Abstract
In a 13-week feeding toxicity study of grape skin extract (GSE) performed previously, 5.0% GSE showed diffuse hypertrophy and basophilia in rat parotid glands. To clarify whether the change in the parotid glands was an adverse effect of GSE, 6-week-old male F344 rats were fed a diet containing 5.0% GSE or were administered a dose corresponding to the dietary concentration via gavage for 4 weeks, and the treatment was stopped for 2 weeks. To ascertain the effect of astringency, other animals were fed a diet containing 5.0% tannic acid (TA) using the same protocol as the GSE feed group. Control groups were fed a basal diet or were administered sterilized distilled water by gavage. In the GSE and TA feed groups, diffuse severe hypertrophy and basophilia in the parotid glandular epithelial cells were observed. Macroscopic, microscopic, and ultrastructural characteristics consistent with cellular hypertrophy was less apparent after the recovery period in both feed groups. In contrast, no changes were observed in the parotid glands of the gavage GSE and control groups at week 4. Based on these findings of parotid hypertrophy without cytotoxicity, the data from this and previous studies suggest that hypertrophy of the parotid glands induced by feeding treatment with GSE is an adaptive non-adverse effect that is reversible upon removal of the sialotrophic agent.
Introduction
It is known that parotid gland hypertrophy in the rat can be induced with some chemicals such as lac color, tannic acid (TA), glyphosate, or isoproterenol. It has been reported that lac color, a food coloring extracted from the resinoid secretion of coccidium larvae, Laccifer lacca KERR, induced diffuse hypertrophy of rat parotid glands in repeated 13-week or 78-week oral dose toxicity studies (Sakamoto et al. 2000). In the hypertrophic parotid glands of rats treated with lac color, the number and size of the secretory granules increased, and these granules were electron-translucent (Fukumori, Sakamoto, and Aoki 2000). In the case of lac color, since there were no ultrastructural changes in the organelles except for the secretory granules of the parotid glands, it was concluded that this reaction in the parotid glands might be an adaptive response (Fukumori, Sakamoto, and Aoki 2000). It is known that adrenergic agents such as isoproterenol and glyphosate induce parotid gland hypertrophy in rodents (Chan and Mahler 1992; Selye, Veilleux, and Cantin 1961). Isoproterenol also causes the development of infarct-like lesions when injected intraperitoneally into animals, which are morphologically similar to coagulative myocytolysis or myofibrillar degeneration (Milei, Nunez, and Rapaport 1978). The degenerative and necrotic effects of isoproterenol on the heart and parotid glands are considered to be adrenergic agonistic effects. Therefore, parotid gland hypertrophy induced by isoproterenol is the results of direct interaction with β-adrenoreceptors located on the basolateral surface of the responsive acinar cells and the induction, accumulation, and secretion of salivary proline-rich proteins (PRPs; Gho, Pena-Neira, and Lopez-Solos 2007). For glyphosate, a systemic, broad-spectrum and post-emergence herbicide, feed treatment including 5.0% glyphosate induced parotid hypertrophy in F344/N rats and B6C3F1 mice in 13-week toxicity studies (Chan and Mahler 1992), and the mechanism of parotid gland hypertrophy by glyphosate was mediated via an adrenergic pathway, which was confirmed by concurrent administration of propranolol, an adrenergic antagonist, resulting in reduction of the severity of parotid gland changes (Chan and Mahler 1992). Thus, the mechanism of parotid hypertrophy induction with isoproterenol and glyphosate is considered to be adaptive but is different from the one for lac color and TA (Gho, Pena-Neira, and Lopez-Solos 2007).
Grape skin extract (GSE) has been used as a food additive for its functional ingredients, polyphenols. GSE is extracted from the pericarp of grapes such as Koshu, Chardonnay, or Riesling with room temperature or lukewarm ethanol. In our previous 13-week study on the subchronic toxicity of GSE, 5.0% GSE induced diffuse severe hypertrophy accompanied with basophilia in the rat parotid glands without any cytotoxic changes (Inoue et al. 2013). This histopathological change was not observed in the submandibular and sublingual glands. In the same subchronic toxicity study with 5.0% GSE, there were no treatment-related differences associated with clinical signs, body weight, food intake, hematological and serum biochemical analysis, or histopathology except for parotid gland hypertrophy in both sexes and calcification in female rat kidneys (Inoue et al. 2013). In another subchronic 3-month oral toxicity study of GSE, there were no treatment-related changes in rats treated with up to 2.5% GSE (Bentivegna and Whitney 2002). However, the 3-month study did not evaluate histopathology in the parotid glands.
Since there was no systemic toxicity except for the parotid gland, the toxicological significance of the parotid gland alteration was unclear, and there was a possibility that the parotid gland hypertrophy induced by GSE might be an adaptive effect. Therefore, the present study was performed to confirm the histopathology of the parotid gland hypertrophy with GSE and to clarify whether the parotid alteration is or is not an adverse effect.
Materials and Methods
Chemicals
GSE (exGrape® Total PPR, a product of GROUPE GRAP′SUD, France) was kindly provided by the Japan Food Additive Association (Tokyo, Japan). This product is a purple powder extracted from red grapes (Vitis vinifera). According to the manufacturer’s information, the product contains total polyphenols (>92% as catechin equivalent), oligomeric proanthocyanidins (OPC, >15% as catechin equivalent), and anthocyanins (>2%). TA (CAS no. 1401-55-4) was purchased from Merck (Germany). For feeding experiments, GSE or tannic acid was mixed well with the powdered basal diet, CRF-1 (Oriental Yeast Co., Tokyo, Japan), at a concentration of 0% (control) or 5.0%. The dosage of GSE and tannic acid was determined based on results from the previous subchronic toxicity study and published report (da Costa et al. 2008), respectively. For gavage experiments, the dose of GSE corresponded to 5.0% in the feeding experiment as calculated from the food consumption volume measured once a week in the feeding experiment.
