Abstract
Introduction
This study investigates the skin-irritating and skin-resorptive effects of drilling fluid (DF) through acute and subacute experiments conducted on adult rabbits and sexually mature male and female rats.
Methods
Acute and subacute experiments were conducted on adult rabbits and sexually mature male and female rats. The acute experiment involved a single exposure to DF, while the subacute study involved repeated dermal exposure. Various physiological, biochemical, and histopathological assessments were performed to evaluate the effects.
Results
The results of the acute experiment demonstrated that DF exhibits a mild skin-irritating effect but causes significant irritation to the ocular mucous membranes in rabbits. In the subacute study, dermal exposure to DF led to notable alterations in the physiological and biochemical status of rats, including reduced food and water intake, decreased body weight gain, and significant changes in hematological and biochemical parameters. An increase in the relative count of certain leukocyte populations was observed, with a statistically significant elevation in absolute eosinophil counts in both sexes. Similar trends were observed for basophils and lymphocytes. Moderate elevations in serum enzyme levels (ALT, AST, ALP, GGT, and LDH) indicated systemic toxicity. Morphological and morphometric analysis of the skin further supported the dermal toxicity of DF, revealing epidermal keratinization, marked proliferation of germinative layer cells, and cellular heterotopia. In the basal layer of the epidermis, cells exhibited signs of intracellular edema. The papillary layer showed moderate focal inflammatory infiltrates, while the reticular dermis displayed edema of fibrous structures, swelling of collagen fibers, and increased fiber thickness.
Discussion
These findings collectively demonstrate that the tested drilling fluid possesses significant dermal and systemic toxicity, indicating potential health risks for mammals upon repeated exposure.
Introduction
Modern oil production technologies extensively utilize a wide range of drilling fluids (DF), which are complex multicomponent systems broadly classified into water-based and oil-based formulations. These fluids typically comprise clay, silica suspensions, a variety of polymers, and numerous chemical additives,1,2 including nanomaterials 3 and, in some cases, asbestos. 4 DF may also contain heavy metals, polycyclic aromatic hydrocarbons (PAHs), petroleum hydrocarbons, and naturally occurring radioactive materials, all of which can pose significant risks to the environment, aquatic life, and human health. 5 Recent studies have highlighted the potential toxicity of nanoparticles present in DF, particularly their harmful effects on aquatic organisms.6,7
Several investigations have assessed the non-carcinogenic and carcinogenic health risks to drilling rig personnel, focusing on chemical exposure through inhalation, ingestion, and dermal absorption.8,9 Among the most frequently reported health issues associated with DF exposure are skin irritation and contact dermatitis, 10 which result from both the intrinsic properties of the fluids and the actions of their chemical constituents. Workers involved in drilling operations are commonly exposed to DF during drilling, sample collection, equipment maintenance, and routine inspections. Occupational exposure in oil production settings has also been linked to disorders of the sebaceous glands. 11 Comedogenic factors in DFs contribute to the pathogenesis of both common acne and oil acne, which share mechanisms such as inflammation, increased sebum production, and excessive follicular hyperkeratosis.
In addition, studies have documented the dermatotoxic effects of crude oil, 12 phthalates, 13 and dioxins, 14 including skin irritation and the development of dermatitis. These agents can impair the skin’s barrier function, rendering it more vulnerable to irritants, sensitizing agents, and microbial invasion.
However, current literature on the general toxic, skin-irritating, and skin-resorptive effects of DF is limited and insufficient to provide a comprehensive understanding of the associated health risks. This is further complicated by the complex and variable chemical composition of DF and the multifactorial nature of dermal reactions, which include both exogenous irritant effects and endogenous physiological responses.
Research aimed at evaluating the dermal toxicity of DF is essential for understanding its systemic effects and for improving the prediction and prevention of adverse skin reactions among oil industry workers.
The objective of this study was to assess the potential toxicity and health risks of DF through evaluation of its skin-irritating and skin-resorptive effects.
Comprehensive experimental studies on mammals will not only make it possible to identify the mechanisms of intoxication development in acute and subacute experiments, but also to provide specific recommendations for improving the working conditions of key worker groups involved in hydrocarbon extraction Based on the results, a set of proposals will be prepared for inclusion in the Sanitary and Epidemiological Requirements for Technological and Related Facilities and Structures Engaged in Oil Operations (Annex 4 to Order No. KR DSM-13 of the Ministry of Health of the Republic of Kazakhstan, dated February 11, 2022) and the Waste Classifier (Order No. 314 of the Acting Minister of Ecology, Geology, and Natural Resources of the Republic of Kazakhstan, dated August 6, 2021).
Moreover, the Health Care Development Concept of the Republic of Kazakhstan until 2026 (Resolution No. 945 of the Government of the Republic of Kazakhstan, dated November 24, 2022) highlights the absence of scientifically validated methodologies within the current practices of the sanitary and epidemiological services. Specifically, there is a lack of systematic approaches for investigating the harmful effects of environmental factors on public health and for establishing cause-and-effect relationships.
Objective of the Study: To assess the dermal toxicity of DF in acute and subacute experimental models.
Materials and methods
Animals and experimental design
To evaluate the acute skin-irritating effects of DF, six adult male rats were used. Additionally, six adult male rabbits were employed to assess the local irritant effects of DF on the ocular mucous membranes. For the subacute toxicity study, sexually mature outbred rats of both sexes (body weight: 180–220 g) were used. Animals were randomly assigned to control (n = 20; 10 males and 10 females) and experimental (n = 20; 10 males and 10 females) groups. Randomization was performed based on body weight, ensuring that intergroup differences did not exceed 10%. The species, strain, sex, and age of animals were standardized across groups. To determine the sample size, several approaches were employed: a regulatory approach based on standard OECD protocols; a statistical approach utilizing a number of equations derived from preliminary studies, including literature sources; and an ethical approach grounded in the principles of the ‘3Rs’ concept.
All animal procedures were conducted in compliance with the ARRIVE guidelines and international regulations governing the ethical use of animals in research, including the EU Directive 2010/63/EU and institutional equivalents such as IACUC protocols. Within the structure of the NJSC «M. Ospanov West Kazakhstan Medical University», a Scientific and Practical Center operates, which conducts scientific research and is responsible for carrying out experimental studies on animals. The center employs qualified scientific personnel, as well as staff responsible for the operation of the vivarium. This team utilizes the necessary tools for the practical application and implementation of the latest technical and scientific innovations related to the «3 Rs» principle.
At the end of the study, animals were euthanized according to ethical standards recommended by the Federation of European Laboratory Animal Science Associations (FELASA), the International Council for Laboratory Animal Science (ICLAS), and the AVMA Guidelines for the Euthanasia of Animals: 2020 Edition. To minimize distress, animals received intramuscular xylazine at a dose of 4 mg per 100 g of body weight, followed by decapitation.
