Abstract
Paraquat (PQ) is a well-known quaternary nitrogen herbicide. The major target organ in PQ poisoning is the lung. Reactive oxygen species (ROS) and inflammation play a crucial role in the development of PQ-induced pulmonary injury. Neopterin is synthesized in macrophage by interferon γ and other cytokines. We aimed to evaluate the utility of neopterin as a diagnostic marker in PQ-induced lung toxicity. Sprague Dawley rats were randomly divided into two groups (sham and PQ), administered intraperitoneally 1 mL saline and PQ (15 mg/kg/mL) respectively. Blood samples and lungs were collected for analyses. Lung injury and fibrosis were seen in the PQ group. Serum total antioxidant capacity, lactate dehydrogenase (LDH), and lung transforming growth factor-1β (TGF-1β) levels were significantly higher than the sham group (in all,
Introduction
Paraquat (PQ, 1-10-dimethyl-40-bipyridylium dichloride) is a quaternary nitrogen herbicide. It is one of the most widely used herbicides and is a highly toxic compound for both humans and animals. Following the acute intoxication, death may occur. 1,2 In serious PQ intoxication, PQ causes aggressive tissue damage in lung, kidney, and liver. The major target organ is the lung in PQ poisoning. 1,2
PQ-induced lung injury is characterized by edema, hemorrhage and hypoxemia, infiltration of inflammatory cells into the interstitial and alveolar spaces, proliferation of fibroblasts and excessive collagen deposition, damage of alveolar epithelial cell, and proliferation of bronchial epithelial cells. 3 It has been accepted that reactive oxygen species (ROS) play a crucial role in the development of PQ-induced pulmonary injury. PQ is a potent ROS inducer and induces the formation of ROS such as superoxide anion and hydroxyl and peroxyl radicals. 1,4–6 In addition to oxidative stress, inflammation has been reported as another mechanism in the progression of lung injury. 7 It has been reported that lung cells produce inflammatory mediators such as cytokines and chemokines in case of oxidative stress. They induce recruitment of inflammatory cells and activation of transcription factors such as nuclear transcription factor-κB (NF-κB). 8 Moreover, NF-κB induces various proinflammatory genes such as inducible nitric oxide synthase and cyclooxygenase-2. It enhances the inflammatory process. 9 Finally, PQ leads to pneumonitis and pulmonary fibrosis. 1
Neopterin, a guanosine triphosphate metabolite, is produced and released by macrophages and monocytes in response to stimulation by interferon-γ and other cytokines. 10–13 Increased serum and tissue neopterin levels accompany immune responses and indicate immunologic T-cell activation. The measurement of neopterin levels is accepted as a biomarker of immunologic activation. 10,13 In addition, neopterin is known as an indicator for severity of some diseases. 10–15 It has been reported that neopterin levels are related to inflammatory processes in rat alveolar epithelial cells. 13
To the best of our knowledge, there are no reports about changes in serum neopterin levels in PQ-induced lung toxicity. In this study, we aimed to investigate whether the neopterin concentrations were elevated in tissues and evaluate the utility of neopterin as a diagnostic marker in PQ-induced lung toxicity.
Materials and methods
Animals
A total of 18 female Sprague Dawley rats (Health Sciences Institute, Gulhane Military Medical Academy, Ankara, Turkey), each weighing between 202 and 255 g were randomly divided into two groups (
Experimental design
A single dose of 15 mg/kg PQ (Sigma-Aldrich Laborchemikalien GmbH, Seelze, Germany) dissolved in 1 mL saline were administered intraperitoneally to animals in PQ group.
16
The animals in the sham group were given intraperitoneal injection of 1 mL saline alone. After the 4-day experimental period, all animals were anesthetized with ketamine (85 mg/kg) and xylazine (12.5 mg/kg) and their thoracic cavities were opened. We collected blood samples from all animals via cardiac puncture, allowed the samples to clot, and removed the serum centrifugation at 1000
Tissue preparation
The frozen lung tissues were homogenized in phosphate buffer (pH 7.4) using a homogenizator (HeidolphDiax 900; HeidolphElektro GmbH, Kelhaim, Germany) on an ice cube. The supernatants were centrifuged at 14,000 r/min for 10 min at 4°C. The supernatants were used for transforming growth factor-1β (TGF-1β) and malondialdehyde (MDA) analyses. The protein content of lung homogenates was measured by the method described by Lowry et al. with bovine serum albumin as the standard. 17
Biochemical assays
Serum lactate dehydrogenase (LDH) activities were measured with a spectrophotometric technique by the Olympus AU-2700 autoanalyzer using commercial kits (Olympus, Hamburg, Germany). Serum LDH activities were expressed in units per liter.
