Abstract
It is important to detect injection site reactions during the nonclinical phases of drug development. However, differentiating between normal changes following needle trauma and changes due to the toxicity of injected drugs can be challenging. Therefore, we used the Sprague-Dawley rat model to evaluate the pathological findings expected following a single subcutaneous injection of normal saline. Rats were subcutaneously administered with normal saline, and the injection sites were examined microscopically. Inflammation was evident in most of the injection sites, mostly in minimal severity. Parakeratosis/epithelial crust was also seen in several sites, and necrosis was observed in a minority of the cases. These findings indicate that needle puncture trauma can present with some degree of inflammation and necrosis. Although limited to a specific time point and strain, this study shows that inflammation following subcutaneous injection can be attributed in part to the needle trauma and not necessarily to the drug itself.
Injection site reactions are one of the most common adverse events seen during and after parenteral administration of different medications (Ramot et al. 2018). In some cases, the rate of injection/infusion site reactions can reach 90%, and in the more severe cases, it can lead to treatment termination (Ramot et al. 2017a). Nonclinical studies using animals are of utmost importance in detecting and evaluating injection site reactions, as they can help in predicting such effects in future use in humans and can sometimes even lead to the termination of the development program of the tested drug when the effects are considered severe (Ramot et al. 2012, Varkony et al., 2009). Therefore, efforts should be made to properly evaluate the reactions observed in the injection sites, including meticulous pathological evaluation and sometimes using imaging modalities to follow the effects over time (Maronpot et al. 2017; Ramot et al. 2017b).
An important part of the evaluation process of injection site reactions is to differentiate between the effects that are related to the trauma caused by the injection procedure itself (Ramot et al. 2016) and effects that should be attributed to the tested compound. Therefore, better understanding of the expected pathological findings following injection trauma is essential.
In this study, we utilized the Sprague-Dawley rat model to test and evaluate the pathological findings following a single subcutaneous injection of normal saline.
A total of 3 male Sprague-Dawley rats, 8-week old, were supplied by Vivo Bio Tech Ltd. (Telangana, India). The animals were singly housed in an environment-controlled room at 22°C ± 3°C with a relative humidity of 30% to 70%. There was continuous air exchange, and the room was artificially lit for 12 hr each day. The study animals were acclimatized for 5 days before treatment initiation.
The study was performed at Eurofins Advinus (Bengaluru, India), which is an Association for Assessment and Accreditation of Laboratory Animal Care International approved laboratory. Animal care practices conformed to the requirements of the Committee for Purpose of Control and Supervision of Experiments on Animals, and the study protocol was approved by the Institutional Animal Ethics Committee of Eurofins Advinus.
Each rat was given a single dose of normal saline (0.9% NaCl in water) by subcutaneous injection at 1.77 ml/kg, with the dose divided into aliquots and administered at multiple sites so that each site received approximately 0.1 ml (the last site received the remaining volume, which may have been slightly less than 0.1 ml). The dose volume and the division into aliquots of 0.1 ml/site were chosen to mimic conditions in which the test compound used in the nonclinical studies is highly viscous and absorption of the test compound is slow. Based on their body weight, two animals received 6 injections and one animal received 5 injections. The injections were given in the dorsal, cervical and thoracic regions. Hairs at the injection site were clipped before administering the injection. Each injection site was marked with an indelible ink for identification at necropsy. Suitable graduated disposable sterile plastic syringes attached with a sterile needle (26½ G) were used for each injection, and the needle remained in place for 40 s.
Rats were observed for clinical signs, including the site of injection, approximately 30 min prior to dosing and 3 hr post dose. All rats were observed for morbidity and mortality once in the morning prior to dosing and once in the evening.
