Abstract
A major disadvantage that may occur in association with atorvastatin (ATV) therapy is elevation of serum transaminases. This study was designed to evaluate the effects of treatment of rats with various doses of ATV (2, 5, and 10 mg/kg/day) on liver function, oxidative stress, and histology and on the severity of acetaminophen (APAP) hepatotoxicity. ATV administration for 21 days resulted in a dose-dependent significant rise in serum activities of alkaline phosphatase, aspartate aminotransferase, and alanine aminotransferase. Only ATV at 10 mg/kg/day decreased reduced glutathione (GSH) levels and superoxide dismutase (SOD) activity, increased malondialdehyde (MDA) levels, and elicited histopathological changes in the liver. In rats challenged with APAP (500 mg/kg), the livers showed centrilobular necrosis with evident oxidative stress and liver dysfunction after 24 h. Rats challenged with APAP after pretreatment with ATV 2 or 5 mg/kg/day showed significantly lower activities of serum enzymes, higher hepatic GSH levels and SOD activities, lower MDA levels and milder histopathological changes compared with rats challenged with APAP after pretreatment with ATV 10 mg/kg/day or without drug pretreatment. In conclusion, the effect of ATV on the liver is dose dependent. Our results showed that ATV, at the highest dose used, induced hepatic lipid peroxidation and injury, suggesting a role for oxidative stress in ATV-induced hepatotoxicity. However, lower doses of ATV attenuated APAP-induced hepatotoxicity via a mechanism related, at least in part, to a reduction of APAP-induced hepatic oxidative stress. These results are of practical interest as both drugs may be used concurrently in clinical practice.
Introduction
There is a strong evidence for the therapeutic benefits of atorvastatin (ATV), a widely used 3-hydroxy-3-methylglutaryl-coenzyme A (HMG-CoA) reductase inhibitor, in the treatment of dyslipidemia with a significant reduction in morbidity and mortality in patients with coronary heart disease. 1,2 Compared with other medications in its class, ATV provides superior efficacy, probably due to prolonged inhibition of HMG-CoA reductase. 3 In addition, several recent clinical trials have shown that patients treated with high doses of ATV had better outcomes than patients treated with placebo or lower doses of ATV. 2,4 However, prolonged use of a high dose of ATV may be associated with significant increases of serum transaminases. 5,6 This change in serum indices may be followed by prolonged cholestasis, hepatitis or marked hepatic dysfunction. 7,8 ATV has thus been added to the list of medications potentially responsible for drug-related hepatotoxicity. 8,9 Therefore, in ATV-treated patients with persistent elevations in serum transaminases, the dosage should be reduced or ATV therapy should be discontinued. 10 The pathophysiology of ATV-associated liver disease is unclear. Some investigators suggest an immunoallergic mechanism, 11 while others suggest a direct toxic mechanism and that the induction of hepatic cytochrome P-450 enzyme system may be central to this adverse reaction to ATV. 12 It has also been demonstrated that coadministration of antioxidants with ATV reduces the risk of raised liver enzymes. 13
Drugs are the most frequent cause of acute liver failure and several mechanisms seem to initiate hepatic cell damage and aggravate ongoing injury processes. For example, depletion of reduced glutathione (GSH), increased production of reactive oxygen species (ROS), and enhanced lipid peroxidation have been proposed as common mechanisms involved in drug-induced hepatotoxicity. 14,15 However, the effect of ATV on the liver has not yet been well characterized, and it is unclear whether ATV-induced elevation in serum transaminases is due to hepatotoxicity associated with its use.
