Abstract
Ethylene glycol (EG), in addition to its neurotoxic and nephrotoxic effects, evokes oxidative stress. The aim of this study was to assess the influence of the ethylene glycol on the biochemical indicators and oxidoreductive balance of patients treated for acute poisoning. The total study group consisted of 56 persons including 26 alcoholics who took EG as a substitute for ethyl alcohol in the course of alcohol dependence syndrome and 30 controls. Severity of poisoning, results of acid-base parameters, biochemical, and toxicological tests as well as biomarkers of the oxidative stress in blood were analyzed during the patients’ hospitalization. The key issue was to assess the oxidative stress and biochemical disturbances caused by EG and the type of treatment applied in the course of poisoning. Significant changes in some parameters were found both at time of diagnosis and after treatment initiation (ethanol as an antidote and hemodialysis). The most important differences included the activity of hepatic parameters (aspartate aminotransferase, AST) and oxidative stress markers like catalase (CAT); correlation of the lipid peroxidation products level (TBARS) with urea concentration has been shown. On the last day of the hospitalization, in some cases, the mutual correlation between the evaluated markers were observed, for example, between alanine transaminase (ALT) and glutathione reductase (GR), and urea concentration and glutathione level (GSH/GSSG). The concentration of ions (H+) had a major impact on the oxidoreductive balance, correlating with the elevated GR and GSH/GSSG levels.
Introduction
One of the factors that determines the effectiveness of treatment is the time of taking diagnosis of EG poisoning—the shorter time, the better chance of success of the therapy. The confirmation of ethylene glycol in blood and results of complementary laboratory tests, as well as the patient’s clinical condition, are factors on the basis of which the decision to start specific therapy is made.8–11 Medical history indicating the consumption of non-commercial alcohol accompanied by high anion gap metabolic acidosis and renal failure, especially if the EG blood concentration exceeds 0.2 g/L, indicates the need for a general clinical evaluation of the patient and implementation of proper therapy.2,12 During EG poisoning, severe high anion gap metabolic acidosis develops with extremely low pH values, bicarbonates concentrations, and base deficiency, as well as excess acid products (BE).10,13 The basic biochemical tests which enable the assessment of liver and kidneys function (activity of transaminases and creatinine or urea concentrations) are also extremely important, in particular to alcohol addicts.14,15
The assessment of xenobiotics effects on biological systems is the basis of toxicological sciences and primarily concerns the study of the mechanisms of their harmful effects. The analysis of the risk of exposure as well as the control and prevention of adverse effects is also the essence of proper diagnosis and treatment of poisoning. Apart from thorough clinical observation, an important role is played by biochemical and toxicological tests. To date, no data have been published that demonstrates the effect of acute EG poisoning treatment on oxidative stress in the group of patients addicted to alcohol. That is why the first aim of the study was to assess the effect of ethylene glycol and ethyl alcohol used as an antidote on the oxidoreductive balance in the course of acute poisoning treatment. And then, we examine the relationship between oxidative stress and biochemical disorders observed during the treatment.
Material and methods
Study groups
Ethylene glycol and control groups.

