Abstract
In this research, we investigated the toxicity responses of rat following a continuous 4 h inhalation exposure of only the head and nose to iron oxide nanoparticles (Fe3O4 NPs, size = 15–20 nm). The rats for the investigation were exposed to a concentration of 640 mg/m3 Fe3O4 NPs. Markers of lung injury and proinflammatory cytokines (interleukin-1β, tumor necrosis factor-α, and interleukin-6) in bronchoalveolar lavage fluid (BALF) and blood, oxidative stress in lungs, and histopathology were assessed on 24 h, 48 h, and 14 days of postexposure periods. Our results showed a significant decrease in the cell viability, with the increase in the levels of lactate dehydrogenase, total protein, and alkaline phosphatase in the BALF. Total leukocyte count and the percentage of neutrophils in BALF increased within 24 h of postexposure. Immediately following acute exposure, rats showed increased inflammation with significantly higher levels of lavage and blood proinflammatory cytokines and were consistent throughout the observation period. Fe3O4 NPs exposure markedly increased malondialdehyde concentration, while intracellular reduced glutathione and antioxidant enzyme activities were significantly decreased in lung tissue within 24-h postexposure period. On histological observation, the lung showed an early activation of pulmonary clearance and a size-dependant biphasic nature of the Fe3O4 NPs in causing the structural alteration. Collectively, our data illustrate that Fe3O4 NPs inhalation exposure may induce cytotoxicity via oxidative stress and lead to biphasic inflammatory responses in Wistar rat.
Keywords
Introduction
Transition metal nanoparticles, especially magnetite iron oxide (Fe3O4) nanoparticles (NPs) have found wide applications in a number of biomedical and bioengineering fields and have been widely used in a variety of in vivo applications such as magnetic resonance imaging, tissue repair, immunoassay, magnetic target drug delivery, cancer therapy, detoxification of biological fluids, cell separation, and hyperthermia. 1 –8 Extensive usage of Fe3O4 NPs in various fields has raised the possibility of human exposure to these nanoparticles especially in the manufacturing process as well as during handling, transport, and at usage. Hence, it is essential to identify whether there are any adverse health effects associated with these nanoparticles exposure. Furthermore, Organization for Economic Co-operation and Development (OECD) has established a Working Party on Manufactured Nanomaterials (WPMN) to concentrate on human health and environmental safety implications of manufactured nanomaterials and included iron nanoparticles as one of the 13 representative manufactured nanoparticles for its use of safety testing and risk assessment of manufactured nanomaterials. 9 A number of recent publications have reported that the Fe3O4 NPs induced toxicity. Exposure of iron nanoparticles to human microvascular endothelial cells increased the cell permeability through reactive oxygen species (ROS) oxidative stress-modulated microtubule remodeling. 10 Similarly, Hussain et al 11 reported a moderate toxic effect of these nanoparticles in rat liver-derived BRL 3A cell line that produced cytotoxicity at 250 µg/ml dose concentration by MTT assay. Intratracheal instillation of Fe3O4 NPs in mice at different dose concentrations induced subchronic inflammatory responses via oxidative stress. 12,13 Whole-body inhalation exposure of iron nanoparticles in mice showed no significant pathological changes following acute exposure, while subacute exposure showed increased inflammation, which is reversible within 3 weeks of postexposure. 14 In contrast, some in vitro studies reported that Fe3O4 NPs is nontoxic as no significant changes were observed in the cells treated with these nanoparticles. 15 –17
Most of the published literature addressing Fe3O4 NPs adverse health effects has used in vitro systems to assess their toxicity. Although it is preferable to investigate the toxicity of nanoparticles with in vitro assays because of their simplicity, being faster, more cost-effective, and pose no ethical problems compared with in vivo studies, researchers found little correlation between in vivo and in vitro toxicity results, especially with nanoparticles. 18 In most cases, toxic responses observed for nanoparticles in vitro studies were not exactly reproduced by in vivo studies. 19,20 Previous studies have shown that the inhalation as the most probable exposure route to assess metal nanoparticle toxicity as inhalation from an occupational standpoint is likely to be one of the most significant routes of exposure of nanoparticles. 21,22 Furthermore, Madl and Pinkerton 23 stated that the inhalation exposure provides a natural way for the delivery of toxicants, deposition and clearance patterns comparable with that in a real world setting, evaluate effects at all levels of the respiratory tract, and results in even distribution of delivered toxicant.
Therefore, in the current study, we made an attempt to assess the acute toxicological effects of Fe3O4 NPs in Wistar rats of either sex exposed by head and nose inhalation route as per OECD 403 guideline 24 with few modifications. In this study, a series of biochemical, hematological, bronchoalveolar lavage fluid (BALF) analysis for cytotoxicity and proinflammatory cytokine responses, and histopathological changes in target organs were assessed after 24 h, 48 h, and 14 days of a single 4-h continuous inhalation exposure of Fe3O4 NPs.
Materials and methods
Fe3O4 nanoparticles
The Fe3O4 NPs with 15–20 nm primary diameter, 99.5% purity, and ≥40 m 2 /g specific surface area was purchased from Nanostructured and Amorphous Materials Inc. (Houston, Texas, USA). These particles were prepared via a hydrothermal method. The average size and crystalline structure of the particles in the dry state was reconfirmed by scanning electron microscopy (SEM; Figure 1(a)) and x-ray diffraction (XRD) analyses, respectively, whereas hydrodynamic particle size and zeta potential measurement (Figure 1(b)) in deionized water (wet state) was performed by dynamic light scattering (DLS) using Malvern Zetasizer 3000HS (Malvern, Bangalore, India). The characterizations of the Fe3O4 nanoparticles were shown in Figure 1 and Table 1.

(a) Scanning electron microscope (SEM) image and (b) Zeta (ζ) potential of iron oxide nanoparticle (Fe3O4 NPs).
Characterization of Fe3O4 NPs

Fe3O4 NPs: iron oxide nanoparticles.
aAccording to the manufacturer.
bUsing dynamic light scattering.
cUsing BET (Brunauer–Emmett–Teller) analysis.
dUsing x-ray diffraction.
Animals
A total of 72 specific pathogen-free Wistar rats of either sex, aged 8 weeks, and weighing 185–200 g were obtained from animal house, International Institute of Biotechnology and Toxicology (IIBAT). Females used in the study were nulliparous and nonpregnant. Animals were allowed to adapt to the animal room conditions for a week period before the initiation of the study. Every three animals with same sex were housed in stainless steel cages and the environmental conditions were set at a temperature of 22 ± 1°C, relative humidity of 54 ± 4%, and a 12-h light/dark cycle. Commercial pellet diet and reverse osmosis water were available ad libitum. After acclimation, animals were divided into experimental and control groups randomly with each group consisted of 36 animals of 18 males and 18 females. The study was performed with the approval of Institutional Animal Ethics Committee (IAEC), IIBAT.
Inhalation exposure
The current inhalation study was conducted with the aid of an inhalation exposure unit (Ch. Technologies, USA). The unit has three major modules: an aerosol generator—Wright Dust Feeder (BGI, Inc.); a rodent head and nose-only inhalation exposure chamber, and aerosol concentration measurement device—gravimetric single filter (BGI, Inc.).
Prior to initiation of the animal exposure, a pilot study was conducted by generating Fe3O4 NPs aerosols continuously for 4 h in an inhalation chamber in the absence of rats to understand the particle size distribution of the generated aerosol. Measurements were made using Mercer–Seven stage cascade impactor (Ch. Technologies, USA). The average actual aerosol concentration of 640 mg/m3 was obtained with mass median aerodynamic diameter (MMAD) and geometric standard deviation (GSD) within respirable range as per OECD 403.
