Abstract
2-Acetylaminofluorene (2-AAF) is a known hepatic carcinogen which leads to tumour formation in rodents. 18-β Glycyrrhetinic acid (18β-GA) derived from liquorice plant has various pharmacological properties such as anti-ulcer, anti-inflammatory, antiviral, hepatoprotective and antioxidant. This study is designed to elucidate the chemopreventive properties of 18β-GA against 2-AAF-induced liver toxicity in Wistar rats and evaluated its effect on inflammatory and tumour promotion marker and activities of different oxidative stress enzymes. Administration of 2-AAF at the dose of (50 mg/kg body weight (b.w.) intraperitoneally (i.p.)) for five consecutive days induces hepatic toxicity, inflammation, oxidative stress and hyperproliferation. Pretreatment with 18β-GA at two different doses (45 and 75 mg kg−1 b.w.) significantly ameliorates 2-AAF-induced increased lipid peroxidation, alanine transaminase and aspartate transaminase, xanthine oxidase activities and activities of phase-II detoxifying enzymes along with the levels of glutathione content. Administration of 18β-GA also significantly restored the expressions of proliferating cell nuclear antigen, cyclooxygenase 2, inducible nitric oxide synthase and nuclear factor κB. Furthermore, histological observations also support the preventive effects of 18β-GA. Our findings suggest that pretreatment with 18β-GA showed potential hepatoprotective effects via attenuation of oxidative stress, inflammation and hyperproliferation.
Introduction
Hepatocellular carcinoma (HCC) is the fifth most common cancer and third common cause of death from cancer worldwide. 1 It is reported that incidence of cancer has been increasing in developing countries, and it has become the fourth leading cause of death from cancer worldwide. 2,3 It is a multistep process that involves different genetic alterations leading to the malignant transformation of the hepatocytes. 4 Several studies have been done to define the pathogenesis of HCC using rodent models and have contributed to the current knowledge of HCC. Many chemically induced experiments have been conducted on rodents to evaluate the chemopreventive potentials of possible drugs. 5–7 2-Acetylaminofluorene (2-AAF) is an aromatic amine and a potent carcinogen that induces tumours in various species and tissues including liver. 8–11 2-AAF shows genotoxic effect by producing free radicals that damages DNA and other macromolecules. 12
Phytochemicals are promoted for the prevention and treatment of many health conditions, including cancer, heart disease, diabetes and high blood pressure. 13 Various fruits and vegetables having diversified and pharmacologically active phytochemicals have shown protective effects in reducing the risk of cancer development. 14,15 Several studies have shown that most of the natural products have the potential to suppress cancer and hence considered as cancer chemopreventive agents. 16 Liquorice is an ancient traditional Chinese medicinal plant, which is used as a conditioning and flavouring agent, and for the treatment of various inflammatory diseases. 17 Glycyrrhizin (GL) is a major bioactive compound obtained from the liquorice root extract that has various pharmacological properties. It is widely used for the treatment of chronic hepatitis C and human immunodeficiency virus infections. 18,19 GL exhibits its pharmacological functions through its biologically active metabolites such as 18-β glycyrrhetinic acid (18β-GA). 19 18β-GA is a pentacyclic triterpenoid and has the property to mask the bitter taste of various drugs. Several pharmacological effects of 18β-GA have also been reported which include anti-ulcer, anti-inflammatory, antiviral, hepatoprotective and antioxidant properties. 20–23 Therefore, this study is designed to investigate the chemopreventive potential of 18β-GA by studying the early markers of oxidative stress and inflammation in the liver.