With this in mind, the aim of this study was to examine the effects of daily treatment with various doses of ATV on liver function, oxidative stress, and histology in rats. In addition, experiments were performed to evaluate the effect of treatment with ATV on the severity of acetaminophen (APAP)-induced acute hepatic injury in rats. This model was chosen due to the wide popularity of APAP and because several studies have identified the pivotal role of GSH in detoxifying the electrophilic reactive metabolite of APAP responsible for its hepatotoxicity. 16,17
Materials and methods
Drugs and chemicals
ATV powder (a gift from Pharo Pharma Co., Egypt) was dissolved in phosphate buffer solution (0.1 M, pH 7.4). APAP powder (Sigma Chemical Co., St Louis, Missouri, USA) was dissolved in propylene glycol. Fresh solutions were prepared at the beginning of each experiment. All the chemicals used for serum and tissue biochemical assays were of the highest analytical grade. All solutions used for enzyme assays were prepared using deionized water to eliminate trace element contamination.
Experimental animals
The experimental protocol of this study was approved by the Animal Care Committee of our institution (Medical Research Institute, Alexandria University, Egypt) and the guidelines for use and care of laboratory animals were strictly adhered to. Adult male albino rats of a local strain weighing between 100 g and 125 g were used for the study. The animals were kept under observation for at least 1 week prior to study with free access to food and water.
Animal grouping and treatment
At the time of experimentation, the rats were randomly divided into eight groups with six rats per group. All drugs (or vehicles) were given orally by gavage.
Control group: control rats received only equivalent volumes of the vehicle (phosphate buffer solution 0.1 M, pH 7.4) orally and were run in parallel.
Three ATV groups: ATV was administered orally to three groups of rats at doses of 2, 5, and 10 mg/kg/day for 21 days. The choice of the doses of ATV was based on data reported in the literature. 18,19 In man, the dose of ATV needed to decrease low-density lipoprotein cholesterol by 30% is 10 mg/day (0.15 mg/kg/day) and the maximum dose used in therapy is 1 mg/kg/day. 20,21 As pharmacokinetic studies in rodents indicate that greater statin doses are needed to achieve similar effective concentrations, 18,22 we tested, in this study, ATV doses 2-, 5- and 10-fold higher than its maximum therapeutic dose. On day 22, all rats of control and ATV groups were treated orally with propylene glycol, the vehicle of APAP.
Three ATV + APAP groups and one APAP group: three groups of rats received ATV (2, 5 and10 mg/kg/day) and a fourth group of rats received phosphate buffer solution (0.1 M, pH 7.4) for 21 days. On day 22, all rats were treated orally with APAP at a dose of 500 mg/kg. The choice of the dose of APAP was based on the results of previous studies. 23,24
In all groups, rats were fasted overnight and decapitated 24 h after the last dose of drug (or vehicle) treatment. Immediately thereafter, trunk blood was collected from each rat and serum was separated. The liver of each rat was removed, blotted from blood, and rinsed with cold saline. All samples were preserved at −80°C until analysis.
Measurements
Serum biochemical assays
Serum concentrations of alkaline phosphatase (ALP), alanine aminotransferase (ALT), and aspartate aminotransferase (AST), as indicators of hepatic injury, were measured using standard diagnostic colorimetric kits (Vitro Scient, Egypt).