Rats were maintained for approximately 24 hr after injection and then euthanized. All injection sites, including the underlying subcutis, were collected at necropsy and preserved in 10% neutral buffered formalin. Two approximately 2- to 3-mm-thick pieces of the fixed tissue samples from each injection site were processed for the routine paraffin embedding technique. Each paraffin block was then serially sectioned to yield at least 20 sections approximately 5-µm thick. These tissue sections were stained with hematoxylin and eosin stain and examined microscopically. Findings were recorded by injection sites, rather than by sections, that is, no matter how many sections contained a finding, it was recorded only once for the injection site. Findings were graded as either minimal or mild based on the 4-scale grading system (minimal, mild, moderate and severe). If a finding was present in more than one section and its grade differed, then it was recorded only once for the injection site and the highest grade was assigned. All rats survived to scheduled termination, and there were no clinical signs in any rat.
Microscopic examination of the 17 injection sites revealed findings that included inflammation (comprising mainly neutrophil infiltration), focal parakeratosis, focal epidermal necrosis, focal necrosis of the dermis, focal degeneration or necrosis of the muscularis, and hemorrhage (Table 1 and Figure 1). These findings were interpreted as secondary to traumatic injury caused by the needle puncture, partly because they were always localized (focal) to a small area and partly because they were often oriented in a line perpendicular to the skin surface. In one instance, findings clearly extended in a narrow line from the epidermis through the dermis and into the subcutis, that is, a needle tract was clearly visible.
Incidence of Injection Site Histopathologic Findings.
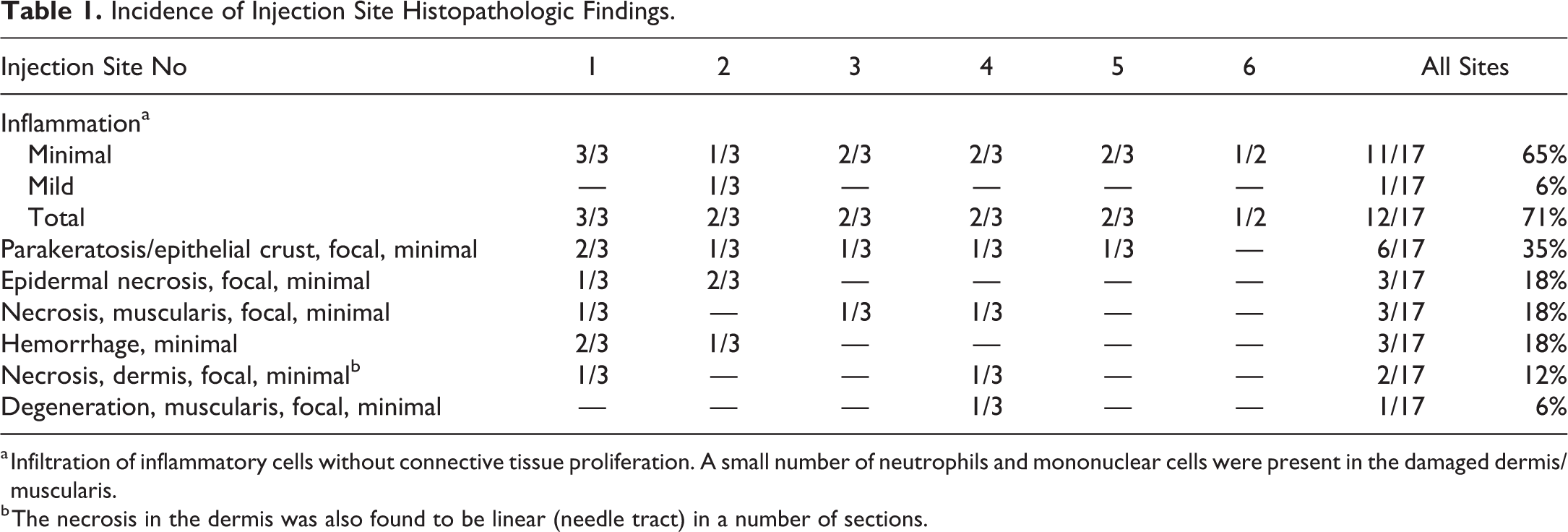
a Infiltration of inflammatory cells without connective tissue proliferation. A small number of neutrophils and mononuclear cells were present in the damaged dermis/muscularis.
b The necrosis in the dermis was also found to be linear (needle tract) in a number of sections.