Based on the pilot study, rats in the experimental group were exposed continuously for a period of 4 h at an average actual aerosol concentration of 640 mg/m3 of Fe3O4 NPs. Similarly, rats in the control group were exposed to clean air. The site at the breathing zone of the rats was assessed periodically for aerosol concentration, aerosol particle size distribution, temperature, relative humidity, and oxygen concentration. The distribution of aerosol particles had an average MMAD of 2.25 µm and an average GSD of 2.56. The dynamics of the chamber had temperature ranging from 21 ± 0.8°C and relative humidity 52–57% at the breathing zone. Oxygen concentration was above 20% all through the experiment. All the dynamics indicate that the aerosols were not static.
Biochemical and hematology analysis
After 24 h, 48 h and 14 days of Fe3O4 NPs inhalation exposure, six animals per sex of each group at respective time points were anesthetized with isoflurane and about 2 ml of blood was collected per animal from the retro-orbital venous plexus into prelabeled EDTA and heparinized vials for hematological and biochemical analysis, respectively. Hematology analysis includes erythrocytes, hemoglobin, hematocrit (HCT), mean corpuscular volume (MCV), mean corpuscular hemoglobin (MCH), MCH concentration (MCHC), platelet count, leukocytes, and leukocyte differential count analyzed using fully automated hematology analyzer (Advia 120, Siemens, USA). Heparinized plasma used for the biochemical analysis was obtained by centrifugation at 1500g for 20 min. Biochemical analysis includes glucose, urea, creatinine, total cholesterol, triglycerides, aspartate transaminase, alanine transaminase, alkaline phosphatase (ALP), total protein (TP), albumin, calcium, and phosphorus analyzed using fully automated biochemistry analyzer (Dimension Xpand plus, Siemens, USA). Serum electrolytes (sodium and potassium) were assayed using electrolyte analyzer (Humalyte, Human GmbH, Germany).
Bronchoalveolar lavage analysis
After blood collection, animals were euthanized with an overdose of sodium pentobarbital (150–200 mg/kg body weight) via an intraperitoneal injection and the lungs of control and treated group rats were lavaged immediately with phosphate-buffered saline for two times. An average of above 90% of the total volume instilled was retrieved on both the times and the amounts did not differ among the groups. The two lavage collections were pooled and centrifuged at 300g for 10 min at 4°C and 3 ml supernatant was collected for biochemical and cytokine assays. Biochemical assays like lactate dehydrogenase (LDH), ALP, and TP were analyzed spectrophotometrically using Dimension Xpand plus (Siemens, USA) clinical chemistry analyzer. The resulting cell pellet was resuspended in RPMI 1640 and was used for the enumeration of cell viability as well as total and differential cell counts. Cell viability was determined with 0.2% trypan blue and total cells were counted in a hemocytometer. Differential cell count slides were prepared by cytocentrifuge and fixed in methanol and were stained with Geimsa stain. A total of 500 cells of each slide were counted under oil-immersion microscope with 1000× magnification for alveolar macrophages (AMs), neutrophils, and lymphocytes, which were identified by their characteristic cell shapes.
Measurement of proinflammatory cytokines by enzyme-linked immunosorbent assay
The concentrations of proinflammatory cytokines (interleukin-1β (IL-1β), tumor necrosis factor-α (TNF-α), and interleukin-6 (IL-6)) in the BALF and plasma were determined using commercially available enzyme-linked immunosorbent assay kits (Ray Biotech, Georgia, USA). Briefly, 100 µl of BALF, plasma, or standard antibodies were added to the respective antibody precoated individual wells of a microplate and incubated overnight at 4°C. The plates were washed and 100 µl biotinylated antibodies was added to each well and was stirred gently before incubating for 1 h at room temperature. The plates were washed again and further incubated with streptavidin–HRP for 45 min before detection with TMB solution. Finally, reactions were stopped by adding 2 M H2SO4, and the absorbance was measured at 450 nm (STATFAX-2100, CPC Diagnostics, India). The amount of cytokine was calculated from the linear portion of the generated standard curve. 25
Measurement of oxidative stress in lungs
One part of the lavaged lung of each animal was thawed and homogenized 10 times (w/v) by homogenizer in ice-cold 0.1 M phosphate buffer (pH 7.4). Aliquots of homogenates from rat lung were separated and used to determine protein, lipid peroxidation, and glutathione. While the remaining homogenates were centrifuged at 15,000g for 60 min at 4°C and the supernatant was then used to assay glutathione peroxidase (GSH-Px), superoxide dismutase (SOD), and catalase activity. Lipid peroxidation, reduced glutathione (GSH), and GSH-Px in lung were assayed as per our previous publication, 26 while SOD and catalase were assayed as per methods described by Kakkar et al and Aebi, respectively.
SOD enzyme activity
SOD activity of the lung tissue was analyzed by the method described by Kakkar et al. 27 Assay mixture contained 0.1 ml of sample, 1.2 ml of sodium pyrophosphate buffer (pH 8.3, 0.052 M), 0.1 ml phenazine methosulphate (186 µM), 0.3 ml of 300 µM nitroblue tetrazolium, and 0.2 ml NADH (750 µM). Reaction started by the addition of NADH. After incubation at 30°C for 90 s, the reaction was stopped by the addition of 0.1 ml glacial acetic acid. Reaction mixture was stirred vigorously with 4.0 ml of n-butanol. Mixture was allowed to stand for 10 min, centrifuged, and butanol layer was separated. Color intensity of the chromogen in the butanol was measured at 560 nm spectrophotometrically and concentration of SOD was expressed as units per milligram of protein.
Catalase enzyme activity
Catalase activity was measured by the method of Aebi. 28 A 0.1-ml of supernatant was added to cuvette containing 1.9 ml of 50 mM phosphate buffer (pH 7.0). Reaction was started by the addition of 1.0 ml of freshly prepared 30 mM H2O2. The rate of decomposition of H2O2 was measured spectrophotometrically from changes in absorbance at 240 nm. Activity of catalase was expressed as units per milligram of protein.
Histopathology
The remaining part of the lung along with the lung-associated lymph nodes (tracheobronchial and mediastinal lymph nodes), liver, spleen, kidneys, thymus, and brain were collected from all animals and preserved in 10% neutral buffered formalin. The lungs were inflated with 10% neutral-buffered formalin before preservation. The organs were processed in vacuum infiltration tissue processor (Sakura Tissue-Tek® VIPTM, Japan), embedded (Sakura Tissue-Tek® TECTM, Japan) with paraffin wax, sectioned (Microm GmbH, Germany) at approximately 5 µm thick, stained (Leica ST5020–Autostainer, Germany) with hematoxylin and eosin, viewed under light microscope (Nikon 50i, Japan), and images were captured through an image analyzer (Q–Imaging systems, Canada). The identity and analysis of the pathology slides were blind to the pathologist.
Statistical analysis
Statistical analyses were conducted with NCSS 2007 software. Data were expressed as mean ± SD (n = 6). Student’s t test was carried out to illustrate the significant difference between the treated groups and the respective control groups and the one-way analysis of variance was carried out to compare the differences among the time points within the group. Levels of significance were represented for each result. The Spearman rank correlation test was used to analyze correlations of proinflammatory cytokines in BALF and blood.
Results
Fe3O4 NPs characterization
Prior to the conductance of acute inhalation exposure, characterization of the Fe3O4 NPs was performed (Figure 1 and Table 1). We employed SEM and DLS techniques for size determination of the Fe3O4 NPs in dry and wet state, respectively, and SEM image showed the morphology of Fe3O4 NPs. The mean size and SD was calculated from measuring over nine random fields of view. The mean ± SD of Fe3O4 NPs was 48.18 ± 7.54 nm. The mean hydrodynamic diameter of the Fe3O4 NPs in deionized water as confirmed by DLS analysis was 651.2 nm and the crystal structure as characterized by XRD was tetrahedral in shape and the zeta (ζ) potential was −15.9. The surface area of Fe3O4 NPs was ≥40 m2/g as stated by the manufacturer.
Biochemical and hematological analysis of blood
None of the animals of either sex at any time tested showed any significant changes in serum biochemical parameters except in triglycerides, which was significantly different on 24 h in male rats (p < 0.05; Supplement Table 1A and B). Similarly, acute inhalation exposure of Fe3O4 NPs did not alter any hematological parameters including erythrocytes, hemoglobin, and HCT in any of the animals at any tested time (Supplement Table 2A and B).
