Abstract
Administration of natural antioxidants has been used to protect against nephrolithiasis. Urolithiasis was induced by ethylene glycol (EG) in Wistar rats. For 4 weeks, group 1 (control) was fed with a standard commercial diet. Group 2 received the same diet with 0.75% of EG. Group 3 received EG plus the diet and water added with antioxidant nutrients and lime juice as the dietary source of citrate (EG + AX). Group 4 same as group 3 with no EG in water. For 8 weeks, group 5 was fed the standard diet with EG in water for the first 28 days, followed by no EG. Group 6 received the diet with EG for the first 28 days, followed by discontinuation of EG and addition of antioxidant nutrients. Group 7 were provided the diet with antioxidant nutrients for 8 weeks. Group 8 received the diet with antioxidant nutrients for 4 weeks, followed by antioxidant nutrients with EG for the next 4 weeks. Blood samples were collected and kidneys were removed. The size and the mean number of crystal deposits in EG-treated groups was significantly higher than the EG-treated groups, added with antioxidant nutrients and lime juice. After 4 weeks, the mean concentration of malondialdehyde in group 2 was higher than the group 3, and significantly lower in group 4; and in groups 7 after 8 weeks, as well. After 8 weeks, supplementation developed less mean number of deposits in group 6 as compared to group 5; and in group 8, the crystal deposits was substantially less than either group 2 or group 5 (EG-treated rats). Elevated concentration of androgens (as promoters of the formation of renal calculi) as a result of EG consumption decreased following antioxidant supplementations. Results showed a beneficial effect of antioxidant and provided superior renal protection on treating and preventing stone deposition in the rat kidney.
Keywords
Introduction
Stone disease is an increasingly common form of renal disease that is associated with crystal deposition in the renal medulla in all cases studied so far. One suggested mechanism for the formation of stones, especially, calcium stones, increased urinary super saturation of stone-forming salts, which leads to homogeneous nucleation in the lumen of the nephron, followed by crystal growth and consequent obstruction in the distal nephron. It is a complex process that is a consequence of an imbalance between promoters and inhibitors in the kidney. Therapy to prevent stones is based on lowering super saturation, using both diet and medication. To reduce the incidence, it must be considered that the urine composition is directly related to diet. 1,2 Treatment for symptomatic stone passage is based on high oral fluid intake in all stone formers. It has been shown that increasing water intake to ensure a urinary volume of approximately 2.5 l/day was associated with reduced urinary super saturation with calcium oxalate (CaOx), and a significant reduction in stone recurrence 3 and fluid intake as fruit juice, specifically orange juice, is also effective in reducing urinary CaOx saturation and increasing urinary citrate excretion. 4 Pharmacological treatment is needed in most recurrent calcium kidney stone formers as well as in specific stone-forming populations 5
Although a complete picture of the pathophysiological mechanisms involved is still unclear, but there is increasing evidence that reactive oxygen species (ROS) and development of oxidative stress (OS) are produced during idiopathic CaOx nephrolithiasis. It is reported that OS, renal epithelial injury, and inflammation are also engaged in idiopathic stone formation which is indicated by the urinary excretion of ROS, products of lipid peroxidation, enzymes indicative of renal epithelial injury as well as many markers of chronic kidney disease and suggested that stone formation can lead to hypertension, diabetes, chronic kidneys disease, and myocardial infarction. 6 The reverse also appears to be true, in that several systemic diseases, such as hypertension, diabetes mellitus, and hypercholesterolemia; infection; antibiotics, chemotherapeutics, and radiocontrast agents; and environmental toxins, occupational chemicals, radiation, smoking, as well as alcohol consumption induce oxidative stress in kidney. 7
