Abstract
Naringenin is a naturally occurring flavanones and has been found to exhibit free radical scavenging, enzyme inhibition, antioxidants, anti-inflammatory, and anticancer activities. Present study was designed to evaluate the protective role of naringenin against benzo[a]pyrene (B[a]P)-induced oxidative stress and pulmonary toxicity. Rats were treated with naringenin at a dose of 100 mg/kg body weight (b. wt.), by oral gavage. B[a]P in a single dose of 50 mg/kg b. wt. was given intraperitoneally. Total protein, total cell counts, lactate dehydrogenase, lipid peroxidation, reduced glutathione, antioxidant enzymes activities, lung histology and expression of nuclear factor kappa B (NF-κB), and cyclo-oxygenase-2 (COX-2) was assessed to evaluate protective effects of naringenin. Histopathological and immunohistochemical studies were also carried out to observe lung toxicity and inflammation. B[a]P administration enhanced the levels of lung injury markers and reduced antioxidant enzymes activities. Naringenin treatment attenuated the levels of oxidative stress by restoring antioxidant enzymes, further improved lung histological damage and significant decrease in inflammatory responses. Naringenin also effectively decreased the expression of NF-κB, and COX-2 induced by B[a]P. These findings suggest that naringenin supplementation is beneficial in maintaining the integrity of alveoli and the epithelium that may be used as a protective agent in B[a]P-induced oxidative stress and lung damage. However, further studies are warranted to elucidate the potential mechanism of action of naringenin.
Introduction
Benzo[a]pyrene (B[a]P), a polyaromatic hydrocarbon, is formed during the process of incomplete combustion of organic matter such as fossil fuel, garbage, and plant decay. 1 It is also present in tobacco smoke including cigarette smoke. 2,3 B[a]P exposure causes structural and physiological changes such as emphysema, inflammation, edema, and surfactants dysfunction in lungs that may lead to development of various chronic lung disorders. 2,4,5,6 Short-term exposure of B[a]P has been used to find out detrimental effects on lung and established as a model for toxicant to study adverse effects on pulmonary system. 2,6
Chemoprevention is an approach which is used to inhibit, suppress, or reverse the process of cancer development by using natural product or synthetic agents to combat oxidative stress and toxicity. 7,8 Phytochemicals and different pharmacologically active agents present in dietary intake of food; vegetables and medicinal plants play an important role in the management of oxidative stress and toxicity caused by chemical toxicants. 9 –12 Flavonoids are phenolic compounds widely present in fruits and vegetables, which is a characteristic of the human diet. They exhibit anti-inflammatory, antioxidant, antimutagenic, anticarcinogenic, and modulation of enzymatic activities. 13 –15 Chemical toxicants cause tissue damage via diverse mechanisms, many of which involve activation of cell survival and apoptosis pathways leading oxidative stress. 16 B[a]P is metabolized by cytochrome P450 1A1 (CYP1A1) into epoxide that attach at DNA to form adduct by producing reactive oxygen species (ROS), which mediate damage to cell structures, including lipids, membrane, and proteins by causing inflammation and oxidative stress. 17 –19
Naringenin (4′,5,7-trihydroxyflavanone) is a naturally occurring plant bioflavonoid found in citrus fruits, tomatoes, cocoa, and tangerines. 20 Naringenin has been investigated for its biological activities such as free radical scavenging, enzyme inhibiting, antioxidants, anti-inflammatory, immune system modulatory, and anticancer properties. 21 –23 It is a potent inhibitor of the B[a]P metabolizing enzyme benzo (a) pyrene aryl hydrocabon hydroxylase (AHH) in in vitro and in vivo studies. 24 Naringenin also affect different oxidative processes associated with chronic degenerative diseases by modulating cytochrome P450-dependent monooxygenase which involved in the metabolism of xenobiotics. 25 These studies demonstrate that naringenin has a promising potential as a preventive agent against oxidative stress and pulmonary toxicity, which is worthy of further investigations. As far as we know, there is no study concerning the effect of naringenin against B[a]P-induced oxidative stress and lung injury. Therefore, the present study was designed to examine the protective potential of naringenin against B[a]P-induced lung toxicity in Wistar rats.
Materials and methods
Chemicals and reagents
Benzo[a]pyrene, naringenin, reduced glutathione (GSH), oxidized glutathione, nicotinamide adenine dinucleotide phosphate reduced (NADPH), nicotinamide adenine dinucleotide phosphate (NADP+), flavin adenine dinucleotide (FAD), ethylenediaminetetraacetic acid (EDTA), thiobarbituric acid, pyrogallol, poly-
Experimental animals
Male Wistar rats (180–200 g), 8–10 weeks old, were used for the experimental study. Animals were obtained from the Central Animal House Facility of Jamia Hamdard (Hamdard University), New Delhi, India. All the animals used in this study were reviewed and approved by Institutional Animal Ethical Committee that is fully accredited by the Committee for Purpose of Control and Supervision on Experiments on Animals, India. Animals were housed in polypropylene cages in groups of six rats per cage. Animals were acclimatized in a room maintained at 25 ± 2°C with 12 h light/dark cycle. Animals were given free access to standard laboratory feed (Hindustan Lever Ltd., Bombay, India) and water ad libitum. Approval ID/project number for this study is 1060.
Treatment regimen
To study the prophylactic/therapeutic effects of naringenin against B[a]P-induced oxidative stress and inflammatory responses in the lungs, animals were randomly allocated into five groups, that is, (I–V), having six animals ( Group I: Served as control group which received distilled water (2 mL/kg body weight (b. wt.) orally) from day 1 to day 14 and also administered a single dose of corn oil (2 mL/kg b. wt., intraperitoneally) at 7th day only. Group II: Served as a toxicant group and B[a]P was administered (50 mg/kg b. wt. intraperitoneally dissolved in corn oil) a single dose at 7th day only. Groups III: Animals were pretreated with naringenin dissolved in distilled water at the dose of 100 mg/kg b. wt., by oral gavage from day 1 to day 7 and thereafter a single dose of B[a]P (50 mg/kg b. wt., intraperitonially) was administered. Group IV: Animals were administrated with B[a]P (50 mg/kg b. wt., intraperitonially) at 7th day, thereafter, treated with naringenin dissolved in distilled water at the dose of 100 mg/kg b. wt., by oral gavage from day 7 to day 14. Group V: Animals received naringenin dissolved in distilled water only at 100 mg/kg b. wt., dose by oral gavage daily up to 14 consecutive days.

