Abstract
Polychlorinated biphenyls (PCBs) comprise a ubiquitous class of toxic substances associated with carcinogenic and tumor-promoting effects as well as neurotoxic properties. Reactive oxygen species, which is produced from PCBs, alters blood–brain barrier (BBB) integrity, which is paralleled by cytoskeletal rearrangements and redistribution and disappearance of tight junction proteins (TJPs) like claudin-5 and occludin. Quercetin, a potent antioxidant present in onion and other vegetables, appears to protect brain cells against oxidative stress, a tissue-damaging process associated with Alzheimer’s and other neurodegenerative disorders. The aim of this study is to analyze the role of quercetin on oxidative stress markers and transcription of transmembrane and cytoplasmic accessory TJPs on cerebrum, cerebellum and hippocampus of female rats exposed to PCBs. Rats were divided into the following four groups. Group I: received only vehicle (corn oil) intraperitoneally (i.p.); group II: received Aroclor 1254 at a dose of 2 mg/kg body weight (bwt)/day (i.p); group III: received Aroclor 1254 (i.p.) and simultaneously quercetin 50 mg/kg bwt/day through gavage and group IV: received quercetin alone gavage. From the experiment, the levels of hydrogen peroxide, lipid peroxidation and thiobarbituric acid reactive substances were observed to increase significantly in cerebrum, cerebellum and hippocampus as 50%, 25% and 20%, respectively, after exposure to PCB, and the messenger RNA expression of TJP in rats exposed to PCBs is decreased and is retrieved to the normal level simultaneously in quercetin-treated rats. Hence, quercetin can be used as a preventive medicine to PCBs exposure and prevents neurodegenerative disorders.
Introduction
Polychlorinated biphenyls (PCBs) are persistent environmental toxicants associated with numerous adverse health effects. 1 PCBs are widely used in electrical industry, as coolants for transformers and capacitors. 2 PCBs are lipophilic and resistant to biological decomposition and can accumulate in higher tropic levels through the food chain. 3 Most of the systemic toxic and biological effects of PCBs are mediated by the aryl hydrocarbon receptor (AhR). Aroclor 1254 is a technical mixture containing almost 70 different PCB isomers and congeners with a molecular mass ranging from 188 to 430 Da, 4 which has been widely utilized in studying PCB toxicity. As far as its neurotoxicity is concerned, Aroclor 1254 has been shown to produce neurochemical alterations in several experimental settings 5 as well as behavioral changes in learning, memory, motor activity and sexual behavior. 6,7 Developmental exposure to PCB mixture or congeners altered the motor activity, neurological development and cognitive function in laboratory animals. 8 Our earlier studies suggest that the PCB increases the reactive oxygen species (ROS) level and decreases the activities of antioxidant enzymes, functional markers like creatine kinase, acetyl choline esterase and membrane bound ATPases in selected rat brain regions. 9 –13
Homeostasis of the central nervous system (CNS) microenvironment is essential for normal functions of the brain and is maintained by the blood–brain barrier (BBB). The BBB is mainly formed by highly specialized endothelial cells (ECs), and it protects the brain from blood-borne substances. At the same time, the BBB ensures the supply of nutrients to the brain by specific transport systems. 14 The BBB acts as a physical and metabolic barrier because a complex tight junction (TJ) system between the adjacent ECs restricts most paracellular movement of ions and solutes across the brain endothelium. 15 TJs of the BBB are composed of an intricate combination of at least three integral membrane proteins (claudins (Cldn), junction adhesion molecules (JAMs) and occludin (Ocln)) and cytoplasmic accessory proteins, such as zonula occludens (ZO)-1, ZO-2 and ALL-1 fusion partner at chromosome-6 (AF6).