Materials and methods
Chemicals
2-AAF, 18β-GA, reduced glutathione (GSH), oxidised glutathione (GSSG), nicotinamide adenine dinucleotide phosphate (NADPH), NADP+, flavin adenine dinucleotide, ethylenediaminetetraacetic acid (EDTA), thiobarbituric acid (TBA), pyrogallol, poly-
Experimental animals
Male albino rats of Wistar strain approximately 8 weeks old (weighing 180–200 g) were used for the experimental study. Animals were obtained from the Central Animal House Facility of Hamdard University, New Delhi, India. All the animals used in this study were reviewed and approved by Institutional Animal Ethical Committee, accredited by the Committee for Purpose of Control and Supervision on Experiments on Animals (CPCSEA), Government of India. Animals were housed in polypropylene cages in groups of six rats per cage. Animals were acclimatized in a room maintained at 25 ± 2°C with 12-h light/12-h dark cycle. Animals were given free access to standard laboratory feed (Hindustan Lever Ltd, Bombay, Maharashtra, India) and water ad libitum. Approval ID/project number for this study is 972.
Treatment regimen
To study the protective effects of 18β-GA against 2-AAF in the liver, 30 male Wistar rats were randomly allocated into five groups having six in each group. A schematic representation of treatment design has been shown in Figure 1.

Schematic representation of treatment regimen.
Group I served as control receiving distilled water (5 ml kg−1 body weight (b.w.)) by oral gavage from day 1 to day 20 and corn oil at the dose of 5 ml kg−1 b.w., intraperitoneally (i.p.) once daily for five consecutive days from day16 till day 20. The animals had free access for water ad libitum.
Group II was administered 2-AAF (50 mg kg−1 b.w. i.p.) for five consecutive days from day16 till day 20 and had free access of water ad libitum and served as a toxicant group.
Groups III animals were pretreated with 18β-GA at the dose of 45 mg kg−1 b.w. by oral gavage from day 1 to day 15, and from day 16 up to day 20 2-AAF (50 mg kg−1 b.w.) was given. The 18β-GA was given 1 h before 2-AAF on days 16–20.
Group IV animals were pretreated with 18β-GA at the dose of 75 mg kg−1 b.w. by oral gavage from day 1 to day 15 and from day 16 up to 20 of 2-AAF(50 mg kg−1 b.w.) was given. The 18β-GA was given 1 h before 2-AAF on days 16–20.
Group V animals received higher dose of 18β-GA alone, that is, 75 mg kg−1 b.w. by oral gavage for 20 days.
All animals were killed by cervical dislocation 24 h after the last injection of 2-AAF and processed for subcellular fractionation; livers were quickly removed and perfused in ice-cold saline. Blood samples were taken for various serological parameters. Liver tissues were taken at the same time for immunohistochemistry, various biochemical and histological parameters.
Biochemical estimation
Tissue processing and preparation of post-mitochondrial supernatant (PMS) was done immediately after killing the animals. For all biochemical estimations, liver tissue, cytosol and serum samples were utilized.
Assay for serum oxaloacetate and pyruvate transaminase activity (ALT and AST)
Alanine transaminase (ALT) and aspartate transaminase (AST) activities were determined by previously described methods.
24
Briefly, each substrate (0.5 ml; 2 mM α-ketoglutarate and either 200 mM
After another 30 min at room temperature, the colour was developed by addition of 5.0 ml of NaOH (0.4N), and the product was read at 505 nm.
Assay for GR activity
GR activity was determined by the method followed by Carlberg and Mannervik. 25 The reaction mixture consisted of 1.65 ml phosphate buffer (0.1 M, pH 7.6), 0.1 ml EDTA (0.5 mM), 0.05 ml GSH (1 mM), 0.1 ml NADPH (0.1 mM) and 0.1 ml of 10% PMS in a total volume of 2 ml. Enzyme activity was quantified at 258°C by measuring the disappearance of NADPH at 340 nm and was calculated as nanomoles of NADPH oxidized per minute per milligram protein using a molar extinction coefficient of 6.22 × 103 M−1 cm−1.