Schematic representation of treatment regimen. Group I: received distilled water (2 mL/kg b. wt., orally) from day 1 to day 14 and a single dose of corn oil (2 mL/kg b. wt., intraperitoneally) at 7th day only; group II: B[a]P (50 mg/kg b. wt., intraperitoneally) in corn oil only once at 7th day. Group III: Naringenin (100 mg/kg b. wt., orally) daily up to 14 consecutive days and B[a]P (50 mg/kg b. wt., intraperitoneally) once at 7th day. Group IV: B[a]P (50 mg/kg b. wt., intraperitoneally) at 7th day and naringenin (100 mg/kg b. wt., orally) from 7th day to 14th day. Group V: received naringenin (100 mg/kg b. wt., orally) daily up to 14 consecutive days. b. wt.: body weight; B[a]P: benzo(a)pyrene.
All animals were sacrificed by cervical dislocation under mild anesthesia of ether 24 h after the last treatment and processed for subcellular fractionation; lungs were quickly removed and perfused in ice-cold saline. Lung tissues were taken at the same time for estimation of lung wet/ dry weight ratio, immunohistochemistry, biochemical, and histological parameters. Biochemical estimations were performed both in bronchoalveolar lavage (BAL) fluid as well as in lung tissues samples.
Tissue processing
After sacrificing the animals, lung tissues were processed for subcellular fractionation. A piece of lung tissue was preserved in 10% neutral buffered formalin for histological observation. A 10% homogenate was prepared in chilled phosphate buffer (0.1 M, pH 7.4) using Polytron® homogenizer (Kinematica, Inc., Switzerland). The homogenized tissue was centrifuged at 10,500
BAL fluid collection
Animals were anesthetized and then euthanized by exsanguinations. Trachea and lungs were exposed by thoracotomy; a cannula was inserted into the trachea and was ligated using a thread. Approximately 25 mL/kg b. wt. of warm (37°C) phosphate-buffered saline (PBS; 0.15 M NaCl, 50 mM phosphate, pH 7.4) was injected into the lungs via a syringe fitted with tracheal cannula to collect BAL fluid.
26,27
PBS was allowed to stay in lungs for 30 s, then retrieved and re-instilled with the help of a syringe. The process was repeated three times with the same solution. The BAL fluid recovered from different animals was centrifuged (300
Protein content and total cell counts
Protein content and total cell counts were estimated in BAL fluid as a measure of edema and inflammation. The concentration of protein in all samples was determined by using the method of Bradford, 1976, in which BSA was taken as standard. 28 Cell pellet obtained after centrifugation of BAL fluid was washed three times with PBS. The washing was performed immediately after the BAL fluid collection. This obtained cell pellet was resuspended in PBS and the cells were counted using a hemocytometer (Neubauer chamber).
Lung W/D weight ratio
Lung wet/dry (W/D) weight was calculated as an index of pulmonary edema, as it is a technique commonly used for assessment of experimental lung injuries. 29 Lung samples of all animals were obtained immediately from the right upper lobe, and the lung weight was measured immediately after its excision (wet weight). The lung tissues were then dried in an oven at 60°C for 72 h and reweighed as dry weight. The W/D weight was calculated by dividing the wet by the dry weight. 30
Assay for LDH activity
Lactate dehydrogenase (LDH) activity was estimated by the method of Kornberg. 31 The assay mixture consisted of sample (0.2 mL), (0.1 mL) 0.02 M NADH, (0.1 mL) 0.01 M sodium pyruvate, (1.1 mL) 0.1 M, pH 7.4 phosphate buffer, and distilled water in a total volume of 3 mL. Enzyme activity was recorded at 340 nM, and activity was calculated as nmol NADH oxidized/min/mg protein.
Assay for LPO level
The assay for microsomal lipid peroxidation (LPO) was done following the method of Wright et al..
32
The reaction mixture in a total volume of 1.0 mL contained 0.58 mL phosphate buffer (0.1 M, pH 7.4), 0.2 mL microsomes, 0.2 mL ascorbic acid (100 mM), and 0.02 mL ferric chloride (100 mM). The reaction mixture was incubated at 37°C in a shaking water bath for 1 h. The reaction was stopped by addition of 1.0 mL 10% TCA. Following addition of 1.0 mL 0.67% thiobarbituric acid, all the tubes were placed in boiling water bath for 20 min and then shifted to crushed ice bath before centrifuging at 2500
Assay for SOD activity
The superoxide dismutase (SOD) activity was measured by the method of Marklund and Marklund. 33 The reaction mixture consisted of 2.875 mL Tris–HCl buffer (50 mM, pH 8.5), 0.025 mL Pyrogallol (24 mM in 10 mM HCl), and 0.1 mL PMS, in a total volume of 3 mL. Enzyme activity was measured at 420 nM and was expressed as units per milligram protein. One unit of enzyme is defined as the enzyme activity that inhibits the auto-oxidation of pyrogallol by 50%.
Assay for catalase activity
Catalase activity was measured by the method of Claiborne. 34 The reaction mixture consisted of 2 mL phosphate buffer (0.1 M, pH 7.4), 0.95 mL H2O2 (0.019 M), and 0.05 mL PMS in a final volume of 3 mL. Changes in absorbance were recorded at 240 nM. Catalase activity was calculated as nanomole H2O2 consumed per minute per milligram protein.
Assay for XO activity
The activity of xanthine oxidase (XO) was assayed by the method of Athar et al.. 35 The reaction mixture consisted of 0.2 mL PMS which was incubated for 5 min at 37°C with 0.8 mL phosphate buffer (0.1 M, pH 7.4). The reaction was started by adding 0.1 mL xanthine (9 mM) and kept at 37°C for 20 min. The reaction was terminated by the addition of 0.5 mL ice-cold perchloric acid (10% v/v). After 10 min, 2.4 mL of distilled water was added and centrifuged at 4000 rpm for 10 min and micrograms of uric acid formed per minute per milligram protein was recorded at 290 nM.
Assay for GR activity
Glutathione reductase (GR) activity was estimated by the method of Carlberg and Mannervick. 36 The assay system containing 1.65 mL phosphate buffer (0.1 M, pH 7.6), 0.1 mL EDTA (0.5 mM), 0.05 mL GSSG (1.0 mM), 0.1 mL NADPH (0.1 mM), and 0.1 mL PMS (10%) in a total volume of 2.0 mL. The enzyme activity was recorded at 25°C by measuring the disappearance of NADPH at 340 nM and was calculated as nmol NADPH oxidized/min/mg protein using molar extinction coefficient of 6.22 × 103/M/cm.
Assay for GSH level
Reduced glutathione (GSH) was determined by the method of Jollow et al..
37
A 1.0 mL sample of PMS was precipitated with 1.0 mL of sulfosalicylic acid (4%). The samples were kept at 4°C for 1 h and then centrifuged at 1200
Assay for GST activity
Glutathione-S-transferase (GST) activity was assayed by the method of Habig et al.. 38 The reaction mixture consisted of 1.475 mL phosphate buffer (0.1 M, pH 6.5), 0.2 mL reduced glutathione (1 mM), 0.025 mL CDNB (1 mM), and 0.3 mL PMS (10% w/v) in a total volume of 2.0 mL. The changes in the absorbance were recorded at 340 nM and enzymes activity was calculated as micromoles of 1-chloro-2,4-dinitrobenzene (CDNB) conjugate formed per minute per milligram protein using a molar extinction coefficient of 9.6 × 103/M/cm.