Animal housing and ethical compliance
All animals were housed under standard vivarium conditions in accordance with the Sanitary Rules for the Arrangement, Equipment, and Maintenance of Experimental Biological Clinics (Vivariums) (Order No. 8.01.004.97, dated June 19, 1997). Animals had unrestricted access to food and water, were maintained under natural light cycles, and were housed at an ambient temperature of 20°C–22°C.
The experimental procedures were conducted in accordance with Guidelines R1.2.31.56-13 (Assessment of Toxicity and Hazard of Chemicals and Their Mixtures for Human Health, Rospotrebnadzor, Moscow, 2014), which conform to international standards, including the OECD Test Guideline No. 420 (Acute Oral Toxicity – Fixed Dose Procedure). Additionally, the study adhered to GOST 33216-2014 (Guidelines for the Maintenance and Care of Laboratory Animals) and the Rules for the Maintenance and Care of Laboratory Rodents and Rabbits, adopted by the Interstate Council for Standardization, Metrology, and Certification (Protocol No. 73-P, December 22, 2014). Selection of experimental animals took into account their specific pathogen-free (SPF) health status, as well as consistency and control of environmental conditions.
To assess the local irritant effect of the DF on ocular tissues, two drops of native DF were instilled into the conjunctival sac of the left eye (OS) of each rabbit using a pipette. The right eye (OD) served as an untreated control. Clinical evaluations were performed at 1, 24, 48, and 72 h post-exposure. Ocular assessments were conducted visually and using an ophthalmoscope (Heine Beta 200 S, Germany, 2017).
For the acute dermal irritation study, native-state DF was applied at a dose of 20 mL/cm2 to a 4 × 4 cm (16 cm2) shaved area on the dorsal surface of each rat. Observations focused on signs of erythema, edema, and other visible dermal reactions at predetermined intervals following exposure.
Dermal application of the DF was performed using the open method with a pipette. A single dose was applied to a designated skin area, and local skin reactions were evaluated at 1, 4, 24, 48, and 72 h post-application. Signs of irritation, including erythema and edema, were assessed visually.
Erythema severity was graded on a scale from 0 to 4 using the colorimetric scale developed by S.V. Suvorov, where: 0 = no erythema, 1 = very slight, 2 = well-defined, 3 = moderate to severe, 4 = severe (beet-red coloration). Edema was also scored on a 0–4 scale based on skin fold thickness: 0 = 0–0.3 mm, 1 = 0.4–0.6 mm, 2 = 0.7–1.0 mm, 3 => 1.0 mm (moderate), 4 = severe edema.
Based on these indicators, the overall severity of the skin-irritant effect was determined.
The skin-resorptive effect of DF was assessed in a subacute 30-days experiment. In the experimental group, native-state DF was applied daily to a shaved 5 × 5 cm area (approximately 5% of total body surface area) on the right dorsal side of the rats, using a pipette and spatula.15,16 The left side, symmetrical to the treated area, served as an untreated control. Hair removal was performed 1–2 days prior to exposure using scissors or electric clippers. The DF was applied at a dose of 20 mg/cm2. The application technique was non-invasive and did not result in mechanical injury to the skin.
Throughout the 30-days observation period, the general condition of the animals was monitored daily, including appearance (fur and skin condition), behavioral signs of toxicity, food and water intake, and mortality. Motor activity (duration and frequency of vertical rearings) and emotional responses (number of grooming episodes) were also recorded. The duration of rearings was measured for 5 min, and their number was recorded over a 1-h period on days 1, 7, and 15 of the study.
Body weight was recorded in all animals prior to the start of the experiment and every 10 days thereafter. Weight gain was calculated and expressed in grams.
At the conclusion of the experiment, animals were euthanized, and biological samples were collected for further clinical, biochemical, and histopathological analysis. The assessment of subchronic dermal toxicity following repeated exposure in a 30-days study is consistent with the OECD Test Guideline No. 410, «Repeated Dose Dermal Toxicity: 21/28-days Study.»
Biochemical and hematological analysis
On day 30 of the experiment, peripheral blood samples were collected for biochemical and hematological evaluation.
Hematological parameters included red blood cell indices: hemoglobin concentration, erythrocyte count, color index, hematocrit value, platelet count, and erythrocyte sedimentation rate (ESR). White blood cell parameters comprised total leukocyte count and differential leukocyte profile, including lymphocytes and monocytes. These analyses were performed using an automated hematology analyzer (Sysmex XN-550 II, Sysmex Corporation, Japan).
Biochemical parameters were assessed to evaluate systemic toxicity and included serum enzyme activities: • Alanine aminotransferase (ALT), • Aspartate aminotransferase (AST), • Alkaline phosphatase (ALP), • Lactate dehydrogenase (LDH), • Gamma-glutamyl transpeptidase (GGT), and • C-reactive protein (CRP).
CRP was included as an inflammatory marker to assess the systemic inflammatory response, given that internal organs — including the liver, heart, kidneys, stomach, and adrenal glands — were also sampled for histological analysis. As a sensitive acute-phase protein synthesized by the liver in response to tissue injury or inflammation, CRP is widely used for monitoring systemic responses.
These measurements were carried out using a fully automated modular clinical chemistry analyzer (Cobas 6000, immunochemical module C 501-1, Roche Diagnostics, Germany).
Histological examination
Microscopic analysis of tissue samples was performed at the Morphological Laboratory of the Histology Department, NJSC West Kazakhstan Marat Ospanov Medical University. Skin tissue was fixed in 10% buffered formalin and processed according to the standard operating procedures of the laboratory.
Histological sections, 4–5 µm thick, were obtained from paraffin-embedded tissue blocks using a rotary microtome (Accu-Cut SRM 200, Sakura Finetek, USA). Sections were stained with hematoxylin and eosin (H&E), both manually and using an automated staining system (BIO-OPTICA-AUS 240, Bio-Optica, Milan, Italy).
Histological evaluation was conducted using a digital light microscope (Axio Lab A1, Carl Zeiss, Germany; registration certificate RK-MT-7 No. 009046, registered on 17.08.2018) at magnifications of ×40, ×100, and ×400. Digital microphotographs were captured using the AxioCam ERc5s camera.
The morphological assessment included evaluation of the structural organization of the epidermis and dermis, as well as the condition of hair follicles. Specific attention was paid to the thickness of the entire epidermis and its layers (stratum corneum, stratum granulosum, and stratum basale), as well as the dermal thickness. Quantitative morphometric analysis was performed using ImageJ software.
Drilling fluid characterization
Samples of the DF used in the study were collected from the Zhanazhol oil field, located in Western Kazakhstan. Chemical composition analysis was conducted in collaboration with the Aktobe Regional Center for Sanitary and Epidemiological Expertise and LLP Geoplasma under a contractual agreement.
The analysis revealed the presence of several hazardous and industrial chemical components, including lead and arsenic. The DF also contained bentonite clay, lime, ferrochrome lignosulfonate, carboxymethylcellulose, caustic soda, and barium sulfate. Additional constituents identified in the drilling sludge included phosphorus oxide, silicon dioxide, calcium oxide, sodium oxide, potassium oxide, and bituminous substances. 17
Statistical analysis
The data obtained from the study were analyzed using methods of descriptive and inferential statistics. Quantitative results are presented as mean values (M) with corresponding standard deviations (SD). Intergroup differences were evaluated using the non-parametric Mann–Whitney U test for independent samples. For parameters assessed over multiple time points, repeated-measures analysis of variance (ANOVA) was applied to determine statistical significance.
