Abstract
Background
Paraquat (PQ) poisoning induces severe acute kidney injury and causes extremely high rate of death. In this study, we aimed to investigate the protective effects of silymarin on PQ-induced acute kidney injury and explore the underlying mechanisms.
Methods
A rat model was established through intraperitoneal injection of PQ. Rats were administrated with saline or silymarin for 3 days. Then, survival rate, physiological parameters, and renal injury score were evaluated. The apoptosis and oxidative stress in kidney tissues were determined through hematoxylin and eosin staining, quantitative reverse transcription polymerase chain reaction, Western blot, and enzyme-linked immunosorbent assays.
Results
Silymarin administration could significantly increase the survival rate of PQ-poisoned rats. It was found that silymarin treatment improved renal function, decreased injury score in kidney tissues, and inhibited the apoptosis and oxidative stress in PQ-induced acute kidney injury through the activating the signaling pathway of Nrf2 and promoting its nuclear translocation.
Conclusion
Silymarin exhibited a protective effect against PQ-induced kidney injury, suggesting that treatment with this flavonoid could be a potential therapeutic agent for the treatment of acute kidney injury.
Introduction
Paraquat (PQ) is one of the most frequently used non-selective herbicides, which has low cost and high efficiency. Therefore, it is widely used in developing countries.1,2 However, this herbicide has extreme toxicity for humans and most animals, causing 20 deaths per million persons worldwide due to the lack of effective drugs.3,4 In mammals, PQ results in very serious multiple organ failure, including liver, lung, heart, brain, kidney, and gastrointestinal tract.4–9 Upon poising, this chemical is distributed in most organs rapidly, with highest concentration detected in kidneys.6,10 The accumulation of PQ in kidney causes severe nephrotoxicity, reduces renal function, and results in further kidney injury. 11 Hence, maintaining renal function is the important therapeutic strategy for patients who suffer from PQ poisoning.
As a potent free radical producer, oxidative stress is one of the major characters of PQ toxicity. Paraquat poisoning accelerates the production of reactive oxygen species (ROS) that subsequently results in tissues injury and cell damage through apoptosis, mitochondrial damage, lipid peroxidation, and inflammatory response. 12 Although antioxidants, such as Vitamin C, ethylenebis tetraacetic acid, and ethylenebis hydroxyphenylglycine showed protective effects on PQ poisoning, the underlying mechanism is still unknown.13,14
Silymarin is a polyphenolic flavonoid extracted from fruits and seeds of milk thistle. It has been reported to have promising effects on PQ poisoning.15–17 Recently, several studies have demonstrated anti-inflammatory and antioxidant activities of silymarin, and no significant adverse effects were observed during the studies.18,19 Zhao et al. reported that silymarin (200 mg/kg) can treat acute lung injury caused by PQ poisoning through regulating the Nrf2 signaling pathway. 17 Ahmad et al. demonstrated that silymarin (100 mg/kg) had a protective value against acute liver injury caused by PQ exposure. 15 However, the protective effects of silymarin on PQ-induced kidney injury remain unclear.
Materials and methods
Reagents
Silymarin (SML2581, ≥95% (high performance liquid chromatography)) and PQ were ordered from Sigma-Aldrich (St Louis, USA), and dissolved in 0.5% CMC-Na (Dalian Biotech, Dalian, China) and saline, respectively.
Animals
Six-to eight-week-old male Wistar rats (200–250 g) were used in this study. Animals were housed under the 12/12 h light/dark cycle standard condition at 23°C with 50% humidity, and free access to food and water. All animal experiments were carried out according to the guidelines created by the University Committee on Use and Care of Animals at Xingtai People’s Hospital.
Paraquat poisoning model and treatments
The rats were intraperitoneally injected with PQ to establish the PQ poisoning model, and saline injected rats were used as controls. Based on the previous studies, 25 mg/kg dose was used in this study.17,20–23 Animals were randomly divided into six groups: (1.) Sham group: rats were exposed to saline; (2.) Sham+SIL-H group: rats were exposed to saline and then treated with high dose of silymarin (200 mg/kg body weight) for 3 days via i.p. injection 1 h after exposure to saline; (3.) PQ group: the rats were exposed to PQ (25 mg/kg bodyweight) and treated with saline; (4.) PQ+SIL-L group: the rats were exposed to PQ (25 mg/kg bodyweight) and treated with low dose of silymarin (50 mg/kg body weight) for 3 days via i.p. injection 1 h after exposure to PQ; (5.) PQ+SIL-M group: the rats were exposed to PQ (25 mg/kg bodyweight) and treated with medium dose of silymarin (100 mg/kg body weight) for 3 days via i.p. injection 1 h after exposure to PQ; (6.) PQ+SIL-H group: the rats were exposed to PQ (25 mg/kg bodyweight) and treated with high dose of silymarin (200 mg/kg body weight) for 3 days via i.p. injection 1 h after exposure to PQ. After 3 days treatment, rats were euthanized by using 10% chloral hydrate (3 mg/kg) for the subsequent expression and regulation analyses.
