Abstract
Curcumin (Cur) has been used extensively in dietary supplement with antioxidant and anti-apoptotic properties. Although dibutyl phthalate (DBP) has adverse effects on the kidney, any association between DBP exposure and the role of Cur is unclear. We tested the hypothesis that exposure to DBP has adverse consequences on renal dysfunction in mice and the potential protective role of Cur in decreasing DBP-induced renal dysfunction via inhibiting oxidative stress and apoptosis. Kidney function, oxidative stress biomarkers, and apoptosis factors as well as Bcl-2 and Bax were investigated. The results showed a marked increase of renal dysfunction, oxidative stress and apoptosis level after DBP exposure compared to the control. While administration of Cur to DBP-treated mice may reduce these adverse biochemical changes compared with DBP-alone group. Overall, these results suggest that oxidative stress and apoptosis are involved in DBP-induced renal disorder, whereas Cur plays a protective role in inhibiting these two pathways.
Introduction
Curcumin (Cur), the principal curcuminoid of turmeric (Curcuma longa), can be extensively used as a herbal supplement, food flavoring, food coloring and cosmetics ingredient.1,2 It has increased in annual sales since 2012, largely due to an increase in its popularity as a dietary supplement. 3 North America is the biggest market for Cur, where sales exceeded US$20 million in 2014, while Europe leads market growth. 4 Recently, the potential renal protective effect of Cur has attracted much attention, as it is nontoxic and exhibits a variety of therapeutic properties, including anti-apoptotic and antioxidant activities.5,6 Certain studies have indicated that Cur may exert free radical scavenging effects on a variety of biological pathways involved in oxidative stress and apoptosis to reduce renal hypertrophy.7,8 Meanwhile, it can improve acute and chronic renal diseases such as diabetic nephropathy, nephrotoxicity, renal ischemia and so on. 9
Dibutyl phthalate (DBP), as one of the main plasticizers, can be immersed in the solution stored in polyvinyl chloride (PVC) medical devices at different concentrations. 10 Clinically, some people, including the blood or urine of dialysis patients and pregnant women, may be exposed to DBP for a long time. 11 Data from epidemiological studies have suggested DBP and its metabolites are associated with a series of adverse reactions in the kidney, and toxicological studies have also confirmed that DBP has potential nephrotoxicity.12–14 Cheng et al. reported that DBP-induced activation of ROS causes renal damage in KM mice. 13 Other studies suggest that diisodecyl phthalate (a homologue of DBP) could induce renal toxicity through apoptosis mediated by oxidative stress. 15
Of concern is that the adverse effects of DBP on the kidney and any association between DBP exposure and the role of Cur remains unknown, this study thus tests the hypothesis that short-term exposure to DBP has adverse consequences on renal dysfunction in mice and the potential protective role of Cur in decreasing DBP-induced renal disorder via inhibiting oxidative stress and apoptosis. To this end, creatinine (Crea) and urea nitrogen (Urea) levels in serum were detected to assess kidney function. Any changes in reactive oxygen species (ROS), malondialdehyde (MDA) and total antioxidant capacity (T-AOC) levels were measured to evaluate oxidative stress. In addition, the expression of extracellular regulated protein kinase1/2 (ERK1/2) and phosphorylated ERK1/2 (p-ERK1/2), apoptosis-related proteins [Cytochrome C (Cyt C), Caspase-9 (Casp-9), Caspase-3 (Casp-3)], as well as Bcl-2 and Bax in renal tissue, were observed to explore the underlying role of Cur in apoptosis pathway.
Materials and methods
Animals and ethics statement
Twenty-eight SPF male Kunming (KM) mice (3 weeks, 17 ± 2 g) were purchased from Hubei Experimental Animal Research Center (Animal license No.: SCXK 2008-0052). The animals were raised under standard conditions: 25 ± 1°C, 50–70% humidity, and 12 h light/dark cycle. Commercial diet and sterilized water were provided
Chemicals and kits
Cur (CAS: 458-37-7) and DBP (>99%, CAS: 84-74-2) were obtained Sigma-Aldrich (St Louis, MO, USA). Cyt C, Casp-9 and Casp-3 ELISA kits were purchased from eBioscience (San Diego, CA, USA). T-AOC colorimetric kit was purchased from Nanjing Jiancheng Bioengineering Institute (Nanjing, China). The rabbit anti-mouse p44/42 ERK1/2 (#4695), Phospho-p44/42 ERK1/2 (Thr202/Tyr204) (#4377), and goat anti-rabbit IgG antibodies were purchased from Cell Signaling Technology (Danvers, Massachusetts, USA). The rabbit anti-mouse Bcl-2 (ba0412) and Bax (ba0315) were purchased from Bioss (Beijing, China).
