Abstract
Paracetamol overdose is common and microRNA (miR)-122 expression is increased with liver injury. We aimed to measure miR-122 in the setting of an abbreviated paracetamol overdose treatment regimen. We compared miRNA expression in patients treated for paracetamol poisoning with an abbreviated 12-h intravenous acetylcysteine regimen (200 mg/kg over 4 h, 50 mg/kg over 8 h) or a 20-h regimen (200 mg/kg over 4 h, 100 mg/kg over 16 h) (NACSTOP trial). miR-122 expression is increased (decreased cycle threshold (Ct) values) with paracetamol liver injury. We assessed miR-122 expression in patients receiving the two acetylcysteine regimens and in a separate group with acute liver injury (ALI). We examined 121 blood samples in 38 patients. After 20 h of acetylcysteine, median alanine transaminase (ALT) was 12 U/L (18, 14) versus 16 U/L (11, 21) (p = 0.17) and median miR-122 Ct was 30.1 (interquartile range (IQR): 28.9, 33.3) versus 31.4 (28.9, 33.9) (p = 0.7) in the NACSTOP abbreviated and control groups, respectively. Median normalized miR-122 Ct after 20 h of acetylcysteine was 2.2 (IQR 1.9, 6.4), 1.1 (0.7, 2.9), 63.9 (2.5, 168), 123.2 (40.9, 207.8) in the NACSTOP-abbreviated, NACSTOP-control, ALI and hepatotoxicity groups, respectively. There was no significant difference in ALT or miRNA between NACSTOP treatment groups and no signal of increased liver injury from an abbreviated 12-h acetylcysteine regimen. These findings suggest that an abbreviated acetylcysteine regimen in low-risk patients who have overdosed on paracetamol is safe. Further study is required to validate this finding utilizing miRNA as a comparative biomarker.
Introduction
Paracetamol overdose is the most common pharmaceutical poisoning in developed countries, and the incidence is increasing in the developing world. 1 It accounts for 50% of overdoses in the United Kingdom alone (100,000 hospitals presentations per annum) and is the most frequent reason for calls to poison information centres in Australia. 2,3 Notably, paracetamol overdose is the most common cause of acute liver failure in developed countries. 4
Acetylcysteine is the antidote used to treat patients at risk of developing hepatotoxicity following paracetamol overdose. When intravenous (IV) acetylcysteine was first administered in the 1970s, 5 a 20.25-h infusion duration was selected as this represented five-elimination half-lives of paracetamol, assuming an elimination half-life of 4 h. This was during a time when serum paracetamol concentration assay was not widely available. Serial testing of paracetamol concentration and liver function tests is now routinely possible and can provide reassurance that hepatic function is preserved when paracetamol concentration has decreased. A two-bag acetylcysteine regimen consisting of 200 mg/kg infused over 4 h followed by a further 100 mg/kg infused over 16 h is associated with a decreased adverse reaction rate to acetylcysteine compared to the three-bag regimen. 6,7 The NACSTOP trial recently evaluated the safety of ceasing this two-bag acetylcysteine regimen after 12 h of treatment when the paracetamol concentration was low (<132 μmol/L or <20 mg/L) and the alanine aminotransferase (ALT) was normal (<40 IU/L) 8 (i.e. individuals with low risk of developing subsequent liver injury) and compared this to a group receiving the full 20-h acetylcysteine treatment. Neither group developed acute liver injury (ALI) when measured by ALT. Other studies have also evaluated shorter acetylcysteine regimens after paracetamol poisoning. 9,10
MicroRNAs (miRNAs or miR) are small non-coding molecules found in plants, invertebrates and mammals that function in post-transcriptional regulation of gene expression. 11 In addition to intracellular functions, miRNAs are secreted by cells into body fluids 12 and are released after organ injury. 13 Specifically, serum concentration of miR-122 increases with liver injury and is more sensitive and specific in detecting this compared to ALT. Conversely, miR-483 expression decreases or remains unchanged with liver injury. 13 ALT is present in hepatocytes but is also found in skeletal and cardiac muscle cells. Therefore, injury to cardiac or skeletal muscle can also cause an increase in ALT. The combination of these two miRNAs has been proposed as a better predictor of liver injury than miR-122 alone or ALT. 13,14 However, there are no studies examining the utility of miRNAs to compare acetylcysteine treatment regimens.
