Abstract
Introduction:
We used immunohistochemistry to investigate the histopathologic effects of hyperbaric oxygen (HBO) on the facial skin of rats.
Material and methods:
Rats in the HBO group (n = 6) were placed in a 20 L HBO chamber at 2.5 atmospheres absolute at 25–26°C with 100% oxygen for 90 min/day for 7 days. Following euthanasia, sections of facial skin were removed for examination.
Results:
Epidermal hyperplasia and degeneration, basal-cell hypertrophy, subepithelial fibrosis, and increased connective tissue were observed in the HBO group. E-cadherin expression was reduced in the epidermis, hair follicles, and sebaceous glands in HBO-treated rats relative to control animals. HBO treatment was associated with vimentin immunoreactivity in fibroblasts, endothelial cells, and the bulbus pilorum of a subset of hair follicles. It also resulted in increased type IV collagen expression within the connective tissue in the hair follicles and sebaceous glands.
Conclusion:
The HBO group demonstrated epidermal hyperplasia and degeneration, basal-cell hypertrophy, and subepithelial fibrosis. In addition, HBO decreased E-cadherin expression, which suggests that HBO may impair intracellular adhesion. Expression of vimentin and type IV collagen was also observed in the dermis. Increased connective tissue, hemorrhage, and mononuclear cell infiltration were observed in the dermis of HBO-treated animals. Thus, HBO has effects on the structures of the epidermis and dermis.
Introduction
Hyperbaric oxygen (HBO) therapy includes the inhalation of 100% oxygen under pressures greater than 1 atm (pressurized between 1.5 and 3.0 atmospheres absolute (AtA)) in a pressure chamber, resulting in significant antioxidant therapy. 1 HBO therapy increases resistance to ischemia through improved tissue oxygenation and has beneficial effects on wound-healing processes. 2 It has been widely used as primary or adjuvant therapy for a variety of conditions including gas embolism, carbon monoxide poisoning, intracranial abscess, necrotizing soft tissue infection, delayed radiation injury, compromised grafts and flaps, acute thermal burn injury, diabetic foot, livedoid vasculopathy, and chronic osteomyelitis. 3,4
E-cadherin is expressed throughout the epidermis, with the exception of the cornified layer, and has been identified as an important adhesion molecule mediating keratinocyte interactions. Epidermal keratinocytes express two types of cadherin known as E-cadherin and P-cadherin, which have different distributions in the epidermis, that is, E-cadherin is found throughout the epidermis, whereas P-cadherin is restricted to the basal-cell layer. 5,6
Vimentin filaments are broadly distributed in many tissues, including mesenchymal cells. 7 The extracellular matrix protein collagen is synthesized by fibroblasts, chondroblasts, osteoblasts, and other cells. The molecular configuration of collagen fibers gives them a tensile strength greater than that of steel. Consequently, collagen imparts a unique combination of flexibility and strength within tissues. Type IV collagen bundles are intertwined with type I collagen bundles, forming fibrils that are continuous with the basement membrane and blood vessel walls. 8 HBO is administered through inhalation and may have adverse effects on the skin surrounding the mouth and nose. It has been successfully applied to the treatment of ischemic reperfusion injury. In this study, we evaluated the histopathologic effects of HBO on rat skin.
Materials and methods
The study protocol was reviewed and approved by the Animal Research Committee of Dicle University, Turkey. Twelve adult Sprague-Dawley rats, each weighing 180–220 g, were used as experimental animals. The animals were group housed (six per cage) under standard conditions (21 ± 2°C) in the Animal Health and Research Center of Dicle University. The rats were randomly divided into HBO and control groups. The rats in the HBO group (n = 6) were placed in a 20 L HBO chamber (2.5 AtA) at 25–26°C with 100% oxygen for 90 min/day for a total of 7 days. The rats were not anesthetized and breathed spontaneously during HBO treatment. The rats in the control group (n = 6) were not given HBO therapy.
Preparation of tissue for light microscopy
At the end of the study, the animals were killed by decapitation approximately 2 h after the end of the final HBO treatment on the 7th day of HBO exposure. The skin surrounding the nasal cavity was removed, and the tissue was prepared for light microscopy. The skin samples were fixed in neutral buffered formalin solution, directly dehydrated in a graded series of ethanol solutions and embedded in paraffin wax. Five-micrometer sections were cut with a microtome (RM2265 rotary microtome; Leica, Germany) and mounted on coated slides. The sections were subjected to Masson’s trichrome staining for observation under a light microscope (Nikon Eclipse 80i, Japan).