Animals and Experimental Design
Five-week-old male F344/DuCrj rats purchased from Charles River Japan Inc. (Kanagawa, Japan) were used in the present study (Figure 1). After a 1-week acclimation period, rats were divided into 5 groups containing 10 (for feeding experiments) or 12 (for gavage experiments) animals. To clarify the dietary effect of GSE, rats were fed a diet containing 5.0% GSE or 5.0% tannic acid for 4 weeks followed by removal of the additives from the diet for 2 weeks to evaluate recovery. To avoid exposure of the chemicals to the oral cavity, rats were administered GSE by gavage for 4 weeks with a 2-week recovery period following treatment. In the control groups, rats were fed a basal diet or were administered sterilized distilled water by gavage. During the experiment, rats for gavage experiments were maintained on a pelleted basal diet CRF-1. All animals in the present study were allowed tap water ad libitum and were housed 3 to 4 animals per polycarbonate cage with sterilized softwood chips as bedding in a barrier-sustained animal room conditioned at 23 to 25°C and 50 to 60% humidity on a 12-hr light/dark cycle during the experiment. After the 4-week treatment or 2-week recovery, 5 or 6 animals in the feeding or gavage experiments, respectively, were anesthetized with isoflurane, weighed, sacrificed by exsanguination from the abdominal aorta, and then all of the salivary glands (parotid, submandibular, and sublingual) were collected.

Experimental design of the present study.
The animal protocols were reviewed and approved by the Animal Care and Use Committee of the National Institute of Health Sciences, Japan.
Histopathology and Immunohistochemistry
Collected salivary glands including the parotid, submandibular, and sublingual glands were fixed with 10% buffered formalin (Wako, Pure Chemical Indusrties,Osaka, Japan). Tissue slices of all of the salivary glands were routinely processed for paraffin embedding, and sections were prepared and stained with hematoxylin and eosin. To confirm the morphological features of the hypertrophic glandular epithelial cells in the parotid gland, periodic acid-Schiff (PAS) staining was also performed using paraffin-embedded parotid gland sections from a feeding experiment. To evaluate cell proliferative activity of the glandular epithelial cells in the parotid glands, nuclear immunoreactivity for proliferating cell nuclear antigen (PCNA) was assessed from a feeding experiment. Deparaffinized and hydrated parotid gland sections were treated with 0.3% hydrogen peroxide in absolute methanol for 15 min to block endogenous peroxidase activity, and the sections were then incubated with mouse anti-PCNA monoclonal antibody (clone PC-10, ×100 dilution; Dako Japan, Tokyo, Japan) at 4°C overnight. Immunodetection was carried out using Nichirei Histofine® Simple Stain MAX-PO (MULTI; Nichirei Biosciences Inc., Tokyo, Japan) following the manufacturer’s protocol with 3,3′-diaminobenzidine as the chromogen.
Electron Microscopy
Tissue blocks (approximately 1 mm3) of the parotid glands in 2 animals from each feed group were fixed in 2.5% glutaraldehyde/0.1 M phosphate buffer (pH 7.4) overnight at 4°C, post-fixed in 1% osmium tetroxide/0.2 M phosphate buffer (pH 7.4) for 2 hrs at 4°C, dehydrated in graded ethanol, and embedded in epoxy resin. Semi-thin sections were cut, stained with toluidine blue, and examined by light microscope to find the object field. Then ultrathin sections were prepared, double stained with uranyl acetate and lead citrate, and examined with a JEM-1400EX (JEOL Ltd., Tokyo, Japan).
Statistics
Values of body weight are expressed as the means ± SD (in Figure 2, the value of mean body weight is expressed only as the mean). Statistical analysis was performed using Pharmaco Analyst software (Three S Japan Co., Ltd., Tokyo, Japan). Values were analyzed by the Bartlett test to evaluate the homogeneity of variance. If the variances were homogenous, Student's t-test was performed. If the variances were heterogeneous, Welch's t-test was performed. For histopathological changes, incidences were compared using the Fisher’s exact probability test. Statistically significant differences (compared with the untreated controls) yielding p values less than .05 or .01 were indicated as * or **, respectively.