ZO-1 was the first TJ-associated protein identified and characterized among cytosolic accessory proteins of TJ. 16 The ZO proteins belong to the family of membrane-associated guanylate kinases (MAGUK), which possess a distinct modular organization and associate peripherally with the cellular membranes. ZO-1 is essential for TJ assembly, and it forms a scaffold complex with other cytosolic accessory proteins, such as ZO-2 and AF6, to anchor integral membrane proteins to actin cytoskeleton. 17,18 Disruption of ZO-1 at TJ has been observed in response to bacterial toxins, 19 drugs, 20 growth factors, 21 cytokines 22 and hypoxia 23 and correlated with increased permeability and/or decreased electrical resistance. In addition, ZO proteins are believed to form a complex for transcription factors and signaling proteins involved in the regulation of cell proliferation and differentiation. 24,25
Relatively less information is available on the role of ZO-2 and AF6 in the regulation of integrity of the brain endothelium. ZO-2 shares three-defined core regions of MAGUK family proteins with ZO-1, which are a Src homology 3 (SH3) domain, a guanylate cyclase and a PDZ (PDZ is an acronym combining the first letters of three proteins – post synaptic density protein (PSD95), Drosophila disc large tumor suppressor (Dlg1), and zonula occludens-1 protein (zo-1) – which were first discovered to share the domain) domain. SH3-domains bind with signaling proteins and cytoskeletal elements. Guanylate cyclase domains are involved in the adenosine triphosphate (ATP)-dependent transformation of guanosine monophosphate (GMP) to guanosine diphosphate (GDP), and the PDZ-domains mediate specific binding to carboxy-terminal cytoplasmic ends of transmembrane proteins. 18 ZO-2 acts as a signaling molecule to communicate the state of cell–cell contact of the TJ. 26 It appears that ZO-2 can modulate these interactions by its influence on the transcription factors c-Jun, c-Fos and C/EBP (CCAAT-enhancer binding proteins). 27 ZO-2 expression decreases in response to several pathological conditions, including amyloid beta treatment, hypoxia, 23 cerebral infarction in spontaneously hypertensive rats 28 and exposure to HIV proteins, such as gp120 29 or Tat. 30
AF6 is a multidomain actin-binding protein that serves as a scaffold protein between transmembrane proteins and the actin cytoskeleton. 31 It interacts with ZO-1, nectin, Eph receptors and the actin-regulatory protein profiling. 31,32 It binds to Ras-like small GTPases and is suggested to be an effector for both Ras and Raf. It has been shown that the activation of Ras can result from the interaction of AF6 with ZO-1. 32 AF6 may regulate the adhesion of leukocytes to extracellular matrix. Indeed, overexpression of AF6 in Jurkat cells increased its adhesion to fibronectin. In contrast, knockdown of AF6 in T-cells results in an enhancement of cell adhesion. 33
Exposure to PCBs can contribute to the induction of neurological disorders including morphological changes in neurons, 34 brain tumor promoting effects 35 and the developmental defects of the nervous system. 36 However, the mechanisms by which PCBs cause these neurotoxic effects are not fully understood. BBB integrity is paralleled by cytoskeleton rearrangements, redistribution and disappearance of tight junction proteins (TJPs), such as claudin-5 (Cldn5) and Ocln. ROS induces enhanced Ca2+ mobilization and inositol (1,4,5)-triphosphate formation in brain ECs via phospholipase C. 37 The organization of TJPs, Ocln and ZO-1, is altered by exogenous ROS in brain ECs. 38 ROS selectively activates signaling cascades involving Ras homolog gene family; member A (RhoA) is a small GTPase protein known to regulate the actin cytoskeleton in the formation of stress fibers. In humans, it is encoded by the gene RhoA, phosphoinositide 3-OH (PI3) kinase and protein kinase B (PKB/Akt), leading to the rearrangements of the actin cytoskeleton and spatial redistribution, disappearance of Ocln and Cldn5, inducing altered BBB integrity. ROS disrupts the integrity of TJ, by transient activation of the PI3 kinase and PKB pathway via RhoA. 39
ROS may activate protein tyrosine kinases in brain ECs, which in turn may lead to tyrosine phosphorylation of TJ proteins. 40 ROS selectively activates signaling cascades involving RhoA, PI3 kinase and PKB/Akt leading to the rearrangements of actin cytoskeleton and spatial redistribution and disappearance of Ocln and Cldn5, inducing altered BBB integrity. ROS induces PKB phosphorylation through RhoA activation. 39 The family of Ras-related small GTP-binding proteins comprises RhoA, Rac1 and Cdc42, which are the regulators of the actin cytoskeleton, and it has been described that the RhoA activation leads to the phosphorylation of Ocln, Cldn5 and TJ reorganization. 41 It has been reported that the Cldn5 can be phosphorylated after the exposure of brain ECs to ROS. 40 The inhibition of Rho activation prevented the ROS-induced cytoskeleton rearrangements and the disappearance of Ocln and Cldn5. It indicates that ROS activates Rho, which subsequently may affect the actin cytoskeleton via PI3 kinase and PKB. Cytoskeleton depolymerization causes the redistribution of TJ molecules. Hence, signaling molecules that control the organization of the actin cytoskeleton may indirectly be involved in the regulation of TJs. 39
Quercetin, a flavonoid, has been shown to have a beneficial role in neuroprotection through strong antioxidant activity. 42 It increases GSH level in brain and maintains spontaneous behavior and cognitive performance. 43 Our earlier studies reveal that quercetin acts against PCB-induced neurotoxicity in cerebral cortex, cerebellum and hippocampus by decreasing oxidative stress via scavenging ROS and brought back the messenger RNA (mRNA) expression of dopamine receptors in male rats. 9 –11 PCB-induced neurodegeneration has also been assessed by histological studies. Our earlier studies proved that the exposure to PCBs decreased serum thyroid hormone profile, follicle stimulating hormone (FSH), luteinizing hormone (LH), testosterone and estradiol in male rats. 44 The present study is aimed to analyze the role of PCB on BBB integrity particularly on female rats. The studies on serum hormonal profiles and other parameters are in progress.
Materials and methods
Experimental design
Experiments were carried out on adult female Wistar rats weighing 180–200 g, which were maintained under standard laboratory conditions in large polypropylene animal cage. Six rats were kept in a cage and all animal had free access to pellet diet (Gold Mohur Ltd., Mumbai, India) ad libitum. Rats were maintained at room temperature (25°C) with normal light cycle (12 h light and 12 h dark) and treated for 30 days.