Moreover, the administration of antioxidants has been used to protect against nephrotoxicity in humans and experimental animals. In the kidney, these treatments are reported to diminish the increase in malondialdehyde (MDA) and the decrease in protective enzyme activity that are induced by chemical and pharmacological agents. 8 As experimental and clinical studies have demonstrated, the most frequently studied natural antioxidants with free radical scavengers to provide superior renal protection are vitamins A and carotenoids, E, C, B6, and antioxidant trace elements such as selenium and zinc (Zn) 8 –15 that can be easily and safety increased in tissues by supplementation. Medical treatment of stone-forming patients using pyridoxine is considered as an effective first-line therapy to decrease hyperoxaluria in patients who develop stones. 16 Zn is believed to have inhibitory effect on CaOx stone formation 17 and is a constituent of the enzymatic antioxidant, superoxide dismutase (SOD). 18 The enhanced activity of antioxidant enzyme SOD can contribute to nephrolithiasis prevention via direct effects on renal epithelial cells. 19 Among dietary factors, evidence exists that boron may have antioxidants and anti-inflammatory properties 20 –22 and seems to have an impact on kidney stone removal, dissolving, or passing out with pain alleviation and cease of hematuria. 23
Moreover, the incidence of urolithiasis has been increasing throughout the past three decades, and the incidence is reported to be three times higher in men than in women, 24 mainly occurring in the third and fourth decades of life when the level of serum testosterone is also the highest. 25
The association between serum testosterone and urolithiasis has yet received only limited attention, the cause of this male predisposition is currently unknown, and the reason for this male predominance is obscure and two possibilities are possibly available, for example, either that male hormones may promote or that female hormones may inhibit kidney stone formation. Although clinical proof for this hypothesis is lacking, recently the relationship of kidney calculi with high plasma total and free testosterone is reported. 26
Overall, it appears that the kidney is a highly vulnerable organ to damage caused by ROS, due to the abundance of long-chain polyunsaturated fatty acids. Antioxidant and reactive oxygen scavengers have been shown to be effective in animals for protecting kidney. Therefore, the aim of the current study was to test the effects of a combination of natural antioxidants such as vitamins A, C, E and B6; Zn and selenium as two essential constituent trace elements associated with adequate amount of antioxidant enzymes production plus boron with a proposed antioxidant capacity against ethylene glycol (EG)-induced nephrolithiasis or kidney calculi.
Material and methods
Male Wistar rats weighing 150–200 g were obtained from the Animal House of Physiology Group, Baqiyattallah University of Medical Sciences. Seven rats in each group (Control vs. seven treatment groups) were weighed and randomly kept in plastic cages in a controlled environment with a 12-h light /12-h dark cycle and a constant temperature (22–25°C) and humidity (55–65%), with free access to food and water. They had access to normal rat chow diet and water for 7 days before the beginning of experimental protocols. Animals were provided clean cages weekly, for either 4 or 8 weeks. All experiments were performed in accordance to the guidelines of the care of animals and approved by the Research and Ethics Committee of Baqiyattallah University of Medical Sciences (permit number: 03-1390).
Diets and animal treatments
Rats in all groups were fed with standard rat chow from Pars Animal Food Co. (Tehran, Iran) and water ad libitum throughout the study. According to the manufacturer, the composition of the standard diet include 9.0 IU vitamin (Vit.) A, 18.0 μg Vit. E, 3.0 μg Vit. B6, 0.20 μg selenium, and 85.0 μg Zn/g dry food, with no added Vit. C and boron. The daily supplementation of the nutrients in the diet or the water consumed by the animals was calculated given that a 150–200 g animal food intake is approximately 20.0 g/day and drinks water at the rate of 10–12 ml/100 g body weight/day. Therefore, the supplementation rate was considered to provide the amounts of antioxidant nutrients approximately five to seven times of the natural daily intake. Accordingly, in the first part of the study (4 weeks study), group 1 (control) was fed a standard commercial diet (rat chow). Group 2 received the same diet with 0.75% of EG (Sigma-Aldrich Co., St Louis, Missouri, USA) in drinking water for 28 days ad libitum to induce the kidney stone formation. Group 3 received the standard diet enriched with 4000.0 μg Vit. E and 1500.0 IU Vit. A for each rat per day added to the diet once a week and provided daily with 5.0 mg Vit. C, 400.0 μg Vit. B6, 20.0 μg selenium, 12.0 mg Zn, and 2.0 mg boron for each rat per day in their drinking water plus 0.75% of EG. Group 4 same as group 3 with no EG in water. Commercial lemon juice as the dietary source of citrate was added to the water at the level of 1 ml/rat/day for all the groups receiving antioxidant nutrients.