Experimental protocol
KM mice were randomly divided into: control group, oral exposure group (50 mg/kg/day DBP), administration group (2.5 mg/kg/day Cur, intraperitoneal injection) and oral exposure + administration group (2.5 mg/kg/d Cur was given before the intragastric administration of 50 mg/kg/day DBP, DBP + Cur), with 7 mice in each group. Dose selection for Cur (2.5 mg/kg/d) is based on a pharmacological report by Bhutani et al. 16 The exposure dosage of DBP (50 mg/kg/d) was selected as the optimized dosage according to Wang et al. 8 Experimental mice received daily treatment for 28 days.
Sampling and testing
The mice were sacrificed after a period of 4 weeks, the serum of the mice was obtained to determine Urea and Crea level by using iMagic-M7 automatic biochemical Analyzer (Shenzhen Cooper Biotechnology Co., Ltd., China). The collected kidney for the preparation of a homogenate was used to determine ROS, MDA, T-AOC, Cyt C, Casp-9 and Casp-3 levels. The fluorescence intensity of ROS in the renal supernatant were determined by DCFH-DA fluorescent assay, at an excitation wavelength of 488 nm and an emission wavelength of 525 nm using an Infinite M200 PRO fluorescence reader (Tecan, Giessen, The Netherlands).
13
MDA contents in renal supernatant were determined by thiobarbituric acid (TBARS) assay, and were calculated as follows: MDA (μmol·g−1 prot) = [6.45(
Western blot
The expression of ERK1/2 and p-ERK1/2 protein in kidney tissues of mice in each group was detected by Western blot. 19 After the frozen mouse kidney tissue was lysed with RIPA lysate, the total protein was extracted, and the protein concentration in the sample was determined by BCA. The protein was separated by 10 µl 10% SDS-PAGE gel electrophoresis, and then transferred to 0.45 µm polyvinylidene fluoride (PVDF) membrane. An anti-blocking buffer was added to shake at room temperature for 1 hour, and then β-Actin (5% skim milk, 1: 10000), p44/42 ERK1/2 (5%BSA, 1:2000) and Phospho-p44/42 ERK1/2 (5%BSA, 1:1000) antibodies were added and sealed overnight at 4°C. The TBST membrane was washed and incubated with sheep anti-rabbit second antibody at room temperature for 1 h. The gray values of β-Actin, ERK1/2 and Phospho-ERK1/2 bands were collected, and β-Actin was used as internal reference. The gray ratios of ERK1/2, Phospho-ERK1/2 and β-Actin of each sample were obtained respectively.
Immunohistochemistry
The renal tissue of tested mice was collected and fixed with 4% paraformaldehyde with 24 h. The sections (5 µm thickness) were then dewaxed and mounted on glass slides. After antigen repair and inactivation of endogenous enzymes, immunohistochemical analyses were performed as described previously using the following antibodies. 20 Bcl-2 and Bax were the first antibodies, and goat anti-rabbit IgG antibodies were the second antibodies. Bcl-2 and Bax expressions were observed under a microscope respectively. Immunohistochemical staining was quantified as a mean density using Image Pro-Plus software (version 6.0; Media Cybernetics, Baltimore, MD, USA).