The animals were divided into the following groups: (1) group I comprises control rats that were injected with corn oil of 80 µl/day intraperitoneally (i.p.) for 30 days as a vehicle; (2) group II comprises rats that received i.p. injection of Aroclor 1254 at a dose of 2 mg/kg body weight (bwt)/day; 13 group III comprises rats that were administered with Aroclor 1254 at a dose of 2 mg/kg bwt/day (i.p.) and simultaneously quercetin was supplemented gavage at a dose of 50 mg/kg bwt 11 and group IV comprises rats that received quercetin by gavage at a dose of 50 mg/kg bwt. 11 The doses of both Aroclor 1254 and quercetin were chosen based upon their activity in the in vivo functional and biochemical studies. 11,13 All animal procedures were approved by our institutional ethical committee (Ref No. IAEC No 01/01/11). After the treatment period of 30 days, rats were killed. The brain was dissected and cerebellum, cerebrum and hippocampus were isolated and analyzed for the mRNA expression of TJPs.
Drugs and chemicals
Aroclor 1254 was purchased from Chem Service (West Chester, Pennsylvania, USA). Quercetin, total RNA isolation (TRI) reagent and primers were purchased from Sigma-Aldrich Private Limited (Bangalore, Karnataka, India). Superscript-III reverse transcriptase was purchased from Invitrogen (Chennai, Tamilnadu, India) and PCR Ready Mix DNA Polymerase was purchased from KAPA-Biosystem (Chennai, Tamilnadu, India).
Oxidative stress markers
Estimation of H2O2
The generation of hydrogen peroxide (H2O2) was assayed by the method of Pick and Keisari. 45 The reaction mixture prepared consists of tissue homogenate, 2.64 ml phosphate buffer, 54 µl horseradish peroxidase, 30 µl of 28 nM phenol red, 165 µl of 5.5 nM dextrose and 600 µl of enzyme source. It was incubated at 35°C for 30 min and terminated by adding 60 µl of 10 N NaOH solution. Horseradish peroxidase converts H2O2 into water and oxygen. This causes the oxidation of phenol red, which forms adduct with dextrose, and shows maximum absorbance at 610 nm. The levels of H2O2 generation were expressed as nanomoles of H2O2 generated per minute per milligram of protein.
Estimation of LPO
Lipid peroxidation (LPO) was measured by the method of Devasagayam and Tarachand. 46 The reaction mixture comprising 0.2 ml of tissue homogenate, 1 ml of 0.15 M Tris-HCl buffer (pH 7.4), 0.3 ml of 10 mM potassium dihydrogen phosphate (KH2PO4) and 1.5 ml of 1% thiobarbituric acid (TBA) were heated for 20 min. The reaction was then stopped by the addition of 10% trichloroacetic acid. Malondialdehyde, an end product of LPO, reacts with TBA to form a pink chromogen (TBA 2-malondialdehyde adduct), and it was measured at 532 nm absorbance with a spectrophotometer. The results were expressed as nanomoles of malondialdehyde (MDA) formed per milligram of protein.
Measurement of TBARS as a marker of LPO
Cerebrum, cerebellum and hippocampus were excised out of the brain and rinsed with cold 0.14 M NaCl, and a part of it was homogenized in 25% ice-cold 50 mM Tris-HCl buffer, pH 7.4. 47 A total of 150 μL of the tissue supernatant of samples was diluted to 500 μL with deionized water. A total of 250 μL of 1.34% TBA was added to all the tubes, followed by the addition of an equal volume of 40% trichloroacetic acid. The mixture was shaken and incubated for 30 min in a boiling water bath. Tubes were allowed to cool to room temperature and the absorbance was read at 532 nm using zero concentration as blank. The results were expresses as nanomoles per milligram of protein. 48
TRI and RT-PCR
Total RNA was isolated from cerebrum, cerebellum and hippocampus using TRI reagent following the method of Chomczynski and Sacchi.
49
Total RNA of 1 µg was subjected to two-step reverse transcriptase-polymerase chain reaction (RT-PCR). (1)
List of primers

ZO-1/-2: zona occludens-1/-2; Ocln: occludin; Cldn5: claudin-5; JAMs: junction adhesion molecules; AF6: ALL-1 fusion partner at chromosome-6.