Estimation of LPO
The assay for microsomal lipid peroxidation (LPO) was carried out following the method of Wright et al. 26 The reaction mixture in a total volume of 1.0 ml contained 0.58 ml phosphate buffer (0.1 M, pH 7.4), 0.2 ml microsomes, 0.2 ml ascorbic acid (100 mM), and 0.02 ml ferric chloride (100 mM). The reaction mixture was incubated at 37°C in a shaking water bath for 1 h. The reaction was stopped by addition of 1.0 ml 10% TCA. Following addition of 1.0 ml 0.67% TBA, all the tubes were placed in boiling water bath for 20 min and then shifted to crushed ice-bath before centrifuging at 2500g for 10 min. The amount of malondialdehyde formed in each of the samples was assessed by measuring optical density of the supernatant at 535 nm using spectrophotometer (Milton Roy 21 D) against a reagent blank. The results were expressed as nanomoles of malondialdehyde (MDA) formed per hour per gram tissue at 37°C using molar extinction coefficient of 1.56 × 105 M−1 cm−1.
Assay for XO activity
The activity of xanthine oxidase (XO) was assayed by the method described by Stirpe and della Corte. 27 The reaction mixture consisted of 0.2 ml PMS which was incubated for 5 min at 37°C with 0.8 ml phosphate buffer (0.1 M, pH 7.4). The reaction was started by adding 0.1 ml xanthine (9 mM) and kept at 37°C for 20 min. The reaction was terminated by the addition of 0.5 ml ice-cold perchloric acid (10% v/v). After 10 min, 2.4 ml of distilled water was added and centrifuged at 4000 r min−1 for 10 min, and microgram of uric acid formed per minute per milligram protein was recorded at 290 nm.
Assay for GST activity
Glutathione-S-transferase (GST) activity was assayed by the method followed by Habig et al. 28 and Athar et al. 29 The reaction mixture consisted of 1.475 ml phosphate buffer (0.1 M, pH 6.5), 0.2 ml reduced GSH (1 mM), 0.025 ml CDNB (1 mM) and 0.3 ml PMS (10% w/v) in a total volume of 2.0 ml. The changes in the absorbance were recorded at 340 nm and enzyme activity was calculated as micromoles of CDNB conjugate formed per minute per milligram protein using a molar extinction coefficient of 9.6 × 103 M−1 cm−1.
CAT activity
Catalase (CAT) activity was measured by the method followed by Claiborne. 30 The reaction mixture consisted of 2 ml phosphate buffer (0.1 M, pH 7.4), 0.95 ml H2O2 (0.019 M) and 0.05 ml PMS in a final volume of 3 ml. Changes in absorbance were recorded at 240 nm. CAT activity was calculated as nanomoles of H2O2 nanomoles per minute per milligram protein consumed.
GPx activity
Glutathione peroxidase (GPx) activity was assayed by the method described by Mohandas et al. 31 The assay mixture consisted of 0.1 M, pH 7.4 sodium phosphate buffer, 1 mM EDTA, 0.2 mM NADPH, 1 mM sodium azide, 1 IU ml−1 GR, 0.25 mM H2O2, and 0.2 m PMS (10% w/v) in a total volume of 2.0 ml. The enzyme activity was calculated as micromoles of NADPH oxidised per minute per milligram protein with the molar extinction coefficient of 6.22 × 103 M−1 cm−1.
Estimation of reduced GSH
Reduced GSH was determined by the methods followed by Athar and Iqbal 32 and Jollow et al. 33 Then, 1.0 ml sample of PMS was precipitated with 1.0 ml of sulphosalicylic acid (4%). The samples were kept at 4°C for 1 h and then centrifuged at 1200g for 20 min at 4°C. The assay mixture contained 0.1 ml filtered aliquot, 2.7 ml phosphate buffer (0.1 M, pH 7.4) and 0.2 ml 5,5′-dithio-bis(2-nitrobenzoic acid) (DTNB; 100 mM) in a total volume of 3.0 ml. The yellow colour developed was read immediately at 412 nm on a spectrophotometer (Perkin Elmer Model Lamda EZ201, USA.).