Tissue biochemical assays
A portion of the kidney/liver was homogenized in 1:5 (w:v) ice-cold 5% solution of trichloroacetic acid in 1 mM disodium ethylenediaminetetraacetic acid. The resulting homogenate was used to measure the GSH content as described by Richardson and Murphy. 25 Briefly, the method is based on the reductive cleavage of 5,5′-dithio-bis-(2-nitrobenzoic acid) (DTNB) by GSH –SH group. The yellow color developed was read immediately at 412 nm and the results were expressed as micromoles of GSH per gram tissue. Another portion of the kidney/liver was homogenized in 1:9 (w:v) ice-cold 1.15% potassium chloride solution. This homogenate was used to measure the extent of lipid peroxidation by the thiobarbituric acid (TBA) method as described by Draper and Hadley. 26 Briefly, the method is based on the reaction of one molecule of malondialdehyde (MDA), the most abundant aldehyde product of lipid peroxidation, with two molecules of TBA at pH 3.5 to yield a pink chromagen that can be detected spectrophotometerically at 532 nm and the results were expressed as nanomoles of MDA per gram tissue. A third portion of the kidney/liver was homogenized in 1:9 (w:v) saline followed by centrifugation at 4000 r/min for 15 min. The clear supernatant was used to measure superoxide dismutase (SOD) activity as described by Marklund and Marklund. 27 Briefly, the method is based on the spontaneous autoxidation of pyrogallol at pH 8.2 resulting in the production of superoxide anion radicals, which in turn enhance autoxidation of pyrogallol with an increase in absorbance at 420 nm. The presence of SOD in the reaction leads to the removal of superoxide anion radicals, thereby inhibiting pyrogallol autoxidation. SOD activity of tissue homogenates was determined by calculating the difference between autoxidation of pyrogallol alone and in the presence of the tissue homogenate that contained SOD. The activity of the enzyme was expressed as units (U) per milligram protein. One unit of SOD activity is defined as the amount of the enzyme that inhibits the rate of pyrogallol autoxidation by 50%. The protein content of the tissue homogenate was measured using the Biüret method based on the ability of protein to form a colored complex with cupric ions in an alkaline medium (Vitro Scient colorimetric kit).
Histological examination
Liver specimens, excised from the rats immediately after killing, were fixed in 10% neutral phosphate-buffered formalin solution. After dehydration, paraffin-embedded hepatic sections (4 µm) were stained with hematoxylin and eosin. Hepatic sections were evaluated for the presence of central vein (CV) congestion, hepatocyte vaculation, cell necrosis, and hepatic fibrosis.
Statistical analysis
The results are presented as mean ± SEM Computer package SPSS 9.0 was used for data management. Analysis of variance (one-way classification) and Scheffé’s multiple comparison procedure were used for the statistical comparisons between groups. Pearson’s test was used for the calculation of the correlation coefficient (r) to evaluate the correlation between variables. Values of p < 0.05 were considered statistically significant.
Results
Effect of ATV treatment with or without an acute challenge with APAP on serum marker enzymes
The effect of ATV on liver function was assessed, in this study, by measuring serum ALP, ALT, and AST activities (Table 1). AST and ALT activities were significantly increased (p < 0.05) compared with control values in rats treated with the smallest dose of ATV (2 mg/kg/day) for 21 days. The serum activity of ALP, in the same group of rats, did not show a similar significant change. In contrast, serum activities of the three biochemical indices were significantly increased (p < 0.05) in both ATV groups treated with 5 and 10 mg/kg/day. In the latter group, serum ALP, AST, and ALT activities were significantly higher than the corresponding values in rats treated with 2 and 5 mg/kg/day. Using the results of the three experimental groups treated with ATV, significant positive correlations were detected between the means of the drug doses administered daily to the rats for 21 days and serum activities of each of ALP (r = 0.786, p < 0.001), AST (r = 0.799, p < 0.001), and ALT (r = 0.800, p < 0.001).
Effect of chronic ATV treatment with or without an acute challenge with APAP (500 mg/kg) on serum activities of ALP, AST and ALT in rats.a

ATV: atorvastatin; APAP: acetaminophen; ALP: alkaline phosphatase; AST: aspartate aminotransferase; ALT: alanine aminotransferase.
aValues shown are mean ± SEM with six rats per group.
b p < 0.05: versus control group.
c p < 0.05: versus ATV 2 mg and 5 mg/kg/day groups.
d p < 0.05: versus all other groups except ATV (10 mg/kg/day) + APAP group.
e p < 0.05: versus the corresponding ATV alone and APAP groups.
f p < 0.05: versus (2 mg/kg/day + APAP) and (5 mg/kg/day + APAP) groups.
