Abstract
Paraquat (PQ) is a nonselective bipyridyl herbicide widely used in agriculture to control weeds, but its accidental, occupational, or intentional exposure in humans is known to cause pneumo- and neurotoxicity which may proves fatal. Oxidative stress is reported as an underlined mechanism of PQ-induced toxicity in alveolar cells, neurons, and astroglia. PQ generates superoxides both through electron transport reaction (ETC) with nicotinamide adenine dinucleotide–dependent oxidoreductase and by the redox cycling via reaction with molecular oxygen. In lungs, it causes edema and inflammation resulting in neutrophils infiltration and subsequent activation of pro-inflammatory cytokines. In the present study, toxicity of subacute oral PQ exposure and effect of resveratrol (Res) and/or tetracycline (TC) on oxidative stress and inflammatory markers in lungs, brain, and liver was studied. Levels of glutathione and malondialdehyde and activities of myeloperoxidase, glutathione peroxidase, and catalase were measured in lungs, brain, and liver. PQ interferes in the function of mitochondrial ETC complexes causing decreased adenosine triphosphate levels, and hence the activities of complexes I and IV were studied in brain tissues. Res, a natural antioxidant, and TC, an antibiotic with its antimicrobial and anti-inflammatory properties, offered significant protection from severe oxidative stress and inflammation and ameliorated the general well-being of mice against the toxic outcome of PQ.
Introduction
Paraquat (PQ; 1,1′-dimethyl-4,4′-bipyridinium dichloride) is a nonselective herbicide used worldwide to get rid of unwanted weeds, but its accidental, occupational, or deliberate exposure is a cause of deep concern due to its high mortality rate (>50%). 1 Severe PQ exposure causes “acute lung injury” due to active polyamine uptake transport systems that concentrate PQ rapidly into type II epithelial cells of alveoli resulting in edema, bronchoalveolar destruction that results ultimately in pulmonary fibrosis. 2 PQ mainly accumulates in the lung (pulmonary concentrations can be 6 to 10 times higher than those in the plasma) and retained even when the blood concentration drops down. 3 Pulmonary fibrosis that develops through the pathological overproduction of extracellular matrix proteins such as collagens, 4 causing respiratory failure, is the most common cause of death from PQ poisoning. Although mechanism of PQ-induced cytotoxicity is not entirely clear, it is known that PQ undergoes a redox cycling reaction (superoxide anion radical), oxidizing nicotinamide adenine dinucleotide phosphate (NADPH) to NADP+, culminating into the production of reactive oxygen species (ROS). 5 NADPH–cytochrome P-450 reductase, 6 NADPH–cytochrome c reductase, 7 and the mitochondrial complex I, also known as nicotinamide adenine dinucleotide (NADH):ubiquinone oxidoreductase, 8 are mainly responsible for the reduction of PQ to form a PQ monocation-free radical (PQ·+). Generation of the superoxide anion radical can lead to the formation of more toxic forms of reduced oxygen, hydrogen peroxide (H2O2), and hydroxyl radicals, culminating in lipid peroxidation, 9 cellular damage, 10 and consequent inflammatory reaction, 11 which proves detrimental to the tissues. Depletion of NADPH, on the other hand, may disrupt important NADPH-requiring biochemical processes such as fatty acid synthesis.
Although pulmonary toxicity is the main area of focus, many studies also indicate PQ toxicity in the central nervous system (CNS). Unlike the exposure to high levels of PQ that mainly produces pulmonary toxicity, prolonged chronic low-level exposure to nonpneumotoxic doses results in the neurotoxicity. The uptake of PQ into the CNS, which is mediated through neutral amino acid transporters at blood–brain barrier, may produce damage to the basal ganglia. Toxic damage to the brain has also been observed in patients who died from PQ poisoning. 12 Autopsy findings in cases of acute PQ poisoning showed cerebral damage with edema, hemorrhage, and neural death. Once in neurons PQ stimulates glutamate efflux initiating excitotoxicity mediated by reactive nitrogen species, particularly peroxynitrite anion (ONOO−), which is also responsible for neurotoxicity. PQ-induced oxidative stress affects both neurons and astroglia alike but it selectively targets dopaminergic neurons in the substantia nigra. There is increasing evidence that PQ-induced neuroinflammation may contribute to dopaminergic pathogenesis.
Resveratrol (Res), a phytoalexin and a natural polyphenol produced by many plants, exhibits various beneficial properties such as antioxidant, antitumor, anti-inflammatory, antiaging, antidiabetic, and ameliorative effects on cardiovascular diseases. 13,14 Res alleviated bleomycin-induced lung injury in rats that otherwise would lead to pulmonary fibrosis. 15 The mechanism of protection by Res against bleomycin lung toxicity is largely unclear, but Kode et al. 16 and Ungvari et al. 17 have explored its effects on the vascular endothelium and lung epithelium, which showed activation of the nuclear factor (erythroid-derived 2)-related factor 2 (Nrf2) pathway. Sirtuin 1 (SIRT1) acts via the deacetylation of a number of crucial transcription factors and has been found associated with various bioprocesses. A decrease in the expression of SIRT1 and Nrf2 has been reported with long-term exposure to PQ. 18 Res is a SIRT1 activator and strongly enhanced SIRT1 expression and attenuated the lung injury due to PQ exposure. Additionally, Res was also reported to upregulate the expression of Nrf2 and glutathione (GSH). It also increases the activity of heme oxygenase 1, superoxide dismutase, and catalase (CAT), and additionally as an antioxidant has shown to deplete the malondialdehyde (MDA) production. 18 This makes the Res a therapeutically important compound against PQ toxicity.