Statistical analysis
All the values were expressed as mean ± SEM of three independent observations. Data were analyzed using one-way analysis of variance followed by post hoc test Student’s Newman-Keul’s test with Graphpad Prism5 software. In all the cases,
Results
Effect of quercetin on body weight of adult female rats exposed to PCBs
The effect of PCBs on change in body weight of rats is shown in Figure 1. No mortality was observed in any of the experimental groups. Maximum weight gain was observed in the quercetin-administered rats when compared with all other groups. There was a gradual and significant (
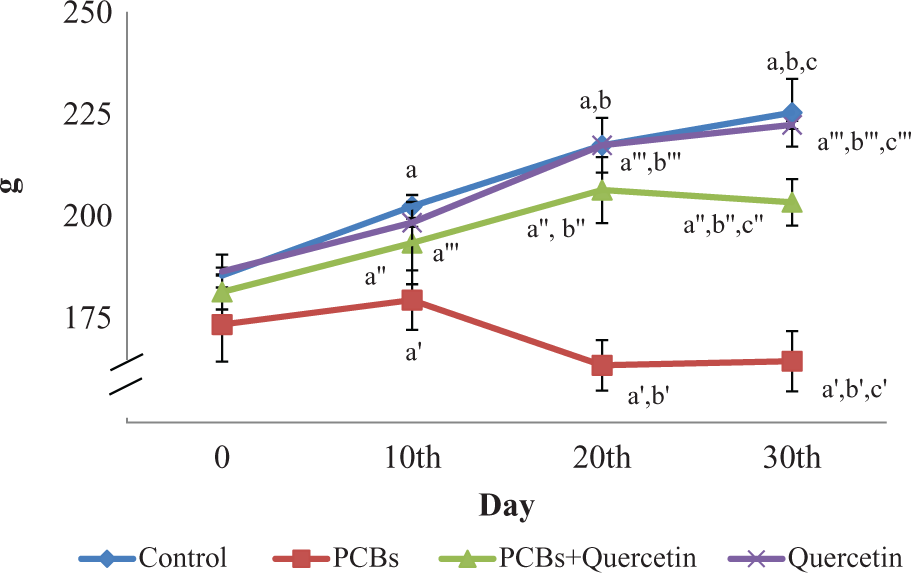
Effect of quercetin on the body weight of adult female rats exposed to PCBs. Each bar represents mean ± SEM of six animals. Statistical significance at
Effect of quercetin on oxidative stress markers in cerebrum, cerebellum and hippocampus of adult female rats exposed to PCBs
The levels of H2O2 generation, LPO and TBA reactive substance (TBARS) content in cerebrum, cerebellum and hippocampal tissue of control and experimental rats were estimated (Figure 2(a) to (c)). The levels of H2O2, LPO and TBARS were increased significantly in PCB-exposed rats in cerebrum, cerebellum and hippocampus as 50%, 25% and 20%, respectively. Quercetin acts as a free radical scavenger and decreased the levels of oxidative stress markers (H2O2, LPO and TBARS) in quercetin alone or in PCB + quercetin-treated animals.
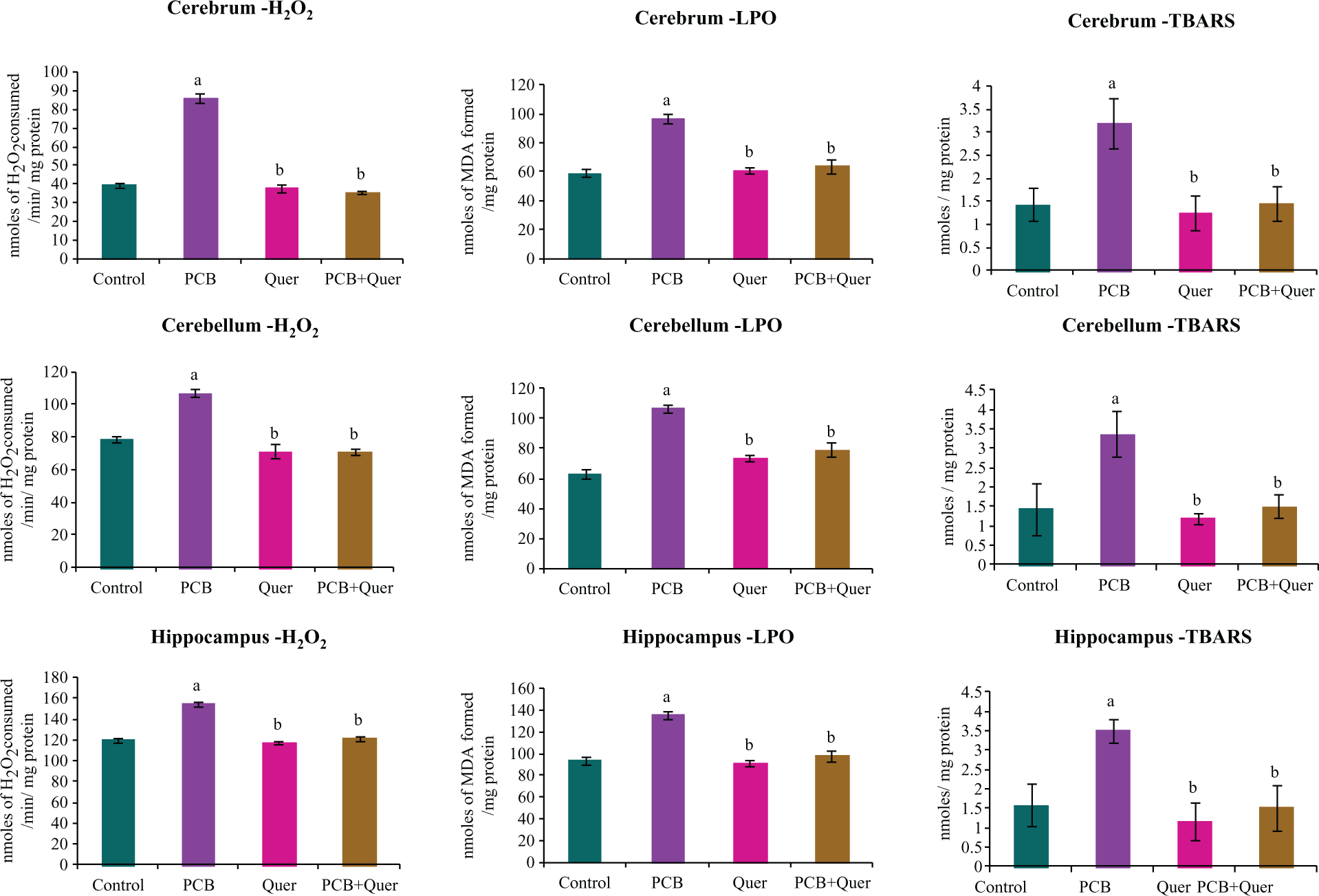
Effect of quercetin on oxidative stress markers in cerebrum, cerebellum and hippocampus of adult female rats exposed to PCBs. Each bar represents mean ± SEM of six animals. Statistical significance at
mRNA expression of TJPs
Figure 3 represents the effect of quercetin on mRNA expression of integral membrane protein Ocln in cerebrum, cerebellum and hippocampus of adult female rats exposed to PCBs. mRNA expression of Ocln was decreased (40%) in the exposure to Ocln, while simultaneous supplementation of quercetin brought the levels back to normal. Exposure to quercetin alone showed no change.

Effect of quercetin on mRNA expression of integral membrane protein Ocln in cerebrum, cerebellum and hippocampus of adult female rats exposed to PCBs. Each bar represents mean ± SEM of three independent observations. Statistical significance at