The lung tissue TGF-1β levels were measured by enzyme-linked immunosorbent assay kit according to the manufacturer’s instructions (Bender MedSystems GmbH, Vienna, Austria). Lung TGF-1β levels were expressed in pictogram per gram protein.
Total antioxidant capacity (TAC) assays were made using the method defined by Celik et al. 18 Serum TAC levels were expressed in micromoles Troloxequivalent per liter.
MDA levels were determined based on the production of thiobarbituric acid reactive substances in the supernatant by the method as described by Al-Fawaeir et al. 19 After the reaction of MDA with thiobarbituric acid, the reaction product was followed spectrophotometrically at 532 nm, using tetramethoxypropane as the standard. The results were expressed in millimole per gram protein.
Serum neopterin levels were measured with a high-performance liquid chromatography device (Agilent Technologies 1200 Series System, Santa Clara, California, USA), using the method defined by Agilli et al.
20
In brief, to 0.1 mL serum, 0.1 mL 100% acetonitrile was added and vortexed. The precipitated protein was removed by centrifugation at 4°C and 10,000
Histopathological evaluation
Specimens were fixed in 10% neutral-buffered formalin. After tissue processing, tissues were embedded in paraffin and 5-µm cut sections were stained with hematoxylin–eosin stain. Histologic sections were examined by light microscopy and evaluated by a pathologist blinded to the study groups. Histological alveolar epithelial injury, edema, hemorrhage, and inflammation were the parameters assessed. Histopathologic parameters were evaluated semiquantitatively relative to sham rats according to a 4-grade scale. Grade 0 indicates no changes; grade 1 shows minimal to mild change; grade 2 signifies moderate change; and grade 3 implies severe change. The extensity of histopathologic changes was also assessed as focal or diffuse.
Statistical analysis
We used Statistical Package for Social Sciences version 15.0 (SPSS Inc., Chicago, Illinois, USA) software to perform all statistical analyses and Mann–Whitney
Results
No deaths were observed in both groups (PQ or only saline). All laboratory parameters that belong to two groups are shown in Table 1.
Laboratory parameters and the degree of lung injury belong to the sham and PQ groups.a

MDA: malondialdehyde; TAC: total antioxidant capacity; TGF-1β: transforming growth factor-1β; LDH: lactate dehydrogenase; PQ: paraquat.
aAll data were expressed as median (minimum–maximum).
bMann Whitney
cχ2 test.
TGF-1β and MDA levels in lung of PQ-treated rats after 4 days were significantly enhanced (
The weights at the beginning of experimental study were not significantly different among the groups showing 224 g (202−242 g) in the sham group and 235 g (218−255 g) in the PQ group (
Representative histopathological pictures of the study groups are seen in Figure 1. In the sham group, all the structures of pulmonary parenchyma and bronchial tree were of normal appearance. Histopathological evaluation of rat lung following PQ revealed that lesions predominantly appeared in parenchymal region. There was inflammatory cell infiltration in interstitium and peribronchiolar areas. Extensive alveolar edema and massive alveolar hemorrhage were observed after PQ treatment. In addition, interalveolar septal thickening and mesothelial proliferation were also prominent. Inflammation was characterized principally by mononuclear inflammatory cell infiltration composed of macrophages in majority. However, neutrophil leucocytes were also observed among mononuclear inflammatory cells.