Immunohistochemical staining
The tissues were fixed in formalin solution and embedded in paraffin wax. Paraffin blocks were cut into sections (4–6 μm thick) on positively charged glass slides. Sections were deparaffinized with xylene and dehydrated by immersion in graded alcohol solutions. Fixed slides were incubated in ethylenediaminetetraacetic acid (pH 8.0; Merck, Germany) for 3–5 min in a microwave oven (750 W) for antigen retrieval. The sections were subsequently incubated for 20 min in 3% hydrogen peroxide (H2O2)/methanol to block endogenous peroxidase activity, and then rinsed in phosphate-buffered saline (PBS) three times (5 min per wash). Next, the sections were incubated in a blocking solution (Goat serum; Invitrogen, Carlsbad, California, USA). The slides were incubated overnight with a primary antibody against E-cadherin (1/100, mouse monoclonal; Santa Cruz Biotechnology, Inc., Santa Cruz, California, USA), type IV collagen (1/500, rabbit monoclonal; Abcam, UK), or vimentin (1/200, mouse monoclonal; Santa Cruz). After washing in PBS, the sections were incubated with horseradish peroxidase-linked streptavidin (Invitrogen). The reaction was visualized by incubating the sections in a solution of 0.1% 3,3′-diaminobenzidine (DAB) and 0.02% H2O2 for 7 min (DAB substrate kit; Invitrogen). Finally, the sections were counterstained with hematoxylin (Sigma, St Louis, Missouri, USA) and covered. Positive immunostaining was defined as the presence of brown chromogen (DAB) at the edge of the hematoxylin-stained cell nucleus and in the cytoplasm or plasma membrane of the cells, as assessed by light microscopy.
Histopathologic scores
All morphological changes, including epidermal degeneration and mononuclear cell infiltration, were noted. Semiquantitative scoring of epidermal degeneration and mononuclear cell infiltration was performed. The intensity of these changes was graded from 0 to 3 (0 = no epidermal degeneration and mononuclear cell infiltration, 1 = faint staining, 2 = moderate staining, and 3 = intense staining).
Statistical analysis
Statistical analysis was performed using Statistical Package for the Social Sciences for Windows Version 15.0 (SPSS Inc., Chicago, Illinois, USA). Data are expressed as the mean ± standard deviation. The Mann–Whitney U test was used for binary comparisons. p Values below 0.05 were considered statistically significant.
Results
Histopathological analysis revealed significant differences in protein expression and distribution between the HBO-exposed and control animals. Compared to the control group, the HBO group showed significant epidermal degeneration and mononuclear cell infiltration (p < 0.01, Table 1). Changes in hair follicle diameter were not significant. Exposure to HBO led to histopathological changes in the skin (Figure 1). Masson’s trichrome staining showed no deterioration of the epidermis and dermis in the control group (Figure 1(a)). However, the HBO group exhibited epidermal hyperplasia and degeneration, basal-cell hypertrophy, subepithelial fibrosis, and increased connective tissue. Hemorrhage and mononuclear cell infiltration were also observed in the dermis in the HBO-treated animals (Figure 1(b)). While expression of E-cadherin was intense in the epidermis and dermis around hair follicles in the control group, it was reduced in the epidermis, hair follicles, sebaceous glands, and surrounding myoepithelial cells in HBO-treated animals (Figure 2). In the dermis, expression of vimentin was intense in fibroblasts, vascular endothelial cells, myoepithelial cells of sebaceous glands, and the bulbus pilorum of some hair follicles (Figure 3). Histopathological examination demonstrated that the connective tissue consisted of prominent collagen fiber bundles. Expression of type IV collagen was mainly observed in connective tissue, basement membranes of hair follicles, epithelia and the basal-cell layer, sebaceous glands, and blood vessels in HBO-treated animals (Figure 4).
Analysis of histopathological changes of skin in HBO group and control groups.