Figure 4 represents the effect of quercetin on mRNA expression of integral membrane protein Cldn5 in cerebrum, cerebellum and hippocampus of adult female rats exposed to PCBs. Cldn expression levels were decreased (40%) upon exposure to PCBs. Simultaneous supplementation of quercetin scavenges the ROS and decreases the degradation of Cldn and brought back to normal. Exposure to quercetin alone showed no change.
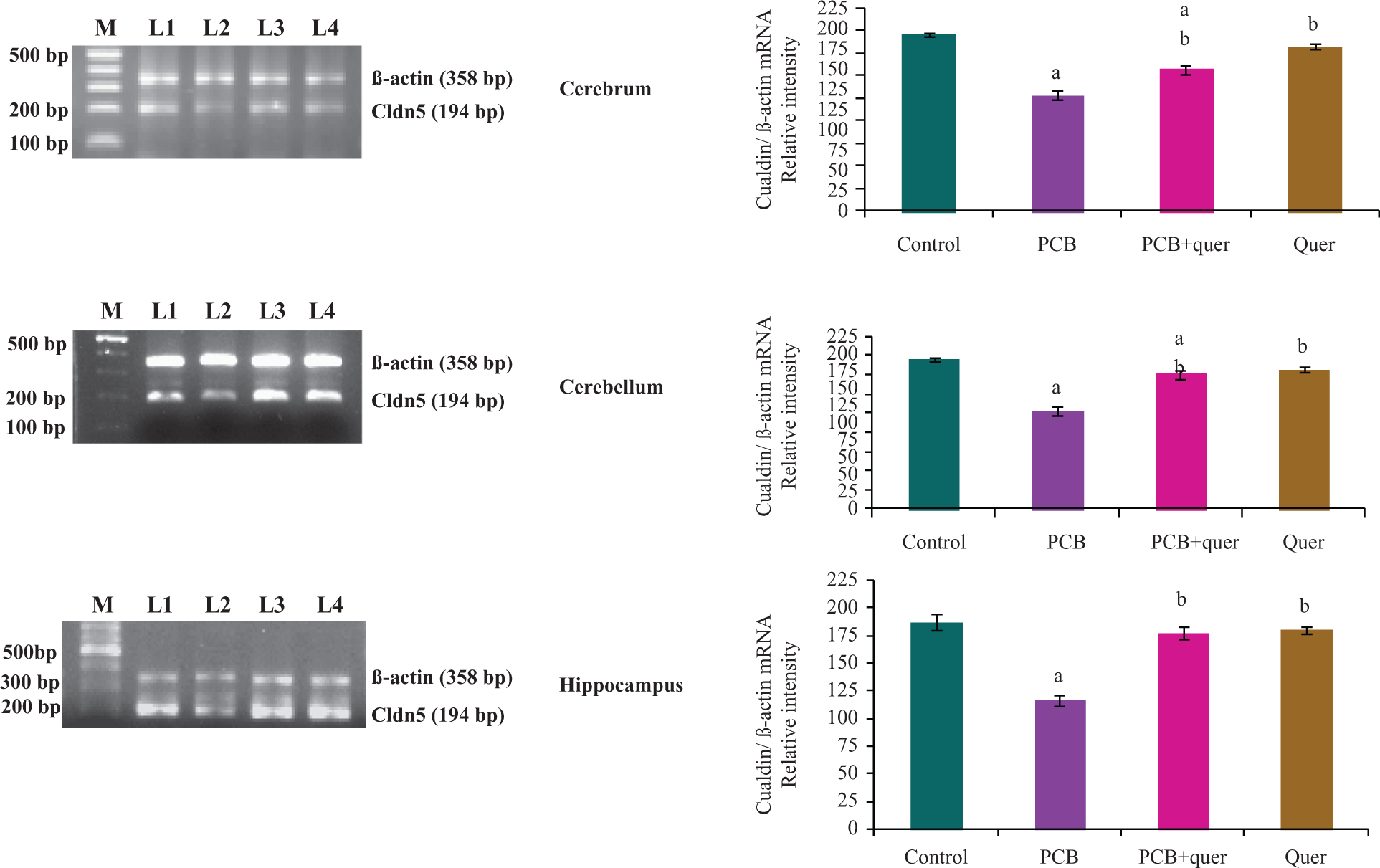
Effect of quercetin on mRNA expression of integral membrane protein Cldn5 in cerebrum, cerebellum and hippocampus of adult female rats exposed to PCBs. Each bar represents mean ± SEM of three independent observations. Statistical significance at
Figure 5 reveals the effect of quercetin on mRNA expression of integral membrane protein, that is, JAM-3 in cerebrum, cerebellum and hippocampus of adult female rats exposed to PCBs. JAM levels were decreased upon exposure to PCBs, while simultaneous supplementation of quercetin brought the levels back to normal. A significant decrease (50%) was observed in cerebellum than in cerebrum (10%) and hippocampus (15%) of the rats exposed to PCBs. Quercetin alone did not show any significant change.

Effect of quercetin on mRNA expression of integral membrane protein JAMs in cerebrum, cerebellum and hippocampus of PCBs exposed adult female rats. Each bar represents mean ± SEM of three independent observations. Statistical significance at
Figure 6 represents the effect of quercetin on mRNA expression of cytoplasmic accessory proteins; zona occludens (ZO; ZO-1 and ZO-2) in cerebrum, cerebellum and hippocampus of adult female rats exposed to PCBs. mRNA expression levels of both ZO-1 and ZO-2 were decreased upon exposure to PCBs, while simultaneous supplementation of quercetin brought the levels back to normal. Quercetin alone did not show any significant change. Similar to JAM, cerebellum showed 50% decrease in both ZO-1 and ZO-2 mRNA expressions after exposure to PCB than other regions.

Effect of quercetin on mRNA expression of cytoplasmic accessory proteins: ZOs (ZO-1 and ZO-2) in cerebrum, cerebellum and hippocampus of adult female rats exposed to PCBs. Each bar represents mean ± SEM of three independent observations. Statistical significance at
Figure 7 shows the effect of quercetin on mRNA expression of cytoplasmic accessory proteins, such as AF6/afadin, in cerebrum, cerebellum and hippocampus of adult female rats exposed to PCBs. mRNA expression levels of AF6 were decreased significantly in PCB-exposed rats in cerebrum, cerebellum and hippocampus as 30%, 50% and 15%, respectively. Quercetin supplementation is brought back only in cerebrum and hippocampus. In cerebellum, quercetin supplementation tried to become normal levels. Quercetin alone treated animals showed normal mRNA expression.