Histopathological changes in lungs and LALN after acute inhalation exposure of Fe3O4 NPs (n = 12, 6 males + 6 females)a

Fe3O4 NPs: iron oxide nanoparticles; LALN: lung associated lymph nodes.
aThe lung and tracheobronchial lymph node tissues were harvested at 24 h, 48 h, and 14 days after acute inhalation exposure of Fe3O4 NPs. Number means the number of positive samples among 12 tested respective tissues.
Biochemical and cytological assessments in BALF
Cell viability was measured to assess the cytotoxic potential of Fe3O4 NPs, when exposed through inhalation route. As shown in Figure 2, a significant decrease (p < 0.001) in cell viability appeared in all the three postinhalation time points with maximum decrease within 24 h of postexposure, where cell viability dropped to about 60% of control animals in either sex. However, a time-dependent recovery was observed with a significant increase (p < 0.001) in cell viability within 48 h of postexposure in males and 14 days of postexposure in female rats (Figure 2).
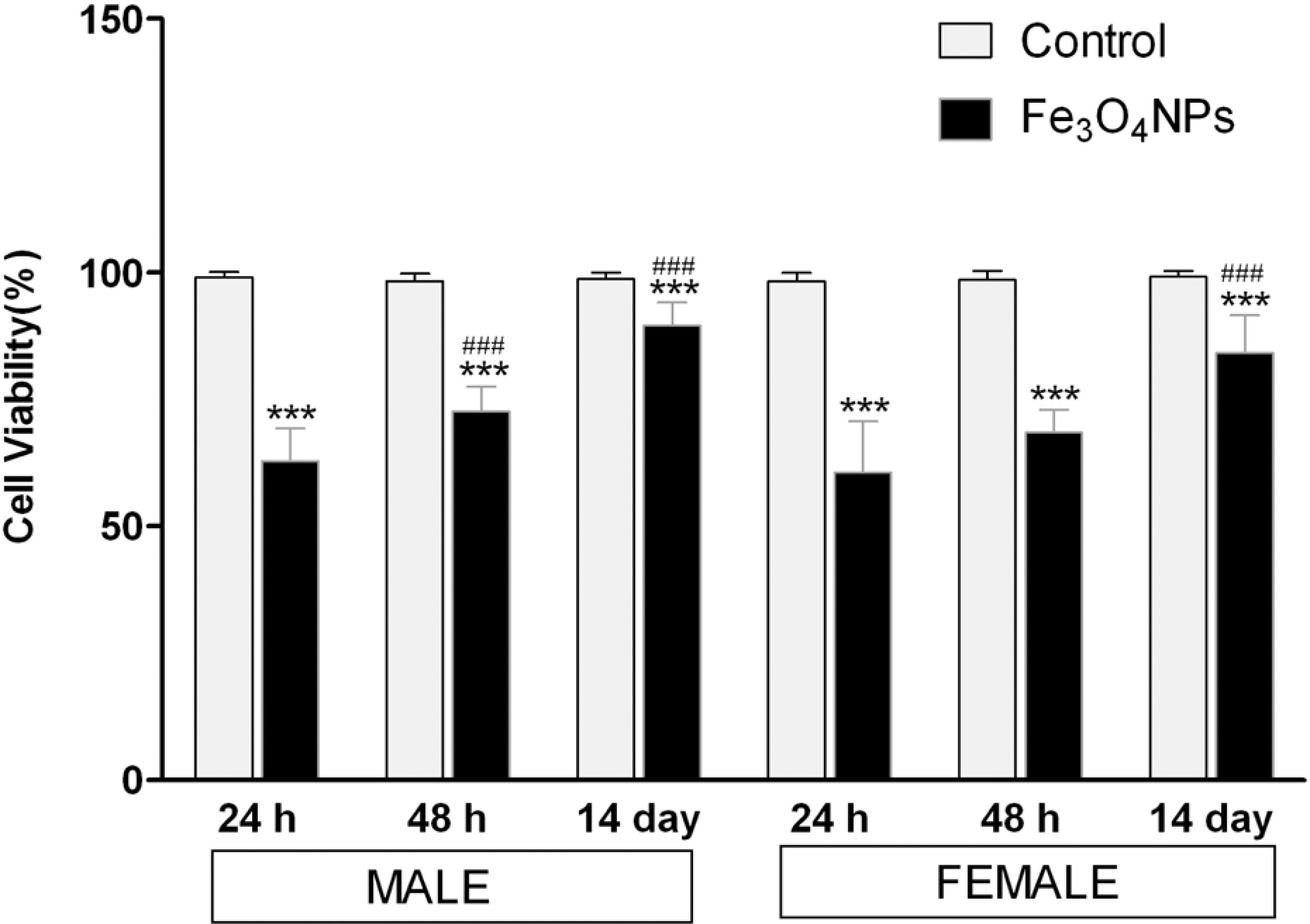
The effect of acute inhalation exposure of Fe3O4 NPs on the cell viability in BALF. BALF was collected from rats at the designated time points of postinhalation exposure of Fe3O4 NPs. Values are mean ± SD; six animals per group. *Significantly different from respective control group: ***p < 0.001. #Significantly different from respective 24 h time point: ### p < 0.001. Fe3O4 NPs: iron oxide nanoparticle; BALF: bronchoalveolar lavage fluid.
As shown in Figure 3, the BALF cells were significantly increased by about 2.5-fold at 24 h of postexposure period in either sex compared with the respective control animals (p < 0.001). However, these cells showed significant decrease (p < 0.05) in a time-dependent manner, as it reached about 1.3-fold increase at the end of 14 days of postexposure period in either sex.
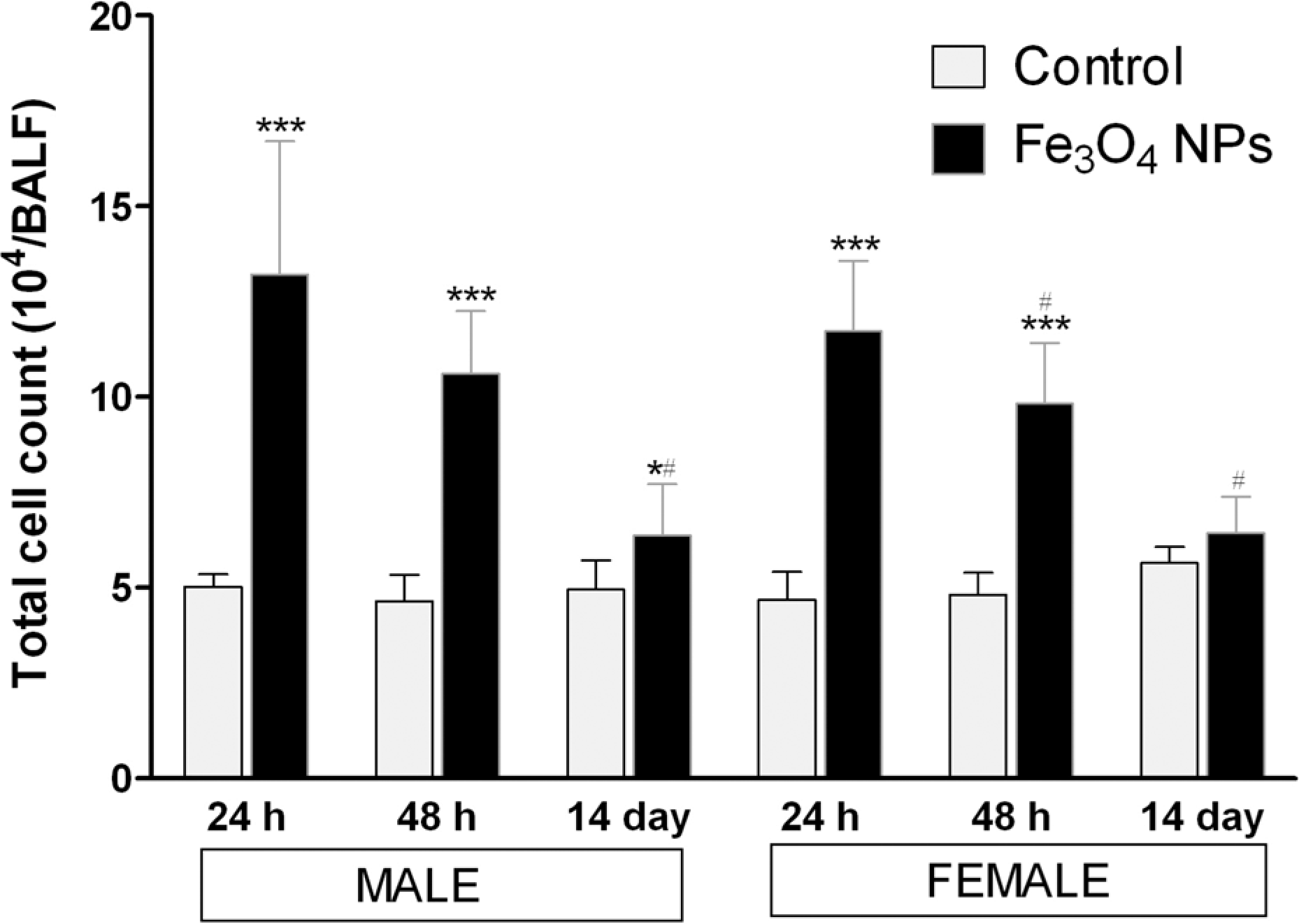
Changes in total cell count in BALF of rats at the designated days of postinhalation exposure of Fe3O4 NPs. Values are mean ± SD; six animals per group. *Significantly different from respective control group: *p < 0.05; ***p < 0.001. #Significantly different from respective 24 h time point: # p < 0.05. Fe3O4 NPs: iron oxide nanoparticle; BALF: bronchoalveolar lavage fluid.