The second part of study lasted for 8 weeks. Group 5 was fed the standard diet with EG in water for the first 28 days, followed by the standard diet with no EG. Group 6 received the diet with EG in water for the first 28 days, followed by discontinuation of EG and addition of antioxidant nutrients. Group 7 were provided the diet with antioxidant nutrients for 8 weeks. Group 8 received the diet with antioxidant nutrients for 4 weeks, followed by antioxidant nutrients with EG for the next 4 weeks. The antioxidant protocol was same as group 3.
To provide the antioxidant nutrients, 300.0 IU (200.0 mg) of a soft gel capsule of Vit. E as
Pharmaceutical Vit. C (ascorbic acid) 250.0 mg tablets (Osvah Pharmaceutical Co., Tehran, Iran), pyridoxine HCl (Vit. B6) 40.0 mg tablets (Ramopharmin Pharmaceutical Lab., Tehran, Iran), selenium (Selenium Amino Acid Chelate) 200.0 μg dietary supplement capsules (Alfa Vitamins Lab., Inc., Doral, Florida, USA), Zn (zinc sulfate) 50.0 mg capsules (Alhavi Pharmaceutical Co., Tehran, Iran), and boric acid (Merck, Germany) as the source of boron was used and added to their drinking water to provide the above-mentioned doses/rat/day, respectively. Lime juice (Mahram Co. Group, Shiraz, Iran) was purchased from local market. Fresh food and water containing the above chemicals was provided three times per week and the consumption was monitored and recorded.
The experimental procedure adopted for the study is detailed as follows.
The first part of the study (4 weeks study); Group 1 or control (CONT): the standard diet and drinking water; Group 2 (EG): same as group 1 + 0.75% of EG in drinking water; Group 3 or treatment (EG + AX): same as group 2 + antioxidant nutrients, boron and lime juice; Group 4 or antioxidants (AX): same as group 3 with no EG in water;
The second part of the study (8 weeks study); Group 5 (EG/EG): the standard diet + EG in water for the first 4 weeks, followed for 4 weeks with no EG; Group 6 or post EG Treatment with AX (EG/-EG + AX): the diet with EG in water for the first 4 weeks, followed by discontinuation of EG and addition of antioxidant nutrients, boron, and lime juice; Group 7 or antioxidants (AX/AX): the diet and water + antioxidant nutrients, boron, and lime juice for 8 weeks; Group 8 or prevention with AX loading (AX/AX + EG): same as group 7, added with EG after 4 weeks.
Four and eight weeks after the experimental periods, rats from all groups were anesthetized for the collection of blood by cardiac puncture with a syringe and needle for further biochemical studies. Rats were restrained from food for 12 h but had access to drinking water. Since some parameters in rats such as steroid hormones are subject to circadian rhythm, blood samples were collected at the peak time in the afternoon between 14.00 and 16.00 h and plasma samples were stored frozen until analysis.
The rats were then killed and both kidneys were excised. Left kidney from each animal was placed and fixed in 10% formalin and dehydrated in a gradient of ethanol, embedded in paraffin, and then cut into 5 μm serial sections. Serial sections were cut and stained by hematoxylin and eosin and von Kossa (for phosphate detection). Nine slides from lateral, middle, and medial sections were selected randomly. The histopathological changes and CaOx crystal deposition at 40× magnifications under light microscope were studied in six fields of each section, composed of three fields in medulla, and three fields in cortex. Aggregations of crystal deposits in the renal tubules were counted in 54 microscopic fields and an average was calculated. The size of crystal deposition was measured using Motic system Image analyzer (Motic Image 2000, 1.2 Micro-Optic Industrial Group Co. Ltd, China) and categorized as zero (no crystal), small (1–10 μm), medium (>10–20 μm), and large (>20 μm).
Commercially available assay kits were used to determine the blood parameter levels.
Total antioxidant status or capacity (TAS) was measured by enzymatic colorimetric method (Rel Assay Diagnostics, Gaziantep, Turkey), with the intra-assay coefficients of variation (CVs) % of 3.6.