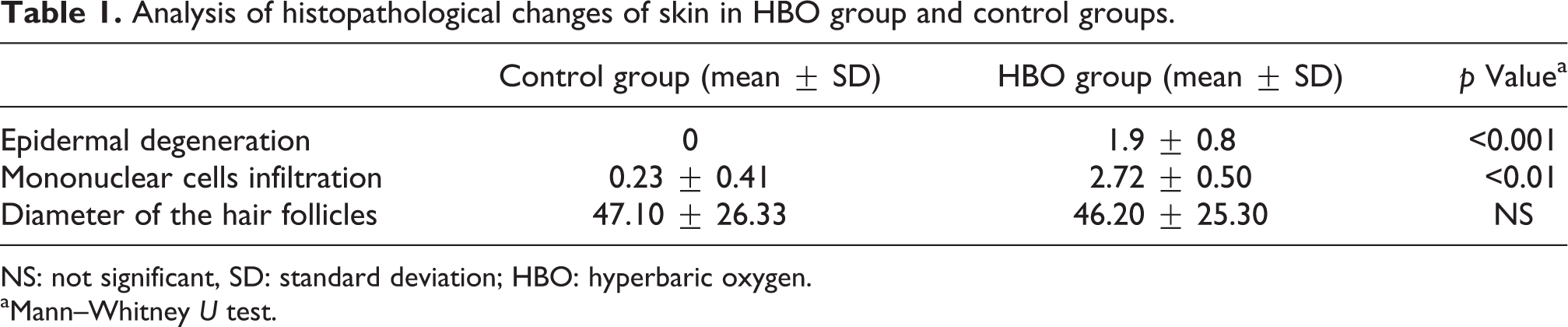
NS: not significant, SD: standard deviation; HBO: hyperbaric oxygen.
aMann–Whitney U test.

Control group. Normal appearance in epidermis and dermis, Masson’s trichrome, bar 50 µm. Degeneration in epidermis, increase in connective tissue, hemorrhaging (red arrow), and mononuclear cell infiltration (yellow arrow), Masson’s trichrome stain, bar 100 µm.
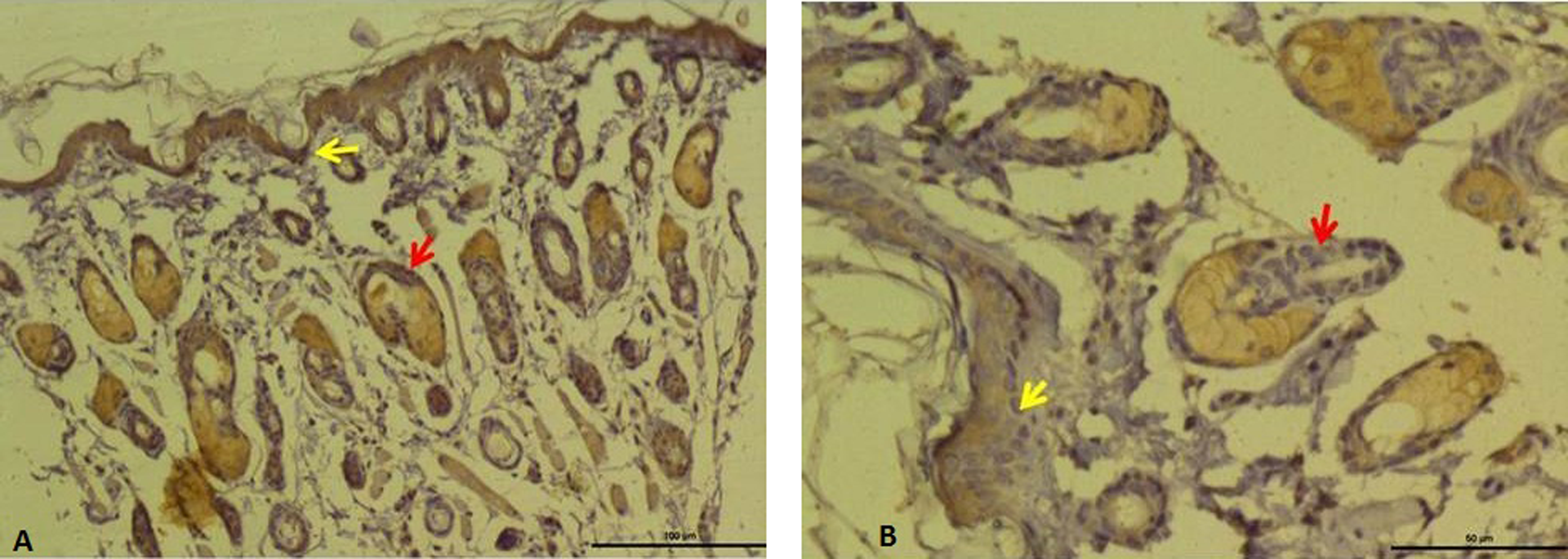
Control group. Intense expression of E-cadherin in epidermis (yellow arrow) and dermis around hair follicles (red arrows), E-cadherin immunoreactivity, bar 100 µm. HBO group, weak expression of E-cadherin in epidermis (yellow arrow) and hair follicle (red arrow), E-cadherin immunoreactivity, bar 50 µm. HBO: hyperbaric oxygen.