Neutrophils, a characteristic indicator of acute inflammation, showed a time-dependent decrease in BALF upon Fe3O4 NPs inhalation exposure. The percentage of neutrophils in treated animals showed a maximum of fivefold increase within 24 h of postexposure and decreased in a time-dependent manner, with females showing promising recovery (p < 0.001) than males (p < 0.05) at the end of 14 days of postexposure period when compared with 24 h of postexposure period. The percentage of AMs was different with neutrophils, which showed a time-dependent recovery in treated animals of either sex. The distribution rate of lymphocytes in BALF showed a weak response when compared with neutrophils with a maximum increase at 24 h of postexposure of Fe3O4 NPs (Figure 4(a) and (b)).

Percentage of macrophages, neutrophils, and lymphocytes in BALF of male (a) and female (b) rats at the designated days of postinhalation exposure of Fe3O4 NPs. Values are mean ± SD; six animals per group. *Significantly different from respective control group: *p < 0.05; **p < 0.01; ***p < 0.001. #Significantly different from respective 24 h time point: # p < 0.05; ## p < 0.01; ### p < 0.001. Fe3O4 NPs: iron oxide nanoparticle; BALF: bronchoalveolar lavage fluid.
As illustrated in Figure 5(a), LDH release, a marker of cell membrane damage, was increased significantly throughout the observation period (p < 0.001). LDH activity in BALF of Fe3O4 NPs shows that the animals of either sex showed a time-dependent decreasing manner and found to be around threefold, 2.0-fold, and 1.6-fold increases when compared with control animals at the end of 24 h, 48 h, and 14 days of postexposure periods, respectively. Similar trends were shown by TP concentration and ALP activity in BALF, which were increased throughout the exposure period, reaching a maximum at 24 h of postexposure (p < 0.001) and decreasing in a time-dependent manner. TP concentration in BALF of Fe3O4 NPs exposed animals of either sex showed around 2.7-fold, 2.0-fold, and 1.4-fold increases when compared with control animals at the end of 24 h, 48 h, and 14 days of postexposure periods, respectively (Figure 5(b)). Similarly, ALP activity in BALF of Fe3O4 NPs exposed animals of either sex showed around 1.5-fold, 1.4-fold, and 1.2-fold increases when compared with control animals at the end of 24 h, 48 h, and 14 day of postexposure periods, respectively (Figure 5(c)).

Changes in LDH (a), TP (b) and ALP (c) concentrations in BALF after acute inhalation exposure of Fe3O4 NPs. BALF was collected from rats at the designated days of postinhalation exposure of Fe3O4 NPs. Values are mean ± SD; six animals per group. *Significantly different from respective control group: *p < 0.05; **p < 0.01; *** p < 0.001. #Significantly different from respective 24 h time point: # p < 0.05; ## p < 0.01; ### p < 0.001. LDH: lactate dehydrogenase; TP: total protein; ALP: alkaline phosphatase; Fe3O4 NPs: iron oxide nanoparticle; BALF: bronchoalveolar lavage fluid.
Proinflammatory response Fe3O4 NPs
The levels of proinflammatory cytokines (IL-1β, TNF-α, and IL-6) released into the BALF in Fe3O4 NPs inhaled rats were increased significantly throughout the observation period (p < 0.001) and reached the maximum level within 24 h of postexposure period in either sex. The increased ratio of IL-1β to the nontreated control group in BALF was 7.8-fold at 24 h, 4.7-fold at 48 h, and 3.5-fold at 14 days of postexposure period in male rats and 7.5-fold at 24 h, 5.2-fold at 48 h, and 2.9-fold at 14 days of postexposure period in female rats (Figure 6(a)). Similarly, TNF-α in BALF reached a maximum at 24 h after postexposure and the level was gradually decreased in a time-dependent manner. The increased ratio to the nontreated control group were 18.1-fold at 24 h, 12.3-fold at 48 h and 5.1-fold at 14 days of postexposure period in males and 19.9-fold at 24 h, 14.5-fold at 48 h and 5.4-fold at 14 days of postexposure period in females (Figure 6(b)). Also, IL-6 reached maximum in BALF at 24 h after exposure and was gradually decreased in a time-dependent manner in either sex. The increased ratio to the nontreated control group was 13.7-fold, 9.1-fold, and 2.9-fold after 24 h, 48 h, and 14 days of postexposure period, respectively, in male rats. Similarly, in female rats the increased ratio to the nontreated control group was 12.8-fold, 8.7-fold, and 2.3-fold after 24 h, 48 h, and 14 days of postexposure period, respectively (Figure 6(c)). Similar time-dependent decreasing trends were observed in the levels of proinflammatory cytokines (IL-1β, TNF-α, and IL-6) in blood as these three proinflammatory cytokines reached a maximum level within 24 h of postexposure and gradually decreased at the end of 14 days of postexposure period. The increased ratio of IL-1β to the nontreated control group in blood was 3.2-fold at 24 h, 2.3-fold at 48 h, and 1.7-fold at 14 days of postexposure period in males and 2.8-fold at 24 h, 2.5-fold at 48 h, and 1.5-fold at 14 days of postexposure period in females, respectively (Figure 6(d)). Similarly, the increased ratio of TNF-α in the blood of treated animals to the nontreated control group were 4.5-fold at 24 h, 3.4-fold at 48 h, and 2.5-fold at 14 days of postexposure period in male rats and 4.8-fold at 24 h, 2.8-fold at 48 h, and 2.5-fold at 14 days in female rats, respectively, (Figure 6(e)). Also, IL-6 reached a maximum at 24 h after treatment and was gradually decreased in a time-dependent manner. The increased ratio to the nontreated control group was 11.3-fold, 8.6-fold, and 4.3-fold after 24 h, 48 h, and 14 days of postexposure period, respectively, in male rats. Similarly, in female rats, the increased ratio to the nontreated control group was 8.9-fold, 8.1-fold, and 4.5-fold after 24 h, 48 h, and 14 days of postexposure period, respectively (Figure 6(f)). As shown in Figure 7, the induction levels of these cytokines exhibited a significant correlation in BALF and blood throughout the observation period. The individual levels of IL-1β and IL-6 in BALF correlated significantly and strongly with respective blood concentrations (r s = 0.8, p ≤ 0.0001 for IL-1β and r s = 0.9, P ≤ 0.0001 for IL-6), while individual TNF-α levels in BALF correlated, but less well with respective blood levels throughout the observation period (r s = 0.5, p = 0.0033).