The aim of this study was to investigate the degree of liver injury and corresponding miRNA concentrations after administration of the standard two-bag 20-h compared to an abbreviated 12-h acetylcysteine regimen used to treat individuals at low risk of paracetamol overdose.
Methods
Study population
A cohort of subjects treated with IV acetylcysteine following single or staggered ingestion paracetamol overdose was enrolled. All subjects during the period from February 2015 to July 2016 were recruited from the NACSTOP trial (Registration ACTRN12615000938505, Australian New Zealand Clinical Trials Registry), 8 from three emergency departments at Monash Health in Victoria, Australia, in a cluster-controlled study design. Patients with a normal ALT (<40 U/L) at presentation and who required acetylcysteine were included in the trial. Patients were treated with IV acetylcysteine if meeting criteria according to the Australian and New Zealand paracetamol treatment guidelines, 15 for example, treatment given for paracetamol concentrations above the nomogram line for single ingestions (1000 μmol/L [150 mg/L] at 4 h post overdose). Those in the intervention group with a low paracetamol (<132 μmol/L or <20 mg/L) and normal ALT after 12 h of acetylcysteine had their acetylcysteine ceased. The control group received the full 20-h course of acetylcysteine (200 mg/kg over 4 h, 100 mg/kg over 16 h), while the intervention group received an abbreviated 12-h acetylcysteine regimen (stopped after at least 12 h of acetylcysteine that is, 200 mg/kg over 4 h, followed by at least half of the 100 mg/kg over 16 h infusion).
Excluded patients included those <16 years old, pregnancy, abnormal ALT on presentation (<40 IU/L), unknown time of ingestion and modified-release paracetamol overdose. Modified-release paracetamol ingestions were excluded because of erratic absorption and delayed peak concentrations of paracetamol. The study protocol conformed to the ethical guidelines of the 1975 Declaration of Helsinki as reflected in a priori approval by the Monash Health Research Ethics Committee. All subjects consented to involvement in the NACSTOP study and subsequent miRNA analysis. A more detailed description of NACSTOP enrolment has been described previously. 8
The primary endpoint of this study was the detection of ALI at 20 h post-initiation of acetylcysteine. ALI was defined as >50 IU/L for this study. Hepatotoxicity was defined as an ALT > 1000 IU/L.
In addition, two comparator groups of patients not involved in the NACSTOP trial were included. These included patients with ALI or hepatotoxicity secondary to paracetamol overdose. Patients were consented for post-treatment analysis of miRNA.
Therefore, the four groups that were studied included: NACSTOP patients (i) abbreviated treatment (12 h) regimen, (ii) full treatment (20 h) regimen (control group) and non-trial patients, (iii) developed ALI or (iv) developed hepatotoxicity. Those developing ALI or hepatotoxicity had acetylcysteine treatment provided beyond 20 h if serum ALT was rising. Treatment was ceased once ALT had peaked and the international normalized ratio (INR) was <2.0.
Full informed consent was obtained from participants prior to inclusion in the study. Venous blood samples were collected at presentation (referred to as ‘pretreatment’), after 12 and 20 h of acetylcysteine treatment in all trial patients. Further samples were collected in those developing liver injury until recovery of liver function was evident.
Serum was separated from blood and stored at −80°C until analysis. Serum was batch analysed for miR-122 and miR-483.
Laboratory analysis
The GenElute Universal Total RNA purification kit (Sigma-Aldrich, NSW, Australia, Cat. No.: RNB100-50RXN) was used to purify total RNA from serum, including miRNAs, according to manufacturer’s instructions. Briefly, 350 μL of RL buffer and 200 μL of 100% ethanol was added to 200 μL of serum sample and mixed using a Vortex machine. The lysate mix was transferred to a filter column and centrifuged for 1 min at 3500 × g. The flow-through was discarded, and the filter column was washed twice with 400 μL of washing solution. To elute the RNA, 20 μL of DNase and RNase free water was added to the centre of the filter column and centrifuged at 200 × g for 2 min and 14,000 × g for 1 min.