All statistical analyses were conducted using standard statistical software, with a significance level set at p < 0.05.
Ethical Approval and Funding
This research was conducted as part of the scientific project IRN AP19676915: Development of Toxicometric Criteria for the Hazard and Toxicity of Drilling Fluids and Cuttings, funded by the Science Committee of the Ministry of Science and Higher Education of the Republic of Kazakhstan.
An approval was obtained from the local bioethics committee of the NJSC «M. Ospanov West Kazakhstan Medical University» dated 15 March 2023, Protocol No. 3 (3/14), which is based on adherence to principles consistent with the requirements of the Institutional Animal Care and Use Committee (IACUC). The principles of IACUC protocols are detailed in the Recommendations of the Eurasian Economic Commission Board dated November 14, 2023, No. 33 «Adilet» «On the Guidelines for Working with Laboratory (Experimental) Animals in Preclinical (Nonclinical) Studies»
Results
The effects of the DF on the mucous membranes of rabbit eyes are presented in Figures 1 and 2. Ocular response to drilling fluid exposure in rabbits. (a) Control eye with no signs of irritation; (b) Mild conjunctival hyperemia observed 24 h post-exposure; (c) Moderate conjunctival hyperemia observed in the experimental group. Ocular responses were observed following exposure to DF in rabbits. (a) Injection of scleral vessels indicating vascular response; (b) Pronounced conjunctival hyperemia observed at 48 h; (c) Conjunctival chemosis with grayish-white discharge in the subconjunctival space.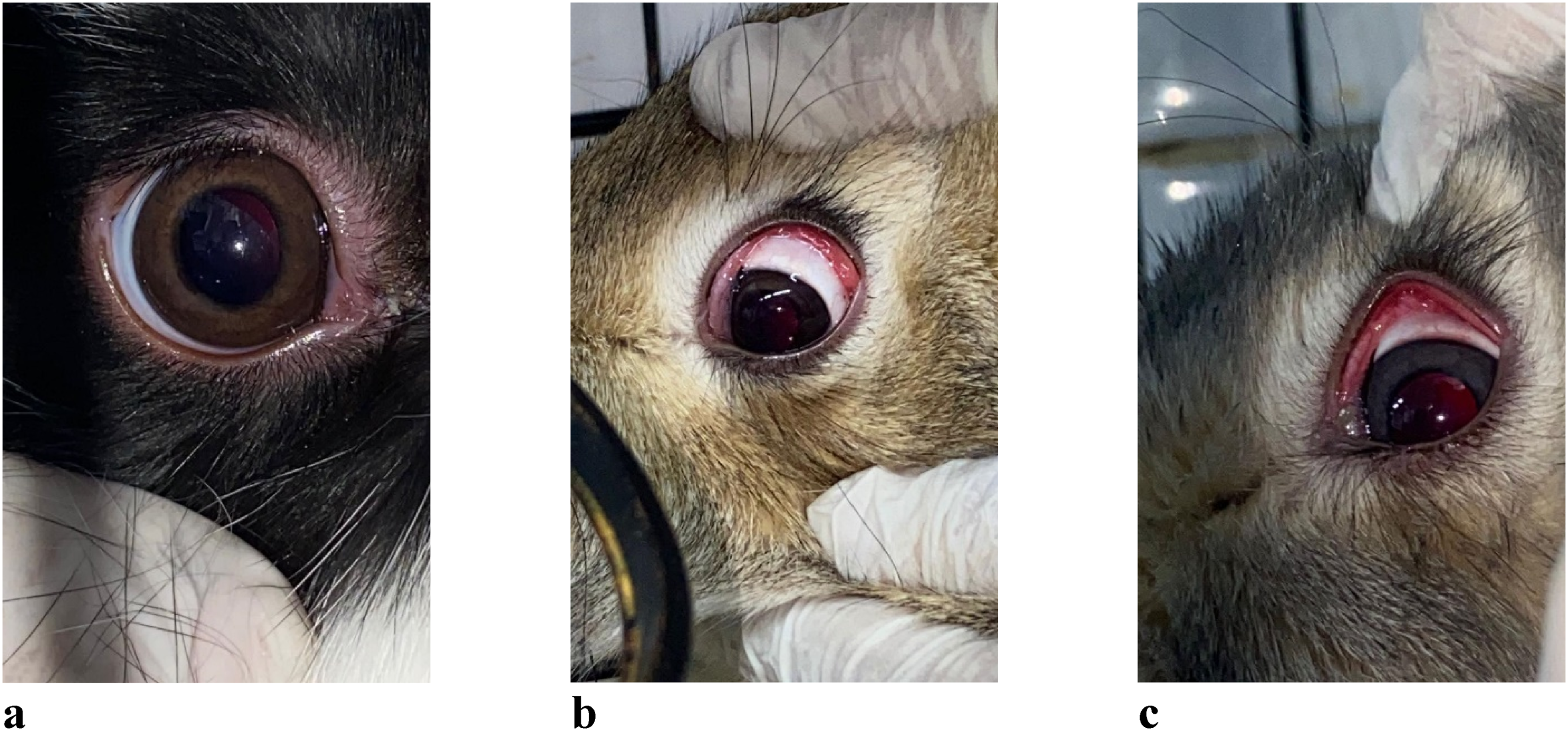

Figure 1(a) shows the right eye (OD) of a control rabbit. No signs of irritation or pathology were observed. The eye appeared normal, with intact eyelid function, unimpeded blinking, and a calm ocular surface.
Following the instillation of DF into the left eye (OS) of experimental animals, no changes were detected in any of the six rabbits at the 1-h mark. However, by 24 h post-exposure, clinical signs of irritation began to appear: • Conjunctival hyperemia was observed in 5 out of 6 rabbits (Figure 1(b) and (c)). • Conjunctival injection was noted in 3 rabbits (Figure 2(g)). • One rabbit developed chemosis, with a grayish-white discharge in the subconjunctival space (Figure 2(e)).
At 48 h post-exposure, 3 rabbits continued to display marked conjunctival hyperemia (Figure 2(d)). No progression to more severe pathology was observed. By 72 h, signs of irritation had subsided in all animals.
A comprehensive ophthalmologic examination was conducted to assess the internal structures of the eye. The following findings were consistent across all subjects: • Full preservation of ocular motility. • A transparent cornea and clear anterior chamber of medium depth. • An intact iris with preserved architecture; the pupil was round, centrally located, and responsive to light. • The fundus appeared normal, with a well-adherent retina. • The optic disc morphology was unchanged compared to the control eye; retinal arteries were narrowed, and veins appeared engorged, though without hemorrhages or exudates.