Assay for GPx activity
Glutathione peroxidase (GPx) activity was assayed by the method of Mohandas et al. 39 The reaction mixture consists of 1.44 mL phosphate buffer (0.1 M, pH 7.4), 0.1 mL EDTA (1 mM), 0.1 mL sodium azide (1.0 mM), 0.05 mL GR (1 IU/mL), 0.05 mL GSH (1.0 mM), 0.1 mL NADPH (0.2 mM), and 0.01 mL H2O2 (0.25 mM), and 0.1 mL PMS (10%) in a total volume of 2.0 mL. Disappearance of NADPH at 340 nM was recorded. The enzyme activity was calculated as nmol NADPH oxidized/min/mg protein using molar extinction coefficient of 6.22 × 103/M/cm.
Histopathological evaluation
The lung tissues were fixed in 10% neutral buffered formalin and then embedded in paraffin wax. Sections (5μM) were cut from the middle lobes of lungs of each group more or less from similar positions. The paraffin-embedded lung tissue sections were deparaffinized using xylene and ethanol. The slides were washed with PBS and permeabilized with permeabilization solution (0.1 M citrate, 0.1% Triton X-100). The deparaffinized sections were stained with hematoxylin and eosin. Lung sections were evaluated at 40× magnification. All microscopic sections were interpreted in a blind fashion with the help of histopathologist. A semi-statistical evaluation of the histological finding in lung was done. Lung slides were evaluated for interstitial edema, bronchopneumonia, inflammatory cells, fibrosis, and acute lung injuries. We used different symbols to design the frequency (the abundance) of the architectural changes in the lungs (Table 1).
Effect of naringenin treatment and B[a]P on semi-score analysis of morphopathological changes in lung tissues.a

−: absence of the changes in the animals of the studied groups; +: change which was rare within a group; ++: change not so often observed in all animals; +++: change which was found in almost all studied animal groups; ++++: change which was often found in all studied animal groups; b. wt.: body weight; B[a]P: benzo(a)pyrene.
aValues are expressed as mean ± SD (
Immunohistochemical detection of COX-2 and NF-κB