As shown in Table 1, serum activities of ALP, AST, and ALT were significantly increased (p < 0.05) in all experimental groups treated with APAP compared with the control group. In rats challenged with APAP after pretreatment with ATV at a dose of 2 or 5 mg/kg/day for 21 days, serum activities of ALP, AST, and ALT were significantly lower (p < 0.05) than the corresponding values in the groups of rats challenged with APAP without prior ATV treatment and with ATV 10 mg/kg/day pretreatment. No significant differences were detected between the latter two groups with respect to the values of serum ALP, AST, and ALT activities (p > 0.05).
Effect of ATV treatment with or without an acute challenge with APAP on hepatic GSH levels
Figure 1 demonstrates hepatic GSH levels after daily treatment of rats with ATV for 21 days with or without an acute challenge with a large dose of APAP. Hepatic GSH levels in rats treated with ATV at a dose of 2 or 5 mg/kg/day did not show any significant change (98% and 91% of control values, respectively, p > 0.05). In contrast, treatment with ATV 10 mg/kg/day caused a significant reduction in hepatic GSH levels to 79% of control levels (p < 0.05). In rats challenged with APAP without prior ATV treatment, hepatic GSH levels were markedly diminished to 52% of control values. In rats challenged with APAP after ATV pretreatment with 2, 5, and 10 mg/kg/day, hepatic GSH levels were found to be significantly decreased to 76%, 69%, and 60% of control values, respectively, (p < 0.05). No significant difference was detected between the APAP group and the group challenged with APAP after pretreatment with ATV 10 mg/kg/day. In contrast, hepatic GSH levels, in the other two groups pretreated with ATV 2 and 5 mg/kg/day, were significantly higher (p < 0.05) than in APAP-challenged rats without drug pretreatment (Figure 1).

Effect of daily treatment with ATV with or without an acute challenge with APAP on hepatic GSH levels in rats. Values shown are mean ± SEM with six rats per group. *p < 0.05: versus control group; # p < 0.05: versus (APAP 500 mg/kg) group; $ p < 0.05: versus (ATV 2 mg/kg/day + APAP) group. ATV: atorvastatin; APAP: acetaminophen; GSH: glutathione.
Effect of ATV treatment with or without an acute challenge with APAP on hepatic SOD activities
The changes in hepatic tissue SOD activity in rats treated with ATV with or without APAP challenge are depicted in Figure 2. Treatment with ATV at a dose of 10 mg/kg/day significantly decreased hepatic SOD activity to 86% of control values (p < 0.05). No significant change in this parameter was observed in rats treated with ATV 2 or 5 mg/kg/day (96% and 91% of control values, respectively). As shown in Figure 2, the activity of hepatic SOD decreased significantly (p < 0.05) in all groups challenged with APAP. In rats challenged with APAP without prior ATV treatment, hepatic SOD showed the greatest reduction in activity (66% of control values, p < 0.05). Treatment with ATV at doses of 2 and 5 mg/kg/day before the acute challenge with APAP significantly improved this decrease in hepatic SOD activity (80% and 76% of control values, respectively), p < 0.05. In contrast, rats treated with ATV at a dose of 10 mg/kg/day before APAP challenge did not show a significant similar improvement in SOD activities as compared to rats challenged with APAP without ATV pretreatment.

Effect of daily treatment with ATV with or without an acute challenge with APAP on hepatic SOD activity in rats. Values shown are mean ± SEM with 6 rats per group. *p < 0.05: versus control group; #
p < 0.05: versus (APAP 500 mg
Effect of ATV treatment with or without an acute challenge with APAP on hepatic MDA levels
The effect of ATV treatment with or without an acute APAP challenge on hepatic tissue MDA levels is demonstrated in Figure 3. Hepatic MDA levels in rats treated with ATV at doses of 5 and 10 mg/kg/day were significantly higher (p < 0.05) than the corresponding values in control rats (119% and 130% of control values, respectively). However, rats treated with the smallest dose of ATV (2 mg/kg/day) did not show a similar significant change in hepatic MDA levels (108% of control values, p > 0.05). In rats challenged with APAP without drug pretreatment or after treatment with ATV at a dose of 10 mg/kg/day, hepatic MDA levels were similarly and significantly increased compared with levels in control rats (182% and 181% of control values, respectively, p < 0.05). Treatment of rats with the lower doses of ATV (2 or 5 mg/kg/day) before the challenge with APAP significantly attenuated (p < 0.05) this increase in hepatic MDA levels (148% and 154% of control values, respectively).