SOD was measured by photometric method (Epigentek, Farmingdale, New York, USA), with the intra-assay CVs % of 1.6.
Glutathione peroxidase (GPx) activity was measured by enzymatic colorimetric method (Enzymatic Colorimetric, Cayman Chemical Company, Ann Arbor, Michigan, USA), with the intra-assay CVs % of 2.1.
MDA content, representing lipid peroxidation, determined by the thiobarbituric acid reactive method was measured by photometric method (Bioassay Systems, Hayward, California, USA), with the intra-assay CVs % and assay sensitivity of 5.5 and 0.1 μM.
Total testosterone (T), free testosterone (FT), dihydrotestosterone (DHT), and estradiol (E2) were analyzed by enzyme-linked immunosorbent assay (ELISA, Diagnostics Biochem, Canada Inc., Ontario, Canada). The intra-assay CVs % and assay sensitivity were 5.1 and 0.022 ng/ml for TT; 3.4 and 0.17 pg/ml for FT; 2.1 and 6.0 pg/ml for DHT; and 4.4 and 1.0 pg/ml for E2, respectively. Sex hormone binding globulin (SHBG) was analyzed using an ELISA kit (Cusabio Biotech Co., Ltd, Wuhan, Hubei, China), with the intra-assay CVs % and assay sensitivity of 6.4 and 1.0 ng/ml.
Statistical analysis
Data are expressed as mean ± SD and a Statistical Package for the Social Sciences (SPSS 18.0), McGraw-Hill, New York, USA) was used to perform all comparisons. One-way analysis of variance (least significant difference post hoc test) and independent sample t-test was used for comparison of the mean of the antioxidant markers and hormones. A p value of ≤0.05 was considered statistically significant for the differences.
Results
The rats in all groups adjusted to the treatments well and consumed food and water normally.
The effect of different treatments on the mean number of crystal deposits in the microscopic fields in the kidney specimens and on the plasma variables including markers of oxidative stress and hormones in different groups is shown in Tables 1 –3.
The size and the number of stone formation and crystal deposit in the cortex and medulla kidney section of the rats in different groups.a

EG: ethylene glycol;
aSmall crystal size: 0–10 µm; medium crystal size: >10–20 µm; large crystal size: >20 µm.