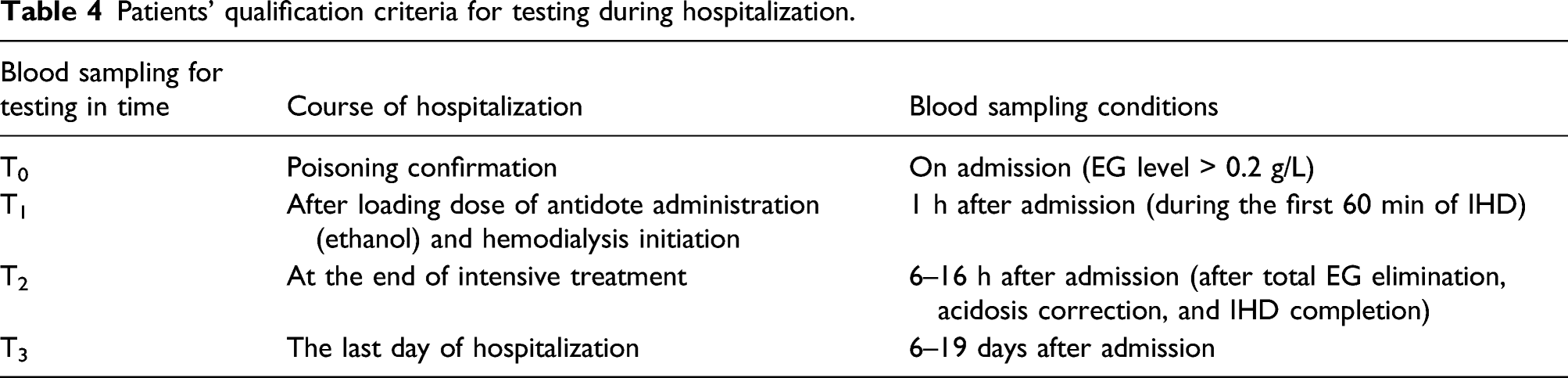
Patients’ qualification criteria for testing during hospitalization.
Blood samples collection
In all measurements, a venous blood (serum fraction and whole blood), sampled into vacutainer tubes (Sarstedt, Germany) was used. For acid-base balance tests, blood was drawn into a gasometrical syringe with lithium heparin and analyses were carried out within 15 min after sampling. For biochemical (aspartate aminotransferase—AST, alanine transaminase—ALT, creatinine—Crea, and urea—Urea) and toxicological (ethylene glycol—EG) tests, blood was collected in vacutainer tubes containing a coagulation activator (silica—SiO2). The concentration of ethyl alcohol (EA) was determined in blood collected in vacuum tubes containing anticoagulant (potassium sodium edetate). The biochemical and toxicological tests were carried out within a maximum of 2 h after sampling. Oxidative stress markers (TBARS, hydroxynenenal—HNE, total glutathione—GSH/GSSG, glutathione peroxidase—GPx, glutathione reductase—GR, S-glutathione transferase—GST, and catalase—CAT) were determined in serum obtained after centrifugation of the blood collected in silica-containing tubes. Before evaluation, serum was stored at −80°C.
Ethylene glycol (EG) and ethyl alcohol (EA) determination
Ethylene glycol and ethyl alcohol were determined by gas chromatography with flame ionization detection. In order to determine EG, before injection, blood with addition of internal standard (1,3-butanediol solution 0.5 g/L in methanol) was deproteinized and dehydrated using anhydrous sodium sulfate. To determine EA, the head-space analysis was used by adding N-propanol solution 1 g/L in water. Due to the differences in the volatility of both alcohols, EG and EA determinations were carried out in two independent analyses using the same capillary column (BAC1: 30 m × 0.32 mm × 1.8 μm; Restek, USA).27–29
The acid-base balance and renal-hepatic functions evaluation
To determine the arterial blood gases (pH defined as −log10(H3O+) and calculated to the concentration of hydrogen ions (H+), bicarbonates—HCO3−, and base excess—BE levels), potentiometry and oximetry methods were used (ABL 800 flex, Radiometer Copenhagen, Denmark). Biochemical tests (AST, ALT, Crea, and Urea) were performed using the biochemical analyzer Cobas Integra 400 (Roche Diagnostics, USA).
Oxidative stress markers’ determination
Lipid peroxidation was measured by means of a spectrophotometric test of thiobarbituric acid-reactive substances (TBARS Assay Kit, Cell Biolabs, USA). Glutathione peroxidase and glutathione reductase activities were evaluated using ready-made kits: Ransel (Randox Laboratories, UK) and Glutathione Reductase Assay Kit (Calbiochem, Germany), respectively. The hydroxynonenal and total glutathione levels were measured using immunoenzymatic methods (HNE Adduct ELISA Kit and GSSG/GSH Assay Kit, Cell Biolabs, USA). The activity of S-glutathione transferase was measured according to the methods of Habig et al. 30 (1974) and Habdous et al. 31 (2002). All results were evaluated using spectrophotometric reader Bio-Tek Instruments Elx-800 (Highland Park, Winooski, Vermont, USA), spectrophotometers: Zuzi Series 4481 (Auxilab, Spain), and Shimadzu A-116 (Shimadzu Scientific Instruments, USA).