These results suggest that while DF exposure can induce mild to moderate transient irritation of the conjunctiva, it does not cause damage to the internal ocular structures under the conditions tested.
Under conditions of acute single exposure, dermal application of the DF to rat skin induced moderate erythema, characterized by a pink-red coloration, corresponding to score 2 on the erythema grading scale. Assessment of skin edema revealed mild swelling, corresponding to score 1, with a measured increase in skin fold thickness of 0.3 mm.
The combined irritation score, calculated as the sum of erythema and edema scores, yielded an average total score of 3. According to toxicological classification standards, 17 this response corresponds to a moderate irritant effect and is classified as Class 2 in terms of skin reaction severity.
The skin-resorptive effects of DF were evaluated in a 30-days subacute experiment based on behavioral, physiological, and clinical indicators, including food and water intake, body weight dynamics, and visible signs of systemic toxicity.
Daily feed consumption in rats (g/day) during the 30-days observation period.

Note. *p < 0.05 indicates statistically significant differences between female groups (Mann–Whitney U test).
**p < 0.05 indicates statistically significant differences between male groups (Mann–Whitney U test).
Daily feed consumption in rats (g/day) during the 30-days observation period.

Note. *p < 0.05 indicates statistically significant differences in day-to-day comparisons within the same group (Mann–Whitney U test).
**p < 0.05 indicates statistically significant differences between control and experimental groups on the same day (Mann–Whitney U test).
Water consumption also declined significantly by day 20. For example:
In male rats, water intake decreased to 46.70 ± 0.48 mL/day in the experimental group versus 49.00 ± 0.67 mL/day in controls (p < 0.001).
In female rats, values were 46.40 ± 0.52 mL/day in the experimental group compared to 49.40 ± 0.70 mL/day in controls (p < 0.001).
Body weight dynamics mirrored the changes in food and water intake. As shown in Figure 3, the most pronounced reductions in body weight were recorded on days 20 and 30 in the experimental group. No statistically significant gender differences were observed in body weight trends. Dynamics of changes in body weight.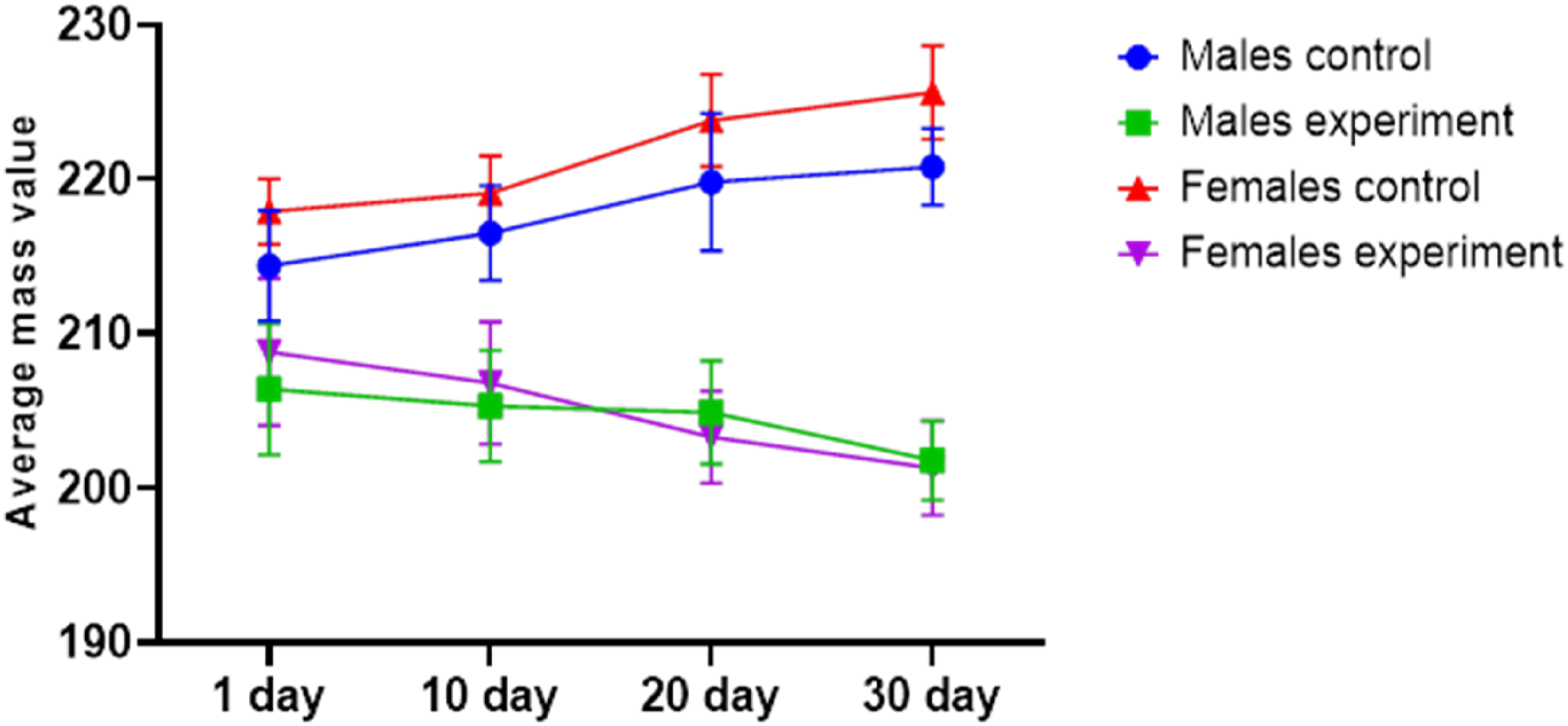
Food and water consumption, which are key regulators of general metabolic processes, directly influence body weight. In conditions of acute or subacute intoxication—particularly when digestive functions and the regulation of feeding behavior are disrupted—body weight dynamics serve as a critical indicator of the animals’ overall physiological status.18,19 In the present study, the observed reduction in food intake and subsequent attenuation in body weight gain among experimental animals can be attributed to these mechanisms.