Effect of EG administration and AX supplementation on plasma parameters in the 4 weeks study groups (mean ± SD).
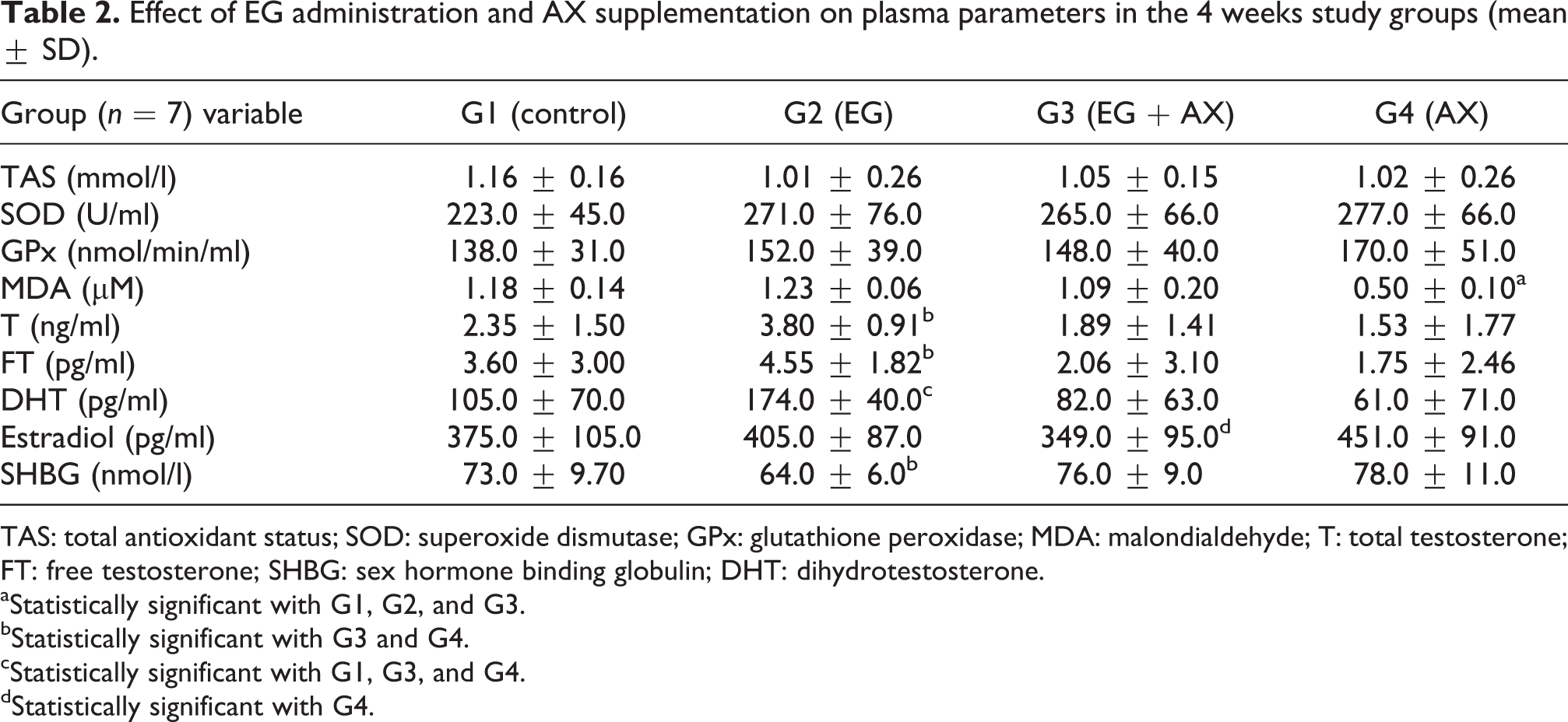
TAS: total antioxidant status; SOD: superoxide dismutase; GPx: glutathione peroxidase; MDA: malondialdehyde; T: total testosterone; FT: free testosterone; SHBG: sex hormone binding globulin; DHT: dihydrotestosterone.
aStatistically significant with G1, G2, and G3.
bStatistically significant with G3 and G4.
cStatistically significant with G1, G3, and G4.
dStatistically significant with G4.
Effect of EG administration and AX supplementation on plasma parameters in the 8 weeks study groups (mean ± SD).

TAS: total antioxidant status; SOD: superoxide dismutase; GPx: glutathione peroxidase; MDA: malondialdehyde; T: total testosterone; FT: free testosterone; SHBG: sex hormone binding globulin; DHT: dihydrotestosterone.
aStatistically significant with G8.
bLower than G5, p = 0.06.
In the 4 weeks study, the mean number of deposits in EG group (G2) was higher than the EG + AX group (G3). Crystal deposition was completely absent in kidneys in the control group (G1) and antioxidant group (G4), as shown in Table 1.
A marked difference in the plasma concentration of MDA was noted between EG group (G2) and EG + AX group (G3; p = 0.07), observing a higher concentration in G2. Also a significant lower concentration in antioxidant group (G4) was noted in comparison with the groups 1–3 (p = 0.000; Table 2).
Furthermore, no other major changes were noted for other markers of oxidative stress.
A higher difference in the plasma concentration of T in EG group (G2) compared with G1 (p = 0.06), followed by a significant reduction with G3 (p = 0.01) and G4 (p = 0.00).
A higher plasma concentration of FT in EG group (G2) decreased in group 3 (G3; p = 0.09) and was significant in G4 (p = 0.05).
The plasma concentration of DHT in EG group (G2) was significantly higher than G1 (p = 0.05), followed by a significant reduction with G3 (p = 0.01) and G4 (p = 0.00).
A higher plasma concentration of E2 in antioxidant group (G4) was significantly higher than EG + AX group (G3); p = 0.05). A significant high plasma concentration of SHBG was noted between G4 with G3 (p = 0.05) and between G3 (p = 0.01) and G4 (p = 0.00) with G2.