Statistical analysis
The obtained experimental data were presented as mean ± standard error of the mean (SEM) and analyzed by GraphPad Prism 5.0 (San Diego, CA, USA). The differences between groups were analyzed for significance by performing a one-way analysis of variance combined with a Tukey test. A
Results
Cur improves DBP- induced kidney dysfunction
The changes of serum Urea and Crea contents in each group are shown in Figure 1. The levels of Urea and Crea in DBP group were markedly higher than those in control group (

Urea, Crea levels in serum of mice in each group (
Cur reduces DBP-promoted oxidative stress
As shown in Figure 2, compared with the control group, the levels of ROS and MDA of renal tissues in the DBP group significantly increased (

The ROS, MDA and T-AOC levels in renal tissue of mice in each group (
Cur alleviates DBP-mediated p-ERK 1/2 levels
Figure 3 shows the western blotting analysis of ERK1/2 and p-ERK1/2 protein expression in kidney tissue of mice, demonstrating a significant up-regulated (
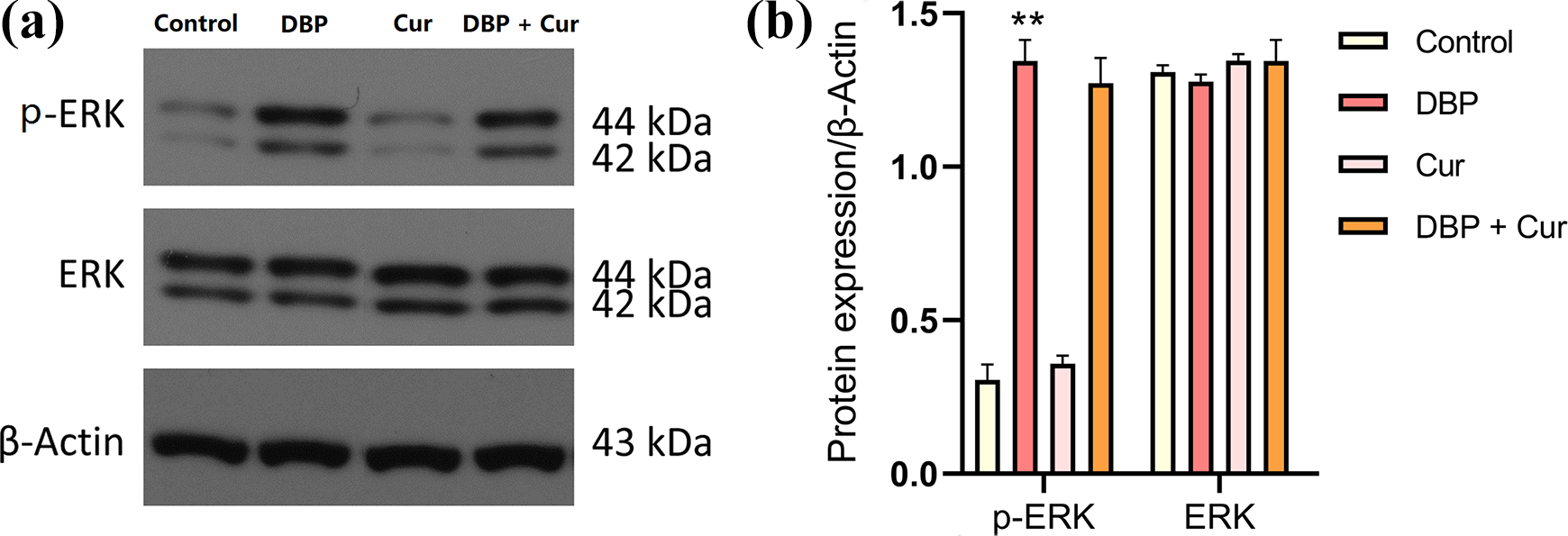
Expression of ERK1/2 and p-ERK1/2 proteins in kidney tissue of mice in each group. (a) Western blot protein bands, (b) ratio of p-ERK1/2/β-Actin and ERK1/2/β-Actin following the different treatments.
Cur decreases DBP-induced apoptotic level
The apoptosis detecting result is shown in Figure 4. Cyt C, Casp-9 and Casp-3 levels in renal tissue were markedly increased in DBP group compared with the control group (
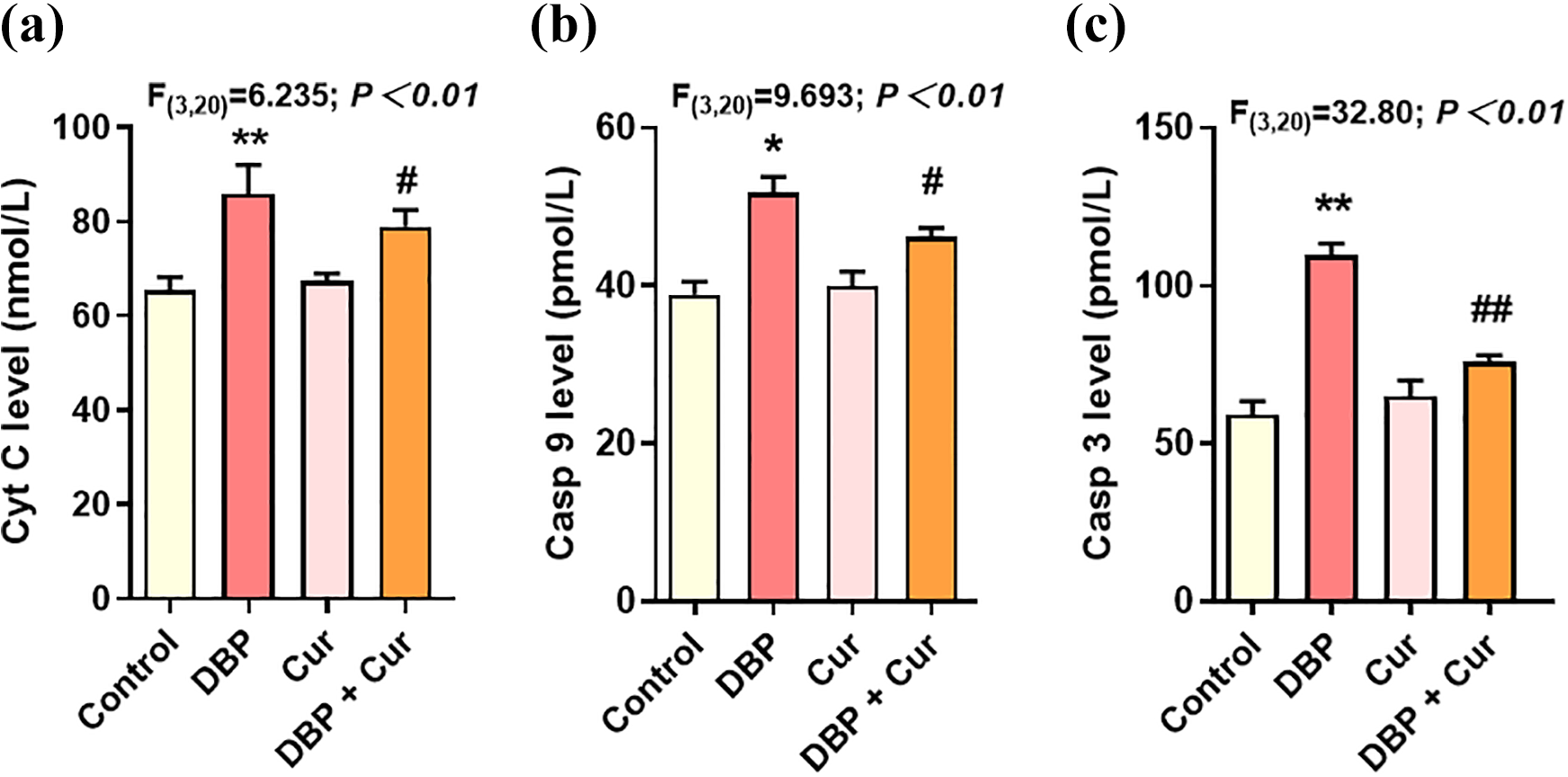
The Cyt C, Casp-9 and Casp-3 levels in renal tissue of mice in each group (