Tetracyclines (TCs)are broad-spectrum antibiotics and exert antimicrobial effects by inhibiting protein synthesis. TC has multiple mechanisms of action including anti-inflammatory, matrix metalloproteinases (enzymes that inhibit collagen and gelatin production) inhibition, and antiapoptotic properties that make it an attractive neuroprotective agent. It has been shown to exhibit several intracellular and extracellular biologic effects that are not related to antibiotic activity. These activities correlate with anti-inflammatory and anticollagenolytic properties, making it an attractive agent to deal with PQ-induced pneumo- and neurotoxicity. 19 Hence, in the present study, we have explored the protective properties of Res and TC either alone or in combination against the PQ-induced lungs and brain toxicity in mice.
Materials and methods
Chemicals
PQ, TC, Res, coenzyme Q1, decylubiquinone, 2,6-dichloroindophenol (DCIP), rotenone, antimycin-A, phosphate buffer saline (PBS), glutathione reductase, NADH, NADPH, and all buffers were purchased from Sigma-Aldrich, Co., St. Louis, Missouri, USA. All other chemicals of extra pure grade were purchased from Merck, Mumbai, India.
Animals
Randomly bred Swiss male mice (35–40 g) from Defence Research and Development Establishment (DRDE) animal house facility were used for the study. The animals were kept in polypropylene cages with sterilized and dry paddy husk as a bedding material. Free access to food (standard pellet diet, Ashirwad Ltd., India) and water were allowed until 2 h before the experiment. The care and maintenance of animals were as per the approved guidelines of the “Committee for the Purpose of Control and Supervision of Experiments on Animals,” India. Food and water were allowed 2 h after the exposure. All animal experimentation procedures were approved by the institutional animal ethical committee.
Treatment
Forty male mice were divided into five groups (
Motor coordination
Behavioral studies included measuring motor coordination in mice. A rotarod (Orchid Scientific, India) with a 3 cm diameter rod was used. The rod was elevated 20 cm above the floor. The animal was made to balance on the constant acceleration of 20 r/min. When the mice fell from the rod, a sensor was activated and the corresponding time in seconds was recorded. For each experimental time, each mouse was subjected to three rotarod trials and the mean value was calculated. Then, the mean value for control mice was calculated and reduction in rotarod performance observed in PQ-treated animals was expressed comparatively to the mean control value.
Evaluation of oxidative stress
Measurement of GSH
Reduced GSH was determined using a fluorimetric method as described by Hissin and Hilf. 20 Briefly, to the 0.5 mL of tissue homogenate (10%, prepared in phosphate ethylenediaminetetraacetic acid (EDTA) buffer), 0.45 mL of phosphate EDTA buffer and 0.5 mL of metaphosphoric acid (25%) were added. Then, centrifuged at 10,000 r/min for 10 min at 4°C. Then, 2.25 mL of phosphate EDTA buffer was added to 0.25 mL of supernatant. Fifty microliters of this was separated and 900 µL of phosphate EDTA buffer and 50 µL of phthalaldehyde were added to this. Mixed well and incubated at room temperature for 15 min. Fluorescence intensity was measured using spectrofluorophotometer (Ex. 350 nm and Em. 420 nm) and values were expressed as millimolars of GSH per gram of tissue.
Measurement of MDA
MDA levels in the tissue were assayed following the method of Ohkawa et al. 21 to observe the amount of lipid peroxidation. Briefly, 10% tissue homogenate (in 1.15% KCl) was prepared and centrifuged for 10 min at 6000 r/min. To 0.1 mL of the above supernatant, 0.2 mL of 8.1% sodium dodecyl sulfate was added, followed by addition of 1.5 mL of 20% acetic acid (pH 3.5) and 1.5 mL of 0.8% thiobarbituric acid. This was followed by boiling at 95°C for 1 h and cooling and centrifugation at 6000 r/min for 15 min. The supernatant was separated and optical density of the supernatant was taken at 532 nm. The values were expressed as nanomoles of MDA per gram of tissue.
Measurement of glutathione peroxidase
The enzyme glutathione peroxidase (GPx) reduces the hydrogen peroxide to water. This was assayed by spectrophotometric method following Flohe and Gunzler’s 22 protocol. Briefly, a 5% tissue homogenate was prepared in potassium phosphate buffer (100 mM, pH 7.0; 1 mM EDTA; 1 mM phenylmethylsulfonyl fluoride (PMSF)). Then, centrifuged at 10,000 r/min at 4°C for 30 min and the supernatant was collected. To 2 µL of supernatant, 298 µL of assay buffer (0.1 M potassium phosphate buffer, pH 7; 1 mM EDTA; 1 mM sodium azide), 100 µL of mixture of reduced GSH and glutathione reductase, 50 µ of NADPH, and 50 µL of hydrogen peroxide (prewarmed at 37°C) were added. Enzyme kinetics was recorded at 340 nm over a period of 3 min at 37°C against air blank and the values were expressed as millimoles of NADPH oxidized per minute per milligram of protein.