Immunohistochemical staining for the detection of PCNA, iNOS, COX-2 and NF-κB
To examine the protective effects of 18β-GA on 2-AAF-induced inflammation and proliferation in liver tissue was assessed by immunohistochemical staining. The liver tissues were fixed in formalin and embedded in paraffin. Sections of 5 µm thickness were cut on to poly-
Histopathological analysis
Fixation, dehydration, infiltration and block preparation. The tumour from livers were excised out and fixed in bovine’s fluid for 24–28 h. The tissues were then rehydrated by passing through graded series of ethyl alcohol (50%, 70%, 90% and 100%) for 1 h in each giving two changes. These were then cleared in xylene (two changes of 1 h each). The cleared tissue was placed for 5 min in xylene-containing molten paraffin wax at 50–60°C for infiltration. Sections were deparaffinized by dipping in xylene and given two changes. These slides were then passed through graded concentrations of ethyl alcohol (30%, 50%, 70%, 90% and 100%) with two changes of 2 min each. Then stained with haematoxylin for 1 min and again washed in running water thoroughly. Slides were then passed through 50% and 70% ethyl alcohol and subsequently put in eosin stain (prepared in 70%, 90% and 100%, 100% + xylene (1:1)) and finally they were given two changes of xylene. All slides were mounted in DPX. They were covered with glass coverslips and kept at room temperature for drying. To avoid any type of bias, slides were coded and examined by histopathologist blinded to the study. Liver sections were evaluated at 40× magnification.
Mast cell staining
For detection of mast cells, liver was fixed in methacarn solution (methanol:chloroform:glacial acetic acid::4:2:1) overnight at 4°C and then fixed in 4% neutral-buffered formalin for additional 24 h. The liver sections of 4 µm were cut from formalin-fixed, paraffin-embedded tissue blocks and mounted on poly-
Estimation of protein
The protein concentration in all samples was determined by the method described by Lowry et al. 34 using BSA as standard.
Statistical analysis
The data from individual groups were presented as the mean ± SD. Differences between groups were analysed using analysis of variance followed by Tukey–Kramer multiple comparisons test, and minimum criterion for statistical significance was set at p < 0.05 for all comparisons.
Results
18β-GA attenuates the serum ALT and AST activity
Effect of 18β-GA on serum AST and ALT levels was observed. It shows significant increase in these parameters in the 2-AAF-treated groups as compared to control (***p < 0.001). It was found that pretreatment with 18β-GA significantly (# p < 0.05; ## p < 0.01) reduces the level of these markers when compared with 2-AAF-treated group (Figures 2 and 3). 18β-GA alone exhibits no significant difference as compared to control.

Effect of 18β-GA on the hepatotoxicity marker AST and ALT. Each value represents as mean ± SE of six animals. Their levels were significantly (***p < 0.001) increased in 2-AAF-treated group II as compared to group I. 18β-GA: 18-β glycyrrhetinic acid; ALT: alanine transaminase; AST: aspartate transaminase; 2-AAF: 2-acetylaminofluorene.