Time-dependent changes in IL-1β, TNF-α, and IL-6 levels in BALF (a–c) and in blood (d–f) after acute inhalation exposure of Fe3O4 NPs. Values are mean ± SD; six animals per group. *Significantly different from respective control group: **p < 0.01; ***p < 0.001. #Significantly different from respective control group: # p < 0.05; ### p < 0.001. IL-1β: interleukin-1β; TNF-α: tumor necrosis factor-α; IL-6: interleukin-6; Fe3O4 NPs: iron oxide nanoparticle; BALF: bronchoalveolar lavage fluid.

Correlation of IL-1β (a), TNF-α (b), and IL-6 (c) levels between BALF and blood after acute inhalation exposure of Fe3O4 NPs. r s: Spearman rank correlation coefficient; IL-1β: interleukin-1β; TNF-α: tumor necrosis factor-α; IL-6: interleukin-6; Fe3O4 NPs: iron oxide nanoparticle; BALF: bronchoalveolar lavage fluid.
Oxidative stress in lung
In order to elucidate the lipid peroxidation induced by Fe3O4 NPs, the malondialdehyde (MDA) concentration in rat lung homogenate was measured. As shown in Figure 8, the MDA levels were increased significantly throughout the observation period (p < 0.001) with a peak within 24 h of postexposure period in either sex compared with the respective control animals. However, treated rats exhibited a time-dependent recovery with a significant decrease (p < 0.001) at the end of 14 days of postexposure when compared with 24 h of postexposure period in either sex.

Levels of MDA content in lung homogenates after acute inhalation exposure of Fe3O4 NPs. Values are mean ± SD; six animals per group. *Significantly different from respective control group: ***p < 0.001. #Significantly different from respective 24 h time point: ### p < 0.001. MDA: malondialdehyde; Fe3O4 NPs: iron oxide nanoparticle.
Following acute inhalation exposure to Fe3O4 NPs, the GSH level exhibited a time-dependent increase in lung homogenate. As illustrated in Figure 9, the GSH levels decreased to a minimum within 24 h of postexposure (p < 0.001) and gradually increased toward the end of 14 days of postexposure period. The antioxidative enzymes, GSH-Px and SOD, showed a significant decrease in their activities in treated lungs up to 48 h of postexposure period and exhibited a time-dependent recovery in the lung of treated animals at the end of 14 days of postexposure period in either sex (Figures 10 and 11), while catalase activity showed a significant decrease throughout the observation in either sex (Figure 12).

Concentrations of GSH in lung homogenates after acute inhalation exposure of Fe3O4 NPs. Values are mean ± SD; six animals per group. *Significantly different from respective control group: *p < 0.05; **p < 0.01. #Significantly different from respective 24 h time point: ### p < 0.001. GSH: glutathione; Fe3O4 NPs: iron oxide nanoparticle.
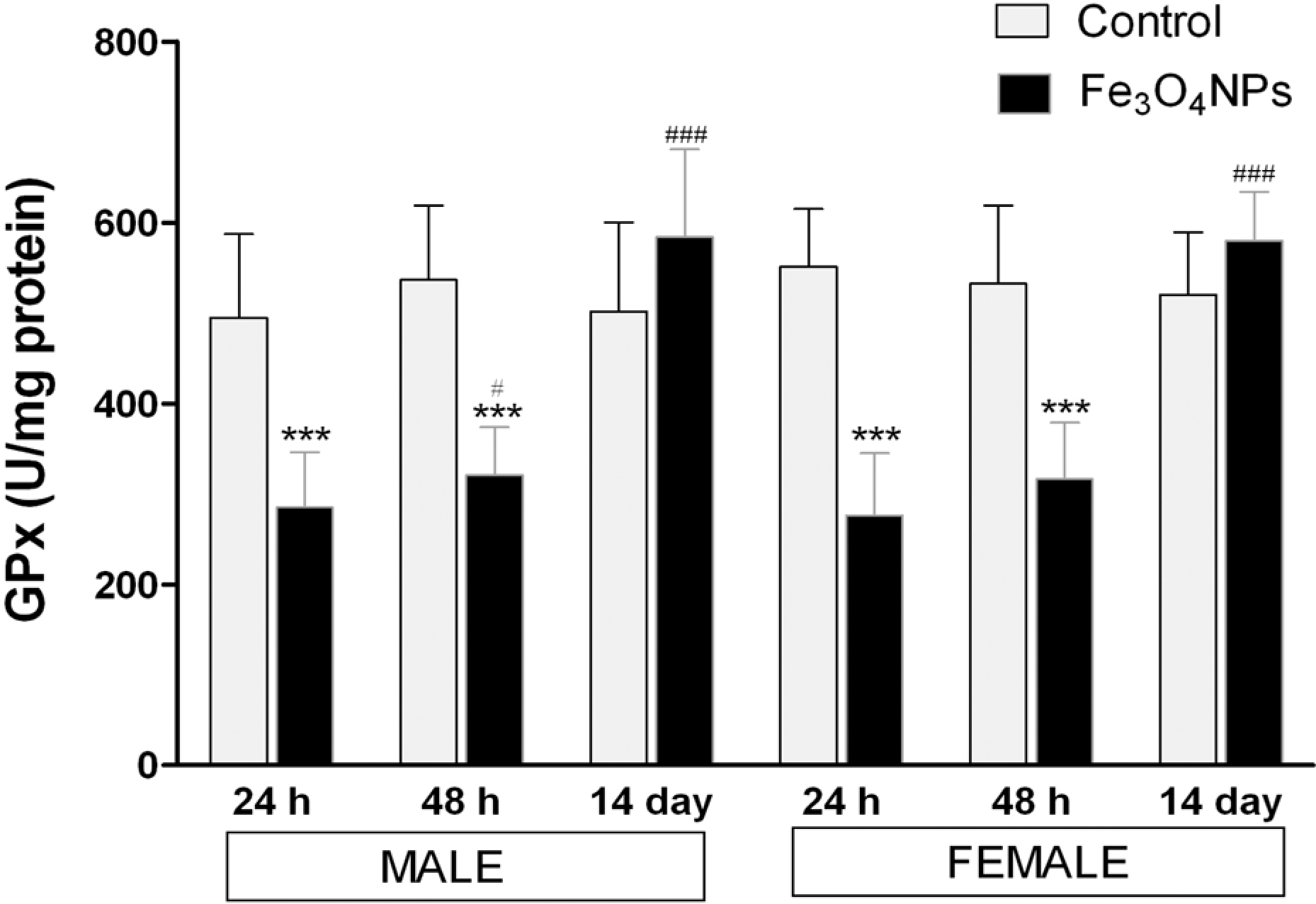
Activity of GSH-Px in lung homogenates after acute inhalation exposure of Fe3O4 NPs. Values are mean ± SD; six animals per group. *Significantly different from respective control group: ***p < 0.001. #Significantly different from respective 24 h time point: # p < 0.05; ### p < 0.001. GSH-Px: glutathione peroxidase; Fe3O4 NPs: iron oxide nanoparticle.

Activity of SOD in lung homogenates after acute inhalation exposure of Fe3O4 NPs. Values are mean ± SD; six animals per group. *Significantly different from respective control group: ***p < 0.001. #Significantly different from respective 24 h time point: # p < 0.05; ### p < 0.001. SOD: superoxide dismutase; Fe3O4 NPs: iron oxide nanoparticle.