Immunohistochemical staining was assessed to examine the protective effects of naringenin on B[a]P-induced inflammation and proliferation in lung tissues. Tissue samples were fixed in formalin and paraffinized sections of 5 μM thickness were cut onto poly (
Statistical analysis
The data from individual groups were presented as the mean ± SD. Differences between groups were analyzed using one-way analysis of variance followed by Tukey–Kramer multiple comparisons test and minimum criterion for statistical significance was set at
Results
Edema and inflammation
B[a]P-treated animals (group II) led to discernible edema in terms of protein content (a
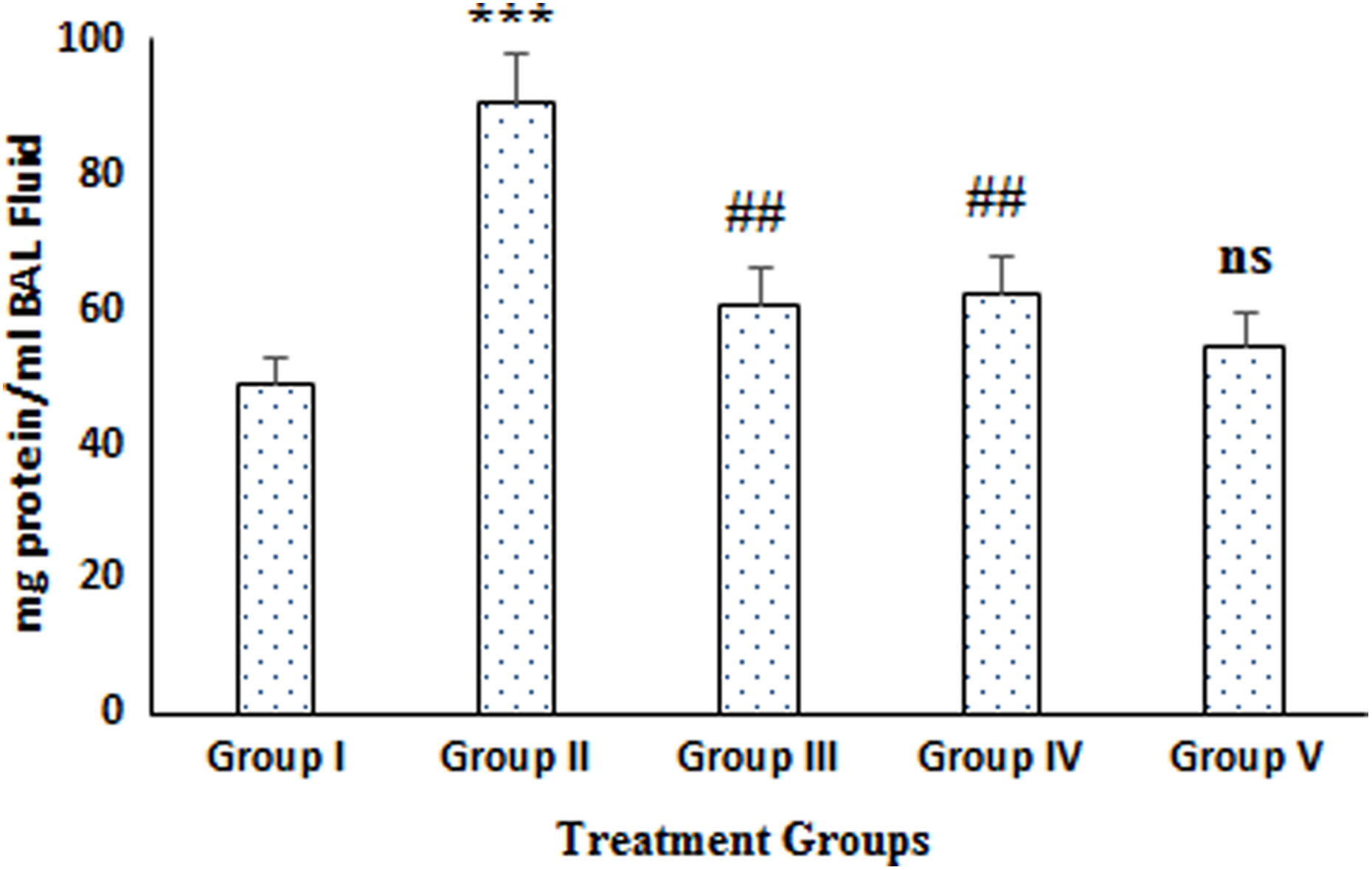
Effect of naringenin treatment and B[a]P on protein concentration in BAL fluid. The results are expressed as mean ± SD of six animals in each group. ***

Effect of naringenin treatment and B[a]P on total cell counts in BAL fluid. The results are expressed as mean ± SD of six animals in each group. ***
Lung wet weight/dry weight ratio
Subsequent to induction of lung injuries with B[a]P, lung W/D weight ratio was much more pronounced (a

Effect of naringenin treatment and B[a]P on lung wet/dry weight ratio. The results are expressed as mean ± SD of six animals in each group. ***
Lung cellular injury markers in BAL fluid and lung tissues
The activity of LDH and LPO level in BAL fluid and lung tissue homogenate in B[a]P-exposed animals (group II) were found to be significantly higher (a

Effect of naringenin treatment and B[a]P on the activity of LDH and LPO level in BAL fluid and lung tissue. The results are expressed as mean ± SD of six animals in each group. ***
Effect on glutathione-depended enzymes level
The levels of GSH and glutathione-dependent enzymes such as GPx, GST, and GR were significantly (a
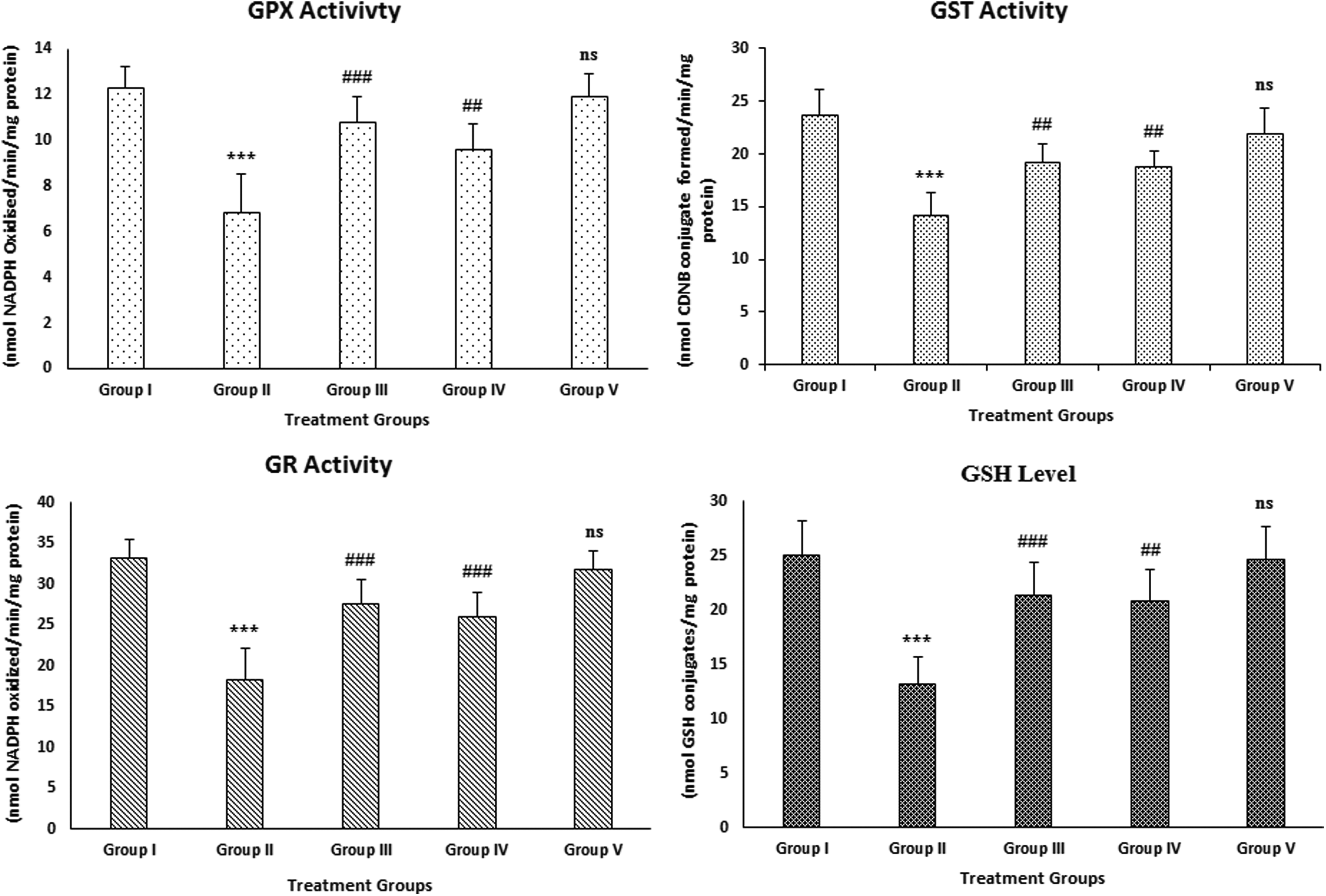
Effect of naringenin treatment and B[a]P on the levels of antioxidant enzymes (GPx, GST, and GR) and GSH in BAL fluid. The results are expressed as mean ± SD of six animals in each group. ***
Effect of naringenin treatment and B[a]P on the levels of activities of GPx, GST, GR, and GSH in lung tissues.