Determination of CAT activity
The activity of CAT enzyme was determined as per the protocol of Aebi. 23 Briefly, 5% homogenate was prepared in potassium phosphate buffer (100 mM, pH 7.0; 1 mM EDTA; 1 mM PMSF) and centrifuged at 10,000 r/min for 30 min at 4°C. To 5 μL of the above supernatant, 1875 μL assay buffer (50 mM Na2HPO4, 50 mM KH2PO4, pH 7.0) and 120 μL H2O2 (0.66 M) were added. Enzyme kinetics was recorded at 240 nm over a period of 1 min. An extinction coefficient of 43.6 M/cm was used to determine the enzyme activity and values were expressed as millimoles of H2O2 degraded per minute per milligram of protein.
Evaluation of inflammation
Myeloperoxidase assay
Myeloperoxidase (MPO) levels in tissue homogenate were measured by using a MPO assay kit (USCN, Life Science Inc., China) following manufacturer’s instructions. Ice-cold PBS (pH 7.2) was used for tissue homogenization. The resulting suspension was sonicated with an ultrasonic cell disrupter, centrifuged at 5000 ×
Interleukins assay
Interleukins (ILs) assay was done using Qiagen Multi-Analyte ELISArray kit using manufacturer’s protocol. Tissue samples preparation was similar to that of MPO. This kit is designed to simultaneously profile the levels of multiple cytokines and/or chemokines using the conventional and simple sandwich-based enzyme-linked immunosorbent assay (ELISA) technique. The 96-well ELISA microplate has been coated with a panel of 12 target-specific capture antibodies, 1 in each 8-well strip allowed to obtain qualitative relative profiling of the sample. Various ILs assayed in this kit were IL-1α, IL-1β, IL-2, IL-4, IL-6, IL-10, IL-12, IL-17α, interferon γ (IFN-γ), tumor necrosis factor α (TNF-α), Granulocyte-Colony Stimulating Factor (GCSF), and Granulocyte-macrophage colony stimulating factor (GMCSF). The results were expressed as nanograms per gram of tissue.
Assessment of mitochondrial integrity and electron transport chain function
Complex I (NADH: ubiquinone oxidoreductase) assay
Complex I is one of the main sites of production of harmful superoxide. Its activity was assayed following the protocol of Janssen et al. 24 Briefly, a 10% brain homogenate was prepared in medium A1 (sucrose 250 mM, Na2EDTA 0.5 mM, Tris 10 mM, pH 7.4) and medium B1 (medium A1, Bovine Serum Albumin (BSA) 70 mg/mL). To the 10 μL of supernatant, 905 μL phosphate buffer (pH 7.8), 6 μL DCIP (0.6 μM/mL), 1 μL antimycin A (0.001 μM/mL), 7 μL decylubiquinone (0.7 μM/mL), and 50 μl BSA were added. Mixed well and after 3 min, 20 μL of NADH (0.2 mM/mL) was added and mixed. Activity was measured quickly at 37°C at every 30 s for 4 min at 600 nm. One microliter of rotenone (1 mM/L) was then added, and activity was again measured at every 30 s for 4 min at 600 nm.
Measurement of cytochrome c-oxidase activity and mitochondrial integrity
Cytochrome c-oxidase, the terminal enzyme of the respiratory chain, was assayed by the cytochrome c-oxidase assay kit (Sigma-Aldrich), following manufacturer’s instructions. This kit is suitable for the detection of mitochondrial outer membrane integrity and for the detection of mitochondria in subcellular fraction.
Measurement of cytochrome c-oxidase activity
Cytochrome c is reduced with dithiothreitol and then reoxidized by the cytochrome c-oxidase. Enzyme dilution buffer (10 mM Tris–HCl, pH 7.0 with 250 mM sucrose) was also used as tissue homogenization buffer. One unit will oxidize 100 μM of ferrocytochrome c per minute at pH 7.0 at 25°C. To 40 μL sample, 50 μL ferrocytochrome c substrate solution, 100 μL enzyme dilution buffer, and 950 μL assay buffer were added. Activity was measured at 550 nm and the values were expressed as nanomoles of cytochrome oxidized per minute per milligram of protein.
Measurement of the outer membrane integrity of mitochondria
The integrity of the outer membrane is assessed by measuring cytochrome c-oxidase activity in mitochondrial membranes in the presence and the absence of the detergent, n-dodecyl-β-
Determination of protein
The total protein of the samples was estimated using the method of Lowry et al. 25
Statistical analysis
The results are expressed as mean ± standard error mean (
Results
PQ is known to incur damage to the dopaminergic neurons in substantia nigra causing loss of dopamine resulting in Parkinson’s like characters. 26 There was a significant effect of PQ intoxication in mice on the rotarod performance, indicating damage to the motor neurons. The rotarod test was performed on day 28 of PQ intoxication to access the locomotor activity and motor coordination in mice. PQ-treated mice showed significant motor deficit and were unable to coordinate on rotarod for long, while treatment with TC either alone or in combination with Res (Figure 1) showed significant improvement in the motor coordination.