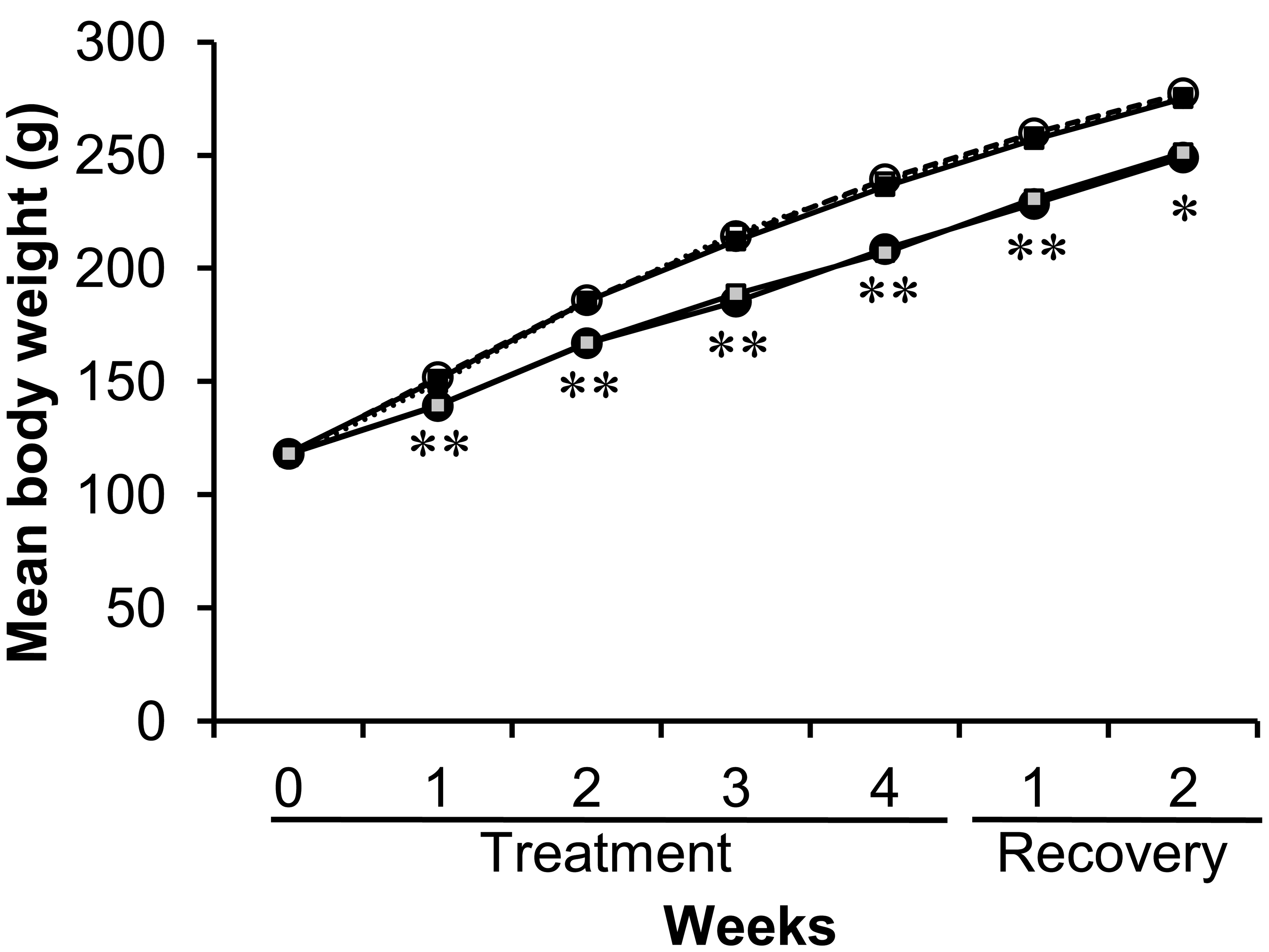
Mean weekly body weight of rats treated with GSE or tannic acid by gavage or feeding for four weeks and the following 2-week recovery period. ^ with a dotted line, control (gavage); •, GSE (gavage); □ with a dotted line, control (feeding); ▪, GSE (feeding); ░, Tannic acid (feeding). *, **: Significantly different from feed or gavage treatment controls at p < .05 and p < .01, respectively (Student’s t-test).
Results
There were no treatment-related clinical signs of toxicity including clear salivation, and all animals survived to the end of the experiment. Mean body weight during the experiment is shown in Figure 2. In the GSE gavage and TA feed groups, mean body weights were significantly lower throughout the dosing (p < .01) and recovery period (p < .05) than the corresponding control during the experimental period.
Macroscopically, enlargement of the parotid glands was observed in the GSE and TA feed groups after the 4-week treatment (Figure 3). In the other groups, including the 4-week gavage treatment and all of the recovery groups, there were no macroscopic changes in the parotid or other salivary glands. Microscopically, diffuse severe hypertrophy accompanied with basophilia without cytotoxic changes in the glandular epithelial cells of the parotid glands was observed in the rats of the GSE and TA feed groups after 4-week treatment (Table 1 and Figure 4). This change became less apparent and the severity and distribution decreased after the recovery period of the GSE and TA feed groups. In contrast, there were no such hypertrophic findings in the parotid glands with rats in the GSE gavage or control groups after the 4-week treatment. Slight basophilic change without hypertrophy was also observed focally in the parotid gland epithelium in the control and gavage treated groups except for the GSE and TA feed groups after the 4-week treatment. To further characterize the hypertrophic parotid glands in the 4-week GSE and TA feed groups, additional histopathological and ultrastructural analysis was performed. In the control feed group, PAS-positive granules were observed in the cytoplasm of parotid glandular epithelial cells (Figure 5). However, the amount of PAS-positive granules clearly decreased in the hypertrophic glandular epithelium in the GSE and TA feed groups.