The pathophysiological basis of chemically induced intoxication is often linked to the development of inflammatory processes in the gastrointestinal tract and impairment in the synthesis and secretion of bile acids, which are essential for the proper absorption of macro- and micronutrients.20,21
Changes in behavioral responses under conditions of subacute dermal-resorptive exposure to DF were assessed by analyzing rearing behavior, specifically the duration and frequency of rearings in experimental animals (Figures 4 and 5). A significant decline in rearing duration was observed by day 15, with partial recovery noted by day 30. These changes were more pronounced in male rats compared to females. Rearing duration. Number of rearings.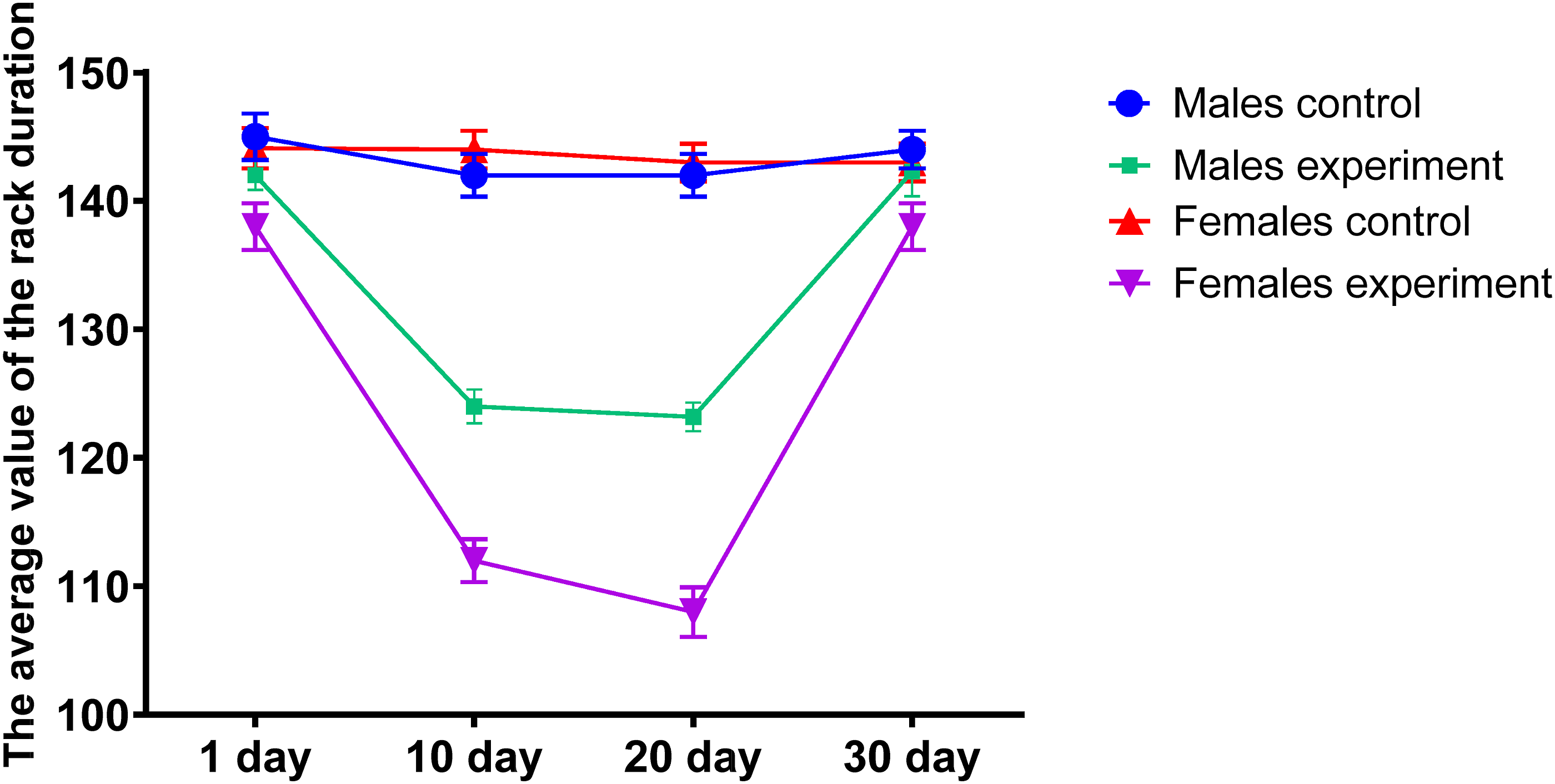

A similar pattern was noted in the number of rearings, which decreased during the mid-phase of the experiment, followed by a gradual restoration of activity. In addition, grooming behavior—a stereotyped form of self-care and an important marker of neurobehavioral function in rodents—was also adversely affected. Experimental animals exhibited both reduced duration and frequency of grooming episodes, as shown in Figures 6 and 7, indicating the negative neurotoxic impact of the DF on behavioral regulation. Duration of grooming. Grooming frequency.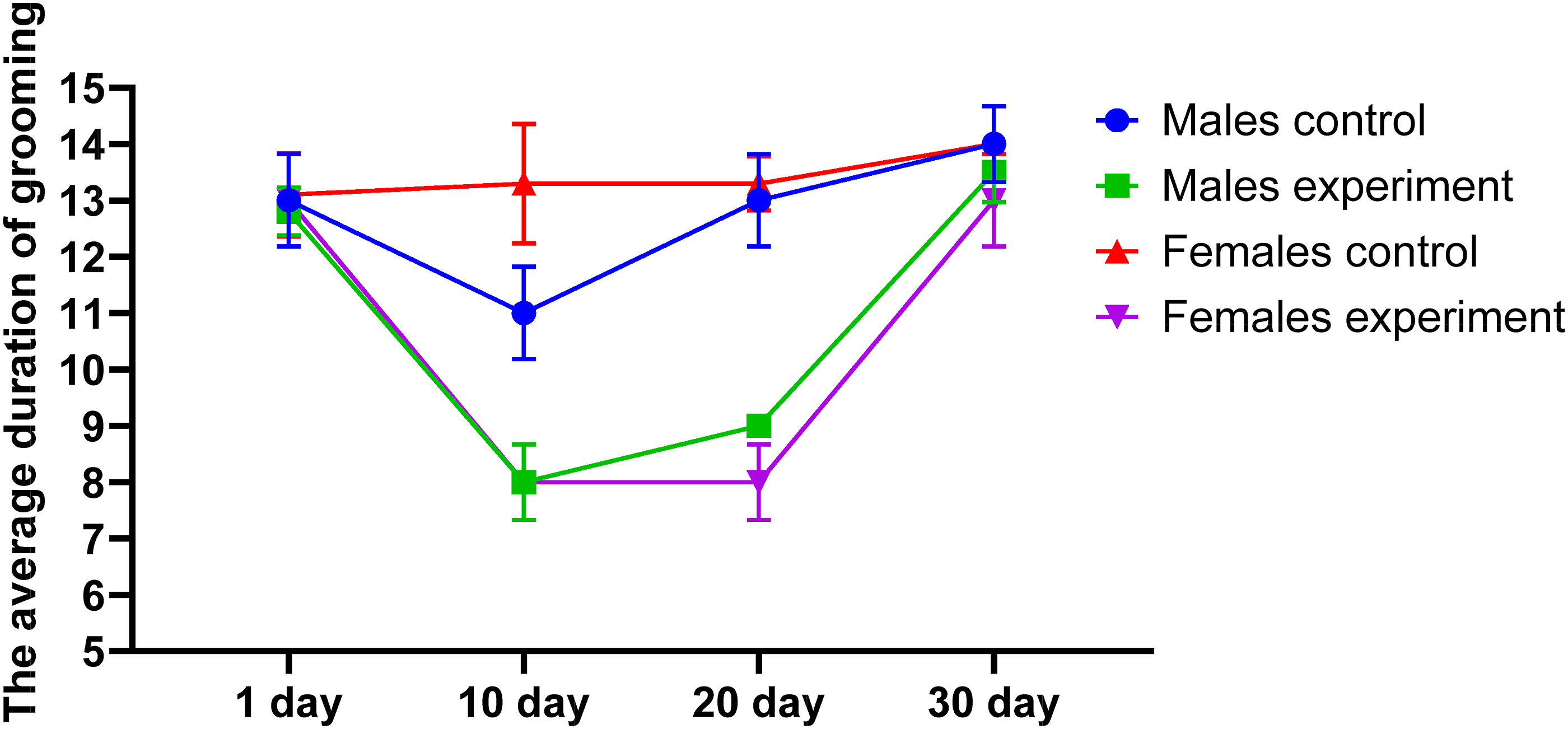

Peripheral blood parameters in female rats (n = 20 per group).