For miRNA detection, total RNA was reverse transcribed with the TaqMan miRNA Reverse Transcription kit (Applied Biosystems, Cat. No.: 4366596, ThermoFisher, Massachusetts, USA) using 0.375 μL of miR-122-5p and miR-483-3p-specific TaqMan primers (Applied Biosystems, Assay No.: 2245 and 2339, respectively, ThermoFisher), 1.5 μL of 10× buffer, 0.125 μL of dNTPs, 0.18 μL of RNase inhibitor, 1 μL of MultiScribe reverse transcriptase and 11.5 μL of total RNA. The complementary DNA (cDNA) mix was incubated for 30 min at 16°C, 30 min at 42°C and 80°C for 5 min.
Real-time quantitative polymerase chain reaction (RT-qPCR) was carried out on the HT7900 RT-qPCR system (Applied Biosystems) using 1 μL of the resulting miRNA cDNA and 9 μL of a RT-qPCR mix including 0.3 μL miR-122 and miR-483 stem-loop TaqMan primers (Applied Biosystems, Assay No.: 2245 and 2339, respectively, ThermoFisher), 5 μL of 2× Sensi-Fast Hi-Rox (Bioline, NSW, Australia, Cat. No.: BIO-82005) and 3.7 μL of DNase and RNase free water. The RT-qPCR plate was incubated for 10 min at 95°C and a cycle (repeated ×40) of 95°C for 15 s and 60°C for 1 min.
Serum miR-122 was normalized to miR-483 13 to account for variations of collection/purification/amplification between samples, using the following calculation (referred to as normalized miR-122 or delta miRNA 14 ): 2−ΔCt (ΔCt = Ct miR-122 – Ct miR-483, Ct: Cycle threshold). Each RT-qPCR sample was carried out in technical duplicates. Ct results are inversely related to miR-expression.
Statistical analysis
Descriptive data are reported with continuous variables reported as median (IQR) unless otherwise stated and compared using Mann–Whitney U test and Kruskal–Wallis analysis of variance by ranks, as appropriate. For subjects with an acute ingestion at a known time, we calculated the ratio between each subject’s paracetamol concentration at presentation (APAPs) and the threshold concentration for treatment based on the time interval post ingestion using the Rumack–Matthew nomogram (APAPt) as customary in risk stratification. 16
miRNA analyses
miR-122 and miR-483 were analysed from each blood sample obtained from subjects in the four groups. These included miRNA at presentation, 12 h and after 20 h of acetylcysteine in the NACSTOP groups. In those that developed ALI or hepatotoxicity, multiple blood samples were taken until ALT had peaked and was improving.
Results
We examined 121 blood samples in 38 patients who consented for miRNA analysis and received acetylcysteine after paracetamol overdose. The median age was 22 years (IQR 18, 32). The majority of patients were female (70%). From the NACSTOP trial; 8 patients (21%) received the abbreviated treatment regimen and 20 (53%) received the full course of acetylcysteine (control group). The two comparator groups were seven (18%) subjects developing ALI and three (8%) developing hepatotoxicity. All received extended acetylcysteine infusions. The APAPs: APAPt ratio was also similar between the NACSTOP groups (1.3 [IQR 1.1, 1.7] in the abbreviated vs. 1.25 [1, 1.6] in the control group, p = 0.38); all had a ratio between 1 and 2. In the ALI group, the APAPs: APAPt ratio was 1.2 (IQR 1, 1.7) for the seven patients who had a paracetamol concentration performed within 24 h of overdose.
Median time to starting acetylcysteine from the reported time of ingestion was 6 h (IQR: 5.5, 12), 6.5 (5.6, 10.5), 7 (5, 12), 24 (12, 31) in the NACSTOP-abbreviated, NACSTOP-control, ALI and hepatotoxicity groups, respectively. Median acetylcysteine infusion duration for the NACSTOP-abbreviated regimen was 13 h (13, 13). This was significantly shorter than the NACSTOP-control (20 h (IQR 20, 20)), ALI (21 h, (21, 21.5)) and hepatotoxicity groups (60 h (48, 80)) (p < 0.0001).
