The authors declared no conflicts of interest.
Funding
This work has been supported in part by the INSPIRE Faculty Fellowship Grant (IFA-12 LSBM-19) to MWK by the Department of Science and Technology (DST), Government of India.