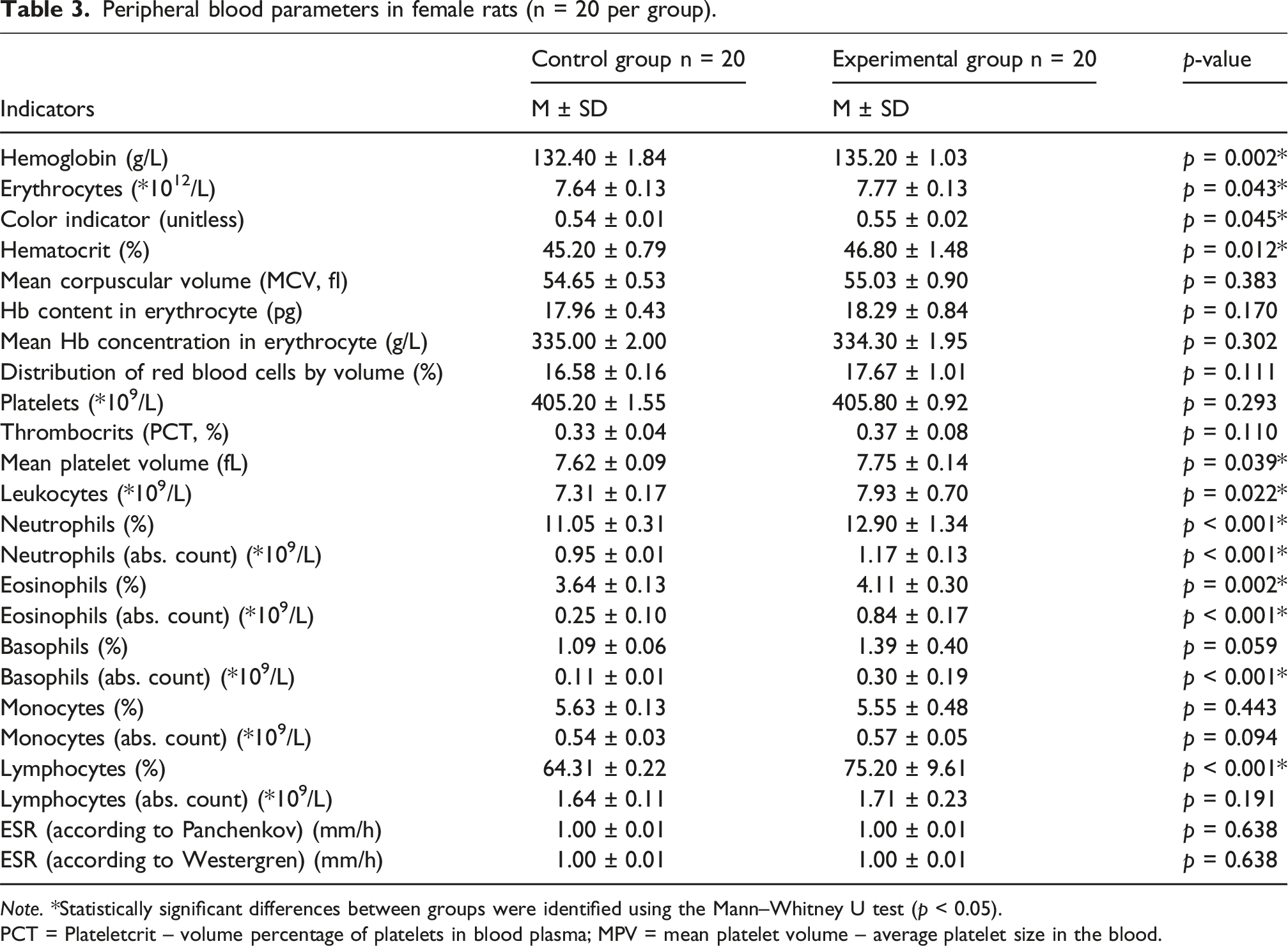
Note. *Statistically significant differences between groups were identified using the Mann–Whitney U test (p < 0.05).
PCT = Plateletcrit – volume percentage of platelets in blood plasma; MPV = mean platelet volume – average platelet size in the blood.
Peripheral blood parameters in male rats (n = 20 per group).

Note. *Statistically significant differences between groups were identified using the Mann–Whitney U test (p < 0.05).
PCT = Plateletcrit – volume percentage of platelets in plasma.
MPV = Mean Platelet Volume – average platelet size in the blood.
In contrast, pronounced alterations were found in the white blood cell (WBC) profile of animals exposed to DF. Notably, there was a significant increase in the relative and absolute counts of specific leukocyte subpopulations in the experimental groups compared to controls. • Neutrophils: ○ Males (experimental): 12.72 ± 0.23 versus control: 11.06 ± 0.23 (p < 0.001) ○ Females (experimental): 12.90 ± 0.31 versus control: 11.05 ± 0.31 (p < 0.001) • Eosinophils (absolute count): ○ Males (experimental): 0.88 ± 0.20 versus control: 0.41 ± 0.09 (p < 0.001) ○ Females (experimental): 0.84 ± 0.17 versus control: 0.25 ± 0.10 (p < 0.001)
Similar trends were observed for basophils and lymphocytes, with elevated absolute counts in both male and female rats from the experimental groups.
These increases in granulocytes and lymphocytes under conditions of subacute dermal exposure to DF are likely due to stress-induced mobilization of immune cells. The elevated leukocyte counts may reflect: • Accelerated release of granulocytes from the bone marrow reserve, • Recruitment of lymphoid cells from the spleen and thymus, • Enhanced immune surveillance and adaptive responses in reaction to chemical stress.
22
The rise in lymphocyte levels, in particular, suggests a compensatory mechanism aimed at maintaining immunological homeostasis during toxic exposure and may serve as a marker of systemic adaptation to xenobiotic-induced stress.
Clinical and biochemical blood parameters in female rats (Mean ± SD).

Note. *Statistically significant differences between the groups were identified using the Mann–Whitney U test.
ALT – Alanine aminotransferase; AST – Aspartate aminotransferase; ALP – Alkaline phosphatase; CRP – C-reactive protein; GGTP – Gamma-glutamyl transpeptidase; LDH – Lactate dehydrogenase.