Cur increases DBP-downregulated Bcl-2 and the Bax/Bcl-2 ratio
Both Bax and Bcl-2 protein was expressed in the cytoplasm (the strong positive place was brown) (Figure 5a). As shown in Figure 5b–c, exposure to DBP significantly increased levels of Bax proteins, and also downregulated Bcl-2 and the Bax/Bcl-2 ratio compared with control group. After Cur treatment, the ratio of Bcl-2 to Bax in DBP + Cur group was significantly higher than that in DBP group (

Immunohistochemical result for Bax and Bcl-2. (a) Immunohistochemical staining (×200), (b) average optical density analysis (n = 3), and (c) the ratio of Bcl-2/Bax.
Discussion
Previous studies have pointed out that DBP is a peroxisome proliferator-activated receptor (PPARs) inducer, and DBP-mediated PPARs activation is considered to be one of the main mechanisms of DBP-induced hepatorenal toxicity. 21 Other studies suggest that DBP could also act as an exogenous oxide to stimulate the body to produce oxidative damage, indicating that the oxidative stress is associated with DBP-induced hepatic and renal damage. 13 This study showed that expose to DBP can up-regulate p-ERK1/2 expression and even increase the apoptosis level, resulting in renal dysfunction or abnormality of the tested mice. Moreover, treatment with Cur, the level of oxidative stress and the expression of apoptotic proteins in renal tissue of DBP-exposed mice decreased, and the Crea and Urea levels in serum tended to be normal. The above results suggest that Cur has a certain protective effect on DBP-induced renal dysfunction through inhibiting oxidative stress and apoptosis (Figure 6).