Median ALT after 20 h of acetylcysteine was 12 U/L (IQR 18, 14) in the abbreviated NACSTOP regimen group, compared to the NACSTOP control group 16 U/L (IQR 11, 21; p= 0.17; Table 1). No patients in the NACSTOP study (abbreviated or control group) developed ALI. Median ALT after 20 h of acetylcysteine was 62 U/L (IQR 49, 87) and 1572 U/L (1249, 2947) in the ALI and hepatotoxicity groups, respectively (Figure 1).
Patient characteristics (abbreviated and control, ALI and hepatotoxicity groups).a
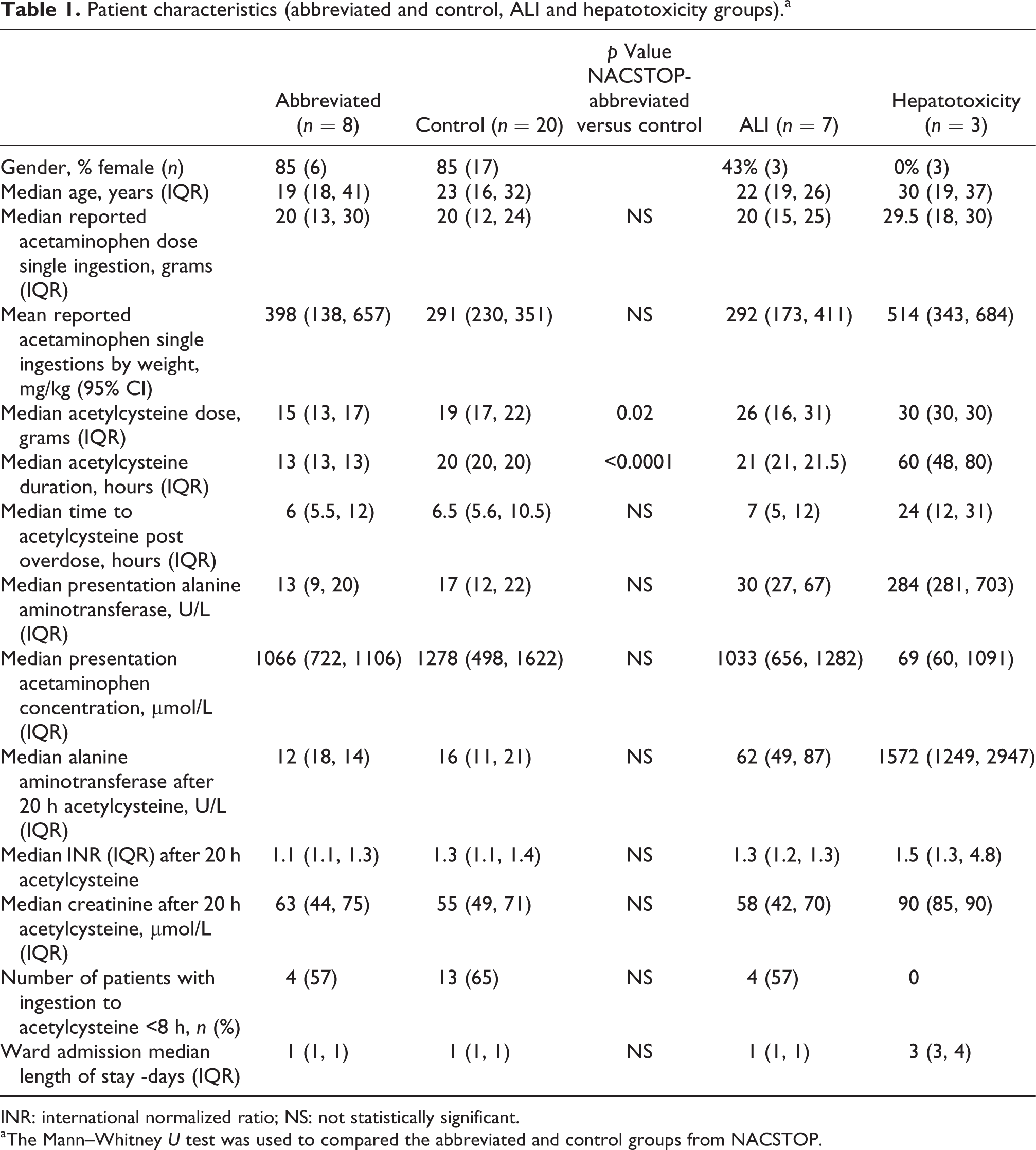
INR: international normalized ratio; NS: not statistically significant.