Clinical and biochemical blood parameters in male rats (Mean ± SD).

Note. *Statistically significant differences between the groups were identified using the Mann–Whitney U test.
ALT – Alanine aminotransferase; AST – Aspartate aminotransferase; ALP – Alkaline phosphatase; CRP – C-reactive protein; GGTP – Gamma-glutamyl transpeptidase; LDH – Lactate dehydrogenase.
In addition to enzyme activity elevation, biochemical analyses revealed increased concentrations of creatinine, bilirubin, cholesterol, and triglycerides in the serum of experimental animals, indicative of systemic metabolic disruption. Notably, despite elevated hepatocellular markers, ALP activity showed a paradoxical decrease, along with a reduction in C-reactive protein (CRP) levels, potentially reflecting a suppression of inflammatory signaling or impaired synthetic liver function under toxic load.
Furthermore, a decline in total protein and serum albumin levels was observed, which may be attributable to impaired protein synthesis in the liver due to hepatotoxic damage. These alterations underscore the sensitivity of blood as a biomarker system, responding promptly to environmental xenobiotic stressors, including dermal exposure to DF.
The observed enzyme profile supports the hypothesis that DF exerts a hepatotoxic effect through the disruption of parenchymal organ function, particularly hepatocytes. The release of indicator enzymes such as LDH, AST, and ALT, commonly associated with hepatocellular injury, along with changes in cholestasis-related enzymes like ALP and the membrane-bound GGTP, reinforces this conclusion. As GGTP is considered more sensitive than ALT or AST in detecting early hepatobiliary disturbances,23,24 its elevated activity further confirms the toxic effect of DF on the liver.
In addition to evaluating eating behavior, behavioral responses, and clinical-biochemical blood parameters, morphological alterations in skin structure were examined following subacute dermal-resorptive exposure to the DF. On day 30 of the experiment, histological analysis revealed a marked polymorphism and reduced quantity of keratohyalin granules within the granular layer, indicating impaired keratinization processes in the epidermis. This impairment resulted in a measurable thinning of the stratum corneum compared to the control group.
Furthermore, significant proliferation of cells in the germinative layers (basal and spinous) was observed, reflecting active reparative regeneration. Cellular heterotopia was detected, as evidenced by the atypical localization of morphologically basal-like cells in the spinous and granular layers of the epidermis. Signs of intracellular edema were present in basal layer keratinocytes, and in the spinous layer, vacuolated cells with pyknotic nuclei were found, indicative of disrupted keratinocyte differentiation and the development of parakeratotic foci.
Chronic DF exposure for 30 days also led to moderate focal inflammatory infiltrates in the papillary dermis. In the reticular dermis, pronounced edema of fibrous structures and the intercellular matrix was observed, accompanied by collagen fiber swelling, increased fiber thickness, and disrupted orientation. These pathological features collectively contributed to an increased thickness of the reticular dermis in experimental animals relative to controls (Figure 8). Histological changes in the rat epidermis following 30-days dermal exposure to drilling fluid: notable proliferation (PP) of the basal and spinous layers of the epidermis, thinning of the stratum corneum (SC), and the presence of light cells (LC) with pyknotic (wrinkled) nuclei, indicative of impaired keratinocyte differentiation. Hematoxylin and eosin staining. Original magnification: ×400.
Changes in the histological structures of the skin on the 30th day of the experiment in female rats.

Note. * indicates statistically significant differences between control and experimental groups, assessed using the Mann–Whitney U test.
Changes in the histological structures of the skin on day 30 of the experiment in male rats.

Note. *indicates statistically significant differences between control and experimental groups, assessed using the Mann–Whitney U test.
Discussion
The findings of our study demonstrate that the DF exhibits a pronounced skin-irritating and skin-resorptive effect. In the acute experiment conducted on rabbits, it was confirmed that the irritant effect on the ocular mucosa is characterized by marked conjunctival hyperemia and vascular injection. In isolated cases, conjunctival chemosis and grayish-white discharge in the subconjunctival space were also observed. Ophthalmological examination revealed no pathological changes in the internal structures of the eye, and the transparency of the cornea, condition of the anterior chamber, iris, pupil reaction, and fundus remained within normal limits.
In the subacute experiment involving male and female rats, the skin-resorptive effects of DF were associated with significant alterations in the general condition of the animals. A notable decrease in food and water intake was observed, which corresponded with a clear downward trend in body weight by the end of the experiment. Importantly, the behavioral responses of the animals were also affected: the number and frequency of rearings decreased significantly by mid-experiment, with a partial recovery observed by day 30. The frequency and duration of grooming episodes—an indicator of emotional and neurological well-being – also declined, with the most pronounced changes recorded on day 15.
Although no sex-specific differences were observed in the dynamics of body weight reduction, gender-based variations were evident in behavioral responses. In particular, male rats in the experimental group exhibited more pronounced changes in rearing duration and grooming behavior compared to females.
The assessment of peripheral blood, along with clinical and biochemical parameters in the subacute experiment, convincingly demonstrates the toxic effects of DF under conditions of skin-resorptive exposure. The observed increase in the total leukocyte count and in specific leukocyte subpopulations (neutrophils, eosinophils, basophils, and lymphocytes) clearly indicates a systemic toxic response to DF.
The damaging effect of DF on membrane-bound enzymes is further supported by alterations in the activity of key detoxification enzymes, including aspartate aminotransferase (AST), alanine aminotransferase (ALT), alkaline phosphatase (ALP), gamma-glutamyl transpeptidase (GGTP), and lactate dehydrogenase (LDH). These changes are indicative of hepatocellular and hepatobiliary system disturbances.
Additional biochemical changes – including decreased levels of total protein and albumin, accompanied by increased creatinine – highlight the adverse systemic impact of DF, likely reflecting impaired protein synthesis and renal function. Moreover, rats in the experimental group exhibited elevated levels of bilirubin, cholesterol, and triglycerides, consistent with metabolic disturbances caused by subacute intoxication.
Sex-related differences in clinical and biochemical blood parameters were also identified. In both control and experimental groups, males exhibited higher levels of total protein, albumin, and enzyme activity compared to females. Conversely, females demonstrated higher levels of alkaline phosphatase activity, creatinine, and bilirubin than males, regardless of group assignment.
The toxic impact of DF on the organs of experimental animals under subacute, skin-resorptive exposure is substantiated by the findings of our morphological and morphometric analyses. By the end of the 30-days experimental period, marked polymorphism and a reduction in the number of keratohyalin granules in the granular layer of the epidermis were observed. These alterations signify disruption of the keratinization process, resulting in a significant thinning of the stratum corneum compared to the control group.
Pronounced proliferation of cells within the germinal layer, as revealed in the subacute experiment, is indicative of reparative epidermal regeneration. In addition, the observed cellular heterotopia—misplacement of cells into atypical epidermal layers—suggests disturbed differentiation processes. These changes collectively reflect the adverse dermal effects of DF.