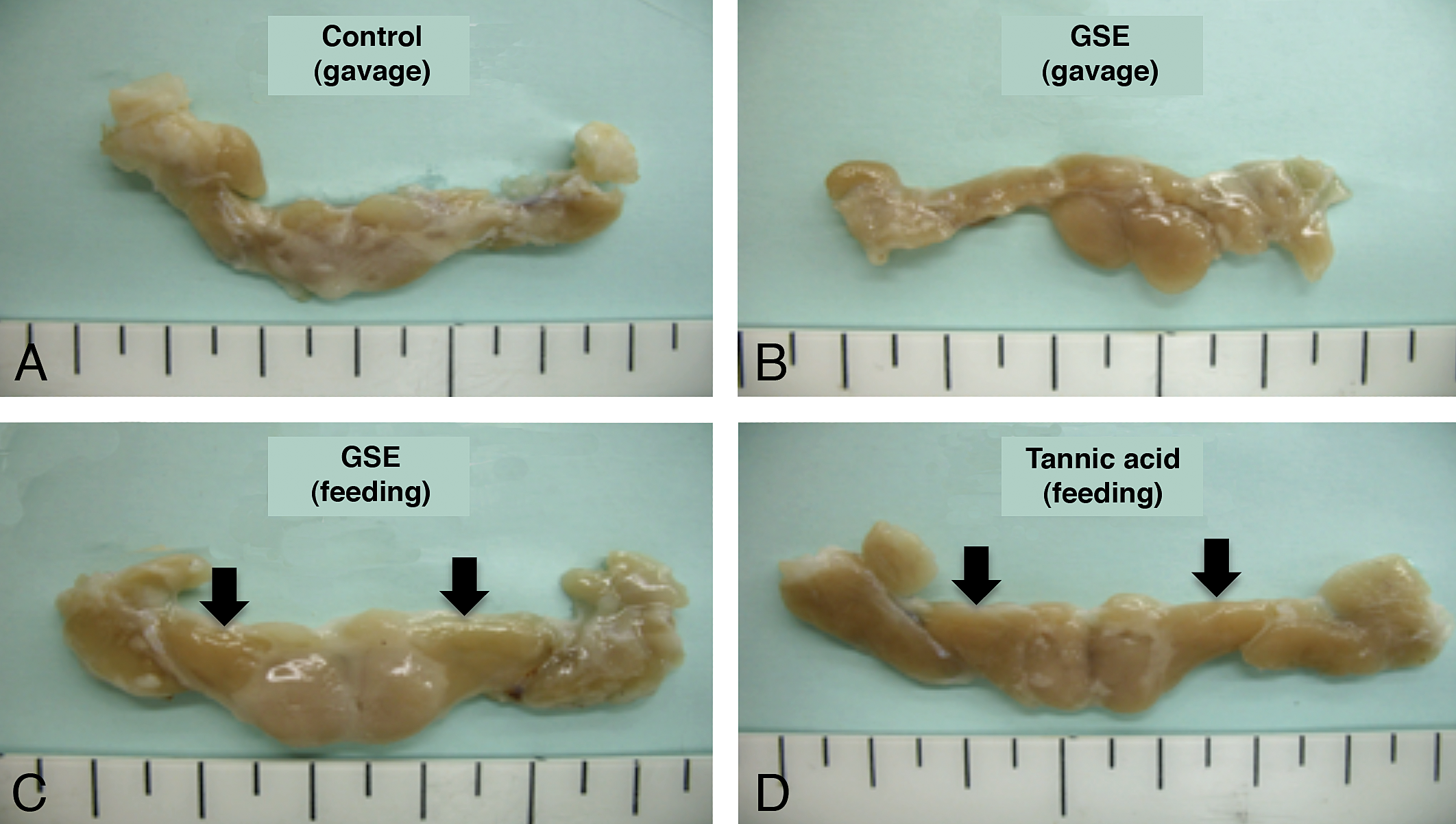
Macroscopic findings of the salivary glands in rats treated with GSE or tannic acid by feeding or gavage for 4 weeks. In the GSE or tannic acid feed groups (C) and (D), enlargement of the parotid glands (arrows) was clearly observed.
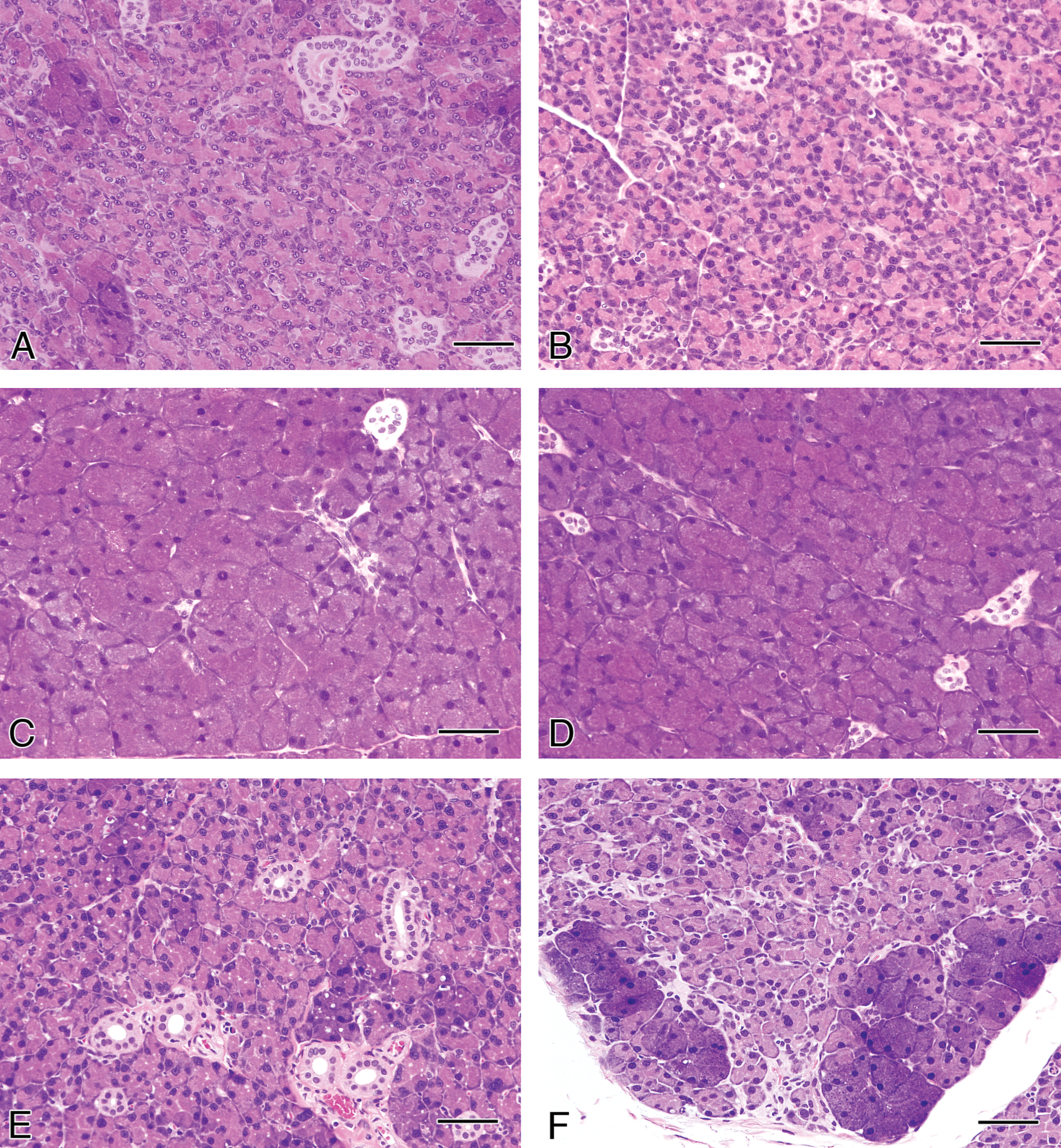
Histopathological findings in the parotid glands from rats treated with GSE or tannic acid by feeding or gavage treatment for 4 weeks or after a 2-week recovery period. Hematoxylin and eosin staining. (A) Control, (B) GSE