In the 8 weeks study, the EG-treated rats (G5) showed substantial persistent crystal deposition even after 4 weeks cessation of EG consumption, whereas antioxidant supplementation as a curative measurement within second 4 weeks period developed less mean number of deposits in G6. However, the number of deposits in the EG-treated rats (G5) after 4 weeks EG abstinence stage or deprivation was lower than 4 weeks EG-treated rats (G2). In other preventive AX loading group (G8), the number of crystal deposits was substantially less than EG-treated rats (compared with G2 or G5; Table 1).
A significant lower plasma concentration of MDA was noted in (G7; AX/AX) compared with (G8) (AX/AX + EG; p = 0.007; Table 3).
The plasma concentration of active steroid hormones (T, FT and DHT in G6, treated with antioxidant nutrients after crystal formation was markedly lower than G5 (EG/-EG; p = 0.06, 0.1, and 0.06, respectively). Similar lower trend was also noted for these hormones in AX loading group (G8) showing less number of crystal deposits.
Discussion
The incidence of urinary stone formation and crystal deposits in rats treated with 0.75% EG implicated that the majority of stones were evenly distributed in the renal parenchyma. No stone or crystal deposition was found either in the control group or in the G4 receiving antioxidants. The results indicate that antioxidant stone preventive agents in G3 significantly decreased CaOx stone formation resulting from free drinking of EG. Also CaOx crystals in different parts of the renal tubules in G3 were clearly smaller in comparison with EG group. It is clearly shown that a combination of excess dietary antioxidants may have a protective effect against free-radical injury in stone formation. The kidney is a highly vulnerable organ to damage caused by reactive oxygen substances, due to the abundance of long-chain polyunsaturated fatty acids. MDA is an end product of lipid peroxidation induced by free radical, and its content could reflect the level of lipid peroxidation in the body. Lipid peroxidation and antioxidant depletion are associated with several pathophysiological conditions, including urinary stone formation. 12,27 The development of tissue injury probably depends on the balance between the generation of ROS and the tissue antioxidant defense mechanism.
Elevated free radical production as shown by increase in MDA concentration due to EG consumption in the formation of nephrolithiasis confirms that renal tissue is under oxidative stress. This hypothesis is strengthened by the report that patients with kidney stones have less activity of antioxidant enzymes with increased lipid peroxidation. 28 Oxidative damage as reflected from the increased level of markers of oxidative injury by higher MDA and decreased TAS and antioxidant enzymes activity such as SOD and GPx level deteriorate kidney structure and functions as observed in calculi-induced rats. Antioxidant and reactive oxygen scavengers have been shown to be effective in animals for protecting kidney.
It is confirmed that Vit. E acts as an excellent antioxidant for the kidney, which is greatly susceptible to oxalate-induced free radical damage and its supplementation maintained the optimal antioxidant enzyme levels to protect renal tubes from peroxidative injury. 14,29 Rubus idaeus, a medicinal plant having a significantly high content of Vit. E has an impressive prophylactic effect on CaOx stones in nephrolithic mice. 30 In the study of free radical-mediated damage to biological systems, the coadministration of vitamin E and selenium to alleviate lung oxidative damage induced by an organophosphorus compound 31 and its cardiotoxicity 32 and in lithogenic rats to decrease the levels of lipid peroxides and the activities of oxalate-synthesizing enzymes, with a concomitant increase in the activities of enzymatic antioxidants and increased levels of nonenzymatic antioxidants 11 confirmed the potential protective effects of selenium and Vit. E.