aThe Mann–Whitney U test was used to compared the abbreviated and control groups from NACSTOP.

ALT versus time post-acetaminophen overdose in NACSTOP abbreviated, NACSTOP control, ALI and hepatotoxicity groups. ALT = 40 is the upper limit of normal range. Time-0 signifies time of ingestion. ALT: alanine transaminase; ALI: acute liver injury.
miR-122 Ct after 20 h of acetylcysteine was 30.1 (IQR: 28.9, 33.3), 31.4 (28.9, 33.9), 29.0 (23.1, 32.4), 23.9 (23.7, 24.9) in the NACSTOP-abbreviated, NACSTOP-control, ALI and hepatotoxicity groups, respectively (Figure 2). There was no significant difference in median ALT or median miR-122 Ct between NACSTOP-abbreviated or NACSTOP-control after 20 h of acetylcysteine (p = 0.7).

miR-122 kinetics versus time after acetaminophen overdose in NACSTOP-abbreviated, NACSTOP-control, ALI and hepatotoxicity groups. Time-0 signifies time of ingestion. ALI: acute liver injury; miR: microRNA.
There was no significant difference in median miR-483 between the four groups (p > 0.05). Median normalized miR-122 Ct after 20 h of acetylcysteine was 2.2 (IQR 1.9, 6.4), 1.1 (0.7, 2.9), 63.9 (2.5, 168), 123.2 (40.9, 207.8) in the NACSTOP-abbreviated, NACSTOP-control, ALI and hepatotoxicity groups, respectively. There was no change in the median normalized miR-122 after 20 h of acetylcysteine between the two NACSTOP trial groups (p = 0.1). Peak normalized miR-122 concentration was higher in those who developed ALI or hepatotoxicity (Figure 3).

Normalized miR-122 (2−ΔCt) kinetics after acetaminophen overdose in NACSTOP-abbreviated, NACSTOP-control, ALI and hepatotoxicity groups. Δ = miR-122 − miR-483. Time-0 signifies time of ingestion. miR: microRNA; ALI: acute liver injury.
Presentation median miR-122 Ct value were 30.1 (IQR: 28.9, 31.5); 30.9 (29.3, 31.5); 28.9 (25.8, 33.6); 26.2 (23.8; 31.6) in the NACSTOP-abbreviated, NACSTOP-control, ALI and hepatotoxicity groups, respectively. There was no significant difference between median presentation miR-122 expression in the NACSTOP groups (p = 0.3). Presentation median normalized miR-122 in the NACSTOP-abbreviated (1.8(IQR: 0.5, 4.4)) and NACSTOP-control (1.8(0.4, 3.6)) groups were significantly lower than those seen in the ALI (5.4 (1.8, 83) and hepatotoxicity groups (34.6 (6.1, 52.1)) (p < 0.04).
Receiver operator curve–area under the curve (ROC-AUC) was 0.65 (95% CI 0.42, 0.89) for miR-122 Ct compared to ROC-AUC of 0.82 (95% CI 0.67, 0.97) for 2−ΔCt (miR-122–miR-483) at presentation for predicting ALI. Sensitivity was 40%, with a specificity of 95% for both presentation miR-122 Ct and normalized miR-122 levels for the development of ALI.
Discussion
Measurement of serum miRNA concentration can be used as a novel method for comparison of acetylcysteine treatment regimens for paracetamol overdose. miR-122 is abundant in the liver and is more sensitive and specific for liver injury than ALT. 13,17 As a result, miR-122 may be a more sensitive marker of liver injury when studying patients treated with various acetylcysteine regimens after paracetamol overdose.