Graphical abstract.
The kidney, an important target organ of DBP, excretes Crea and Urea, thus Crea and Urea levels are two key indexes for quantitatively assessing renal function.15,22 A significant increase of Crea in serum indicates glomerular filtration dysfunction. High serum Crea value, which mostly means renal abnormality. As Urea is the main end product of the kidney, the serum urea level increases, indicating that the renal function is impaired accordingly. 22 The results showed that Urea and Crea levels of DBP-exposed mice increased markedly, suggesting that DBP could induce renal dysfunction, however these alternations were significantly reduced after treatment with Cur, which is consistent with the results conducted by Cheng et al. 13
This study further confirmed that DBP can significantly increase the level of ROS in renal tissue, while the accumulation of ROS will change the homeostasis of cell membrane to increase the content of MDA and reduce the antioxidant capacity, resulting in more serious tissue damage. In addition, ROS produced during oxidative stress participates in the signal transduction pathway as an upstream signal molecule, resulting in pathological changes and organ dysfunction.23,24 Several researches have reported that Cur has an antioxidant activity against oxidative stress. 25 After treatment with Cur, the levels of ROS and MDA in kidney tissue of mice exposed to DBP were downregulated, and the antioxidant capacity was improved. Samarghandian et al. provides an evidence that Cur can prevent restraint stress-induced oxidative damage in the brain, liver and kidney of rats, suggesting that Cur may be useful agents against oxidative stress in the tissues. 25 Our results also showed that oxidative stress was involved in the renal dysfunction caused by DBP, and Cur could play a protective role in blocking ROS accumulation.
ERK1/2 is a member of the mitogen-activated protein kinases (MAPK) family in eukaryotes, and its phosphorylation may play a crucial role in cellular oxidative stress pathway. 26 Previous studies have shown that DIDP (a homologue of DBP) as an extracellular signal can activate p38MAPK and cause corresponding post-transcriptional biological effects. 27 Therefore, the renal dysfunction caused by DBP may be associated with the ERK1/2 activation. 13 Increasing evidence has also pointed out that excessive activation of p-ERK1/2 is involved in the occurrence and development of kidney-related oxidative damage. 28 The results showed that the p-ERK1/2 level in renal tissue of DBP-exposed mice was higher than that in control group, whereas treatment with Cur, p-ERK1/2 level in DBP + Cur group tended to decrease to protect renal tissue from oxidative damage.
As reported, the release of Cyt C across the outer mitochondrial membrane into cytoplasm led to the apparent increased ratio of Cyt C cytosolic/mitochondria and activated Cyt C-caspase-3 cascade activation, eventually causing the cell apoptosis. 29 Cyt C activates casp-9 and through a cascade of caspases the apoptosis is executed by active casp-3. Furthermore, Cyt C release is induced by expression of the pro-apoptosis gene Bax, the anti-apoptosis gene Bcl-2 blocks the release of Cyt C and activation of caspases. 30 Bcl-2 is an anti-apoptotic while Bax enhances apoptosis, the decrease ratio of Bcl-2: Bax promotes apoptosis. The results showed that Bax and Bcl-2 levels in renal tissue of DBP-exposed mice increased, but the relative expression of Bax was higher, suggesting that there was a tendency to promote apoptosis, which was consistent with Li et al.’s study that DBP induce apoptosis by regulating the PI3K/Akt/Bcl-2 signaling pathway in rat insulinoma cells. 31 After treatment with Cur, the increase of bcl-2 level and the decrease of bax in DBP + Cur group indicated that the resistance of cells to apoptosis was enhanced, suggesting that Cur could inhibit apoptosis.
Conclusions
Taken together, the protective effect of Cur on the kidney of DBP-exposed mice may be related to its ability to block the formation of free radicals induced by DBP. On the other hand, the protective effect of Cur may also be associated with the inhibition of the high expression of p-ERK1/2, Bax and Casp-3 in the apoptosis pathway. In view of the complexity of the pharmacological effects of Cur, the precise protective mechanism of Cur to DBP-induced renal dysfunction remains to be further studied.
Highlights
Continuous 28-day exposure to DBP can cause renal dysfunction in tested mice.
Oxidative stress and apoptosis are involved in DBP-induced renal dysfunction.
Cur plays a protective role in DBP-induced renal dysfunction by inhibiting oxidative stress and apoptosis.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was funded by the Health and Family Planning Commission Scientific Research Project of Hubei Province (WJ2017Z027).