To evaluate the association between serum antioxidant levels and the prevalence of kidney stones, the likely role of oxidative tissue damage in the pathophysiology of stone disease is demonstrated. Lower levels of α-carotene, β-carotene, β-cryptoxanthin, and Vit. A deficiency are associated with a history of kidney stones and may indicate a role for these antioxidants in prevention. 10,11,33 The activities of hepatic glycolate oxidase and glycolate dehydrogenase were markedly enhanced in Vit. A and Vit. B6-deficient rats, 34 and pyridoxine is known to be an effective first-line therapy to decrease hyperoxaluria in patients who develop stones. 16 Ascorbic acid, as well as other radical scavengers, and the antioxidant enzymes decreased in urolithic condition, suggestive of the active involvement of free radical-mediated lipid peroxidation-induced membrane damage in kidney. 35 Vit. C is a major water-soluble antioxidant and acts as the first defense against ROS in whole blood and plasma. At 24 h after extracorporeal shock wave lithotripsy, patients given antioxidants had significantly reduced mean serum concentration of MDA and higher levels of serum ascorbic acid that protect these patients from short-term renal injury. 36 However, high Vit. C supplementation either 1 g or 2 g for 3 days may increase urinary oxalate excretion and the risk of CaOx crystallization in calcium stone-forming patients. 37 The efficacy of antioxidant Vits A, E, and C alone and in combination is well known 15,38–39 and have been shown to be effective in animals for protecting kidney. 7
It is reported that Zn and magnesium (Mg) have inhibitory effect on CaOx stone formation 17 and low concentrations of Zn, Mg, and manganese in stones appear to make them resistant to shockwave lithotripsy (SWL) fragmentation. 40 After Zn supplementation, plasma Zn and antioxidant power increased; and lipid peroxidation products decreased in the Zn-supplemented patients, compared with the placebo group. 41 This antioxidant power seems to be effective for protecting kidney. It is speculated that alterations in steroids by boron, and the reduction of cytokines with a possible change in urine calcium concentration may have an impact on the prevention of stone formation or removal. 23 Therefore, it was decided to add boron to the supplemented complex used in this study. Other recent proposed properties of boron such as anticarcinogenic properties, 20,42–43 antioxidant capacity 21–22,44 and against brain oxidative stress 45 support a protective role for boron in the treatment of urolithiasis.
Data suggest that lemon juice as a source of citrate has a protective activity against urolithiasis and large deposits of CaOx crystals were not present in rats treated with either lemon juice. 46
The protective role of citrate is reported to reduce urinary super saturation of calcium salts by forming soluble complexes with calcium ions and by inhibiting crystal growth and aggregation. Furthermore, citrate increases the activity of some macromolecules in the urine (e.g. Tamm–Horsfall protein) that inhibit CaOx aggregation and seems to be able to reduce the expression of urinary osteopontin. 47
In addition, findings suggest that oral antioxidant therapy prior to lithotripsy may reduce the severity of long-term renal injury caused by the shock waves. 48 Despite the decrease in MDA level, treatment with antioxidant nutrients after EG consumption led to significant decrease in the androgen levels, as well. It indicates that it protected against EG oxidative stress and the induced kidney tissue damage.
Urolithiasis is a multifactorial disorder influenced by both intrinsic and environmental factors. Among the studied factors, male gender with three times higher incidence of urolitlithiasis than female gender is considered as a risk factor, but the influence of sex hormones on urinary stone formation remains undetermined. Since the association between serum testosterone and urolithiasis has yet received only limited attention, the pathogenesis of this male predisposition still remains to be elucidated and thus the reason for this male predominance remains apparently obscure. Therefore, with respect to hormonal influences, although the pathophysiologic mechanisms of some disorders, such as hyperparathyroidism, have been elucidated, the actions of other hormones on urolithiasis remain elusive.