Statistical analysis
The assessment of significant differences between the results obtained in poisoned and healthy group was carry out using Mann–Whitney U test (variables without normal distribution) and Student’s t-test (variables with normal distribution). In the absence of equality of variances, the test with independent estimation of variance was used. The value of p was assumed to be 0.05, and the result was considered significant when p < 0.05. The correlations between the results of acid-base balance, biochemical, and toxicological tests with parameters of oxidative stress were investigated using the Spearman rank test. The comparisons between poisoned and control groups were performed using the ANOVA test (Tukey’s post-hoc test). The statistical evaluation was performed using GraphPad software (GraphPad Software Inc., USA).
Results
Toxicological, biochemical, and arterial blood gases results
The presence of ethylene glycol in blood was confirmed upon admission to hospital (T0). Its average concentration was 0.95 g/L (max: 1.40 g/L, min: 0.28 g/L). One hour after admission (T1), the EG concentration was reduced (on average by 25%), which indicates its effective elimination after administration of ethanol and hemodialysis. EG was not found in subsequent time points (T2 and T3).
Ethylene glycol and ethyl alcohol blood levels in group of poisoned patients.

Table 5 shows the results of acid-base balance and renal-hepatic blood parameters in control and poisoned groups. The measurements (T0) were taken in patients addicted to alcohol at the time of admission to hospital. In comparison to the control group (C) (p < 0.05), reduced bicarbonates (−280%) and pH (−300%) concentrations were indicated. These changes corresponded with elevated concentration of hydrogen ions (H+) (+306%) and several-fold increase of creatinine (+401%) and urea (+191%) levels, which indicates metabolic acidosis and renal failure during poisoning. Particularly, the average ALT and AST activities at admission (58 ± 43 U/L and 107 ± 181 U/L) were several times higher than in the group C (25 ± 10 U/L and 29 ± 8 U/L), which may indicate, among others, earlier liver cell damage due to chronic ethanol consumption, typical for alcohol-addicted patients.
Acid-base balance parameters and renal-hepatic values in poisoned group during the treatment in comparison to the control and each time measurement (mean ± SD).

significant statistical differences at the level p < 0.05.
Oxidative stress markers
Oxidative stress parameters in poisoned group during the treatment (T0–T3) in comparison to control (mean ± SD).

significant statistical differences at the level p < 0.05.
Substantial differences in catalase and glutathione reductase activities were indicated, especially in T2, after the end of specific treatment. Catalase activity differed significantly in T2 compared to the results obtained in T1 and T3 (188.96 U/L vs. 402.39 and 129.78 U/L). Poisoning (T0) and administration of the antidote (T1) resulted in a several-fold increase in CAT activity compared to the other two measurement points. Similar trends were noted for glutathione reductase. Its activity in T0 was higher by over 50% than the measurements taken during and after completing the treatment. Insignificant, slight differences were indicated in GPx activity, whose elevated values, not exceeding 40%, were found only in T2 (after completing the treatment). No changes were found in GST activity. In the case of other markers of oxidative stress, significant differences in HNE concentration were found, which, after administration of the antidote, was higher (+70%) compared to the values indicated at admission. Similarly, in case of the catalase, the EG effect (T0) and ethanol administration (T1) caused a several-fold increase in HNE concentration compared to the other two measurements (T2 and T3). Analyzing TBARS levels, their values were ten times higher during and after completing the treatment. The significant time differences were also found in the concentration of total glutathione. EG (T0) caused temporary reduction of GSH/GSSG concentration, which in subsequent measurements (T1 to T3) were about 100% higher in comparison to the values indicated in T0, but did not differ significantly from the control values.