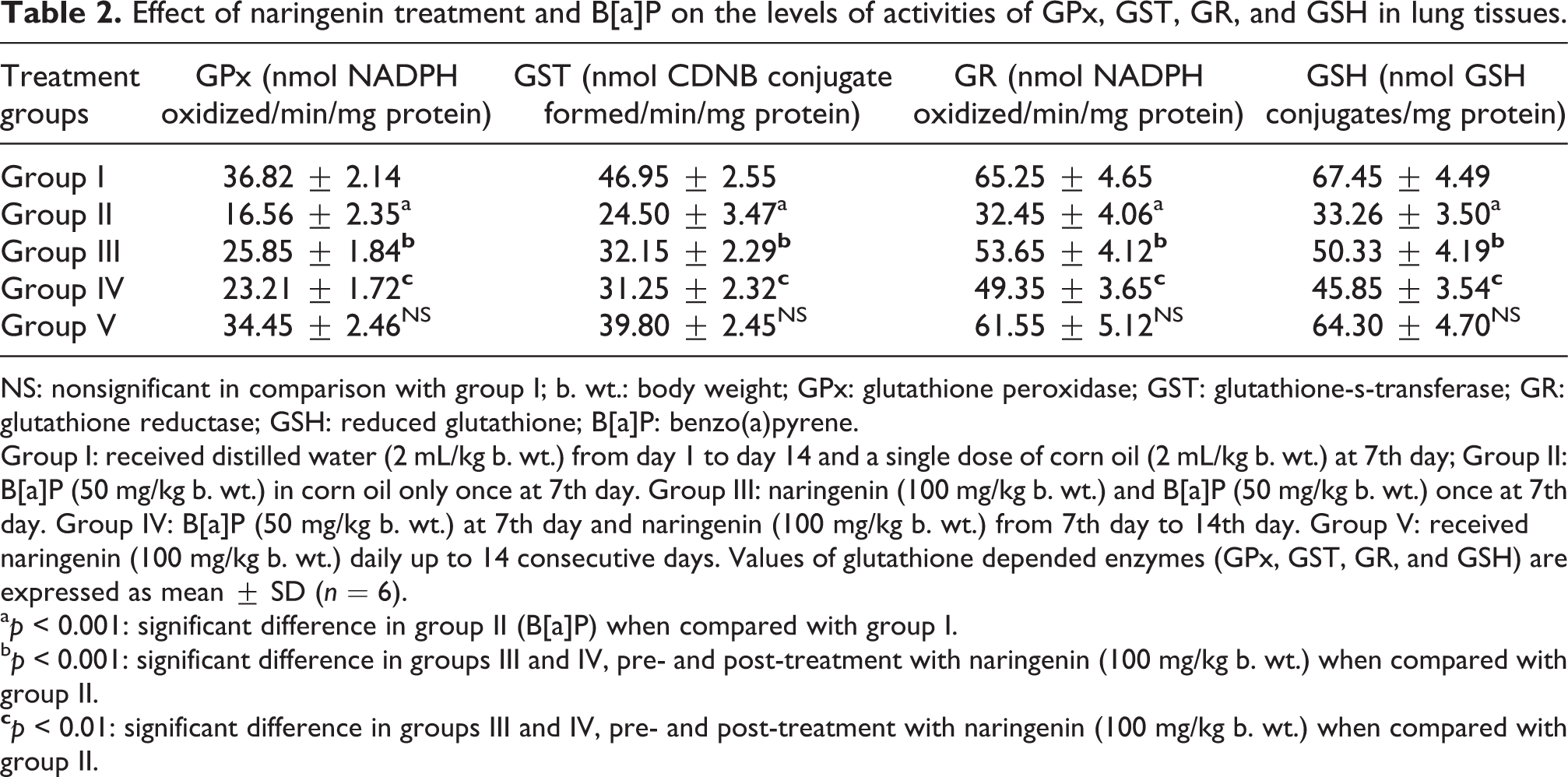
NS: nonsignificant in comparison with group I; b. wt.: body weight; GPx: glutathione peroxidase; GST: glutathione-s-transferase; GR: glutathione reductase; GSH: reduced glutathione; B[a]P: benzo(a)pyrene.
Group I: received distilled water (2 mL/kg b. wt.) from day 1 to day 14 and a single dose of corn oil (2 mL/kg b. wt.) at 7th day; Group II: B[a]P (50 mg/kg b. wt.) in corn oil only once at 7th day. Group III: naringenin (100 mg/kg b. wt.) and B[a]P (50 mg/kg b. wt.) once at 7th day. Group IV: B[a]P (50 mg/kg b. wt.) at 7th day and naringenin (100 mg/kg b. wt.) from 7th day to 14th day. Group V: received naringenin (100 mg/kg b. wt.) daily up to 14 consecutive days. Values of glutathione depended enzymes (GPx, GST, GR, and GSH) are expressed as mean ± SD (
a
b
Effect on the activities of antioxidant enzymes
The effect of naringenin treatment on B[a]P-induced depletion in the activities of antioxidant enzymes, that is, catalase and SOD, was evaluated and results were obtained. We have observed that there was a significant (a
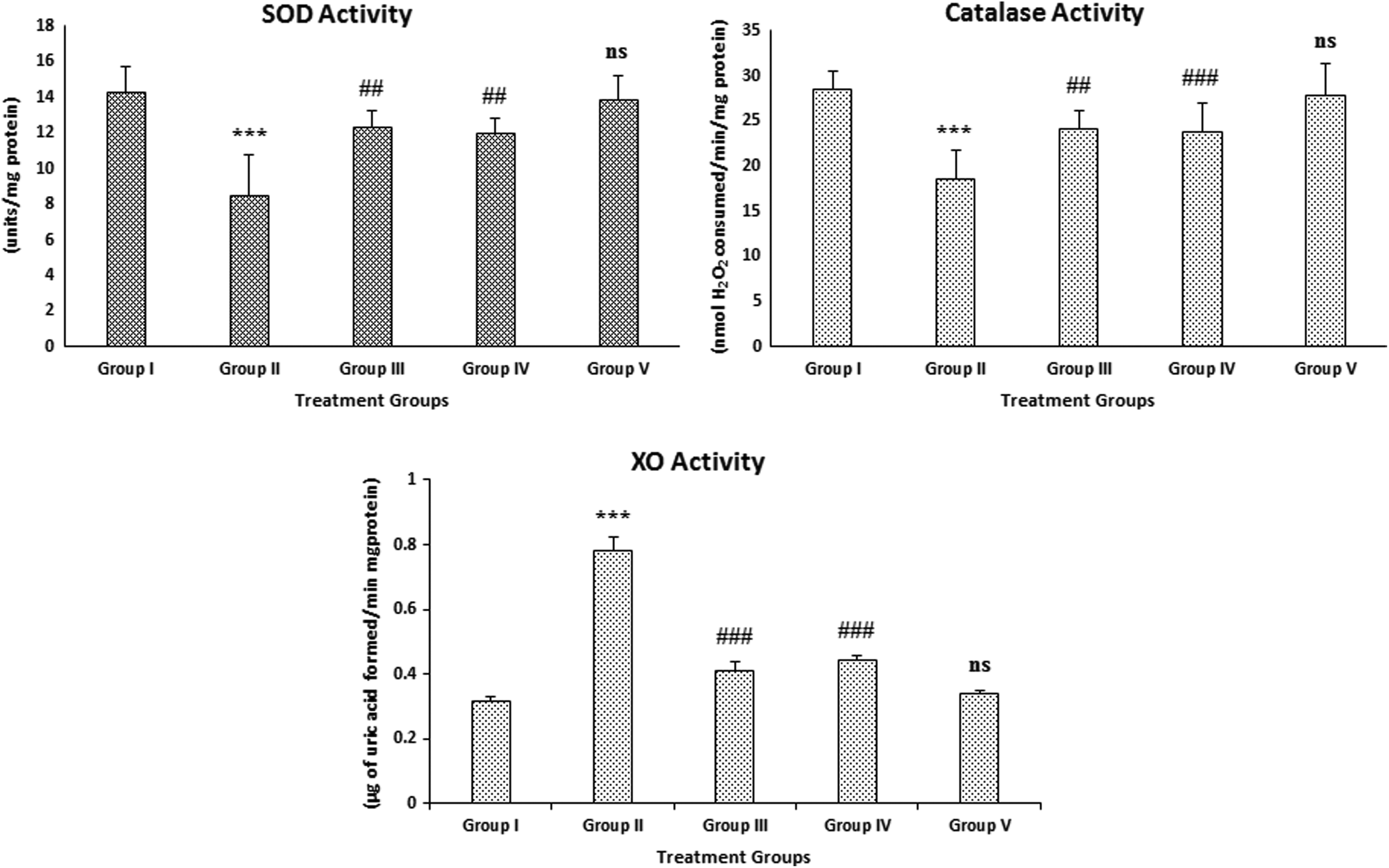
Effect of naringenin treatment and B[a]P on SOD, catalase, and XO activities in BAL fluid. The results are expressed as mean ± SD of six animals in each group. ***
Effect of naringenin treatment and B[a]P on the levels of activities of catalase, SOD, and XO in lung tissues.