Effect of paraquat on motor coordination of mice. Paraquat significantly impairs the motor coordination of mice as seen on rotarod task. Cotreatment of resveratrol and tetracycline helped to improve the motor coordination compared to the treatment given alone. Values are expressed as mean ± SE. Significance was determined by one-way analysis of variance, followed by Student–Newman–Keuls test. *Significant changes are expressed in comparison with control and also # in comparison with PQ-treated mice (
The toxicity of PQ is a consequence of generation of superoxide anion, which can lead to the synthesis of more toxic ROS such as hydroxyl radicals and hydrogen peroxide. 27 To examine the efficacy of TC and Res in PQ-induced oxidative stress in mouse brain, liver, and lung, CAT, GSH, GPx, and MDA activity were measured. Following PQ exposure, the levels of potential antioxidant enzyme CAT (Figure 2) and GPx (Figure 3) were significantly lower as compared to the control. At the same time, the significant decrease in the levels of reduced GSH (Figure 4) and corresponding rise in the levels of MDA (Figure 5), a marker of lipid peroxidation, show severe oxidative stress. Treatment with TC and TC with Res significantly improved the toxic outcome of the PQ-induced oxidative stress. Treatment with Res and TC helped to improve the levels of oxidative stress markers in almost all the tissues.
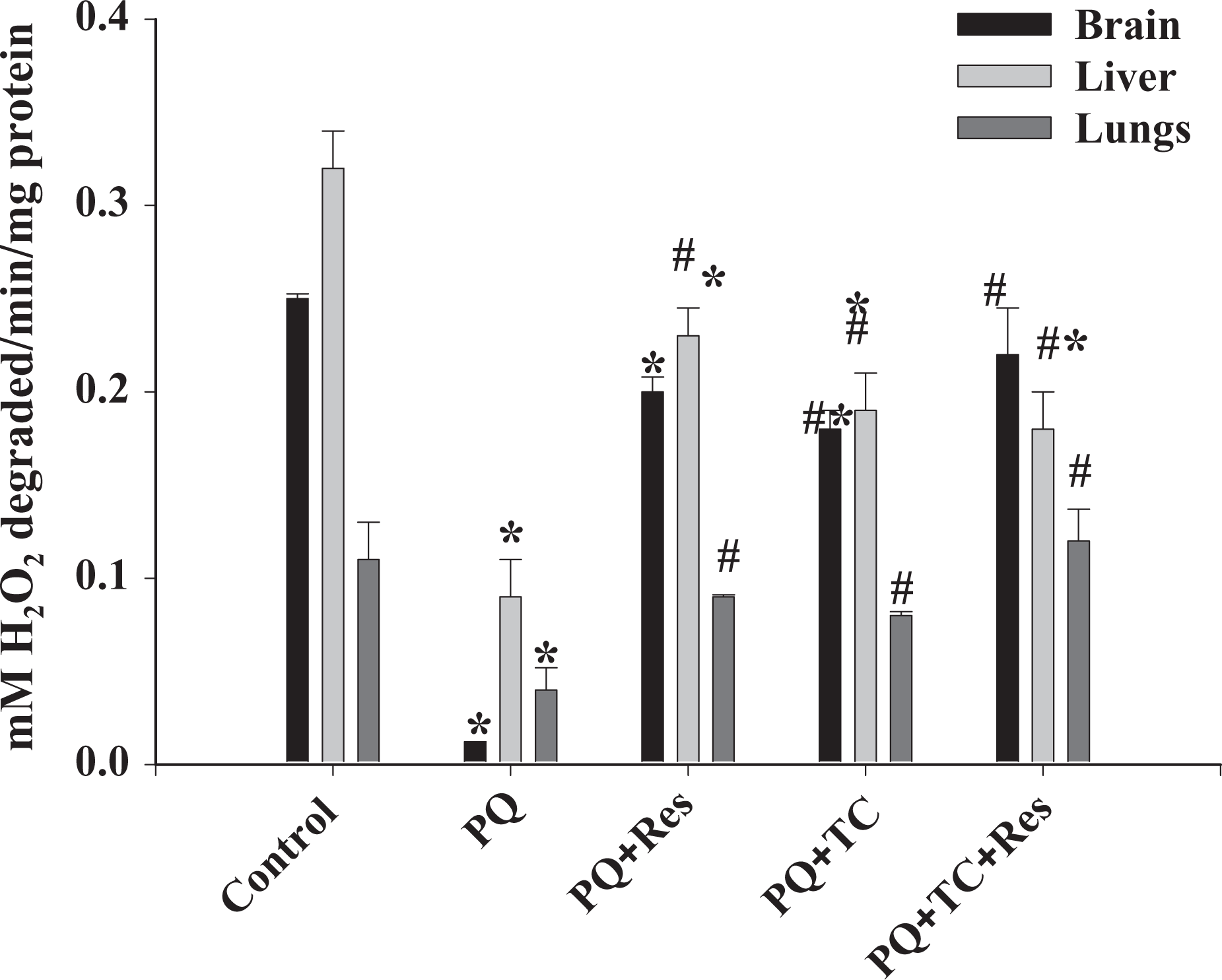
Effect of paraquat (PQ) on catalase activity. Paraquat significantly inhibited the activity of catalase. Tetracycline and resveratrol individually or in combination significantly protected catalase activity in the presence of PQ. Values are expressed as mean ± SE Significance was determined by one-way analysis of variance, followed by Student–Newman–Keuls test. *Significant changes are expressed in comparison with control and also # in comparison with PQ-treated mice. Brain and liver:

Effect of paraquat (PQ) on glutathione peroxidase (GPx) activity. Paraquat significantly inhibits the activity of GPx. GPx activity was significantly protected by the individual treatment of each drug. However, a combination was also significantly effective in protecting GPx activity in all three tissues. Values are expressed as mean ± SE. Significance was determined by one-way analysis of variance, followed by Student–Newman–Keuls test. *Significant changes are expressed in comparison with control and also # in comparison with PQ-treated mice. Brain,
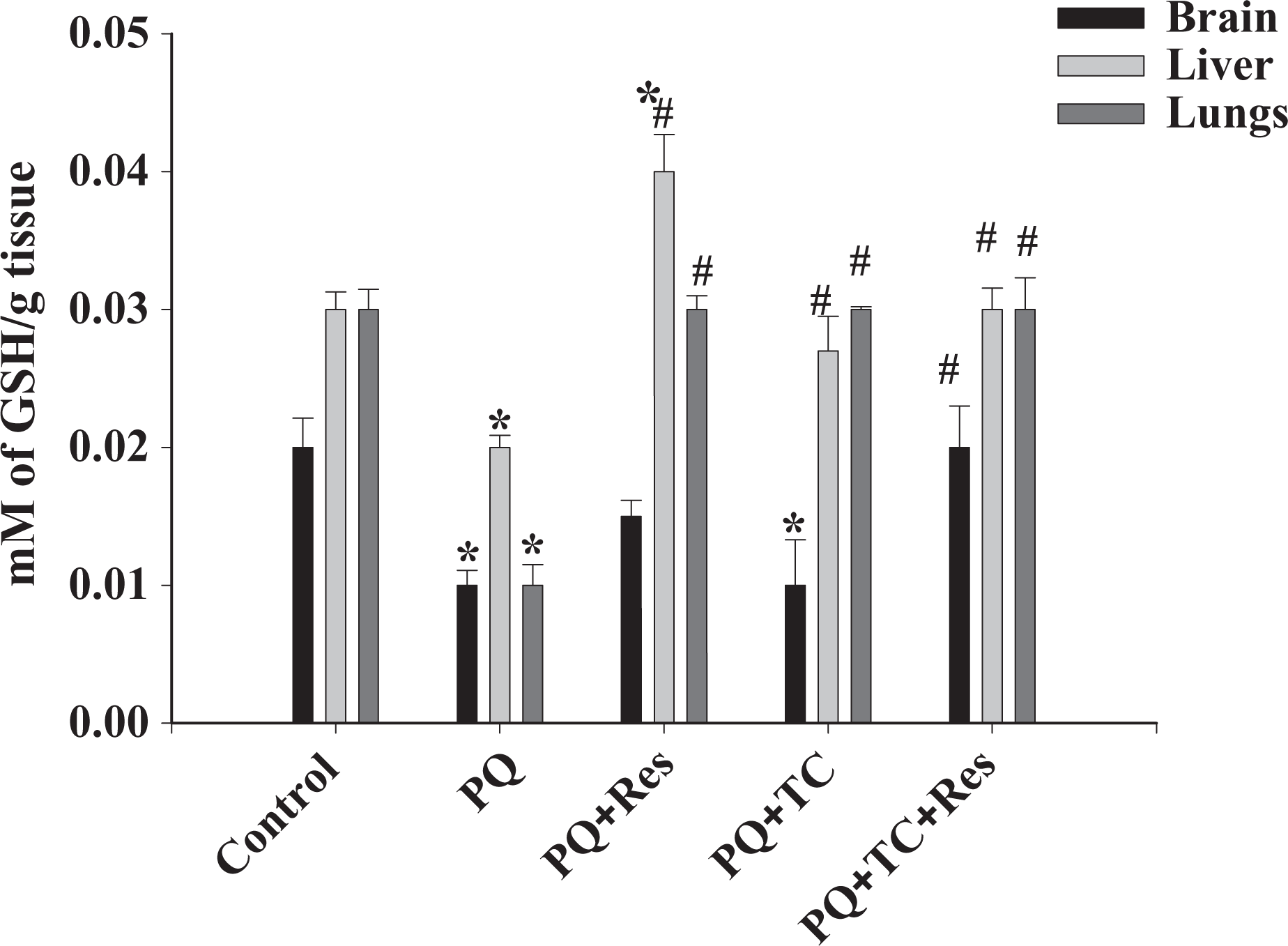
Effect of paraquat (PQ) on GSH level. Paraquat significantly reduced the GSH content in all three tissues. Tetracycline and resveratrol showed significant protection from oxidative stress resulting in increase in GSH levels. Values are expressed as mean ± SE. Significance was determined by one-way analysis of variance, followed by Student–Newman–Keuls test. *Significant changes are expressed in comparison with control and also # in comparison with PQ-treated mice. Brain,

Effect of paraquat (PQ) on the MDA levels. Paraquat significantly increased the MDA levels. Tetracycline and resveratrol showed significant protection from oxidative stress–induced lipid peroxidation resulting in decreased levels of MDA. Values are expressed as mean ± SE. Significance was determined by one-way analysis of variance, followed by Student–Newman–Keuls test. *Significant changes are expressed in comparison with control and also # in comparison with PQ-treated mice.