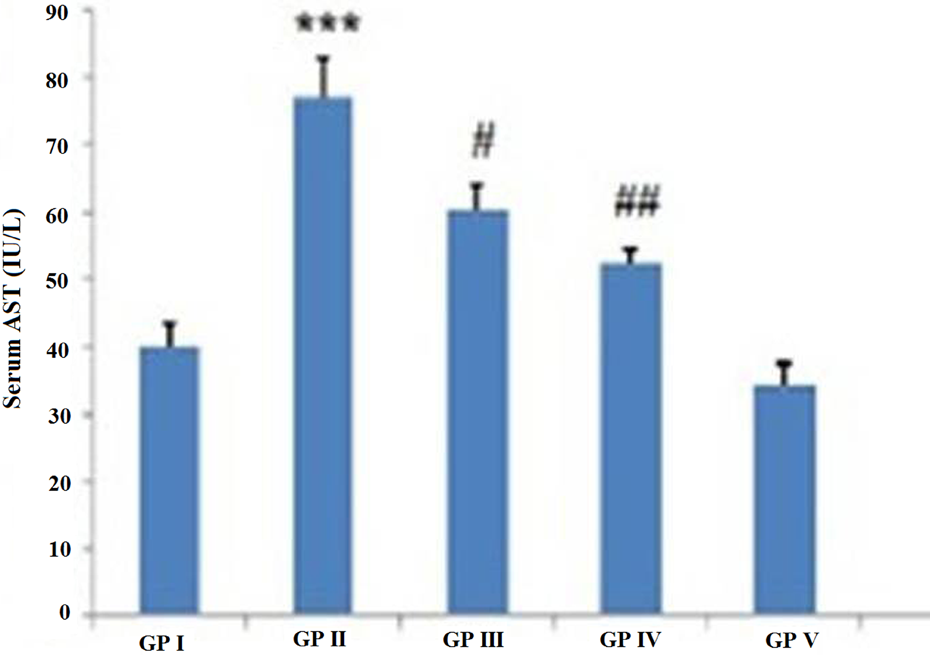
18β-GA pretreatment attenuates the levels of AST and ALT significantly (# p < 0.05, ## p < 0.01) in groups III and IV as compared to 2-AAF alone-treated group. 18β-GA: 18-β glycyrrhetinic acid; ALT: alanine transaminase; AST: aspartate transaminase; 2-AAF: 2-acetylaminofluorene.
18β-GA pretreatment decreased MDA formation
The level of MDA was measured to demonstrate the oxidative damage on LPO of 2-AAF induced liver injury in Wistar rats. MDA level significantly (***p < 0.001) increased in the 2-AAF-treated group II when compared with control (group I). Low (45 mg kg−1 b.w.) and high (75 mg kg−1 b.w.) dose of 18β-GA pretreatment in groups III and IV significantly (## p < 0.01; ### p < 0.001) prevent membrane damage when compared with 2-AAF-treated group II (Figure 4). No significant difference was observed in the MDA level between control and high dose of 18β-GA alone-treated group.

Effect of 18β-GA pretreatment on LPO (MDA level) in hepatic injury induced by 2-AAF. Data were expressed as mean ± SE of six animals and measured in nanomoles of MDA formed per gram tissue. MDA level was significantly (***p < 0.001) increased in 2-AAF-treated group II as compared to control group I. 18β-GA pretreatment significantly (## p < 0.01, ### p < 0.001) decreases the level of MDA content in groups III and IV as compared to group II. 18β-GA: 18-β glycyrrhetinic acid; LPO: lipid peroxidation; MDA: malondialdehyde; 2-AAF: 2-acetylaminofluorene.
Effect of 18β-GA pretreatment and 2-AAF on the XO activity in liver
The activity of XO was significantly (***p < 0.001) enhanced in toxicant group as compared to control. 18-βGA pretreatment groups III and IV (# p < 0.05; ## p < 0.01) shows decreased activity of XO as compared to group II. No significant change was observed in group V as compared to group I (Figure 5).

Effect of 18β-GA pretreatment and 2-AAF on XO activity in liver. Each value represents as mean ± SE of six animals and measured as micrograms of uric acid formed per minute per milligram protein. 2-AAF leads the significant elevated level (***p < 0.001) of XO in group II as compared to group I. 18β-GA-treated groups III and IV significantly (# p < 0.05, ## p < 0.01) decrease the level of enzyme activity as compared to group II. 18β-GA: 18-β glycyrrhetinic acid; XO: xanthine oxidase; 2-AAF: 2-acetylaminofluorene.
Effect of 18β-GA supplementation on CAT activity
The activity of CAT was significantly depleted (***p < 0.01) in group II as compared to group I. 18β-GA pretreatment at the dose of 45 mg kg−1 b.w. significantly (# p < 0.05) enhanced the activity of CAT in group III as compared to group II. The higher dose of 18β-GA (75 mg kg−1 b.w.) also significantly enhance (## p < 0.01) CAT activity in group IV as compared to group II. No significant difference in the activity of CAT was found between groups V and I (Figure 6).