Survival analysis
Rats were administered silymarin at low, medium and high concentrations as mentioned above (1 h after exposure to PQ) for three consecutive days, once a day. The day of PQ poisoning was the first day. Followed up for 19 days to observe the death of rats (
Urine and serum assays
Three days after PQ poisoning, serum and urine were collected. Blood urea nitrogen (BUN) and serum creatinine concentrations were determined by the urea and creatinine assay kits (Beyotime, Shanghai, China). The urine concentration of neutrophil gelatinase-associated lipocalin (NGAL) was measured by the rat NGAL enzyme-linked immunosorbent assay (ELISA) kit (Beyotime, Shanghai, China).
Histological analysis
Kidney tissues from rats with 3 days of treatment (Sham, PQ, and PQ-SIL-H,
TUNEL assay
TUNEL assay was performed to assess the apoptotic status in kidney tissues of rats with different treatments. Kidney tissues from rats with 3 days of treatment (Sham, PQ, and PQ-SIL-H,
Oxidative stress assays
Three days after PQ poisoning, the kidney tissue samples from Sham, PQ, and PQ-SIL-H groups (
Gene expression analysis
The total RNA of kidney tissues from Sham, PQ, and PQ-SIL-H groups (
SOD1 reverse: 5′-TTG TTT CTC ATG GAC CAC-3′;
Western blot
Kidney tissues from Sham, PQ, and PQ-SIL-H groups (
Statistical analysis
All statistical analyses in this study were carried out by using the Prism 8.0 software. One-way analysis of variance method with a post hoc test were used to analyze the differences between groups. The data were represented as mean ± standard deviation (SD).
Results
Silymarin reduces death caused by PQ poisoning
To investigate the protective role of silymarin against PQ-induced acute kidney injury, we firstly performed survival analysis. As shown in Figure 1, less than 10% of rats survived after 15 days of PQ treatment, which indicated its extreme toxicity, whereas with the co-treatment of different doses of silymarin (PQ+SIL-L, PQ+SIL-M, PQ+SIL-H), the survival rates of PQ-treated rats were increased dose-dependently. Both medium and high doses of silymarin could markedly increase the survival rate of PQ-poisoned rats. It’s worthy to note that the survival rate of rats in Sham+SIL-H group was compatible to that in Sham group, which indicated that there was no side effect and toxicity of silymarin. The survival data implied that silymarin had a protective role on PQ poisoning. Survival rate of rats in indicated groups. Rats were administered with PQ (25 mg/kg) by intraperitoneal injection and/or treated with different doses of silymarin for 3 days. 
Silymarin ameliorates the PQ-induced kidney damage
We collected serum and urine samples from rats (3 days after PQ/silymarin treatment) and measured the pathological indicators of kidney damage. The concentrations of BUN and serum creatinine were significantly elevated after PQ exposure; however, silymarin co-treatment could decrease the PQ-induced creatinine and BUN levels significantly (Figure 2(a) and (b)). Moreover, the urine NGAL level, another kidney injury marker, was significantly elevated by PQ treatment and reduced by silymarin co-treatment (Figure 2(c)). We further analyzed the renal injury score through histological examination. PQ exposure significantly induced kidney injury, which could be ameliorated by the silymarin co-treatment (Figure 3(a) and (b)). Assessment of serum and urine parameters of rats in indicated groups. Serum creatinine (a), blood urea nitrogen (BUN, b), and urine neutrophil gelatinase-associated lipocalin (NGAL, c) among different groups at 3 days after PQ poisoning. Data were presented as mean ± SD. Silymarin attenuated PQ-induced histopathological changes of renal tissues. Three days after PQ poisoning, the renal histological changes were determined by H&E staining (a), and the quantification of renal injury score (b). The arrows indicated degenerative glomeruli, degenerative tubuli, necrosis, and hemorrhage. Data were presented as mean ± SD. 