Influence of treatment applied, biochemical, and acid-base disturbances on oxidative stress markers
The correlation between selected oxidative stress markers and biochemical disturbances was indicated (Spearman’s correlation analysis). At admission (T0), the effect of EG concentration and elevated urea concentration on the TBARS level and AST activity as well as the CAT level were found. No other relationship was found between biochemical parameters disturbances and oxidoreductive balance. After initiation of the treatment, the effect of EA and elevated AST activity still influenced the CAT level. Higher AST activity correlated with lower CAT values. On the last day of hospitalization (T3), a relationship between some oxidative stress markers and biochemical parameters was found. The significant relationship between ALT and GR activities was noted like an increase in ALT activity correlated with GR activity. Urea concentration affected glutathione concentration (increased urea concentration with decreased total glutathione values). The concentration of hydrogen ions (H+) and HCO3− level had a major impact on the oxidoreductive balance at that time. A significant effect of the concentration of hydrogen ions and HCO3− on the oxidoreductive balance has been demonstrated. Increased concentration of (H+) correlated with higher GR activity. In addition, higher hydrogen ions also correlate with the glutathione level.
Discussion
For many years, a non-decreasing number of hospitalizations due to acute intoxications by non-food alcohols like ethylene glycol have been observed among the patients addicted to ethanol. 32 Most importantly, neurotoxicity and kidney degeneration induced by metabolic changes of ethylene glycol also affect the oxidoreductive imbalance during poisoning.33–35 Likewise, alcohol abuse leads to antioxidant system disorders associated with the production of reactive oxygen species that generate the oxidative stress.7,19 Hereby, their combined effects, associated with excessive alcohol consumption and EG intoxication, may also increase the oxidation system degeneration during the poisoning treatment. So far, no data have been published regarding the occurrence of oxidative stress in ethyl alcohol addicts who have been diagnosed and treated for acute ethylene glycol poisoning. So, in our article, we evaluate the oxidoreductive imbalance generated not only by EG, but also by the treatment used, such as ethanol administration during the hemodialysis procedure. In addition, we assess the correlation between oxidative status and liver and kidney disorders observed during EG intoxication.
In the EG group, chronically addicted to ethyl alcohol, poisoning was confirmed in range of toxic concentrations (0.95 ± 0.96 g/L). As expected, results carried out at the time of admission revealed metabolic acidosis on the base of reduced pH values (elevated hydrogen ion concentrations (H+)) and decreased levels of BE and HCO3− in blood. The determined values of serum renal parameters (Crea and Urea) indicated developing system failure, well documented in numerous case reports of accidental or intentional EG ingestion. 36 The hepatic enzymes’ (ALT and AST) activities were several times higher at the time of admission compared to the controls. The relationship between elevated liver markers and ethanol abuse is well established. The alcohol-induced liver damage is caused by the direct toxic effects of acetaldehyde and oxidative imbalance, leading to oxidative stress but there are no data that would confirm hepatotoxicity caused by EG intake in ethyl alcohol addicts.37,38 It is believed that all observed changes may not depend directly on alcohol addiction, but also on the ethanol concentration used during the treatment. The ethanol blood level (0.27 ± 0.88 g/L) determined on admission confirmed its previous consumption, although its level was not significant for the study. Nevertheless, higher EA concentration before hospitalization might be an additional factor disturbing the oxidoreductive balance as well as the ethanol abuse confirmed in the EG group.
The main purpose of the study was also to assess the impact of a given ethanol concentration (as an antidote during IHD) on selected laboratory parameters. Analyzing the time from intoxication to the end of treatment, it was found that the acid-base balance parameters were leveled and eventually returned to the reference value. Analyzing the parameters of renal function, dynamic growth was noticed, which was also maintained after administration of the antidote during hemodialysis, but statistical evaluation showed no significant differences between the obtained results. Nevertheless, levels of both renal parameters differed significantly from the values obtained in the C group. A fourfold blood measurement in the EG group provided detailed data on the oxidoreductive balance in the course of EG poisoning, indicating significant differences generated during the treatment.