gavage, (C) GSE feeding, (D) Tannic acid feeding, (E) recovery GSE after feeding, and (F) recovery tannic acid after feeding groups. Bars = 50 μm.
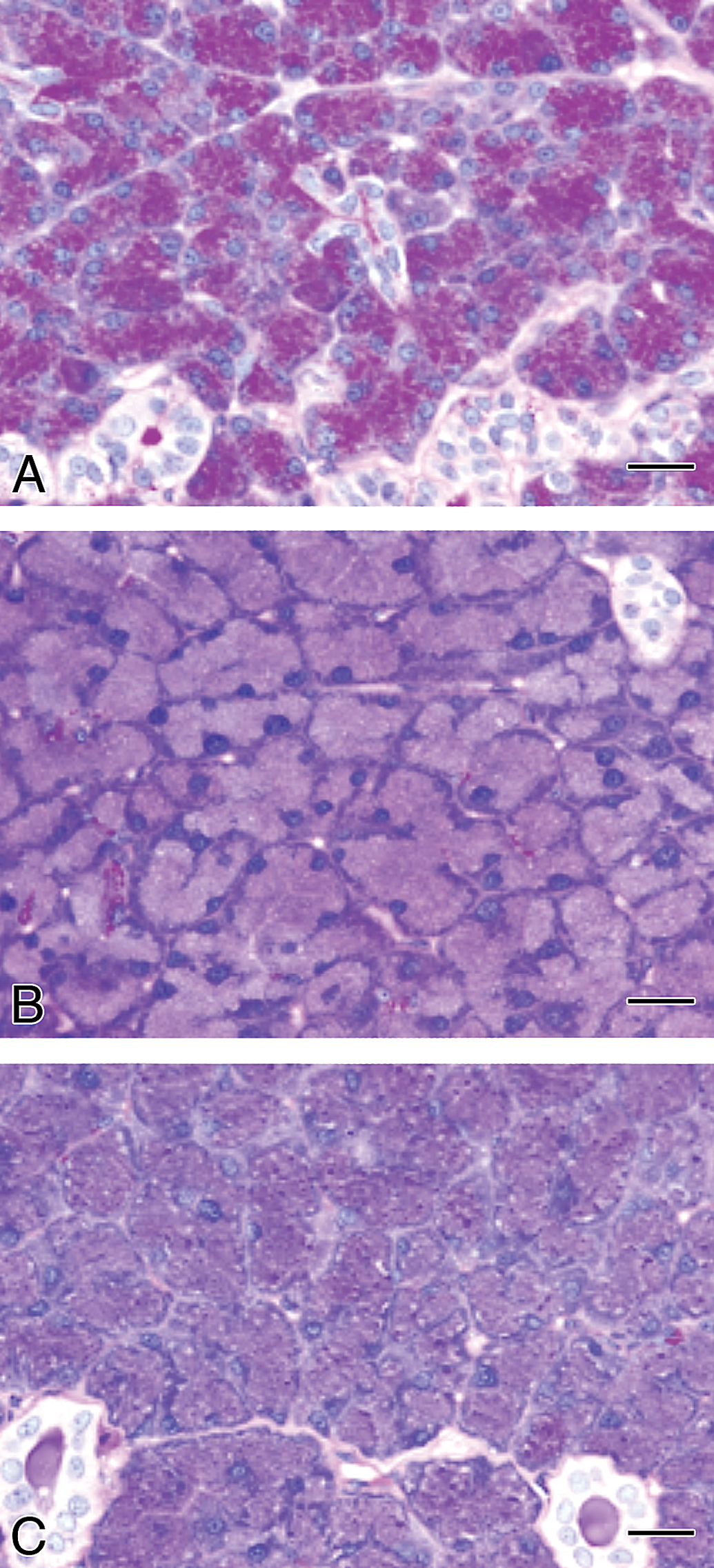
Histopathological findings in the parotid glands from rats treated with GSE or tannic acid by feeding treatment for 4 weeks. PAS staining. (A) Control, (B) GSE, and (C) tannic acid groups. PAS-positive granules in the glandular epithelial cells observed in controls clearly decreased from feeding treatment with GSE or tannic acid. Bars = 20 μm.