Our findings are supported by evidence from other toxicological studies. For instance, the bioaccumulation of chemical constituents of DF, such as heavy metals and hydrocarbons, in the tissues of the earthworm Aporrectodea longa has been associated with moderate histopathological alterations in the digestive tract. 25 Similarly, exposure to DF containing petroleum-based emulsifiers induced structural abnormalities in the gills and liver of Tilapia guineensis fingerlings, including inflammation, cellular degeneration, and necrosis. 26
The toxicity and ecological risks posed by DF and drilling sludges remain insufficiently investigated and are often the subject of debate. In Kazakhstan, where the majority of hydrocarbon extraction is carried out by foreign enterprises, chemical reagents used for DF preparation are largely imported. Consequently, their toxicological profiles and environmental safety data are frequently unavailable or incomplete.
DFs are inherently complex, multicomponent mixtures,1,27 typically comprising clay minerals, siliceous agents, various polymers, and numerous chemical additives – including metal oxide nanoparticles.6,28 Alarmingly, chrysotile asbestos, a well-known carcinogen, has been reported as a DF additive in some cases.7,29
DFs are generally classified into three types based on their base fluid: water-based (WBDF), oil-based (OBDF), and synthetic-based (SBDF) DF. OBDFs tend to exhibit significantly higher toxicity and environmental hazard due to their elevated content of aromatic hydrocarbons.30,31 The presence of heterocyclic analogs of polycyclic aromatic hydrocarbons in oil-based formulations further contributes to a synergistic toxic effect. 8
DF, due to their complex multicomponent composition, should meet stringent safety criteria for both environmental and human health. However, in the vast majority of cases, DFs lack formal sanitary-epidemiological and toxicological-hygienic certification. The available scientific literature on this subject is highly fragmented, with most studies being superficial, descriptive, and lacking in comprehensive toxicological assessments. Critically, there is an evident gap in robust scientific investigations specifically addressing the skin-irritating and skin-resorptive effects of DFs and drilling waste.
Dermal exposure to a wide range of industrial chemicals, including DFs, poses a substantial occupational health risk. The skin of personnel in direct contact with drilling operations may be exposed not only to freshly prepared DFs but also to spent DF and waste products. Cutaneous absorption of chemical agents can result in both local effects, such as inflammation, irritation, and sensitization, and systemic toxicity following percutaneous penetration. 32
Previous studies have shown that skin contact with petroleum products leads to irritation and dermatitis due to disruption of the skin barrier, making it more vulnerable to secondary exposures from other irritants, allergens, and pathogens. 33 The potential for adverse dermatological and systemic outcomes correlates with the chemical composition of the DF and the nature of its additives. Reported symptoms in exposed individuals include not only contact dermatitis but also headache, nausea, ocular irritation, and coughing. 34
The potential and risk of adverse effects from DF, associated with hazardous components and additives, are also accompanied by the development of contact dermatitis 35 ; other effects include headache, nausea, eye irritation, and coughing. As is well known, the skin is a complex and dynamic organ composed of an outer epidermis and an inner dermis, whose functions extend far beyond serving as a mere barrier to the external environment. The functions of the skin include not only moisture retention, tactile sensation, and thermoregulation, but also regulation of endocrine activity, vitamin D synthesis, immunological effects, as well as biotransformation of xenobiotics. 15 Occupational chemical exposure to the skin can lead to numerous disorders that adversely affect human health and work capacity. There are three types of chemical interactions with the skin: direct contact, immune-mediated effects, and systemic exposure. 16 Essentially, the skin serves as a signaling interface between xenobiotics and the body.
The study and assessment of skin irritation is a regulatory requirement in the safety evaluation of industrial and consumer products. 36
Evaluation of dermal irritant effects is a regulatory prerequisite in the toxicological assessment of industrial and consumer products. 34 Given that the skin of laboratory animals such as rats, guinea pigs, and rabbits is generally more permeable than that of humans,32,37 the use of these models in studies of DF toxicity is both scientifically valid and relevant for risk assessment. In fact, for certain substances, the dermal route of exposure can pose a greater toxicological risk than oral ingestion. This is attributed to the skin’s role as the largest organ of the body, with a complex immune architecture comprising interacting networks of innate and adaptive immune cells. 38
Furthermore, during ontogeny, the skin may be subjected to cumulative environmental stressors, including chemical pollutants. For oil industry workers, these exposures are compounded by additional occupational hazards such as ultraviolet radiation, heavy physical exertion, and adverse microclimatic conditions. These factors can further modulate skin physiology and exacerbate dermal responses to chemical insults. 39
Conclusion
The findings of this study underscore the necessity of experimental approaches to accurately determine the degree of hazard and toxicity posed by DF, given their highly variable composition, which changes depending on the level of fluid degradation and reuse. The experimental data obtained in mammalian models provide compelling evidence of both skin-irritating and skin-resorptive effects of DFs. Alterations in feeding behavior, decreased body weight gain, disruptions in behavioral responses, and marked changes in hematological and enzymatic profiles in a subacute setting clearly point to the systemic toxicity of DFs.
Morphological and morphometric changes observed in both superficial and deep layers of the skin further substantiate the dermal toxicity of DFs under skin-resorptive exposure conditions. These findings highlight the importance of conducting comprehensive studies that consider the toxicodynamic properties of DFs, along with interspecies and sex-specific sensitivity to their effects. This study contributes to bridging a critical gap in scientific knowledge by detailing the mechanisms of DF toxicity and illustrating variations in biological response by sex and species. 40
Importantly, the identification of toxicological risk factors provides a scientific basis for understanding potential health determinants that may affect the well-being of exposed individuals. Assessing the toxicity and hazard level of DFs is crucial for informing the development of appropriate sanitary, hygienic, and environmental measures. These include the implementation of engineering and technical controls aimed at minimizing skin contact with DFs and drilling waste, thereby improving occupational safety and working conditions for oil industry personnel.
The knowledge generated from this research can be instrumental in forecasting and assessing medical and environmental risks in oil extraction zones and in shaping effective health protection strategies for at-risk worker populations.
Footnotes
ORCID iDs
Ethical approval
The study was conducted in accordance with the Declaration of Helsinki and approved by the Institutional Local Commission on Bioethics of the West Kazakhstan Medical University (meeting No. 3 (3/14) dated 15 March 2023).
Authors contributions
Conceptualization, A.M., Zh.K., S.B., U.S and S.S..; Methodology, A.M., Zh.K. and S.S.; Formal Analysis, Zh.K., S.B.; Investigation, A.Zh., A.K., K.Zh.; Data Curation, A.Zh. and G.Y.; Writing – Original Draft Preparation, A.M., A.K., G.Y. and U.S.; Writing – Review and Editing, A.Zh., Zh.K., K.Zh., S.B. and S.S.; Supervision, A.M.; Project Administration, A.M.; Funding Acquisition, A.M. All authors have read and agreed to the published version of the manuscript.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The research was funded by the Science Committee of the Ministry of Science and Higher Education of the Republic of Kazakhstan (Grant No. AP19676915).
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
Data are available from the corresponding author upon request.