Group I represents control group. Group II represents 2-AAF-treated group. Group III represents prevention group treated with low dose (45 mg kg−1 b.w) of 18β-GA in addition of 2-AAF. Group IV represents high dose (75 mg kg−1 b.w.) of 18β-GA and 2-AAF. Group V represents high dose 18β-GA alone-treated group. Effect of 18β-GA pretreatment on hepatic CAT activity. Each value represents mean ± SE of six animals and measured as nanomoles of H2O2 consumed per minute per milligram protein. CAT activity was significantly decreased (***p < 0.001) 2-AAF alone-treated group as compared to control group. 18β-GA pretreatment groups III and IV significantly (# p < 0.05, ## p < 0.01) increased CAT activity when compared with only 2-AAF-treated group. 2-AAF: 2-acetylaminofluorene; 18β-GA: 18-β glycyrrhetinic acid; CAT: catalase; b.w.: body weight.
Effect of 18β-GA pretreatment on GSH level
The level of GSH was significantly (c p < 0.01) decreased in the 2-AAF-treated group II as compared to group I. GSH level significantly (d p < 0.05; e p < 0.01) increased in 18β-GA pretreatment groups III and IV when compared with group II. No significant difference was observed between groups I and V (Table 1).
Effect of 18β-GA and 2-AAF on the activities of GPx, GST, GR and reduced GSH on the liver of rats.a

2-AAF: 2-acetylaminofluorene; 18β-GA: 18-β glycyrrhetinic acid; GSH: glutathione; GPx: glutathione peroxidase; GST: glutathione-S-transferase; GR: glutathione reductase; D1: dose 1; D2: dose 2; NADPH: nicotinamide adenine dinucleotide phosphate; CDNB: 1-chloro-2,4-dinitrobenzene; DTNB: 5,5′-dithio-bis(2-nitrobenzoic acid); NS: non-significant.
aValues of GSH-dependent enzymes (GPx, GST, GR and GSH) are expressed asmeans ± SE (n = 6).
b p < 0.001: significant when compared with group I.
c p < 0.05: significant when compared with group I.
d p < 0.05: significant when compared with group II.
e p < 0.01: significant when compared with group II.
Effect 18β-GA on GSH-dependent enzyme
18β-GA caused a significant depletion in the level of GPx (b p < 0.001), GST (b p < 0.001) and GR (c p < 0.01; Table 1) in group II as compared to group I. Pretreatment with 18β-GA significantly (d p < 0.05, e p < 0.01) restored all the antioxidant enzyme level at both doses (45 and 75 mg kg−1 b.w.). We found that there is no significant difference in the activity of these enzymes between control group and 18β-GA alone-treated groups.
Effect of 18β-GA on the histology of the liver
Tissue section of 2-AA-treated group revealed marked changes (40× magnifications) when compared with the control group animals. Cytoplasmic granulation with large hyperchromatic nuclei and vacuolar degeneration are found around the central vein in liver tissue of group II animals, while 18β-GA pretreatment protects the liver architecture against 2-AAF-induced alteration in groups III and IV (Figure 7).

Effect of 18β-GA pretreatment against 2-AAF-induced histological alteration on the liver of Wistar rats. Histopathological profile of group I shows normal cellular architecture (indicated by arrows), whereas group II shows large hyperchromatic nuclei and vacuolar degeneration around the central vein (indicated by arrows) of the liver tissue. 18β-GA supplementation on both doses D1 and D2 in groups III and IV prevent vacuolar degeneration around the central vein. Group V shows normal histology as observed in group I (original magnification ×40). 2-AAF: 2-acetylaminofluorene; 18β-GA: 18-β glycyrrhetinic acid.