Effect of quercetin on mRNA expression of cytoplasmic accessory protein (AF6) in cerebrum, cerebellum and hippocampus of adult female rats exposed to PCBs. AF6: ALL-1 fusion partner at chromosome-6. Each bar represents mean ± SEM of three independent observations. Statistical significance at
Discussion
PCBs are the persistent organic pollutants that exhibited various toxic effects in animals and exposed human populations. The molecular mechanisms of PCB toxicity have been attributed to the toxicological properties of its metabolites, such as hydroquinone, formed by cytochrome-P-450 oxidation. 50 PCBs cause neurodegeneration via the production of ROS. The brain is especially vulnerable to oxidative damage for several reasons. The activities of catalase, superoxide dismutase (SOD) and glutathione peroxidase (GPx) are low in all the brain regions. Furthermore, the nerve cells are rich in polyunsaturated fatty acids that are especially sensitive to the attack by free radicals, which may lead to LPO and membrane impairment and which may lead to neurodenegerative diseases. 13 PCBs cause neurodegeneration via the production of ROS. 51 Since brain has low catalase, SOD and GPx activities, its membrane lipids are rich in polyunsaturated fatty acids, which are especially sensitive to free radical-induced LPO. 52 Hence, the brain is more vulnerable to PCBs-induced neurodegeneration via ROS. This study prevails that H2O2, LPO and TBARS levels were significantly high after exposure to PCB. There are evidences suggesting that the oxidative stress induced by PCBs is due to the interaction of these compounds with AhR and activation of the cytochrome P450 1A superfamily. 53 Previous studies in our laboratory also suggest that PCBs–quinones undergo redox cycling with the formation of ROS, thus becoming a major source of oxidative stress in male Wistar rats. Earlier studies in our laboratory demonstrated that the exposure to PCB decreased mRNA expression of Cu/Zn, SOD and GPx-4 as well as histology of brain regions. 51 PCB-induced oxidative stress decreases the activities of antioxidant enzymes and disrupts the functional parameters in ventral prostate, testicular Leydig cells, sertoli cells and selected brain regions, 10,11,54 –58 and it can damage the cellular elements.