Effect of daily treatment with ATV with or without an acute challenge with APAP on hepatic MDA levels in rats. Values shown are mean ± SEM with 6 rats per group. *p < 0.05: versus control group; # p < 0.05: versus (APAP 500 mg/kg) group; $ p < 0.05: versus (ATV 2 mg/kg/day + APAP) and (ATV 5 mg/kg/day + APAP) groups. ATV: atorvastatin; APAP: acetaminophen; MDA: malondialdehyde.
Effect of ATV treatment with or without an acute challenge with APAP on liver histology
Histological examination of liver sections from rats after daily treatment for 21 days with different doses of ATV revealed dose-dependent changes (Figure 4). Compared with normal liver architecture (Figure 4(a)), ATV treatment at a dose of 2 mg/kg/day resulted in minimal congestion of CVs and dilated sinusoids (Figure 4(b)). After a dose of 5 mg/kg/day, mild congestion and inflammatory changes in portal tracts with ballooning degeneration of some hepatocytes were detected (Figure 4(c)). After a dose of 10 mg/kg/day, coagulative necrosis and vacuolization were evident in many hepatocytes (Figure 4(d)).

Photomicrographs of rat liver sections after daily treatment with different doses of ATV for 21 days (H&E, bar = 50 µm). (a) Photomicrograph of a liver section from a control rat showing normal liver architecture. (b) An ATV (2 mg/kg/day)-treated rat liver section showing minimal CV congestion and dilated sinusoids (+). (c) An ATV (5 mg/kg/day)-treated rat liver section showing mild congestion and inflammation in PT area with ballooning degeneration (→) in some hepatocytes. (d) An ATV (10 mg/kg/day)-treated rat liver section showing coagulative necrosis (▴) and vacuolization (v) in many hepatocytes. ATV: atorvastatin; CV: central vein; PT: portal tract; H&E: hematoxylin and eosin.
As shown in Figure 5, examination of liver sections from rats treated with APAP at a dose of 500 mg/kg demonstrated the characteristic centrilobular hepatic necrosis with evident nuclear disintegration of hepatocytes and cellular infiltration around the CV (Figure 5(a)). However, examination of liver sections from rats challenged with APAP after ATV pretreatment at a dose of 2 or 5 mg/kg/day revealed mild congestion and cellular infiltration in the portal tract area (Figure 5(b) and (c)). These changes were less marked than in rats challenged with APAP without ATV pretreatment (Figure 5(a)). In rats challenged with APAP after treatment with ATV at a dose of 10 mg/kg/day, the extent of hepatic damage was more severe with ballooning degeneration of most hepatocytes (Figure 5(d)) compared with rats pretreated with the smaller doses of ATV.