NS: nonsignificant when compared with group I; b. wt.: body weight; SOD: superoxide dismutase; XO: xanthine oxidase; H2O2: hydrogen peroxide; B[a]P: benzo(a)pyrene.
Group I: Received distilled water (2 mL/kg b. wt., orally) from day 1 to day 14 and a single dose of corn oil (2 mL/kg b. wt.) at 7th day; Group II: B[a]P (50 mg/kg b. wt.) in corn oil only once at 7th day. Group III: Naringenin (100 mg/kg b. wt.) and B[a]P (50 mg/kg b. wt.) once at 7th day. Group IV: B[a]P (50 mg/kg b. wt.) at 7th day and naringenin (100 mg/kg b. wt.) from 7th day to 14th day. Group V: Received naringenin (100 mg/kg b. wt.) daily up to 14 consecutive days. Values of catalase, SOD, and XO activities are expressed as mean ± SD (
a
b
c
Effect on XO activity in lung tissues
The activity of XO was significantly (a
Histological analysis of the lungs
Analysis of lung tissue sections of B[a]P-treated group (group II) revealed marked changes (40× magnifications) when compared with group I. B[a]P administration caused severe destruction of alveolar architecture and congested and thickened alveolar epithelium. Cytoplasmic granulation with large hyperchromatic nuclei and vascular degeneration were found in lung tissues of group II animals. Microscopic examination also showed severe pulmonary edema, acute congestion, and interstitial pneumonia with inflammatory cells infiltration. Pre- and post-treatment with naringenin (groups III and IV) showed protection of the lung architecture against B[a]P-induced alterations in group II. There were no significant changes in only naringenin-treated animals (group V) as compared to group I (Figure 8). The qualitative analysis of different architectural changes in lung tissues of different treatment groups of animals has been summarized in Table 1. These findings were further correlated with the cytotoxicity marker (LDH) levels in BAL fluid and lung tissues which support the results of the other parameters of the present study.
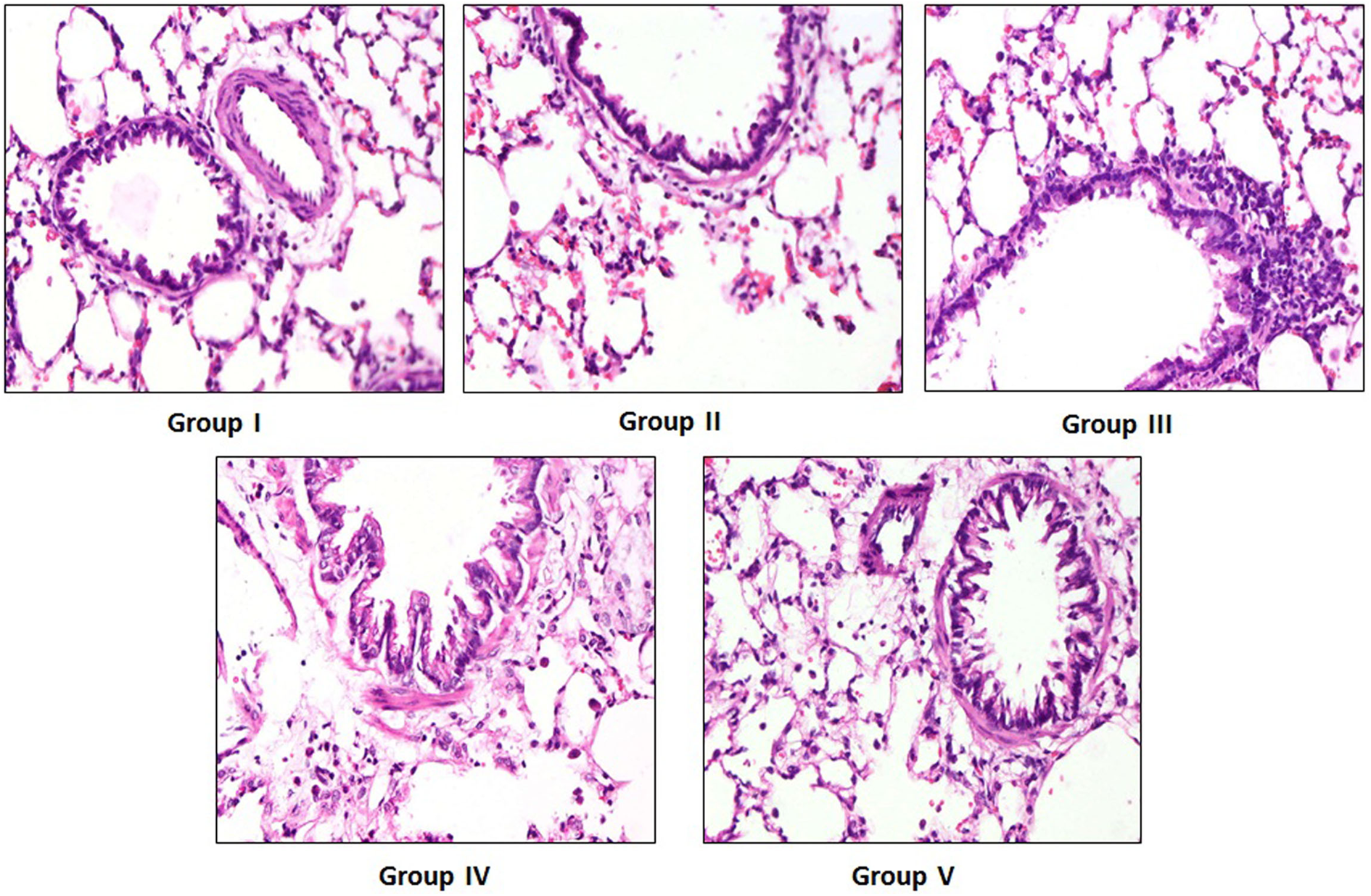
Effect of naringenin treatment and B[a]P on histoarchitecture of lung tissue. Photomicrographs of lung sections depicting vehicle-treated control group (group I), B[a]P-treated group (group II; 50 mg/kg b. wt.), pre- and post-treatment with naringenin (groups III and IV; 100 mg/kg b. wt.), and treated with naringenin only (group V; 100 mg/kg b. wt.). The hematoxylin and eosin-stained sections exhibited normal histoarchitecture with mild inflammatory cells infiltration in group I, while group II showed distorted alveolar architecture and thickened alveolar epithelium with intense inflammatory cells infiltration. Pre- and post-treatment with naringenin (100 mg/kg b. wt.) significantly attenuated the B[a]P-induced histopathological changes in groups III and IV as compared to group II. There was no significant difference between the histology of group V in comparison to group I. Original magnification: 40×, scale bars, 50 µM. b. wt.: body weight; B[a]P: benzo(a)pyrene.