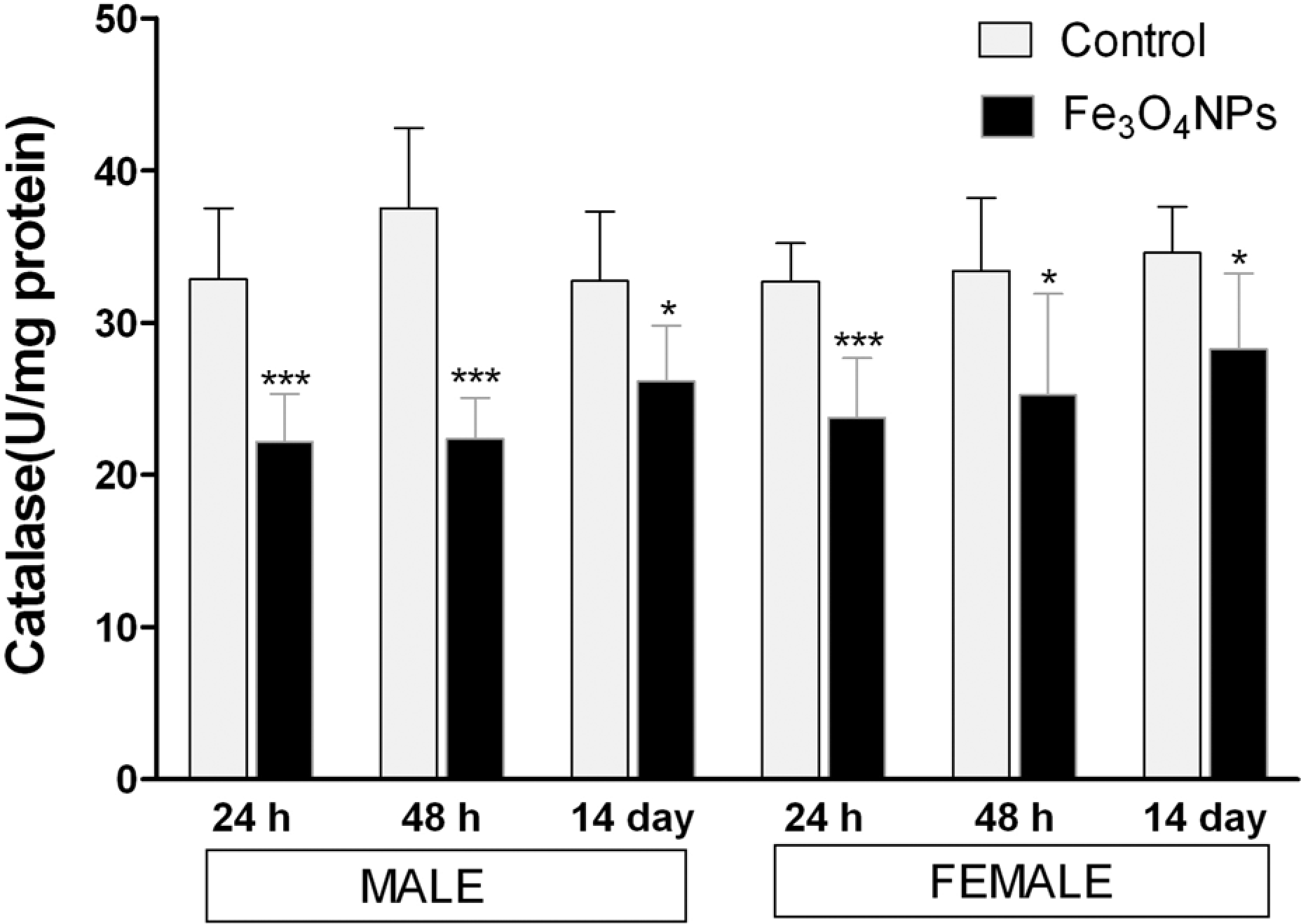
Activity of catalase in lung homogenates after acute inhalation exposure of Fe3O4 NPs. Values are mean ± SD; six animals per group. *Significantly different from respective control group: ***p < 0.001. Fe3O4 NPs: iron oxide nanoparticle.
Histopathological changes
As shown in Table 2 and Figure 13, on histological examination, the Fe3O4 NPs could be identified in all the three postexposure periods, that is, 24 h, 48 h, and 14 days and appeared as dark brown, irregularly round-to-oval shaped, smooth, discrete small, and large agglomerated masses. The small agglomerated masses were predominantly phagocytosed by the AMs (Figure 13(c)) and the large masses were deposited as acellular free form on the luminal surface of bronchi, bronchiole, and alveoli. The pulmonary interstitium revealed the NPs in both phagocytosed and nonphagocytosed form at all postexposure periods with inflammatory changes described below. Appearance of AMs overloaded with the phagocytosed discrete single to multiple irregularly (Figures 13(b) and (c)) gobular intracytoplasmic small agglomerated masses of NPs were observed in the airways and interstitium early at 24 h of postexposure period along with nonphagocytosed free Fe3O4 NPs in alveolar interstium (Figure 13(d)). Emigration and translocation of these masses to the intrapulmonary lymphoid tissues (BALT) and to the extrapulmonary tracheobronchial lymph nodes (lung-associated lymph nodes (LALN)) were also observed at this postexposure period (Figure 13(e) and (f)). Both these findings were consistent in all the exposed animals at all postexposure periods (Figure 13(m)). Fe3O4 NPs induced minimal-to-moderate, multifocal alveolitis accompanied with perivascular cuffing of mononuclear inflammatory cells were noticed at 48 h and persisted up to 14 days of postexposure period (Figure 13(g)). The lesion was characterized by the presence of both phagocytosed and nonphagocytosed Fe3O4 NPs, moderate alveolar wall hyperplasia with variable interstitial inflammatory reaction and infiltrated masses of mononuclear inflammatory cells in the alveolar lumen (Figure 13(k)). After the 14 days of postexposure, except the alveolitis, four of six males and five of six females showed multifocal foreign body type low-grade granulomatous inflammation surrounding the agglomerated masses of Fe3O4 NPs (Figure 13(l)). However, there were no signs of early fibrosis observed in any of the observation period. The above examination shows that nanoparticle-induced histological changes were found to be consistent in either sex. Other extrapulmonary organs that were examined did not show any Fe3O4 NPs-induced toxic responses or the existence of these NPs.

Histopathology of the lungs and the tracheobronchial lymph nodes at 24 h, 48 h, and 14 days after acute inhalation exposure of Fe3O4 NPs (haematoxylin–eosin stained). (a) Control lung (×400); (b) Fe3O4 NPs laden AMs (arrow) in alveoli on 24 h of postexposure (×1000); (c) phagocytosed Fe3O4 NPs in bronchiolar lumen (arrow) on 24 h of postexposure period (×1000); (d) deposition of nonphagocytosed free Fe3O4 NPs (arrow) in alveolar interstitium on 24 h of postexposure period (×1000); (e) Fe3O4 NPs (arrow) in BALT on 24 h of postexposure period (×1000); (f) translocation of Fe3O4 NPs as free agglomerated mass (white arrow) and as intracytoplasmic phagocytosed particles (yellow arrow) in the tracheobronchial lymph nodes on 24 h of postexposure period (×1000); (g) perivascular cuffing of mononuclear inflammatory cells (thin arrow) and minimal alveolitis (block arrow) in the lung on 48 h of postexposure (×200); (h) rare disruption of Fe3O4 NPs laden AM (arrow) on 48 h of postexposure period (×1000); (i) persistence of Fe3O4 NPs laden AMs in alveoli (arrow) on 14 days of postexposure (×1000); (j) persistence of deposited nonphagocytosed free Fe3O4 NPs on the bronchiolar lumen (arrow) on 14 days of postexposure period (×1000); (k) alveolitis characterized by alveolar wall hyperplasia and accumulation of mononuclear inflammatory cell in alveolar space on 14 days of exposure (×100); (l) multifocal low-grade granulomas with Fe3O4 NPs laden AMs (arrow) in pulmonary parenchyma at 14-day postexposure (×400); (m) presence of Fe3O4 NPs in the tracheobronchial lymph nodes (arrow) on 14-day of postexposure period (×1000). Fe3O4 NPs: iron oxide nanoparticle; AMs: alveolar macrophages; BALF: bronchoalveolar lavage fluid.