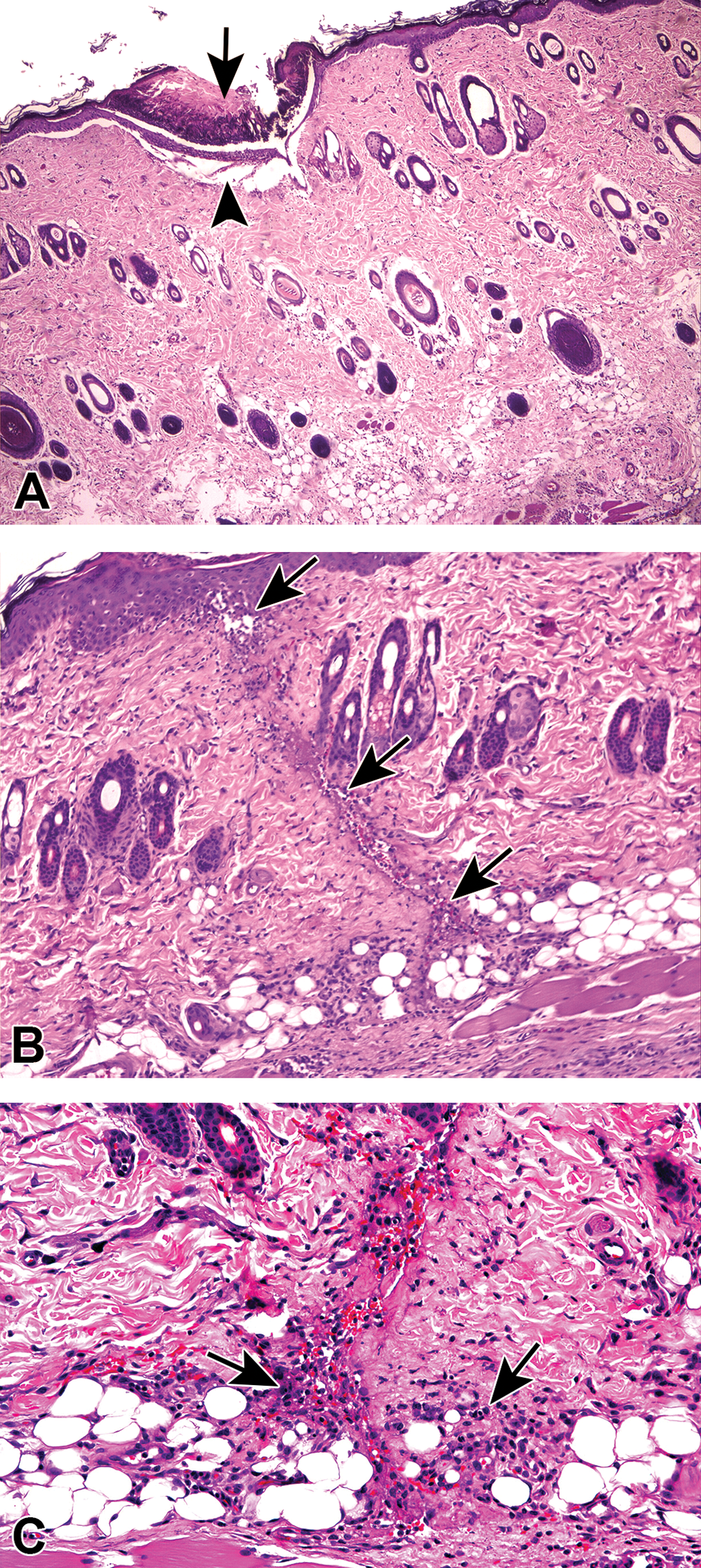
(A) Example of a skin site subjected to needle trauma injury, following single injection with saline 1.77 ml/kg, ≤0.1 ml, using a sterile needle (26½ G). The needle remained in place for 40 s. Histopathological evaluation after 24 hr demonstrated the presence of focal crust formation (long arrow), with necrosis of the underlying epidermis (short arrow). Hematoxylin and eosin. Original objective 4×. (B) Example of a skin site subjected to needle trauma injury, following single injection with saline 1.77 ml/kg, ≤0.1 ml, using a sterile needle (26½ G). The needle remained in place for 40 s. Histopathological evaluation after 24 hr revealed the presence of needle tract-associated edema and inflammation (arrows) extending from the epidermis to the subcutis. Note the perpendicular orientation of the injury from the skin surface. Hematoxylin and eosin. Original objective 10×. (C) Higher magnification of the case presented in (B), showing dermal and subcutaneous inflammation (arrows). Note the small quantity of neutrophils and mononuclear cells present in the damaged area. Hematoxylin and eosin. Original objective 20×.