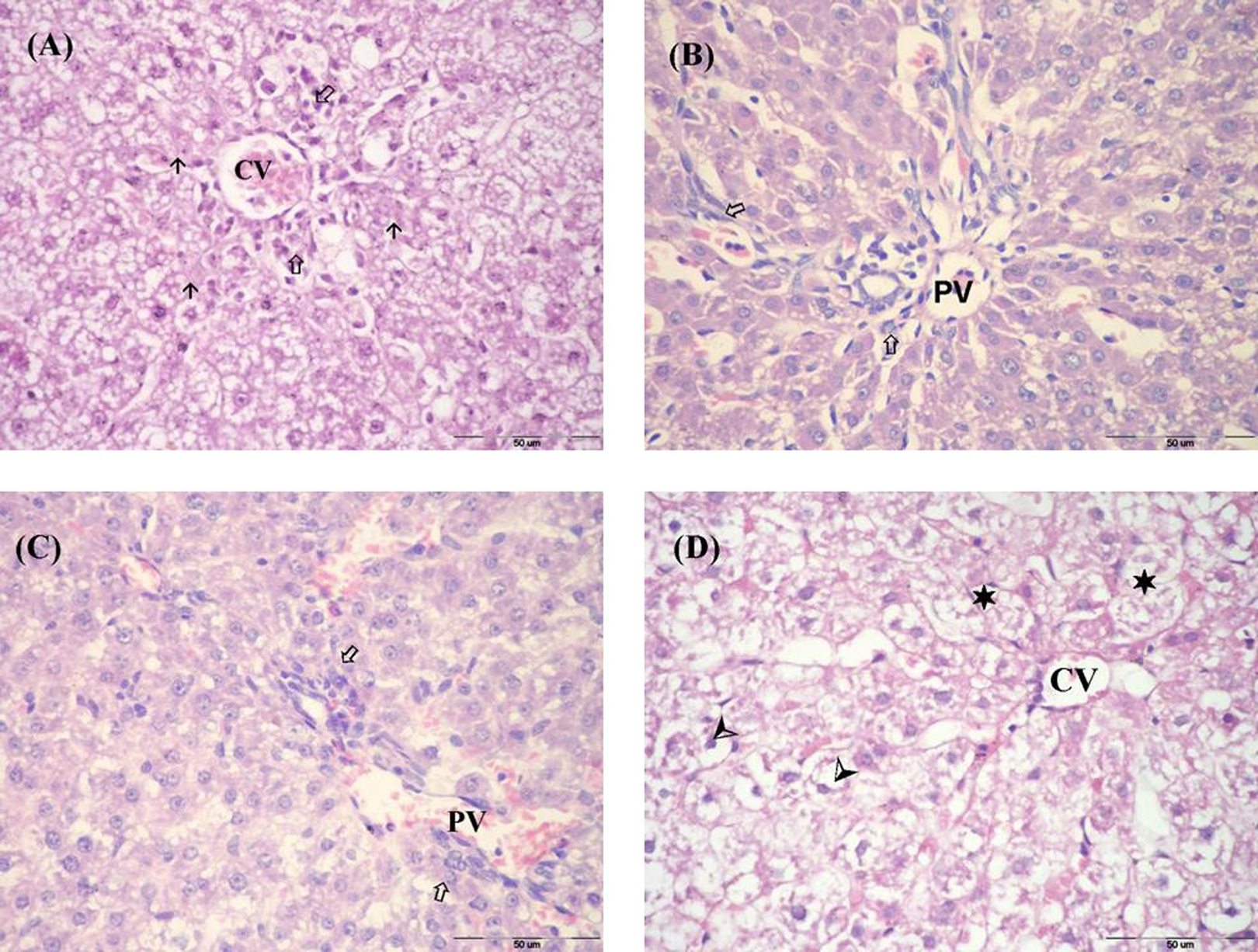
Photomicrographs of liver sections of rats challenged with APAP (500 mg/kg) after daily treatment with different doses of ATV for 21 days (H&E, bar = 50 µm). (a) A liver section from a control rat challenged with APAP showing centrilobular necrosis (↑), cellular infiltration (▴) and CV congestion. (b) A liver section from a rat pretreated with ATV (2 mg/kg/day) before the challenge with APAP showing mild congestion and cellular infiltration (▴) around PV branches. (c) A liver section from a rat pretreated with ATV (5 mg/kg/day) before the challenge with APAP showing congestion in PV branches and cellular infiltration (▴) in the portal tract area. (d) A liver section from a rat pretreated with ATV (10 mg/kg/day) before the challenge with APAP showing cloudy swelling (*) and ballooning degeneration (→) in most hepatocytes. APAP: acetaminophen; ATV: atorvastatin; CV: central vein; PV: portal vein; H&E: hematoxylin and eosin.