Discussion
The objective of this study was to assess the acute toxic potential of Fe3O4 NPs when exposed via head and nose only inhalation in Wistar rat. The majority of published works investigating pulmonary effects of nanoparticles in vivo have been conducted utilizing routes with limited biological relevance (e.g. intratracheal instillation, intraperitoneal injection, and intrapharyngeal aspiration). Here, we conducted this study by aerosol inhalation exposure that has the greatest relevance toward human exposure to nanoparticles.
In the current study, rats were exposed by head and nose inhalation route at a maximum attainable concentration of Fe3O4 NPs (640 mg/m3) for 4 h according to OECD 403 guideline and a series of biochemical, hematological, BALF analysis for cytotoxicity and proinflammatory cytokine responses, and histopathological changes in target organs, especially lung, were assessed after 24 h, 48 h and 14 days of postexposure periods. This time points were selected based on our previous work on acute inhalation exposure of CeO2 NPs, where we observed maximum nanoparticle induced cytotoxicity and proinflammation within the span of 48 h of postexposure and continued up to 14 days of postexposure. 26 The dosage of 640 mg/m3 at which animals were exposed seemed to be high and may be unlikely in biomedical setting, where these magnetite particles were widely used that exposures would occur at such high concentrations. However, it is also important to know the acute adverse effect at higher dosage of nanoparticles for the worst case of exposure. 29 Hence, we conducted the current study with the maximum attainable concentration to study the adverse effects exhibited by these Fe3O4 NPs in rats.
Before initiating any toxicity study with nanoparticles, the characterization becomes more extensive and indispensable. 30 –32 Among all attributes for characterization, size is the most important criterion that differentiates a nanoparticle for functionality. Hence, we employed SEM and DLS techniques for the size determination in dry and wet state, respectively. As shown in Table 1, there existed the differences in size of these nanoparticles in dry state measured by the manufacturer and us by TEM and SEM techniques, respectively. The differences in these size variations can be attributed due to the different techniques employed by the manufacturer and us. Although the rats were exposed to Fe3O4 NPs aerosols in the absence of media, DLS was employed to study the agglomeration or aggregation nature of these particles in distilled water and the mean hydrodynamic size was approximately around 13 times larger than the size in dry state. This suggests that these nanoparticles were tended to agglomerate. It has been known that the toxicity of nanoparticles is closely related to the surface area, and it may be expected that small-sized particles with more surface area are more toxic than large-sized particles. 33 Hence, the BET surface area of ≥40 m2/g reported by the supplier may be considered as one possible reason of these Fe3O4 NPs-induced pulmonary toxicity in rats.
None of the rats in the current study showed morbidity, mortality, or any clinical signs of toxicity. No significant changes in blood biochemical and hematological parameters were observed except in triglyceride, which was significantly different on 24 h in male rats (Supplement Tables 1 and 2). Although significant difference exits keeping in view of broad reference limits in rats for triglycerides, this changes may not be attributed for Fe3O4 NPs exposure.
There are many in vitro and in vivo studies to date stating that nanoparticles generate ROS leading to oxidative stress and can cause cell death. 34 –37 As shown in Figure 2, a significant decrease in cell viability of BALF up to14 days of postinhalation exposure of Fe3O4 NPs was observed suggesting that nanoparticle induced cytotoxicity (Figure 2). This was further supported by biochemical assays in BALF. The activity of LDH along with the concentration of TP significantly increased in the particle-exposed animals compared with those of the controls, throughout the observation period (Figure 5(a) and (c)). LDH, a cytoplasmic enzyme is used primarily as a cell injury indicator, 38,39 while TP as an indicator of injury to the alveolar epithelial-capillary barrier. 40 As shown in Figure 5(c), ALP increased in BALF up to 48 h of postexposure period (Figure 5(c)). ALP is a leaking enzyme specific to type-II alveolar epithelial cells and its elevation indicates the injury of type-II lung epithelial cells and damage to these alveolar cells that can led to irreversible pulmonary inflammation and fibrosis in the lung. 38,40 To date, the exact mechanistic basis of this cytotoxicity induced by nanoparticles is unclear, together our results suggests that the reduction of cell viability is due to increased cellular stress due to Fe3O4 NPs exposure that results in increased microvascular permeability leading to cell mortality, as indicated by increased levels of LDH, TP, and ALP in BALF. However, the severity of cell death tends to decreased in a time-dependent manner as we observed an increase in cell viability up to 84–90% in treated groups of either sex at the end of the 14 days of postexposure suggesting the activation of cellular repair system from oxidative damage induced by acute exposure of these nanoparticles.
Lung inflammation has been identified as a major concern for inhalation exposure of NPs. 41 Hence, we assessed the total and differential cell counts recovered from BALF to understand the role of inflammation in Fe3O4 NPs inhalation exposure. As shown in Figure 3, total leukocytes recovered from BALF in animals exposed acutely to Fe3O4 NPs were significantly different from control rats throughout the exposure period. These total cell count represents a primary indicator of the degree of inflammatory response to nanoparticle exposure. 38,39,42 Furthermore, a drastic recruitment of neutrophils and lymphocytes within 24 h of exposure of these nanoparticles in BALF suggests the severity of acute inflammation in lungs induced by these particles. In contrast, Lay et al 43 have shown that Fe3O4 particles have very low solubility, were less capable of producing ROS, and did not cause inflammatory responses after intratracheal instillation to rats. This may be due to low solubility of Fe3O4 particles compared with iron metal, which have a greater propensity to dissolve in the interstitial and phagolysomal fluids. 14 However, in our study, Fe3O4 NPs enhanced inflammatory response as evidence by increasing in percentage of neutrophils. Also, there is increasing evidence suggesting that redox-active transition metals associated with particles can induce proinflammatory responses in lung cells. 44,45 These results shows that on one hand Fe3O4 NPs significantly induced cytotoxicity as evidence by decrease in cell viability and increase in LDH and protein leakage in BALF. On the other hand, Fe3O4 NPs induced severe inflammation in lung as evident by drastic increase in the total and differential cell counts recovered from BALF within 24 h of postexposure. These findings indicate the pulmonary self-defense mechanism initiated through the induction of the particle-induced inflammatory response.
As shown in Figure 6, concentrations of proinflammatory cytokines, that is, IL-1β, TNF-α, and IL-6 in BALF and blood of Fe3O4 NPs-exposed animals significantly increased within 24 h of postexposure period and remained significantly higher than the control animals throughout the observation period (p < 0.001). Levels of IL-1β, TNF-α, and IL-6 in BALF reached a maximum in BALF within 24 h of postexposure period, as the increasing ratio to the control level were 7- to 8-fold, 18- to 20-fold, and 12- to 14-fold, respectively, in either sex. Previous studies have shown that inflammation in the lung may be initiated by phagocytosis of nanoparticles by AMs, which in turn stimulate the release of these proinflammatory cytokines that plays a major role in the development and progression of inflammation, migration of leukocytes to sites of inflammation, egression into alveolar space and injury. 13,46 –51 Although statistically significant, the concentrations of these proinflammatory cytokines in BALF and blood exhibited a time-dependent decrease following initial rapid increase on 24 h of postexposure as the induction levels were only about 3- to 3.5-fold, 5- to 5.4-fold, and 2- to 3-fold increase in IL-1β, TNF-α, and IL-6, respectively, compared with controls in BALF on 14 days suggesting the recovery of the exposed animals. Similar changes in proinflammatory cytokines were observed by Park et al 13 after intratracheal instillation of Fe3O4 NPs in mice. Another noteworthy finding in the current study was the significant correlation of these inflammatory cytokines in BALF and blood toward Fe3O4 NPs exposure (Figure 7). It was highly evident that these cytokines are more sensitive toward nanoparticle-induced toxicity compared with regular blood biochemical and hematological parameters. However, minor differences exist in the propensity of the correlation among these cytokines in the current study, as IL-1β and IL-6 levels in BALF and blood correlated significantly and strongly, while TNF-α levels in BALF and blood correlated significantly, but less well. Nevertheless, the correlation coefficient (r s) of these cytokines in BALF and blood were between 0 and 1, suggesting that these cytokines tend to increase or decrease together. 52 Based on the current study and our previous work on CeO2 NPs, 26 circulation levels of cytokines in blood may be used as indispensable biomarker for nanoparticles-induced pulmonary toxicity.