ROS may propagate the initial attack of lipid membranes of the brain to cause LPO. It is one of the manifestations of oxidative damage and it has been found to play an important role in the toxicity of several xenobiotics. 59 In the present study, in female rats, PCB increases ROS level by increased LPO, H2O2 and TBARS concentrations in cerebrum, cerebellum and hippocampus. Oxidative modification of proteins in vivo may affect a variety of cellular functions involving proteins: receptors, signal transduction mechanisms, transport systems and enzymes. It could also contribute to secondary damage to other biomolecules.
The BBB is anatomically situated at the level of the cerebral microvascular capillary endothelium and it regulates the blood–brain exchange. The BBB is primarily formed by specialized brain ECs, which form a tight seal due to the presence of well-developed TJs that impede the entrance of circulating molecules and immune cells into the CNS. 60 The TJs limit passive paracellular movement of solutes, ions and water across the BBB. This barrier is variable and physiologically regulated, and its disruption contributes to human diseases. 61 There are ample evidences that PCBs cause neurotoxicity in humans. The predominant mechanisms include alteration in TJ of human brain microvascular ECs, intracellular-signaling process and thyroid hormone metabolism. 62 The loss of TJ proteins is commonly observed in neuroinflammatory and neurodegenerative disorders that are frequently associated with stroke, 63 Alzheimer’s disease, 64 HIV-1 encephalitis 65 and traumatic brain injury. 66 Disruption of TJ in brain ECs can increase serum protein extravasation through impaired BBB and lead to the development of vasogenic brain edema. PCBs exposure induces disruption of TJ in human brain microvascular ECs, which may lead to perturbation of the BBB integrity. 67
The brain is particularly sensitive to oxidative damage because of its high levels of unsaturated lipids as well as a high rate of oxidative metabolism. ROS (oxy radicals) are generated continuously in nervous system during normal metabolism and neuronal activity. 68 Oxidative stress is a contributing factor for the alteration caused in neurodegenerative processes. 51 PCB-induced toxic manifestations may be associated with the production of free radicals. Schilderman et al. 69 demonstrated that PCBs induce ROS which can damage the cellular elements in its target organs such as liver, spleen, seminal vesicles, prostate and epididymis. The ROS alter BBB integrity, which is paralleled by cytoskeleton rearrangements, redistribution and disappearance of TJ proteins 39 (ROS), which are highly reactive molecules, are produced during monocyte migration and contribute to BBB injury and subsequent inflammation in the brain. 70 In the present study, there is a decreased expression of TJ protein in the microvascular ECs in the cerebrum of PCB-treated rats.
TJs of the BBB composed of an intricate combination of integral membrane proteins (Ocln, Cldns and JAMs) mediate cellular interaction between brain ECs and play a major role in TJ functioning and cytoplasmic accessory proteins (ZO (ZO-1 and ZO-2) and AF6), providing a link between transmembrane TJ proteins and the actin cytoskeleton by participating in intracellular signaling. This study prevails that the mRNA expression of TJPs in cerebellum, cerebrum and hippocampus significantly decreased upon exposure to PCB. The evidence suggests that the PCB involves in the alteration of TJ of human brain microvascular ECs, intracellular-signaling process and thyroid hormone metabolism. 62 The ROS alter BBB integrity, which is paralleled by cytoskeleton rearrangements, redistribution and disappearance of TJ proteins. 39 ROS, which are highly reactive molecules, are produced during monocyte migration and contribute to BBB injury and subsequent inflammation in the brain. PCBs-induced disruption of blood–brain homeostasis occurs predominantly by means of ROS-induced TJ disruption. The organization of TJ proteins Ocln and ZO-1 is altered by exogenous ROS in brain ECs. 38 ROS selectively activate signaling cascades involving RhoA, PI3 kinase and PKB/Akt leading to the rearrangements of actin cytoskeleton and spatial redistribution and disappearance of Ocln and Cldn5, inducing altered BBB integrity. 39 Previous studies show neuronal damage as well as alteration in neuronal morphology of cerebral cortical layer, pyramidal cells of hippocampal layers and the Purkinjee cellular layer in cerebellum after exposure to PCB. 56 It may be due to the enhanced free radical generation in PCB-exposed animals. Pyknotic nuclei with predominant perineuronal spaces were observed in cortical layer due to enhanced LPO. PCBs could also affect cerebellar development through complex actions on thyroid hormone system. 71 Anbalagan et al. 72 proved that the rats exposed to PCBs showed decreased thyroid hormone levels. In the present study, down regulation of both integral membrane proteins and cytoplasmic accessory proteins mRNA expression was observed in the PCBs-treated group. The possible mechanism behind this is the ROS-induced disruption.