Our findings along with other studies provide evidence for the influence of sex hormones on the pathogenesis of CaOx stone. Finlayson postulated that lower serum testosterone levels may contribute to the protection of women and children against oxalate stone disease. 49 In contrast, men have been found to have higher mean of oxalate levels than women, 50 while it is reported that plasma oxalate concentration and kidney CaOx deposition is increasing by androgens, but decreasing by estrogens. 51 Menopausal women might have an increased potential in this respect as compared to premenopausal women and estrogen supplementation may be helpful in the avoidance of nephrolithiasis. 52 The lower risk of stone formation and stone recurrence in estrogen-treated postmenopausal women may be due to the lower urinary saturation of stone-forming salts. 53
Data suggest that testosterone can promote and estrogen may inhibit CaOx stone formation in EG-treated rats, 54 likely because of their effects on oxalate synthesis and oxidative stress. 55 Testosterone appears to promote stone formation by suppressing osteopontin expression in the kidneys and increasing urinary oxalate excretion. Estrogen appears to inhibit stone formation by increasing osteopontin expression in the kidneys and decreasing urinary oxalate excretion. 56
In this study, after EG exposure a positive trend was observed between high plasma androgen concentrations with a rise in MDA and incidence of kidney stones, indicating a potential role for the gonadal steroids in the pathogenesis of urolithiasis in male rats. Testosterone is known to increase the hepatic levels of glycolic acid oxidase (GAO), an important enzyme in the metabolic pathway for urinary oxalate synthesis resulting in hyperoxaluria, which in turn may be responsible for the increased predisposition to CaOx urolithiasis. Urinary oxalate excretion increased 12.8-fold after 4 weeks of EG treatment and it was concluded that DHT was partially responsible for the observed exaggerated hyperoxaluria. 57
Following EG administration in rats in G2, DHT was significantly higher than other three groups, indicating to be involved in crystal formation along with increase in oxidative stress and lipid peroxidation. Moreover, FT in G2 had similar trend and was significantly higher than G3 and G4, indicating its contribution in crystal formation, as well.
In patients with nephrolithiasis serum T and FT revealed to be twice as healthy men. The levels of serum T and TT were 13.29 ± 4.79 ng/ml and 63.23 ± 28.58 pg/ml in patients, and 7.30 ± 0.82 ng/ml and 35.59 ± 24.91 pg/ml in healthy men, respectively. 26
Other reports confirm a positive relationship between high plasma androgen concentrations and incidence of kidney stones, indicating a potential role for the gonadal steroids in the pathogenesis of urolithiasis in male sex. 58 –60 It is reported that administration of exogenous testosterone influences the lipid peroxidation and carbonyl stress and decreases the antioxidant defense in the testes. 61
Moreover, supplementation of vitamin E (α-tocopherol) and 17β-estradiol (E2) in ovariectomized diabetic rats resulted in an increase in antioxidant enzymes, Vit. A, and β-carotene levels but a decrease in MDA levels played a role in preventing renal disorders. 62
These results demonstrate that nutrient supplementation decreases EG-induced CaOx crystal deposition in the kidney by enhancing the antioxidant defense mechanism; therefore, our complex would be considered in the treatment of kidney stone formation and may benefit individuals with current kidney stone disease. In most experimental protocols, crystal-inducing drug and the therapeutic agents are administered, simultaneously. In real life, patients actually seek medical treatment after appearance of common initial symptom of nephrolithiasis such as pain and hematuria or after medical confirmation of renal calculi diagnosis.
Hence, in the 8 weeks study, antioxidant supplementation as a curative measurement after 4 weeks calculi induction represents a design as a curative protocol which developed less number of kidney stone deposits and size in G6 compared with the EG-treated rats (G5).
Moreover, in other preventive AX loading group (G8), followed by 4 weeks EG administration, the number of crystal deposits was substantially less than the corresponding groups receiving EG (e.g. EG-treated rats (G2) or (G5)).
Results showed a beneficial effect on curing and preventing stone deposition in the rat kidney. These results provide a scientific rational for preventive and treatment roles of antioxidant nutrients complex in kidney stone disease.
Conclusion
Oxidative stress and active involvement of free radical-mediated lipid peroxidation-induced membrane damage associated with antioxidant imbalances are considered as an important mechanism involved in many pathological conditions and have been implicated in the pathogenesis of several systemic diseases such as crystal attachment and aggregation resulting in nephrolithiasis. Restoration of antioxidant levels is capable of preventing CaOx crystal deposition by reducing accumulation of the lipid peroxidation products and androgens and to prevent free radical-inducing renal injuries.
Overall, the current study data indicated that administration of a combination of natural antioxidants showed beneficial effects on prevention and elimination of CaOx calculi in the rat kidney. It seems that the effect of the selected nutrients on prevention and disruption of the kidney stones may be, at least, in part due to its antioxidant effects.
Moreover, it may be concluded that supplementation of the antioxidants contribute to the total antioxidative capability that could be considered as a healthier option and an effective strategy to protect against the deposition of CaOx stones in the kidney.
Footnotes
Conflict of interest
The authors declared no conflicts of interest.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