The severity of lipid peroxidation processes was assessed based on measured TBARS and HNE levels in the patients’ blood. After antidote administration, we observed a sharp decrease of the TBARS level (by 60%) compared to the value at the time of admission, and then a dynamic, several dozen times increase on the last day of hospitalization (4.35 vs 54.34 μ
For the assessment of oxidative stress, the activities of selected enzymes involved in antioxidative defense were determined, including glutathione peroxidase (GPx), S-glutathione reductase (GR) and S-glutathione transferase (GST), catalase (CAT), and glutathione (GSH/GSSG) level. There were no significant changes in GST activity during the treatment, which may be due to its low participation in the antioxidative processes and stable GSH/GSSG concentration. In contrast to GST, GR activity gradually decreased from diagnosis to the end of treatment. This may confirm its active participation in antioxidant protection in poisoning, not excluding beneficial effects of the implemented treatment (administration of antidote during hemodialysis). CAT activity, high at the beginning of poisoning, slowly decreased until the last day of hospitalization, which indicates a slower consumption of CAT reserves than GR. In available studies conducted by Kukiełka and Cederbaum, it was shown that in ethylene glycol intoxication, catalase as an inhibitor of the conversion of EG to formaldehyde plays an important role in protecting the body against the adverse effects of oxidative stress caused by EG. 4 The activity of the last analyzed GPx remained at a similar level during hospitalization; a slight decrease was found only at the time of antidote administration and hemodialysis implementation.
In addition, the work assessed the impact of biochemical disorders, acid-base balance, and the implemented treatment on the body’s antioxidant defense status. The implemented treatment, with ethyl alcohol given and during the hemodialysis, was reflected in elevated AST and CAT activities. It has been shown that higher AST and CAT activities can be associated with the role or function of catalase in the antioxidant processes, with simultaneous liver damage. 44 Significance for the study was a comparison of the results obtained during the treatment with the measurements conducted on the last day of the hospitalization. A positive correlation between ALT and GR activities as well as between urea concentration and glutathione concentration were indicated. The level of ions (H+) also substantially affected the oxidoreductive balance. On the day of the patient’s discharge, higher (H+) ions correlated with increased GR activity as well as with elevated glutathione levels. Moreover, higher concentrations of hydrocarbons (HCO3−) correlated with the growth of GR activity. After the end of treatment, in the area of biochemical and organ changes, the significant statistical correlation was also found between the urea concentration and TBARS level, which meant that together with the growth of urea concentration (renal dysfunction), the indicator level was raised too (increased number of lipid peroxidation products).
In summary, significant changes in some biochemical and oxidative parameters were found, both at time of diagnosis and after treatment initiation (ethanol as an antidote and during hemodialysis). The most important differences including the activity of hepatic parameters (ALT and AST) and oxidative stress markers like catalase and glutathione reductase, correlation of the lipid peroxidation products level with urea concentration and hydrogen ions and hydrocarbons levels with GR activity have been shown. EG and its degradation products are cytotoxic compounds for cells and cause severe oxidative stress, among others through lipid peroxidation induction, and influence antioxidant enzymes, which are the body’s natural antioxidant barrier. The lipid hydroperoxides initiate and accelerate the process of lipid peroxidation, exerting a detrimental effect on the cell membrane, which loses its integrity. The results obtained in our studies confirm occurrence of oxidoreductive imbalance in the organism of alcohol-dependent patients with acute poisoning with ethylene glycol. Selected markers of oxidative stress reflect these changes in a varied way, especially after starting detoxification therapy.
The results presented in this paper provide new information and broaden the knowledge about the occurrence of oxidative stress in acute ethylene glycol intoxication as one of the important mechanisms of toxic effects in alcohol addicts. The obtained results may contribute to the improvement of treatment and establishing the correlation between the assessed markers and the clinical picture of poisoning. Some of the tested parameters can be used as a prognostic factor in assessment of its severity and the course of treatment of EG intoxication. All the identified changes indicate dynamic stimulation of the oxidative system and their dependence on biochemical changes and organ damage.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Note
BMI—body mass index (< 16.0—starvation, 16.0–16.99—emaciated, 17.0–18.49— underweight, 18.5–24.99—normal value, 25.0–29, 99—overweight, 30.0–34.99—obesity I degree, 35.0–39.99—obesity II degree, and ≥ 40.0—obesity degree III).