Control group. Distribution of vimentin expression in dermis, vimentin immunoreactivity, bar 50 µm. Intense expression of vimentin-positive fibroblast cells (yellow arrow) and endothelial cells of vessels (red arrow) and the myoepithelial cell of sebaceous gland and bulbus pilorum of some hair follicles (yellow arrows) in the dermis (green arrow), vimentin immunoreactivity, bar 50 µm.

Control group. Distribution of type IV collagen expression in dermis and hypodermis, type IV collagen immunoreactivity, bar 50 µm. Expression of type IV collagen was mainly observed in connective tissue, basement membranes of hair follicles (red arrow), epithelia and basal-cell layer sebaceous glands, and blood vessels (yellow arrows), type IV collagen immunoreactivity, bar 50 µm.
Discussion
HBO therapy is generally safe and well tolerated. Many of the known side effects of HBO are mild and easily reversible. Adverse effects such as reversible myopia, symptomatic otic barotrauma, pulmonary barotrauma, and pulmonary oxygen toxicity and seizures caused by central nervous system oxygen toxicity can occur in rare cases. 9 HBO stimulates angiogenesis in radiation-damaged tissue as a result of the diffusion of a steep oxygen gradient across the affected area. 10 Lemarie et al. 11 reported that HBO treatment may increase the accumulation of lipid peroxidation products in free skin grafts. Khandelwal et al. 12 administered HBO therapy immediately following a surgical procedure in a 43-year-old patient with a complete scalp degloving injury, and concluded that HBO therapy may facilitate tissue survival in this type of injury and may be a viable adjunct treatment option. Zamboni et al. 13 demonstrated that HBO treatments are associated with beneficial recovery of axial skin flaps when applied in both the acute recovery phase and during postoperative recovery; they reported that HBO therapy enhances collagen deposition, which may promote capillary growth and improve tissue oxygen perfusion.
In this study, degenerative basal-cell hyperplasia and hypertrophy were observed in the epidermis of HBO-treated animals. Diffuse hemorrhage and increased leukocyte infiltration were also present. E-cadherin is expressed by the airway epithelium, and previous reports have suggested that it mediates the response to allergens through interactions with regulatory T cells and dendritic cells that suppress the production of allergenic mediators. 14 Intracellular junctions are major contributors to the epithelial barrier, and are composed of E-cadherin-mediated intercellular attachments. Damage to the epithelium may result in the loss of E-cadherin expression in the cell membrane and intercellular contacts. 5,6,15 E-cadherin expression differed significantly between the HBO and control groups. The importance of tight junction proteins such as E-cadherin in skin barrier function is widely appreciated. Loss of E-cadherin expression may result in the loss of epidermal cells. Moreover, in rats treated with HBO, the loss of E-cadherin may result in a loss of epidermal structural integrity.
Heid et al. 16 reported vimentin immunoreactivity in the hair follicle bulbs of mammalian skin. Mesenchymal tissues, such as those found in the dermal sheath of hair follicles, express intermediate vimentin filaments. The integrity of the hair follicles is essential to the health of the skin. In this study, vimentin immunoreactivity was seen in dermal stromal cells and extended into hair follicles and sebaceous glands. Inflammation of dermal cells, fibroblasts, and sebaceous glands occurs in HBO-treated animals and is correlated with vimentin filament expression.
Type IV collagen is the primary collagen isoform in the skin basement membrane and the main component of the dermal–epidermal junction. Structural examination of collagen IV can differentiate subepidermal blistering diseases, including bullous pemphigoid, acquired epidermolysis bullosa, anti-epiligrin cicatricial pemphigoid, and bullous lupus erythematosus. Collagen IV is an important mediator of wound healing and embryogenesis. 8 In this study, increased collagen IV deposition was observed in hair follicles, sebaceous glands, the epidermis, and basement membranes in HBO-treated animals. Changes in collagen IV deposition likely influence the integrity of the basement membrane. Interpretation of this study is limited by the relatively small number of animals studied and the use of a single HBO dosage.
Conclusions
A small number of previous studies have examined the effects of HBO on the facial skin of rats. In this study, the HBO group demonstrated epidermal hyperplasia and degeneration, basal-cell hypertrophy, and subepithelial fibrosis. In addition, HBO decreased E-cadherin expression, which suggests that HBO may impair intracellular adhesion. Expression of vimentin and type IV collagen was observed in the dermis. Increases in connective tissue, hemorrhage, and mononuclear cell infiltration were observed in the dermis of the HBO-treated animals. These results suggest that HBO may cause some pathological changes in the structures of the epidermis and dermis. Further large-scale experimental trials may clarify the relationship between changes in the skin and HBO therapy.
Footnotes
Authors’ note
Conflict of interest
The authors declared no conflicts of interest.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