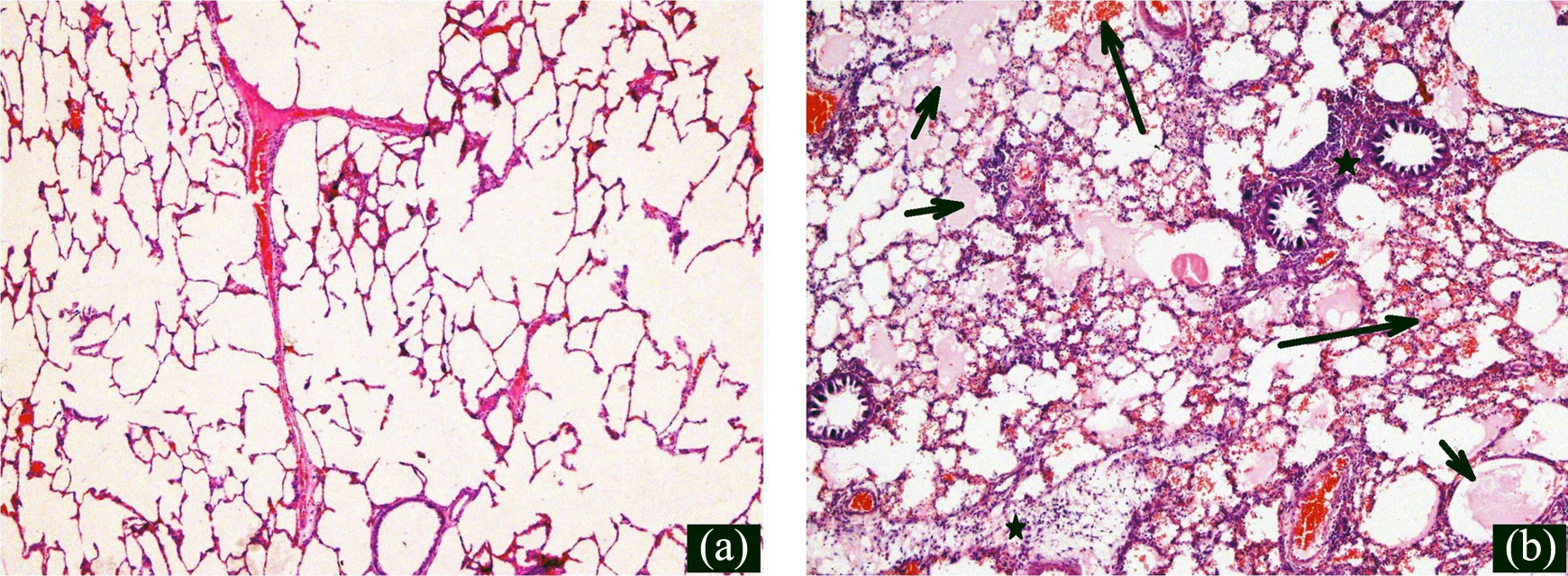
(a) In the sham group, all the structures of pulmonary parenchyma and bronchial tree were of normal appearance. (b) Histopathological evaluation of rat lung following PQ revealed that the lesions predominantly appeared in parenchymal region. There was inflammatory cell infiltration in interstitium and peribronchiolar areas (star). Extensive alveolar edema (short arrow) and massive alveolar hemorrhage (long arrow) were observed after PQ exposure (×100 magnification, hematoxylin and eosin stain). PQ: paraquat.
In the PQ group, serum neopterin levels showed a positive correlation with LDH activities (
Spearman’s ρ correlation coefficients belong to laboratory parameters in the PQ group.

PQ: paraquat; TGF-1β: transforming growth factor-1β; MDA: malondialdehyde; TAC: total antioxidant capacity; LDH: lactate dehydrogenase.
Discussion
The lung is considered as the primary target organ for PQ toxicity due to the accumulation of PQ. 1,2,21 PQ can cause severe lung injury and fibrosis characterized by edema, hemorrhage, interstitial inflammation, proliferation of fibroblasts and enhanced collagen synthesis in humans and experimental animals. 22,23 PQ-induced lung toxicity involves two factors, direct injury by oxygen free radicals and indirect injury by inflammatory cells and fibroblasts. 24
Various studies have reported that ROS have a pivotal role in PQ-induced lung toxicity. 3,25–27 PQ comes in direct contact with oxygen in the lungs. 21 PQ induces the production of ROS and then they interact with membrane lipids. 5 Finally, lipid peroxidation products like MDA occur. Peroxidation of lipids is a causative factor for cell membrane damage. 25 Thus, PQ administration may damage alveolar cells. The formation of highly toxic free radicals such as superoxide anion and hydroxyl radical leads to lipid peroxidation in the cell membrane after exposure to PQ. 21 In this study, we estimated cellular damage with LDH assay. LDH may be a credible determiner of cellular injury after PQ exposure. Serum LDH activities were significantly increased in the PQ group. Our results have shown that PQ significantly increased the lung MDA levels, the typical product of lipid peroxide. Our results compliant with the results of other studies. 2,22,27
PQ leads to some changes in antioxidant system and oxidative damage. Following exposure to PQ, serum TAC levels were decreased. In addition, lung MDA levels were increased. They are used to determine the degree of oxidative stress. Decreased TAC levels and increased MDA levels reflect imbalance between endogenous antioxidant system and oxidative stress. Park et al. have reported that PQ decreases TAC in lung tissues. 26 On the other hand, it has shown that the administration of antioxidants such as quercetin and pyrrolidine dithiocarbamate effectively inhibits lipid peroxidation in lung tissues after exposure to PQ. 26,28
ROS generated by PQ also increase neutrophil adhesion and infiltration. 3 Inflammatory cells and their products such as cytokines, chemokines, and growth factors play a crucial role in PQ-induced pulmonary injury and fibrosis. 29,30 Inflammation contributes to PQ-induced pulmonary damage. In addition, oxidative stress also stimulates fibrogenic responses by promoting production and secretion of inflammatory and profibrogenic factors from macrophages and other lung cells. 31
On the other hand, TGF-β which is a powerful fibrogenic growth factor also promotes pulmonary fibrosis. 32 It has been reported that the expression of TGF-β was significantly increased in the mouse lung after exposure to PQ. 32 The messenger RNA levels of TGF-β were significantly increased in PQ-induced lung toxicity. 32,33 Alveolar macrophages secrete TGF-β in the lungs after exposure to PQ. Activated macrophages also synthesize several cytokines such as tumor necrosis factor-α, interleukin (IL)-1α, IL-1β, IL-6, interferon-γ, and TGF-α that mediate an enhanced fibroproliferative response. 24