Discussion
The results of this study show that daily treatment with a high dose of ATV induced hepatic oxidative stress, histopathological changes and a marked increase in serum biochemical indices of liver injury. Our findings, also, show that treatment with low doses of ATV exerted a partial hepatoprotective effect in a model of acute liver injury induced by a large dose of APAP.
There is increasing evidence that hepatotoxicity is a potential adverse effect of a large number of drugs. 6,28,29 More and more evidence has accumulated suggesting that ROS produced during drug metabolism or by metabolites may be important in the development of drug-induced hepatotoxicity. 14,15 The potential harmful effects of these species are controlled by the endogenous cellular antioxidant defense system. 30,31 GSH is the major nonenzymatic antioxidant in cells that guards against oxidative injury by reducing hydrogen peroxide (H2O2) and scavenging ROS and nitrogen species. 15,32 SOD is one of the important enzymatic antioxidants in cells and is considered the first-line of defense against oxygen free radicals as it catalyzes the breakdown of superoxide anions into H2O2. 33 When the generation of ROS in cells impairs antioxidant defenses or exceeds the ability of the antioxidant defense system to eliminate them, oxidative stress results leading to tissue injury. 34,35
ATV is a widely used drug with proven therapeutic benefits in the treatment of dyslipidemia. 2,6,10 However, independent of its cholesterol-lowering properties, a significant elevation in serum transaminases (greater than three times the upper limit of normal) was observed in patients involved in clinical studies with ATV. 4,6 In this study, a significant rise in serum biochemical indices of liver dysfunction (ALP, AST, and ALT) was observed in rats treated with ATV. This effect was dose dependent as indicated by the significant correlations between the administered doses of ATV and serum ALP, AST, and ALT activities. These findings are consistent with other reports that the risk of a significant rise in serum transaminases while using statins, and in particular ATV, is dose dependent. 10,36 The observed rise in serum enzymes was, most probably, due to hepatotoxicity associated with ATV use, particularly at its high dose, and not merely due to greater leakage of cellular enzymes secondary to the drug effect on the structure of hepatic cellular membranes. 7 This interpretation is supported by the findings, in this study, that daily treatment with the high dose of ATV (10 mg/kg/day) decreased hepatic GSH levels and SOD activities, increased hepatic MDA levels, and elicited prominent changes in liver histology. These results indicate that ATV, at the highest dose used, increased lipid peroxidation and produced tissue injury in the liver, suggesting a role for oxidative stress in ATV hepatotoxicity.