Effect of 18β-GA on the expression of PCNA, iNOS, COX-2 and NF-κB
Immunohistochemical analysis (original magnification 40×) of PCNA, iNOS, COX-2 and NF-κB has been shown in Figures 8, 9, 10 and 11, respectively. There are more number of cells showing expression of these markers in group II as indicated by brown colour as compared to group I. 18β-GA pretreated (both doses 45 and 75 mg kg−1 b.w.) groups III and IV show reduced number of cells having the expression of these proteins as compared to group II. There is no significance difference observed in groups I and V.

Effect of 18β-GA and 2-AAF on the PCNA in the hepatic tissue. Photomicrograph represents the expression of PCNA in various experimental animals. Brown colour indicates the expression of PCNA and bluish colour indicates the haematoxylin staining. Group I represents untreated group with no expression. Group II shows intense brown colour as compared to group I. 18β-GA supplementation on both doses D1 and D2 in groups III and IV shows less brown colour as compared to group II. No significant difference was observed between groups V and I. 2-AAF: 2-acetylaminofluorene; 18β-GA: 18-β glycyrrhetinic acid; PCNA: proliferating cell nuclear antigen.

Effect of 18β-GA and 2-AAF on the expression of iNOS in the hepatic tissue. Photomicrograph represents the expression of iNOS in various experimental animals. Brown colour indicates the expression of iNOS and bluish colour indicates the haematoxylin staining. Group I represents untreated group with no expression. Group II shows high expression as compared to group I. 18β-GA administration on both doses D1 and D2 in groups III and IV shows less expression as compared to group II. No significant difference was observed between groups I and V. 2-AAF: 2-acetylaminofluorene; 18β-GA: 18-β glycyrrhetinic acid; iNOS: inducible nitric oxide synthase.

Effect of 18β-GA and 2-AAF on the COX-2 in the hepatic tissue. Photomicrograph represents the expression of COX-2 in various experimental animals. Brown colour indicates the expression of COX-2 and bluish colour indicates the haematoxylin staining. Group I represents untreated group with no expression. Group II shows high expression as compared to group I. 18β-GA administration on both doses D1 and D2 in groups III and IV shows less expression as compared to group II. No significant difference was observed between groups I and V. 2-AAF: 2-acetylaminofluorene; 18β-GA: 18-β glycyrrhetinic acid; COX-2: cyclooxygenase-2.

Effect of 18β-GA and 2-AAF on the expression of NF-κB in the hepatic tissue. Photomicrograph represents the expression of iNOS in various experimental animals. Brown colour indicates the expression of iNOS and bluish colour indicates the hematoxylin staining. Group I represents untreated group with no expression. Group II shows high expression as compared to group I. 18β-GA administration on both doses D1 and D2 in groups III and IV shows less expression as compared to group II. No significant difference was observed between groups I and V. 2-AAF: 2-acetylaminofluorene; 18β-GA: 18-β glycyrrhetinic acid; iNOS: inducible nitric oxide synthase.
Effect of 18β-GA on mast cell infiltration
In 2-AAF-treated group II, there is high density of mast cell in the portal tract and fibrous septa of the liver section. Mast cells density found in lesser extent in groups III and IV as compared with group II, while there is no mast cell infiltrations in liver section of groups I and V (Figure 12).

Effect of 18β-GA and 2-AAF infiltration on mast cells. There is high mast cell density in group II. Pretreatments with 18β-GA attenuated the mast cell infiltration in groups III and IV as compared to group II (×40 magnification). There is no mast cell infiltration in the liver section of groups I and V. 2-AAF: 2-acetylaminofluorene; 18β-GA: 18-β glycyrrhetinic acid.