Neopterin, a guanosine derivative, is an inflammatory by-product. 13 Plasma or urine neopterin concentration is a sensitive and specific marker cellular immunity and reflects the activation of the macrophage/monocyte line cells. 11 In this study, serum neopterin levels were significantly high in the PQ group. In severe inflammation and sepsis, both serum neopterin levels and endogenous nitric oxide formation are elevated. 34 It has been reported that intraperitoneal injection of neopterin stimulates granulopoiesis during inflammatory processes. 35 PQ may activate alveolar macrophages in the lung. Then, activated alveolar macrophages may secrete neopterin. Elevated serum neopterin levels may trigger the development of pulmonary fibrosis via TGF-1β. It has been suggested that neopterin plays an important role in the modulation of ROS-mediated processes and activates the ROS-sensitive transcription factor NF-κB. 36 Schobersberger et al. have shown that neopterin also induces apoptosis in the rat alveolar epithelial cell line L2. 37 Elevated neopterin levels are correlated with the severity of disease and predict a poor prognosis in patients with inflammatory disorders such as sarcoidosis, lung cancer, tuberculosis, and sepsis. 36,38 The relationship between PQ toxicity and neopterin levels is an indirect one and as such could not be specific enough to determine PQ toxicity. But, neopterin is more likely helping to show extend or severity of pathology due to PQ toxicity just like the above-mentioned pathologies. To show the correlation between rising neopterin levels and severity of inflammation consequent of PQ exposure, further studies should be carried out.
Histological examinations showed that PQ administration induces inflammatory cell infiltration in interstitium and peribronchiolar areas. In addition, it leads to alveolar edema and alveolar hemorrhage. Eventually, it severely damages alveolar cells. In addition, increased TGF-1β, MDA, LDH, and neopterin levels and decreased TAC levels were accompanied by pathological changes in lung tissue.
In conclusion, the results in the present study clearly demonstrated that single dose PQ significantly increased MDA, TGF-1β, neopterin, and LDH levels and reduced TAC levels. These findings suggested that oxidative stress, reduction of antioxidant capacity, and inflammation play a crucial role in the PQ-induced lung injury. Neopterin is not specific for PQ poisoning. But, the current study is one of the first studies about neopterin in an animal model of the PQ poisoning. Although this elevation in serum neopterin levels may show the severity of tissue injury due to PQ intoxication, further studies may be performed to clarify the role of neopterin with different doses of PQ.
In addition, as our study was carried out for a short period of time, it was not adequate to evaluate fibrosis. Intoxication with PQ causes lung fibrosis in the chronic stages in rats. To evaluate lung fibrosis, at least 2 or 3 weeks study is recommended. Therefore, we did not evaluate the lung fibrosis in animals. For this purpose, chronic PQ groups also may be added to further studies.
Footnotes
Acknowledgments
The authors would like to express their sincere appreciation to FAVOR (FMF Arthritis Vasculitis and Orphan Diseases Research/![]() ) Web registries at Gulhane Military Medical Academy, Institute of Health Sciences, Ankara, Turkey, for their supports in epidemiological and statistical advisory and invaluable guidance for the preparation of the manuscript.
) Web registries at Gulhane Military Medical Academy, Institute of Health Sciences, Ankara, Turkey, for their supports in epidemiological and statistical advisory and invaluable guidance for the preparation of the manuscript.
Conflict of interest
The authors declared no conflicts of interest.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