When evaluating the changes that are observed in the injection site, the toxicologic pathologist often faces the challenge of differentiating between the effects that should be attributed to the tested compound and the effects that are the result of the administration process (Lilbert and Mowat 2004; Morton et al. 1997). Therefore, better understanding of the expected pathological changes following injection of normal saline, which has a very little irritation potential and is often used as a vehicle (Lilbert and Mowat 2004), could greatly facilitate such distinction in nonclinical animal studies. Such differentiation is not limited only to animals, and discerning between mechanical injury related to the injection and compound-related effects is also a challenge in clinical daily practice in humans (Hillerup, Jensen, and Ersboll 2011).
The use of a needle to inject materials subcutaneously will inevitably result in a puncture wound. The skin layers that are expected to be affected depend on the depth of injection: the epidermis and the dermis will almost certainly be involved, and in deeper injections, the subcutis and even the underlying muscle layer and nerve fascicles can also be injured (Selander, Dhuner, and Lundborg 1977). When the needle is retracted, the space that it has occupied will either collapse, with the surrounding tissue returning to its original orientation, or this space will be filled with a mixture of intracellular and extracellular fluid, the article injected, hemorrhage from adjacent damaged blood vessels, and lymph from damaged lymphatic vessels. In either case, the wound edges will be closely apposed and held relatively immobile, allowing the puncture wound to heal by first intention (Nicks et al. 2010).
Wound healing is a complex process composed of several stages. It begins with homeostasis at the location of the wound, followed by an inflammatory stage that progresses to the proliferation of the matrix and the epithelium and culminates in the formation of a scar tissue (Gupta and Kumar 2015). During this process, necrotic tissue is degraded by scavenger cells or phagocytized.
The wound healing stages can be divided according to the time following the injury (Woo, Ayello, and Sibbald 2007). Within the first hours or days, the microscopic findings during wound healing are expected to manifest microscopically as focal necrosis and inflammation, possibly with hemorrhage and early epidermal proliferation (Braiman-Wiksman et al. 2007).
The histopathologic findings at the injection sites in the current study correspond to the ones expected approximately 24 hr after subcutaneous injection of a small volume of innocuous material, consisting of inflammation, focal necrosis, hemorrhage, and epidermal insult manifesting as parakeratosis/epithelial crust (Table 1). Taken together, these observations indicate that needle puncture trauma is a “histological microwound” that follows the normal stages of wound healing.
Although limited to a specific strain, sex gender, time point, needle gauge (26½), and minimal saline volume, this study can help differentiating between trauma-induced injury and drug-induced damage in nonclinical examinations. It shows that inflammation can be expected in the site of injection in most of the cases and that normal histologic observations can also include necrosis of the different skin layers, although in a minority of the cases. Nevertheless, the use of proper controls in the context of the specific study that is being undertaken is mandatory, and the interpretation of the results should always be made based on the findings observed in the control group.
Footnotes
Author Contribution
All authors (YR, KK, SR, HK, JD, AN) contributed to conception or design; data acquisition, analysis, or interpretation; drafting the manuscript; and critically revising the manuscript. All authors gave final approval and agreed to be accountable for all aspects of work in ensuring that questions relating to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Declaration of Conflicting Interests
The author(s) declared no potential, real, or perceived conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