Microscopical changes in the epithelium of the parotid glands in F344 rats treated with GSE or tannic acid for 4 weeks.
* and ** Significantly different from the corresponding control at p < .05 or p < .01, respectively.
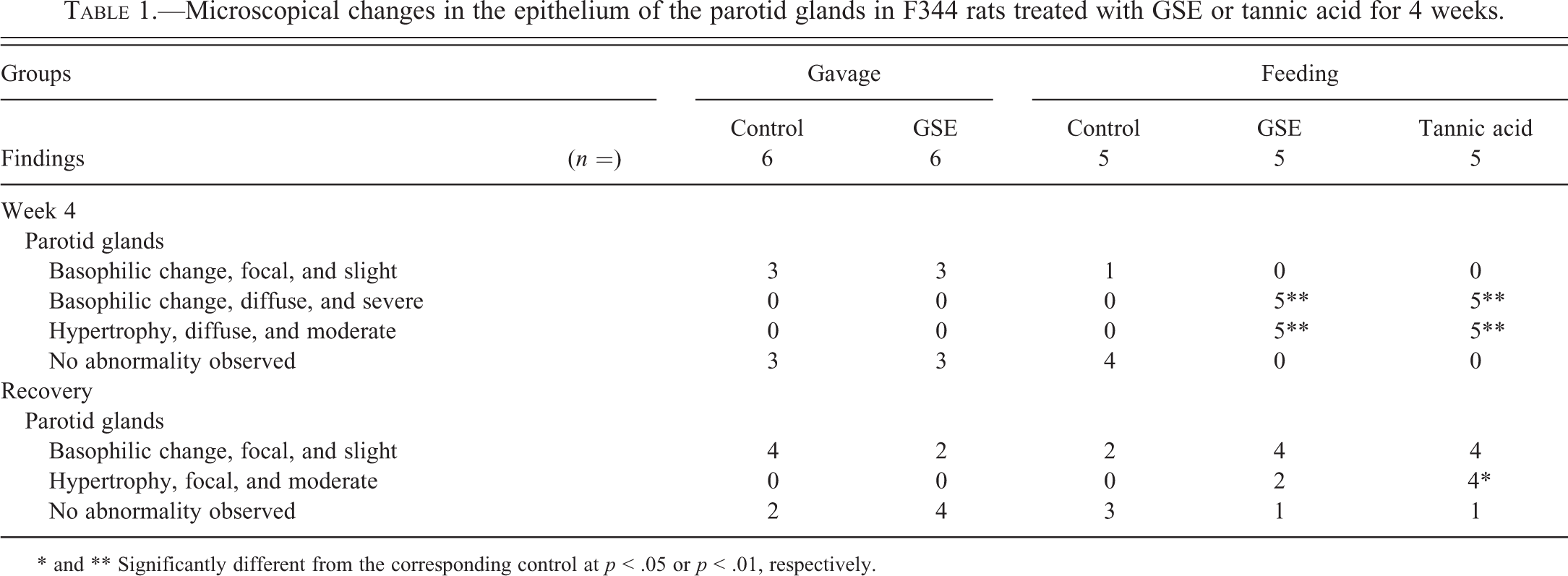
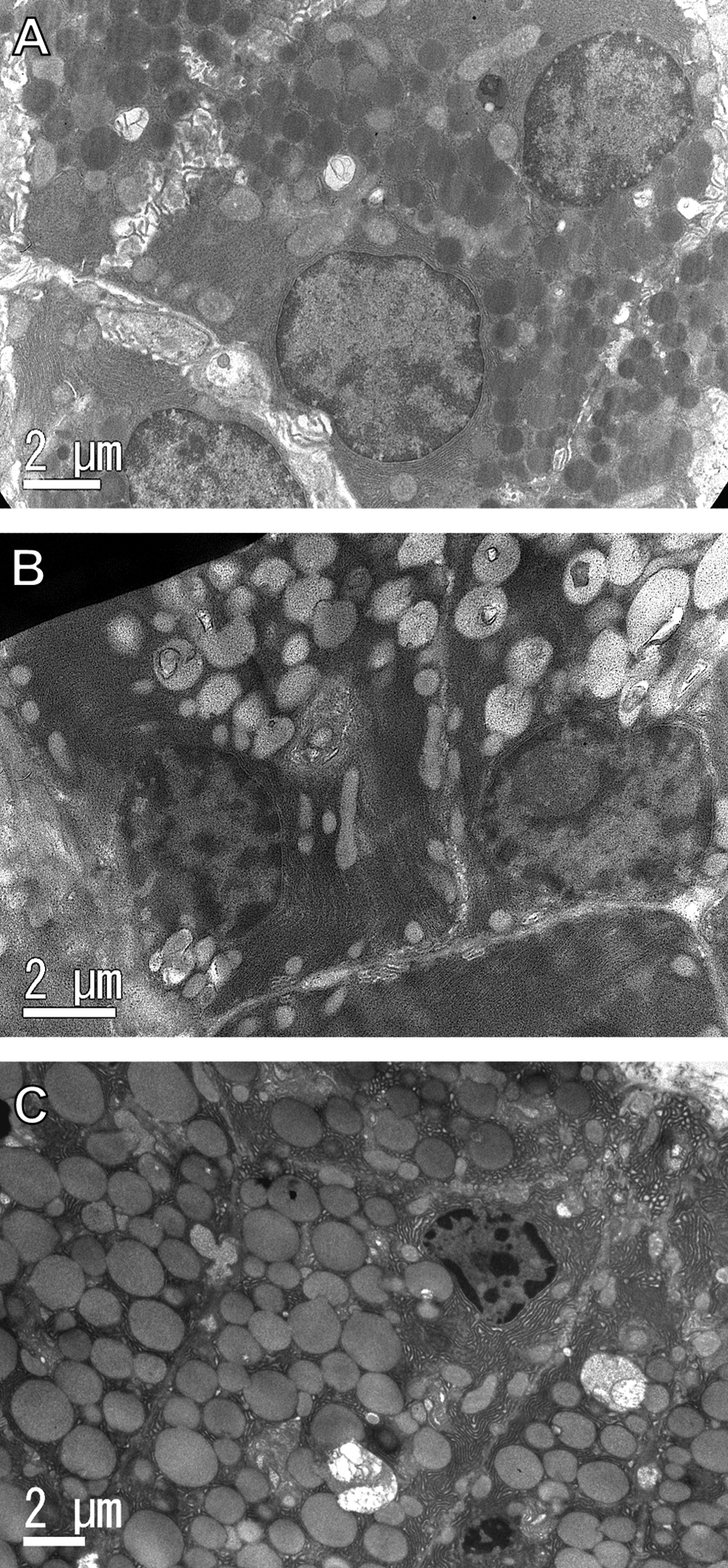
Ultrastructurally, the number and size of secretory granules appeared increased, and the electron density in the secretory granules decreased in the glandular epithelial cells of the parotid glands in the GSE and TA feed groups compared with the controls (Figures 6 and 7). In addition, rough endoplasmic reticulum appeared increased among the increased secretory granules in the GSE and TA feed groups. There was no damage or changes indicating degeneration or necrosis by GSE or tannic acid treatment in any organelles.