Although the exact mechanism related to nanoparticle-induced toxicity is still not known, previous published data have indicated that assessment of oxidative stress as the most valid criteria for evaluating nanoparticle toxicity, as ROS generation by nanoparticles may lead to possible adverse biological effects by reacting with DNA and proteins directly, or could react with lipids to generate MDA that could subsequently react with DNA, proteins, or other lipids. 34,35,53 Furthermore, when particles enter the cell, they could induce oxidative stress by disturbing the balance between oxidant and antioxidant processes. 54 Hence, in our study, we assessed the levels of MDA, a major lipid peroxidation product, GSH a free radical scavenger, and antioxidant enzymes such as GSH-Px, SOD, and catalase activity in the lung, which are considered as critical factors that affect oxidative stress statue. Acute inhalation exposure of Fe3O4 NPs induced a significant increase in MDA concentrations in lungs of exposed animals compared with control throughout the study (p < 0.001), indicating the induction of lipid peroxidation by these particles (Figure 8). These increase in lipid peroxidation resulted in membrane damage of cells as indicated by LDH release in BALF on Fe3O4 NPs exposure. Also, MDA is a proven mutagen and carcinogenic compound that reacts with DNA to form adducts to deoxy guanosine, deoxy adenosine, and deoxy cytidine leading to DNA damage 55,56 suggesting the Fe3O4 NPs exposure may triggers cell apoptosis. As shown in Figure 9, a significant depletion of GSH content in the lungs of Fe3O4 NPs exposed rats were observed up to 48 h exposure, demonstrating a condition of nanoparticle-induced oxidative stress in the cells, which may arise due to imbalance in the ROS formation and antioxidant defense system of cells. GSH plays an important role in the elimination of free radicals generated in the body, and the intracellular GSH redox homeostasis is strictly regulated to govern cell metabolism and protect cells against oxidative stress. 13 The activities of antioxidant enzymes, such as GSH-Px, SOD, and catalase, in these lung tissues significantly decreased up to 48 h of postexposure (Figures 10 to 12). The dysfunction of these antioxidant enzymes can aggravate the oxidant damages induced by these nanoparticles. A recovery in GSH content with the association of antioxidant enzyme activities after 14 days of exposure suggests the recycling of oxidized GSH to reduced GSH by the cell defense system via a coupled reaction involving GSH reductase and glucose-6-phosphate dehydrogenase. 57 The significant increase in lipid peroxidation product MDA associated with the depletion of GSH content and antioxidative enzymes activity up to 48 h of postexposure indirectly reflected the extent of injury in the lung tissues of rat induced by Fe3O4 NPs. However, in the present study, the recovery in GSH content and antioxidant enzyme activities in the lung were not accompanied by the MDA levels after 14 days of exposure, as there exists its significant increase throughout the observation period suggesting that the cellular repair system of oxidative damage was able to reduce nanoparticle induced ROS rapidly, but could not decrease lipid peroxidation significantly after Fe3O4 NPs exposure.
Although no sexual dimorphism was observed in the majority of cytotoxic, inflammatory, and oxidative stress throughout the observation period, there exit minor statistical differences especially in total cell count in BALF, as females are comparable with controls, while males are not at the end of 14 days of postexposure. Similarly, lymphocyte population in BALF of males is comparable with untreated animals on 48 h of postexposure, while in females, it is different. In addition, in most cases, there exists a sexual difference in the recovery of tested inflammatory and oxidative stress parameters. These findings warrant further investigations to study weather these differences were incidental or not.
Common mechanisms associated with the clearance of particulate material in the lung include the mucociliary escalator and phagocytosis by AMs. 58 In the present study, the histopathological observations had depicted all these clearance mechanisms at all of postexposure periods. Although the appearance of phagocytosis and the pulmonary clearance mechanism have been noticed early at 24 h of postexposure period, the persistence of nonphagocytosed free and agglomerated Fe3O4 NPs was observed throughout the observation period (Table 2 and Figure 13) in the pulmonary region. This strongly supports the hypothesis that nanoparticles are retained in the lungs to a greater extent than their fine sized counterparts and are hard to clear. 23 The role of AMs in the clearance of NPs after inhalation exposure in rats has revealed that the NPs are much less phagocytosed by AMs than large particles. 59,60 The appearance of low grade well distributed foreign body type granulomatous inflammation on 14 days of postexposure period can be a local mechanism that sequesters the insoluble Fe3O4 NPs to avoid further movement of the NPs and the resultant damage to the pulmonary parenchyma. The alveolitis accompanied with the perivascular cuffing of mononuclear inflammatory cells which was found early at 48 h of postexposure could be due to the triggered mononuclear phagocyte system and also due to the fact that the ultra fine NPs are more potent in inducing the acute inflammatory response in comparison with their fine or large particle size. 61 –63 The existence of both alveolitis and granuloma formation in same pulmonary parenchyma at 14 days of postexposure period suggests the size-dependant biphasic nature of the Fe3O4 NPs, where the large agglomerated masses were followed by the granulomatous inflammation and the small size or the ultra fine NPs elicits direct damage to the alveolar wall and sequels in to the nongranulomatous inflammatory reaction. Furthermore, the pulmonary inflammatory response as evident by increase in proinflammatory cytokines in BALF and blood and granuloma formation can also be attributed due to deposition of agglomerated Fe3O4 NPs in the lungs. 64
Apart from mucociliary escalator and phagocytosis by AMs, the lymphatic system has long been recognized as an additional pathway for pulmonary particles clearance. The intra- and extra-pulmonary lymphatic structures carry, store, and, in many instances, process a fraction of the alveolar deposit, participating in this way in the lung defense against inhaled particles. 58 Similarly, in our study, we observed the emigration and translocation of Fe3O4 NPs either in free or as macrophage laden form in both BALT and LALN of the exposed animals.
Previous published data have reported the extrapulmonary translocation of the nanoparticles either through the systemic circulation or direct translocation from the nasal epithelium to the brain through olfactory nerve. 65 –71 However, in our study, we did not find the traces of Fe3O4 NPs in any of the extrapulmonary organs, which can be attributed due to short observation period and very slow rate of pulmonary clearance of the nanoparticles. This was further supported by cytotoxicity, proinflammation, oxidative stress, and histological changes induced by Fe3O4 NPs confined only to the lung and no biochemical and hematological alterations in plasma or histological changes in other extrapulmonary organs is observed. It has also been experimentally shown in rats and humans that translocation rates of inhaled ultrafine particles to extrapulmonary organs after a single exposure are generally below 5%. 72 –75 All these pathological changes, combined with the biochemical alteration in BALF and blood indicated a lung injury and inflammation as a result of acute inhalation exposure of Fe3O4 NPs.
In summary, the current study of acute exposure of Fe3O4 NPs in rats via 4 h continuous nose-only inhalation exposure resulted in significant lung responses, including lung inflammation, cytotoxicity, lung injury, induction of microgranuloma, and release of proinflammatory cytokines. Furthermore, a significant correlation between proinflammatory cytokines of BALF and blood was observed; hence, circulation levels of cytokines in blood may be used as indispensable biomarker for nanoparticles-induced pulmonary toxicity. However, in the present work, we provided the acute inhalation toxicity information of these particles in rat; further repeated exposure studies at lower dose levels may be required to assess the exact cumulative and mechanistic elucidation of Fe3O4 NPs induced toxicity.
Footnotes
Acknowledgements
The authors thank B. Siva Prakash and M. Sai Kumar for their contribution to the laboratory work.
Declaration of Interest
The authors report no declarations of interest
Funding
This work was supported by IIBAT management.