As stated earlier, PQ poisoning causes severe lung injury. This is mainly due to the PQ-induced pulmonary inflammation and fibrosis caused due to PQ-induced oxidative stress, inflammatory, and fibrotic reactions. 28 PQ caused increase in the production of inflammatory and fibrogenic cytokines such as TNF-α, IL-1β, and IFN-γ (Table 1). In addition, PQ also caused rise in neutrophils accumulation in lungs and brain as shown by the increased presence of MPO in both the tissues (Table 2). Taken together, PQ caused oxidative stress and inflammation in both brain and lungs. Res with its antioxidant activities caused decrease in oxidative stress. TC with its anti-inflammatory activities along with Res helped to reduce the inflammation (Tables 1 and 2).
Cytokines (ng/g tissue) in brain and lung homogenates.a

PQ: paraquat; IL: interleukin; IFN-γ: interferon γ; TNF-α: tumor necrosis factor α.
aValues are expressed as mean ± SE.
bSignificantly different from the respective control data. A
cSignificantly different as compared to paraquat-treated group. A
Myleoperoxidase (ng/g tissue) activity in brain and lung homogenates.a

PQ: paraquat; Res: Resveratrol; TC: Tetracycline.
aValues are expressed as mean ± SE.
bSignificantly different from the respective control data.
cSignificantly different as compared to paraquat treatment.
Chemical structure of PQ is very similar to the active form of 1-methyl-4-phenyl-1,2,3,6-tetrahydropyridine (MPTP), a neurotoxin known to cause parkinsonism in animals, 29 hence it is hypothesized to inhibit the complexes of the electron transport chain (ETC). Owing to its high oxygen consumption and mitochondrial activity, ETC complexes inhibition study of the brain tissues helps to delineate the PQ-induced neurotoxicity. Mitochondria are a major organelle for oxygen consumption and in turn ROS production in the cells. As reported in many previous research papers, mitochondria are a major target of PQ and damage to mitochondria contributes to oxidative stress induced by PQ. Complex I (NADH: ubiquinone oxidoreductase) is the first and the most complex of the three energy-transducing enzyme complexes of the ETC in mitochondria. Subacute PQ exposure causes significant damage to mitochondria. PQ showed inhibition of the rotenone-sensitive complex I activity (Figure 6), but in the presence of TC and/or Res, the complex I activity is almost unchanged, indicating severe oxidative stress also cause the inhibition of the complex I subunits. PQ was also found to be responsible for the inhibition of complex IV in brain tissue mitochondria. A decrease of approximately 45% complex IV activity was observed in the PQ-exposed mice brain as compared to control (Figure 7). A significant number of mitochondria (approximately 50%) showed damaged outer membrane due to PQ treatment (Figure 8). Res but not TC (alone) was found to be effective to maintain the complex IV activity (Figure 7) and integrity of the outer membrane (Figure 8).

Effect of paraquat (PQ) on the complex I activity of the electron transport chain in brain homogenates. PQ significantly inhibits the complex I activity. Tetracycline and resveratrol showed significant protection of complex I activity. Values are expressed as mean ± SE. Significance was determined by one-way analysis of variance, followed by Student–Newman–Keuls test. *Significant changes are expressed in comparison with control and also # in comparison with PQ-treated mice (
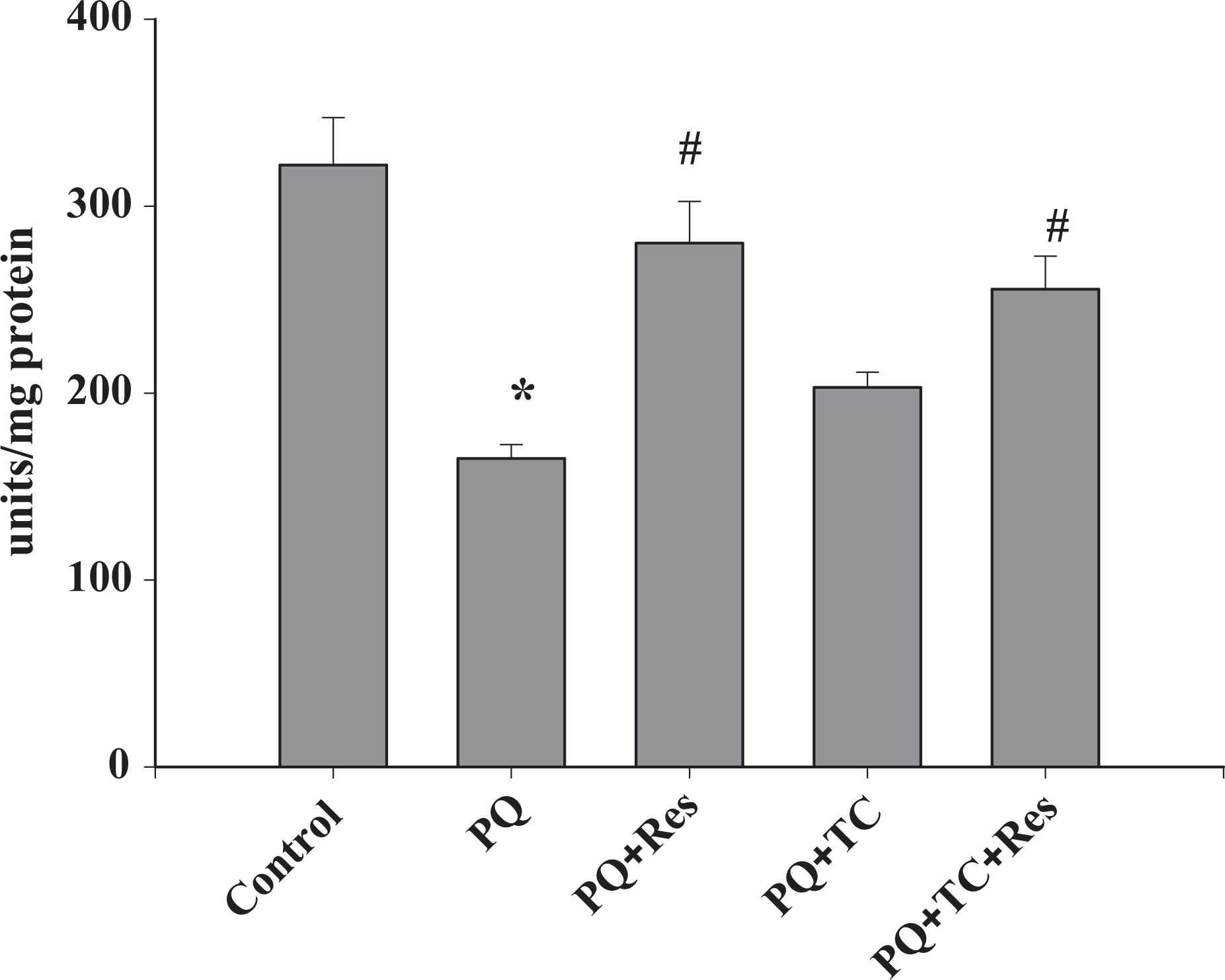
Effect of paraquat (PQ) on the ETC complex IV activity of brain tissue. PQ significantly inhibits the complex IV activity. Combined treatment of tetracycline and resveratrol significantly protected complex IV activity although tetracycline alone was not effective. Values are expressed as mean ± SE. Significance was determined by one-way analysis of variance, followed by Student–Newman–Keuls test. *Significant changes are expressed in comparison with control and also # in comparison with PQ-treated mice (