Ultrastructure of the glandular epithelial cells of parotid glands from rats treated with GSE or tannic acid by feeding for 4 weeks. (A) Control, (B) GSE, and (C) tannic acid groups. Number and size of secretory granules appear increased with feeding treatment with GSE or tannic acid compared with controls.
Ultrastructure of glandular epithelial cells of parotid glands from rats treated with GSE or tannic acid by feeding for 4 weeks. (A) Control, (B) GSE, and (C) tannic acid groups. In addition to the increase in the number and size and the decrease in electron density of secretory granules, the number of rER appears increased with feeding treatment with GSE or tannic acid compared with the controls. These changes were prominent with tannic acid.
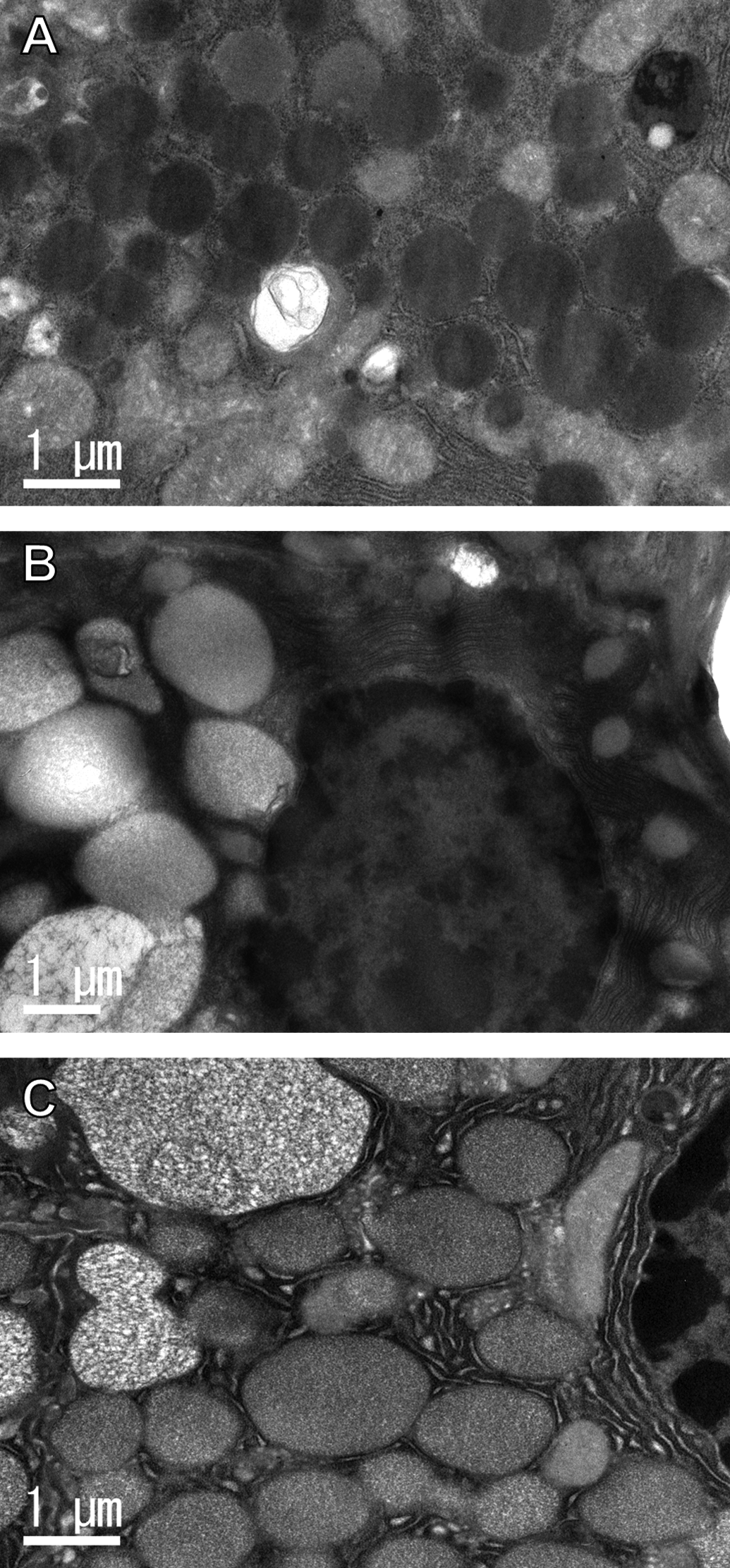
In the hypertrophic parotid glands of the GSE and TA feed groups, no PCNA-positive glandular epithelial cells were observed (Figure 8).