Discussion
Chemoprevention is one of the promising and appealing strategies to combat cancer either by delaying or suppressing the process. Several studies have shown the chemopreventive potential of medicinal plants. 35,36 Phytochemicals having antioxidants properties are regarded as one of the most attractive strategies for cancer control; epidemiological data show that natural compounds and medicinal plants have efficiently inhibited tumour development in various organs, including the liver. 37,38 Hepatocellular carcinoma is one of the few human cancers that have vast potential of prevention. There are several causative agents for the induction of liver cancers such as viral infections and hepatitis. 39
18β-GA is obtained from the liquorice root which is used as medicine in China to reduce the clinical manifestations of chronic hepatitis C infections. 19 This study signifies that pretreatment with 18β-GA showed protection against 2-AAF-induced oxidative stress, inflammation and hyperproliferation in liver of Wistar rats. The elevated level of ALT and AST is associated with the liver toxicity; these transaminases are released into the circulation from cytoplasm after cellular damage 40 and become the damage marker of liver. Pretreatment with18β-GA shows the protection and maintains structural integrity of liver tissue which is evident from significant reduction in serum ALT and AST activities.
Human beings are constantly exposed to several factors that cause oxidative stress. Active oxygen species may be involved in carcinogenesis through two possible mechanisms, namely the induction of gene mutations that result from cell injury and the effects on signal transduction and transcription factors. Which mechanism it follows depends on factors such as the type of active oxygen species involved and the intensity of stress.
41
LPO is a marker of oxidative stress and plays an important role in cellular damage which in turn may ultimately lead to disease development. There was remarkable elevation in MDA level in 2-AAF treatment group which is in agreement with the previous findings.
42
GSH is a tripeptide antioxidant enzyme. Peroxidation of lipid membrane is protected by GSH, conjugating with the electrophile such as 4-hydroxy-3-nonenal, formed during LPO and thus gets depleted in this conjugation reaction.
43
Pretreatment with 18β-GA potentially attenuated the 2-AAF-induced elevated MDA and depleted level of GSH. Furthermore, it was also observed that 2-AAF administration enhances the activity of XO.
44
XO reduces O2 to superoxide anion radical
Cell proliferation rate is an important marker of cancer development 46 and plays an important role in liver carcinogenesis. Here we have analysed the PCNA, an important marker of cell proliferation. PCNA expression has been associated to the late G1 as well as early S phase of cell cycle. 47 Our result suggested that the extent of cell proliferation in hepatic tissue due to the 2-AAF administration was suppressed by 18β-GA. Immunohistochemical analysis revealed high expression of PCNA in the preneoplastic liver tissue from the animals of group II when compared with control. 18β-GA-treated group remarkably decreased the number of PCNA positive cells. It shows that 18β-GA suppresses abnormal proliferation of initiated hepatocytes. Studies have also shown that inflammation plays a potential role in development of most of the chronic diseases including cancer. 48 Blockade of inflammation mediating intracellular signalling pathway is now considered as a road map for the development of molecular target-based chemopreventive agent. 49 The modulation of COX-2 and iNOS-mediating signalling pathway has been considered a new approach for preventing carcinogenesis. 50 This study reveals that 18β-GA suppresses the expressions of COX-2 and iNOS in rat liver through downregulation of NF-κB expression.
Studies have shown that 18β-GA also has anti-inflammatory properties. 51 Various research findings demonstrate that mast cell play an important role in the development of hepatocellular carcinoma. 52,53 Immunohistochemical finding shows that mast cell density was significantly higher in the liver section of toxicant group as compared to the control group. 18β-GA pretreatment potentially suppresses mast cell infiltration in the portal tract and fibrous septa of groups III and IV. Therefore, we can conclude that 18β-GA may be used as a hepatoprotective agent as it has shown strong modulatory potential against 2-AAF-induced inflammation, hyperproliferation and generation of reactive oxygen species (ROS) via induction of antioxidant enzyme, suppression of ROS and depleted level of tumour promoter marker. However, further studies are warranted to elucidate the exact protective mechanism of 18β-GA.
Footnotes
Acknowledgements
Author SS is thankful to the Indian Council of Medical Research (ICMR), New Delhi, India, for providing fellowship to the first author (SKH) to carry out this work.
Conflict of interest
The authors declared no conflicts of interest.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