Effect of naringenin on the expression of COX-2 and NF-κB in lungs
The immunohistochemical evaluation of COX-2 and NF-κB revealed that there were more number of immunopositive cells showing higher expression of these proteins as indicated by brown color in B[a]P-treated animals (group II) when compared with control animals (group I). Naringenin pre- and post-treated animal groups (III and IV) showed reduced number of immunopositive cells having the less expression of these proteins as compared to group II. Though, there were no significant differences observed in the immunostaining of these proteins in group V as compared to group I (Figures 9 and 10).

Effect of naringenin treatment and B[a]P-induced COX-2 expression. Photomicrographs of lung sections depicting immunohistochemical staining of COX-2 vehicle-treated control group (group I), B[a]P-treated group (group II; 50 mg/kg b. wt.), pre- and post-treatment with naringenin (groups III and IV; 100 mg/kg b. wt.), and treated with naringenin only (group V; 100 mg/kg b. wt.). For immunohistochemical analyses, brown color indicated specific immunostaining of COX-2 and light-blue color indicated nuclear hematoxylin staining. The lung section of B[a]P-treated group (group II) had more COX-2 immunopositive staining, as indicated by brown color, as compared to the control (group I), while pre- and post-treatment of naringenin in groups III and IV reduced COX-2 immunostaining as compared to group II. However, there was no significant difference in COX-2 immunostaining in group V as compared to group I. Original magnification: 40×, scale bars, 50 µM. b. wt.: body weight; B[a]P: benzo(a)pyrene; COX-2: cyclo-oxygenase-2.
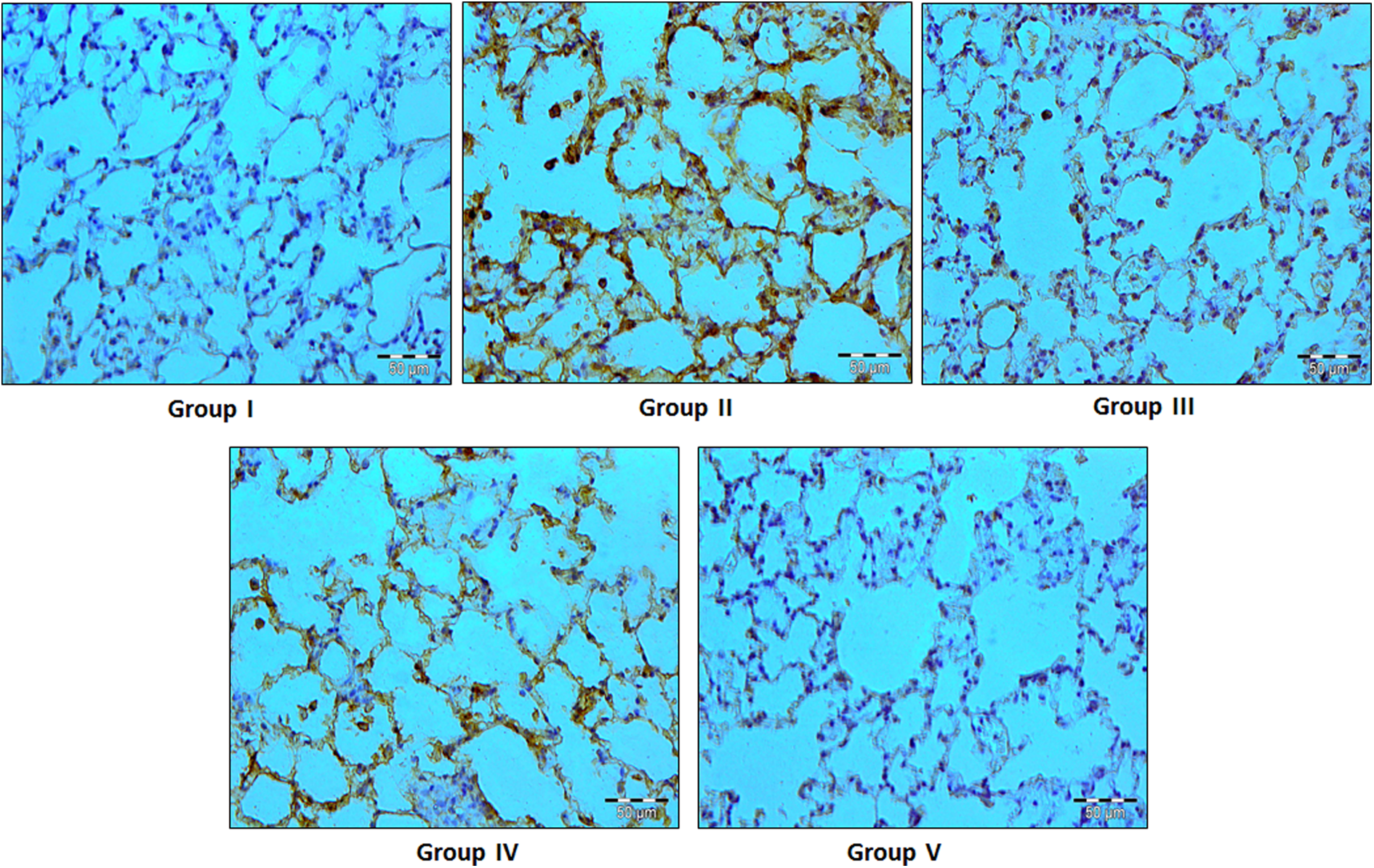
Effect of naringenin treatment and B[a]P-induced NF-κB expression. Photomicrographs of lung sections depicting immunohistochemical staining of NF-κB (group I) vehicle-treated control group, (group II) B[a]P-treated group (50 mg/kg b. wt.), (groups III and IV) pre- and post-treatment with naringenin (100 mg/kg b. wt.), and (group V) treated with naringenin only (100 mg/kg b. wt.). For immunohistochemical analyses, brown color indicated specific immunostaining of NF-κB and light-blue color indicated nuclear hematoxylin staining. The lung section of B[a]P-treated group (group II) showed more NF-κB immunopositive staining which is indicated by brown color in comparison with group I, while pre- and post-treatment of naringenin in groups III and IV reduced NF-κB immunostaining as compared to group II. However, there was no significant difference in NF-κB immunostaining in group V when compared with group I. Original magnification: 40×, scale bars, 50 µM. b. wt.: body weight; NF-κB: nuclear factor kappa B; B[a]P: benzo(a)pyrene.