Effect of paraquat (PQ) on the integrity of the outer membrane of the brain mitochondria. PQ is responsible for significant damage to the outer membrane of the mitochondria. Tetracycline and resveratrol significantly protected mitochondrial integrity either alone or in combination. Values are expressed as mean ± SE. Significance was determined by one-way analysis of variance, followed by Student–Newman–Keuls test. *Significant changes are expressed in comparison with control and also # in comparison with PQ-treated mice (
Discussion
PQ is one of the most clinically significant herbicides in terms of morbidity and mortality. PQ intoxication causes many pathological events that primarily involve fibroblast migration and proliferation, amplified collagen synthesis in various tissues including lungs, liver, kidneys, and venules, mediating by lipid peroxidation and overexpressing of fibrogenic cytokines. 30 PQ intoxication is more frequently fatal than poisoning caused by other pesticides in the absence of any specific antidote(s). Studies of Okonek et al. 31 and Gaudreault et al. 32 have questioned the efficacy of gut lavage, use of activated charcoal, hemodialysis, and hemoperfusion, as treatments for PQ poisoning. Previous studies have also shown that treatment with cyclophosphamide and methylprednisolone showed reduced mortality rate in PQ patients, but at the same time some studies disagreed with these results. 33 Moreover, most treatments used for PQ poisoning are not effective, suggesting an immediate necessity for novel therapies. 34
Mitochondria, being an important organelle of oxidative phosphorylation, are therefore a major site of ROS production. Mitochondria are a major target of PQ2+ and damage to mitochondria or inhibition of the enzyme complexes of the ETC disrupts the steady state of oxygen utilization and ROS production. 35 PQ-induced mitochondrial damage and PQ redox cycling forming a vicious cycle further contributes to oxidative stress. It has been observed that the loss of Nrf2 in cells further exacerbated PQ-induced ROS production and mitochondrial toxicity, supporting the notion that ROS and mitochondrial damage are the major mechanisms by which PQ causes lung damage and fibrosis. In a recent study by Czerniczyniec et al., 36 it has been shown that acute PQ exposure causes changes in mitochondrial oxygen consumption, indicating altered activities of the enzymes involved in ETC. Owing to its remarkable structural similarity to MPP+, PQ has been hypothesized to inhibit complex I of ETC, 37 although this hypothesis has been questioned later 38 but finally, a report from Keeney et al. 39 suggested that in the brain of human Parkinson’s disease (PD) patients, some subunits of the complex I are oxidatively damaged, resulting in misassembling and functional impairment of complex I, resulting in its inhibition. One of the main functions of complex I is the transport of electrons across the mitochondrial inner membrane via oxidation of the NADH coupled to reduction of ubiquinone. 40 The inhibition of complexes I and IV activity and the redox cycling reactions results in the reduction of molecular oxygen (O2) to superoxide radical (O·−2). This superoxide radical thus generated is subsequently converted to H2O2 either spontaneously or through the actions of superoxide dismutase, 36 resulting in a severe oxidative stress. The direct action of PQ on the respiratory chain inhibiting complex I and complex IV of ETC causes an important decrease in mitochondrial membrane potential and a decrease in state 3 respiratory rates, culminating in an impaired mitochondrial function. 36 Palmeira et al. 41 and then Gomez et al. 42 suggested that high concentrations of PQ2+ may exert direct oxidative damage toward all ETC complexes nonselectively, leading to an impaired mitochondrial output. This may be due to oxidative damage and compromised function of the ETC complexes as a consequence of the close physical proximity to the site of mitochondrial ROS production by PQ2+ and ETC complexes. After PQ poisoning, if the oxidative stress and the mitochondrial dysfunction persist, it results into the induction of different death signaling pathways in the cells. The literature review so far suggests that this is an important phase to apply a pharmacological rescue against PQ-induced toxicity in order to reduce the potential alveolar and neuronal deaths.