The great interest in lipid peroxidation stems primarily from the observation that increasing lipid peroxidation is particularly important not only as this indicates higher levels of oxidative stress but also because the biological products of lipid peroxidation can be quite reactive and, therefore, damaging to the tissues. For example, MDA, as a product of lipid peroxidation, can cause further oxidative tissue injury as it has the capability to interact with and bind to protein molecules, potentially rendering a vital protein to be nonfunctional. 37 In addition, MDA can induce oxidative stress by targeting mitochondrial complexes and thereby disrupting the proper flow of electrons through the electron transport chain with a subsequent increase in ROS generation. 38 Profound GSH depletion, especially in the mitochondria, can sensitize to further stress on mitochondria resulting in oxidative tissue injury. For example, a large body of evidence indicates that APAP hepatotoxicity, the leading cause of acute liver failure in the Western countries including the United States and the United Kingdom, 39,40 is mediated by its metabolism by the hepatic cytochrome P-4502E1 to an electrophilic metabolite, N-acetyl-p-benzoquinoneimine, which attacks and covalently binds to cellular proteins. 23,41 Following low doses of APAP, this metabolite is efficiently detoxified by GSH and can, upon over dosage, cause hepatic GSH depletion, lipid peroxidation, and centrilobular necrosis. 17
In this study, the livers of rats treated with APAP at a dose of 500 mg/kg showed the characteristic centrilobular hepatic necrosis 24 h after drug administration. This toxic effect was associated with a marked increase in serum biochemical indices of liver dysfunction (ALP, AST, and ALT activities) and changes in hepatic oxidative stress markers (a significant decrease in hepatic GSH levels and SOD activities, and a significant increase in MDA levels). These results are in agreement with those previously reported by other investigators who studied APAP-induced hepatotoxicity in experimental animals. 17,23,24
Interestingly, APAP-induced hepatic dysfunction, oxidative stress, and histological changes were significantly attenuated, in our study, in rats pretreated with the lower doses of ATV. Rats challenged with APAP after daily treatment with ATV at a dose of 2 or 5 mg/kg/day for 21 days showed significantly lower activities of serum ALP, AST, and ALT, significantly higher hepatic GSH levels and SOD activities, and significantly lower MDA levels compared with rats challenged with APAP without ATV pretreatment. This partial protective effect was confirmed by the histological findings of more severe hepatic necrosis in the APAP group (Figure 5(a)) compared with rats pretreated with ATV at a dose of 2 or 5 mg/kg/day prior to the challenge with APAP (Figure 5(b) and (c)). A similar effect was not observed in rats treated with the high dose of ATV (10 mg/kg/day) before the challenge with APAP (Figure 5(d)). In the latter group, the values of serum ALP, AST, and ALT activities and hepatic SOD activities and GSH and MDA levels were close to the corresponding values observed in rats challenged with APAP without drug pretreatment.
Our study findings that treatment of rats with low doses of ATV effectively reduced APAP hepatotoxicity may be due to more than one mechanism. Since oxidative stress and inflammation are involved in the pathogenesis of APAP hepatotoxicity, 14,16 the free radical scavenging activity of the o-hydroxy and the p-hydroxy metabolites of ATV 42 and/or the direct anti-inflammatory effect of low doses of ATV 43,44 may explain, at least in part, the hepatoprotective effect of ATV low doses against APAP hepatotoxicity observed in this study. However, pretreatment of rats with the high dose of ATV (10 mg/kg/day) failed to reduce the severity of APAP hepatotoxicity. The present results showed that this high dose of ATV per se induced hepatic oxidative stress with evident hepatocellular necrosis at the end of the treatment period. As a result of this effect, the capacity of the liver to form the reactive toxic metabolite of APAP would be expected to be reduced in rats challenged with APAP after pretreatment with this high dose of ATV. Thus, the severity of hepatic injury observed in this group of rats was dependent on both the severity of hepatic damage induced by the high dose of ATV and the degree of impairment of formation of APAP toxic metabolite. Further studies powered to examine the effect of treatment with high doses of ATV on APAP hepatic metabolism would be of interest.
Thus, the major findings of this study suggest that oxidative stress plays a role in the pathophysiology of ATV-induced hepatotoxicity and the results demonstrate a dose-dependent effect of ATV on the liver. Daily treatment of rats with a high dose of ATV induced hepatic oxidative stress, cellular injury, and dysfunction. In contrast, low doses of ATV reduced the hepatic oxidative stress and tissue injury induced by APAP. The importance of the high-dose results to humans is unclear because the daily high dose of ATV administered to the rats was 10-fold higher than its maximum daily therapeutic dose. 20,21 However, the results of the effects of ATV low doses are not without practical interest with regard to the hepatotoxic potential of APAP as both drugs may be used concurrently in clinical practice, in part, because of the availability of APAP alone and in a number of combinations of prescription and non-prescription drugs and, in part, because of the widespread use of ATV in the treatment of dyslipidemia.
Footnotes
Conflict of interest
The authors declared no conflicts of interest.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