Discussion
Lung is the most susceptible organ to the injuries of exposure of industrial and environmental toxicants in terms of high vasculature, large surface area, and its direct interaction with the atmosphere. 40 In the present study, we have observed the protective/therapeutic potential of naringenin against B[a]P-induced oxidative stress and lung toxicity in Wistar rats. Benzo[a]pyrene B[a]P, a well-known environmental contaminant found in diet, tobacco smoke, automobile exhaust, forest fire, and other combustion processes, is one of the most commonly studied carcinogen. 41,42 B[a]P-induced lung toxicity is well documented. However, the exact mechanism of action is still unclear. It may be due to ROS generation that leads to the condition of oxidative stress. 2,6,17,42
Natural products and their derivative phytocompounds are being increasingly recognized as useful complementary treatments for various human diseases which is evident from their antifungal, anti-inflammatory, antioxidant, and anti-cancerous activities. 43,44 These are regarded as one of the most attractive strategies to efficiently inhibit toxicity of different chemicals in various organs. Naringenin is an active flavanone, which is used as traditional medicine in China. 45 It has been reported to have beneficial bioactivities, including anti-oxidative, anti-inflammatory, and anti-tumorigenic activities. 46,47 This study was carried out to elucidate the effect of naringenin on B[a]P-induced lung toxicity in rats and to assess its role in the modulation of the inflammatory pathway.
Analysis of BAL fluid components is very important in identifying specific disease markers such as inflammation, acute injury, asthma, infections, and fibrosis. 48,49 Exposure to chemical toxicants increases permeability of the alveolar capillary barrier which could be measured in terms of the total protein content of the BAL fluid. 50 Animals exposed to B[a]P showed increased protein content and a massive infiltration of inflammatory cells which reflected increase in total cell count in BAL fluid indicating its role in lung injuries. Naringenin was effective in minimizing total protein and total cell counts in BAL fluid which is supported by previous work where it has been reported to substantially subside activated inflammatory cells in BAL fluid as well as in lung tissues. 23 Lung W/D weight ratio which reflects the lung water content and edema was also increased in B[a]P-treated animals indicating pulmonary edema and inflammation. However, naringenin treatment improved this ratio significantly toward the normal. Similar action of naringenin is reported against lipopolysaccharide-induced acute lung injuries in rats. 51
The plasma membrane is normally impermeable to LDH, a cytosolic enzyme present within cells. However, damage to the cell membrane results in subsequent leakage of LDH into the extracellular fluid. LDH and LPO are crucial markers of oxidative stress and play important role in cellular damage which in turn may ultimately lead to disease development including cancer. 52 Inflammatory cells in the lung tissue may also contribute to the increase in LDH activity which reflects acute lung injury. Therefore, LDH release along with levels of MDA was studied as a marker of cell injury. Our result showed that there was remarkable elevation in LDH activity and MDA level after B[a]P treatment which was depleted with naringenin treatment suggesting its protective effect on lung epithelium cells that is also in agreement with previous findings. 53
ROS and endogenous antioxidant species remain in equilibrium in intracellular environment, when this equilibrium disturbs it leads to oxidative stress. The generation of ROS in lungs is enhanced after exposure to numerous exogenous chemical and physical agents. 54,55 Antioxidant enzymes defend against exogenous toxic injury by augmenting the defense against ROS via scavenging of free radicals. 56 In the current study, we have found that the administration of B[a]P caused oxidative stress because of substantial production of ROS. The activities of different antioxidant enzymes, namely, catalase, SOD, and glutathione-dependent enzymes (GPx, GR, and GST) and GSH level were significantly depleted in B[a]P-treated animals as compared to control which supports the involvement of oxidative stress in pathophysiology of B[a]P-induced oxidative stress and pulmonary toxicity. However, naringenin administration enhanced activities of these antioxidant enzymes by scavenging ROS in the form of superoxide radicals, peroxy radicals, peroxides, and singlet oxygen. The data of previous studies has shown that B[a]P administration enhances the activity of XO, which reduces O2 to superoxide anion radical (O2•−) and consequently produce oxidative stress. 53 In our study, naringenin also depleted XO activity showing its protective effect.
The development of lung inflammation is coordinated by the activation, expression, and secretion of numerous pro-inflammatory mediators from the lung parenchyma and vascular cells including cytokines, leukotriene, and adhesion molecules. 57 Blockade of inflammation mediating intracellular signaling pathway is now considered as a road map for the development of molecular target-based chemopreventive agents. 58,59 The modulation of COX-2 mediating signaling pathway has been considered a new approach for preventing oxidative stress, inflammation, and toxicity. 60 In this study we have analyzed the COX-2 and NF-κB expression which is an important markers of inflammation. 61 Activated NF-κB transactivate COX-2 expression, hence its inhibition is a valuable marker for regulation of various inflammatory diseases. Naringenin has been demonstrated as a potent inhibitor of nuclear factor kappa B (NF-κB) to interfere with the generation of pro-inflammatory cytokines. 62,63 Our findings showed that naringenin suppresses the expressions of COX-2 through downregulation of NF-κB in B[a]P-induced enhanced expression. Immunohistochemical analysis revealed the high expression of COX-2 in lung tissues from the animal of group II, that is, B[a]P-treated animals when compared to control. Naringenin-treated groups remarkably decreased the expression of COX-2 and NF-κB which evident that it suppressed inflammation that initiated lung injuries. Histological findings further substantiated the protective effects of naringenin against B[a]P-induced damage of alveolar architecture and thickened alveolar epithelium with intense inflammatory cell infiltration in the lung tissues. Naringenin treatment effectively reverted histological changes demonstrating a protective role against B[a]P-induced oxidative stress and lung injury.
Conclusion
The results of the present study demonstrate that oxidative stress, lung injury markers, antioxidant enzymes, and downregulation of the expression of inflammatory markers are closely concomitant with B[a]P-induced toxicity. Naringenin shows the protective efficacy against B[a]P-induced oxidative stress and lung toxicity possibly via induction of antioxidant enzymes, subsidized inflammatory responses, and suppression of ROS-mediated damage in lung tissues. However, further examinations emphasizing on molecular mechanism involved in modulatory action of naringenin on lung toxicity are required to be carried out.
Footnotes
Acknowledgements
The author (Dr Sarwat Sultana) is thankful to Indian Council of Medical Research (ICMR), New Delhi, India, for providing fellowship to one of the authors (Dr Rashid Ali) to carry out this work.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