Immunohistochemistry for PCNA in the parotid glands of rats treated with GSE or tannic acid by feeding for 4 weeks. (A) Control, (B) GSE, and (C) tannic acid groups. In the controls, some of the nuclei in the ductular and glandular epithelial cells were positive, while hypertrophic glandular epithelial cells from the GSE or tannic acid groups were negative for PCNA. Bars = 20 μm.
Discussion
In the present study, parotid gland hypertrophy induced by repeated feeding treatment with GSE diminished after withdrawal of the treatment, and the parotid change was not observed in the gavage group under conditions that did not expose the oral cavity to GSE.
Results from the present study demonstrating that hypertrophy in the parotid gland was not induced by gavage treatment with GSE and that hypertrophy without cytotoxicity was almost completely resolved following the 2-week recovery period indicate that parotid gland hypertrophy induced by GSE is reversible and that parotid changes are caused by direct exposure of the oral cavity to GSE. Although additional experiments to examine adrenergic agonistic reactions of GSE were not performed in the present study, the results described above clearly demonstrate that parotid gland hypertrophy induced by GSE is an adaptive and reversible sialotrophic response and is dissimilar to exaggerated pharmacological or toxicological responses noted with adrenergic agonist such as isoproterenol or glyphosate, respectively. Due to the morphological character of the parotid gland and the components of GSE, some polyphenols might induce hypertrophy and basophilia in parotid glandular epithelial cells. Tannic acid and catechin, known as bitter taste components (Narukawa et al. 2011; Lesschaeve and Noble 2005; Da costa et al. 2008), also induce basophilia with or without parotid gland hypertrophy (Da costa et al. 2008 and our unpublished data). Therefore, the production and secretion of saliva might increase and the components of saliva might change after direct exposure of chemicals including astringent or bitter components in the oral cavity resulting in hypertrophy and basophilia in the glandular epithelial cells of the parotid glands. The ultrastructural findings and decreased PAS-positive granules in the parotid glands of the GSE and TA feed groups might be the evidence for production increase and component changes in saliva, respectively. It was reported that topically treated TA into the mouse mouth induced isoproterenol-induced salivary polypeptides in the saliva and parotid tissue, markers for mouse parotid hypertrophic growth, indicating that TA is one of non-catecholamine sialotrophic agents (Gho, Pena-Neira, and Lopez-Solos 2007). Therefore, changes of saliva components and parotid hypertrophy by GSE might occur in the same mechanism as TA. In the present study, the glandular hypertrophy was not observed in other salivary glands. Based on the fact that one α-amylase isoform was overexpressed in the saliva of mice treated with tannin-enriched diets which also induced parotid gland hypertrophy (Da costa et al. 2008), indicated that GSE might induce the same α-amylase isoform as tannin-enriched diets only in the parotid gland, not in the other salivary glands.
After the recovery period, a slight focal basophilic change in the parotid glands was observed in some of the animals in all of the groups including the controls. This alteration was also observed in both the gavage and feeding control groups during the treatment period. According to a published report, it is believed that basophilic foci of the parotid gland observed in rats and mice with or without chemical treatment occur spontaneously (Chiu and Chen 1986). Therefore, a slight focal basophilic change in the parotid glands observed in the present study might be spontaneous, similar to findings from the published report and our previous subchronic toxicity study of GSE (Inoue et al. 2013).
In the GSE gavage and tannic acid feed groups, a decrease in the mean body weight was observed. Generally, loss of body weight is caused by decreased food intake or from direct toxicological effects of chemicals in organs. In the present study, although the growth rates of animals in the GSE gavage and tannic acid feed groups were lower, daily food intake (g or kg body weight per day) in these groups was higher than in the control groups (data not shown), suggesting an increased nutritional requirement. Additionally, other morphological changes except for the parotid hypertrophy were not found after treatment with either GSE or TA. Therefore, the reason for body weight depression could not be determined in the present study, and changes in body weight may not be a factor in the induction of parotid hypertrophy. In case of lac color, the parotid hypertrophy did not accompany with significant body weight changes in rats treated with up to 5.0% lac color (Sakamoto et al. 2000), indicating that the parotid gland hypertrophy induced by orally treated chemicals does not always need body weight changes.
Based on macroscopic, microscopic, and ultrastructural findings consistent with parotid acinar hypertrophy and absence of cytotoxicity, the data from this and previous studies suggest that hypertrophy of the parotid glands induced by feeding treatment with GSE is an adaptive non-adverse effect that is reversible upon removal of the sialotrophic agent.
Footnotes
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Abbreviations
Acknowledgments
We thank Ms. Ayako Saikawa and Ms. Yoshimi Komatsu for their technical assistance in conducting the specimen preparation for histopathological analysis.