Res is a naturally occurring antioxidant compound with multiple health-promoting effects. 43 Res was shown to inhibit PQ-induced oxidative damage, expression of profibrogenic cytokines and growth factors, and transformation of fibroblasts into myofibroblasts effectively. 35 This wide range of biological effect may be explained in part by the antioxidant properties of Res, and the activation and expression of SIRT1 are postulated to be a key event in the pathophysiology of Res. 44 A major mechanism in the cellular defense against oxidative stress is the activation of the Nrf2 antioxidant response element signaling pathway. 45 Previous studies have demonstrated that Nrf2 is important in PQ-induced lung injury; PQ can inhibit the Nrf2 expression in vitro and in vivo. 46 Additionally, overexpression of Nrf2 can ameliorate PQ-induced lung injury and cell apoptosis. 47 The function of the Nrf antioxidant pathway is controlled by multiple factors, including the acetylation–deacetylation of Nrf2. Kawai et al. 48 demonstrated that SIRT1-mediated deacetylation of the Nrf2 protein terminated the transcription of antioxidant genes in vitro. As reported by Li et al., 18 an increase in the SIRT1 expression levels by Res was associated with a high level of Nrf2. Furthermore, Res-induced protection of vascular endothelium involves activation of Nrf2. 17 Additionally, upregulation of the enzyme activities of heme oxygenase-1, superoxide dismutase and CAT and reduced GSH levels were upregulated following Res administration. 18 As observed in the present study, Res protects against the oxidative stress either alone or in combination with TC against PQ-induced oxidative stress in lungs, liver, and brain.
TCs are reported to have remarkable neuroprotective properties in models of cerebral ischemia,
3
spinal cord injury,
49
PD and Huntington’s disease,
50
amyotrophic lateral sclerosis,
51
and multiple sclerosis.
52
However, contradictory experimental results and failures in human trials have also been reported.
53
Activation of microglia, a common feature of most of the neurodegenerative diseases, leads to the release of pro-inflammatory mediators and other injury response factors that ultimately compromise neuronal and oligodendroglial viability. Minocycline (a TC), which shows considerable anti-inflammatory properties, inhibits (i) microglial activation and proliferation, (ii) the induction of caspase-1 and iNOS following ischemic insults, exposure to MPTP, or excitotoxicity, and (iii) the induction of cyclooxygenase 2 secondary to ischemia.
54
TCs also known to inhibit caspase-dependent and independent cell death induced by different stimuli such as glutamate, H2O2, thapsigargin, a mixture of TNF-α and cycloheximide, 3-nitropropionic acid (a mitochondrial toxin), and etoposide (a DNA-damaging agent) in vitro.
55
TCs are the bacteriostatic (antibacterial) agents that bind to the 30 S ribosomal subunit of bacteria and inhibit protein synthesis. Second-generation semisynthetic TCs such as minocycline and doxycycline are presently being used as typical antibiotics in humans. Additionally, their efficacy in treating multidrug-resistant infections and ability to penetrate the blood–brain barrier along with good clinical safety record makes them a potential drug candidate against various human diseases.
56
Although previous articles showed that TC could prevent PQ-induced oxidative stress in
In conclusion, PQ causes a series of serious impediments including oxidative stress and inflammation in lungs and other vital organs including brain. PQ·+ causes inhibition of ETC complexes in the brain to further exacerbate the toxicity. Results of the present study show that antioxidant properties of Res and antibiotic and anti-inflammatory properties of TC offered considerable protection against the toxic effect of subacute doses of PQ in mice. Further studies are required to consider this combination as the supportive treatment regimen for PQ poisoning.
Footnotes
Acknowledgement
The authors are thankful to the Director, DRDE, for permitting to use the facility for this work.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