There was no significant difference between median miR-122, normalized miR-122 or ALT in patients receiving abbreviated (12 h) versus the full (20 h) treatment measured after 20 h of acetylcysteine initiation. In addition, no patients in either of these groups developed an ALT > 40 U/L. Therefore, there was no signal of liver injury resulting from a shortened acetylcysteine regimen. Patients enrolled in the intervention arm of the NACSTOP trial received a much shorter duration of acetylcysteine compared to the control group. This study provides some provisional data suggesting that reducing the duration of acetylcysteine infusion in selected patients at low risk of liver injury can be safe. Reduction of treatment time has the potential to decrease hospital medical length of stay, earlier mental health assessment for deliberate self-poisoning patients and possibly also improve access for other patients presenting to the emergency department. 18
Twenty-hours post acetylcysteine initiation, normalized miR-122 were lower in the low-risk patients from the NACSTOP trial compared to the comparator subjects who developed ALI or hepatotoxicity. There was no significant difference in normalized miR-122 or ALT concentration between NACSTOP groups 20 h after initiation of acetylcysteine.
On presentation, normalized miR-122 concentration was significantly higher in those who subsequently developed ALI or hepatotoxicity compared to those who did not. In addition, normalized miR-122 levels were more accurate than miR-122 in predicting ALI on presentation. Other studies support the observation that the combination of miR-122 with miR-483 is a more sensitive marker for the development of ALI (85% vs. 62%, at 90% specificity) than miR-122 alone but has a similar ROC-AUC (0.97 vs. 0.95) when predicting the development of ALI on assay of presentation blood. 13 However, the sensitivity of miR-122 alone was 84% and 94% (for specificity 95%) in a larger study. 17 Identifying patients at greater risk of ALI may trigger changes to management, such as larger acetylcysteine doses and/or prolonged duration of infusion, with the aim of mitigating severity of liver injury.
In patients developing hepatotoxicity, previous studies have shown that both miR-122 and the normalized miR-122 peak (10–20 h) earlier in comparison with ALT and could signify cessation of liver injury progression hours before a downward trend in liver aminotransferases. 13,14 This is in keeping with the few hepatotoxic patients in our study.
In addition, miR-122 and normalized miR-122 were dynamic over time, especially in the ALI and hepatotoxicity groups. This likely reflects the degree of liver injury occurring at that time point. This highlights the need to assess miR-122 over multiple times points if using the biomarker to decide risk of further liver injury or changes to management (e.g. increase in acetylcysteine dosing).
There were several limitations of this study. The study was not randomized at the individual subject level, and a cluster design for the study was chosen to minimize protocol violations and crossover. The primary endpoint was based on biochemical test results and hence unlikely to be biased. The sample size in this study was small but appropriate for an initial exploratory study of miR-122 over time comparing acetylcysteine regimens. While larger studies are required to investigate clinical efficacy of varying acetylcysteine regimens, measuring miR-122 expression can be a useful tool to assess biochemical outcomes of treatment. Other limitations include detection by RT-qPCR may exhibit batch variations. However, all samples in this study were assayed in a single batch. Hence, this reduced the risk of variation.
Conclusions
In this study, there was no significant difference in miRNA between NACSTOP patients, therefore no signal of increased liver injury from an abbreviated 12-h acetylcysteine regimen in selected low risk patients treated for paracetamol overdose. These data complement the normal aminotransferase results. Normalized miR-122 is a better predictor of ALI than miR-122 alone on presentation. These findings suggest that an abbreviated acetylcysteine regimen in low-risk patients who have overdosed on paracetamol is safe. Larger studies including the use of miRNA to assess for liver injury are warranted.
Footnotes
Acknowledgements
We acknowledge the Monash Health Translational Precinct Research Platforms for access to the RT-qPCR instruments.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: AW has received a NHMRC Early Career Fellowship ID 1159907. This work was funded in part by the Australian Research Council (140100594 Future Fellowship to M.P.G.); and the Morson Taylor Award and Grant from the Australasian College for Emergency Medicine.