Khan and Thomas 73 showed that the PCB inhibits tryptophan hydroxylase (TPH) activity, which is accompanied by the reduced 5 hydroxy tryptophan (5HT) concentrations in rat brain regions. PCB decreases TPH activity by a reduction in the enzyme protein inhibition of its biosynthesis or enhanced degradation to inhibit enzyme activation. They suggested that PCBs neurotoxicity may involve oxidative damage to TPH protein resulting in the reduction of TPH activity and 5HT concentration. In this present study, decreased junction proteins may be due to the inactivation of TPH protein by increasing LPO generation.
Quercetin is believed to work via electron donation to directly detoxify free radicals such as the highly toxic hydroxyl radicals. Quercetin is a strong oxygen radical scavenger and also a good metal chelator. In vitro studies have suggested that the quercetin has a potent inhibitory activity against the production of nitric oxide and tumor necrosis factor in lipopolysaccharide-stimulated Kupffer cells. 74 Quercetin was shown to scavenge superoxide in ischemia reperfusion injury. 75 Quercetin prevents oxidative stress-mediated diseases and inflammatory disorders. The 4-oxo group and 2,3-double bond in the c-ring of quercetin are thought to play an important role in their neuroprotective effects in repeated cerebral ischemic model. 43
Thus, toxicity of PCBs impairs TJP at the level of transcription by means of PKB and PI-3 kinase-signaling pathway related to ROS production. This PCB-induced neurotoxicity is minimized by quercetin, an antioxidant flavonoid. The antioxidant activity of flavonoids has been demonstrated by their abilities to scavenge free radicals 76 by inhibiting enzymes such as lipoxygenase and cycloxygenase and by chelating metal ions. 77 Selvakumar et al. 58 demonstrated that quercetin treatment can attenuate PCB-induced oxidative stress in the hippocampus of experimental animals. Quercetin exerts important role in providing indirect protection against free radical injury by stimulating antioxidant enzymes. The degeneration of cells was reduced in simultaneous administration of quercetin with PCB-exposed rats. In the present study, quercetin seems to be capable of protecting PCB-induced neurotoxic effects and maintains the TJP at the metabotrophic level.
Our earlier studies have proved that the PCBs induce ROS that can damage the cellular elements in its target organs such as brain, liver, spleen, kidney, seminal vesicles, prostate, epididymis, sperm, testicular sertoli and Leydig cells. 54 –59 Decreased antioxidant enzymes have also been studied in all the regions of brain in PCB-treated animals, indicating an increase in oxidative stress. PCB-induced neuronal damage on male rats has also been studied. However, the present study is focused on female rats. The neuroprotective role of quercetin in male rats has also been studied recently. 9 –11 Further study on female rats is warranted to understand whether quercetin prevents neuronal damage in brain regions. The present study infers that the quercetin protects PCB-induced BBB integrity and oxidative stress.
In the present study, disruption of TJ integrity due to PCB induction can occur from the loss of protein expression and/or disorganization. Dietary quercetin may exert its actions via the alteration of protein kinase activities and sequential-signaling processes. This may enable quercetin to prevent the disintegration of TJ structure and to enhance the expression, localization and interaction of TJ proteins.
Oxidative damages affect the expression, localization and organization of TJ proteins, particularly, Ocln, ZO1 and Cldns. 78 Oxidative stress is able to trigger signaling pathways in TJs regulation involving mitogen activated protein kinases (MAPKs), especially extracellular signal regulated kinases 1/2 (ERK1/2), P38 and c-jun NH2-terminal kinase (JNK), Protein kinase C (PKC) and phosphodiesterase. 79 Kevil et al. 80 studied that oxidative stress-induced hyperpermeability is related to the phosphorylation of Ocln and ZO1 at the tyrosine residues, down regulation of Ocln and activation of MAPK signaling pathways. The action of quercetin has been linked to a number of enzymes involved in the proliferation and signal transduction pathways including PKC, tyrosine kinase, PI3 kinase, nuclear factor kappa B (NFkß) and MPK family. Chuenkitiyanon et al. 79 studied that the protective effects of quercetin might involve the altered MAPK activities, in particular the decrease in p38 MAP signaling on epithelial and endothelial barriers.
PCBs have epigenetic effects on a wide variety of cellular and physiological functions. PCBs reduce DNA methylation in hippocampal neurons. 81 Further studies are needed. Study on the involvement of protein kinase C and MAPK-signaling pathway after quercetin supplementation is warranted.
Conclusion
PCBs disrupt the gene expression of transmembrane TJPs (Ocln, Cldn5 and JAM) and cytoplasmic accessory TJPs (ZO-1, ZO-2 and AF6) on cerebrum, cerebellum and hippocampus. Quercetin scavenges the PCBs-induced ROS, thereby preventing the transmembrane TJPs and cytoplasmic accessory TJPs in cerebrum, cerebellum and hippocampus from degradation. Protein expression of all these TJ proteins will prove the PCB disruption in a more authentic manner.
Footnotes
Conflict of Interest
The authors declared no conflicts of interest.
Funding
The financial assistance is obtained from the University Grants Commission-Research Fellowship in Science for meritorious students (UGC-RFSMS) program (to KS).