Silymarin reduces PQ-induced cell apoptosis in renal tissues
TUNEL assay result showed that PQ exposure could cause distinct cell apoptosis in kidney tissues compared to that in Sham rats, while PQ-induced apoptosis was significantly decreased in rats with silymarin co-treatment (Figure 4(a) and (b)). Consistently, the protein level of cleaved caspase-3, an apoptotic biomarker, was markedly increased after PQ administration and decreased with silymarin co-treatment (Figure 4(c) and (d)). These results suggested the anti-PQ poisoning effect of silymarin might be mediated by the inhibition of renal cell apoptosis. Silymarin attenuated PQ-induced cell apoptosis of renal tissues. (a), Representative TUNEL staining of renal tissues (magnification, ×400) and the quantification of TUNEL positive cells per field (b). 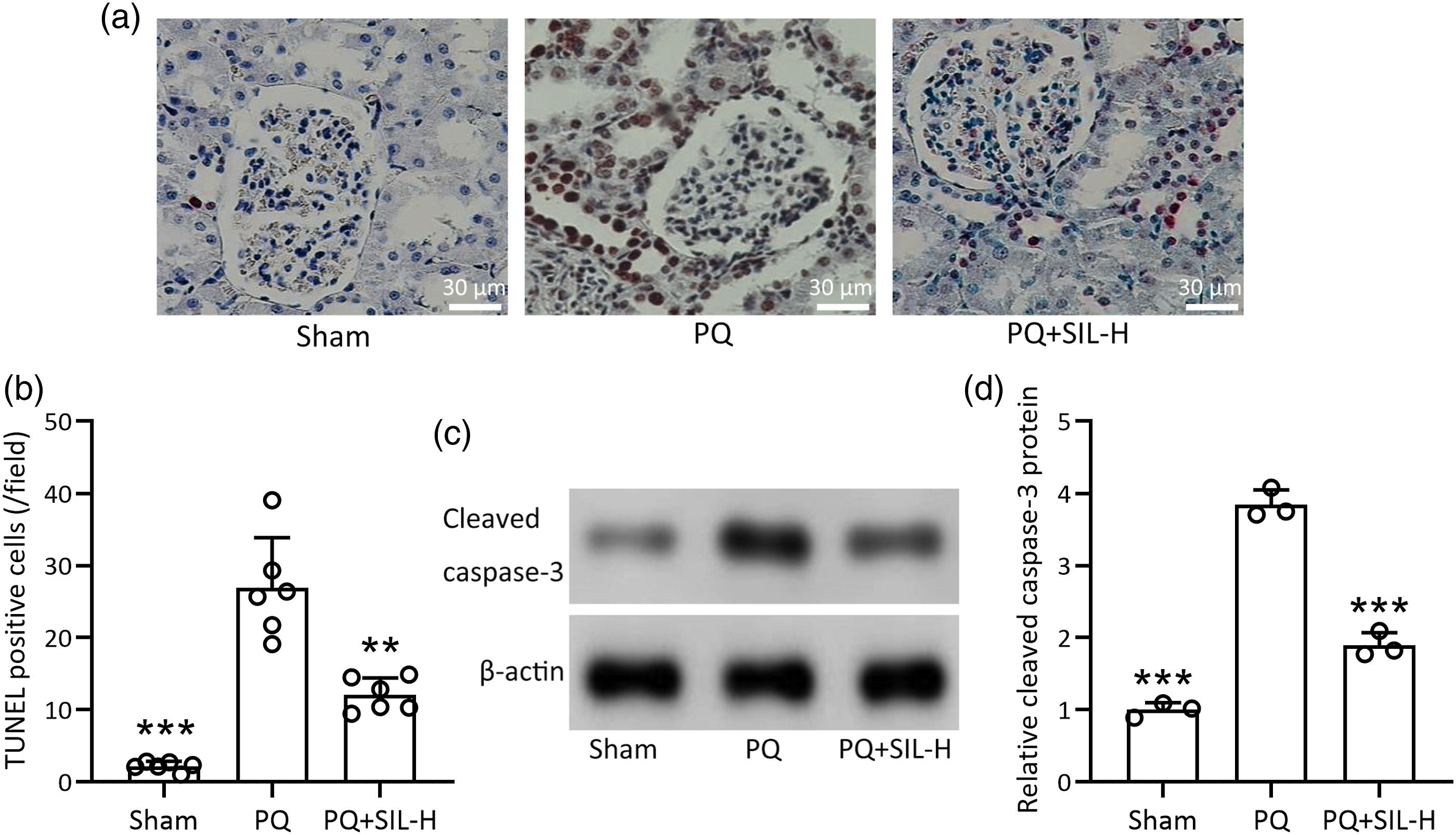
Silymarin attenuates the PQ-induced oxidative stress
Oxidative stress is one of the major features of PQ-induced tissue injury.
24
To evaluate the antioxidant effect of silymarin on acute kidney injury, we measured the concentration and activity of multiple oxidative markers in kidney tissues, including MDA concentration, SOD activity, CAT activity, and the GSH/GSSG ratio. Silymarin co-treatment was able to significantly decrease the PQ-induced MDA elevation in kidney tissues (Figure 5(a)). Moreover, PQ exposure inhibited the activities of SOD and CAT, and decreased the ratio of GSH/GSSG. In contrast, silymarin co-treatment could partially rescue the activities of SOD (Figure 5(b)) and CAT (Figure 5(c)), and the ratio of GSH/GSSG (Figure 5(d)). Silymarin attenuated PQ-induced oxidative stress in renal tissues. Three days after PQ poisoning, the MDA concentration (a), SOD activity (b), CAT activity (c) and the GSH/GSSG ratio in kidney tissues were evaluated. Data were presented as mean ± SD. 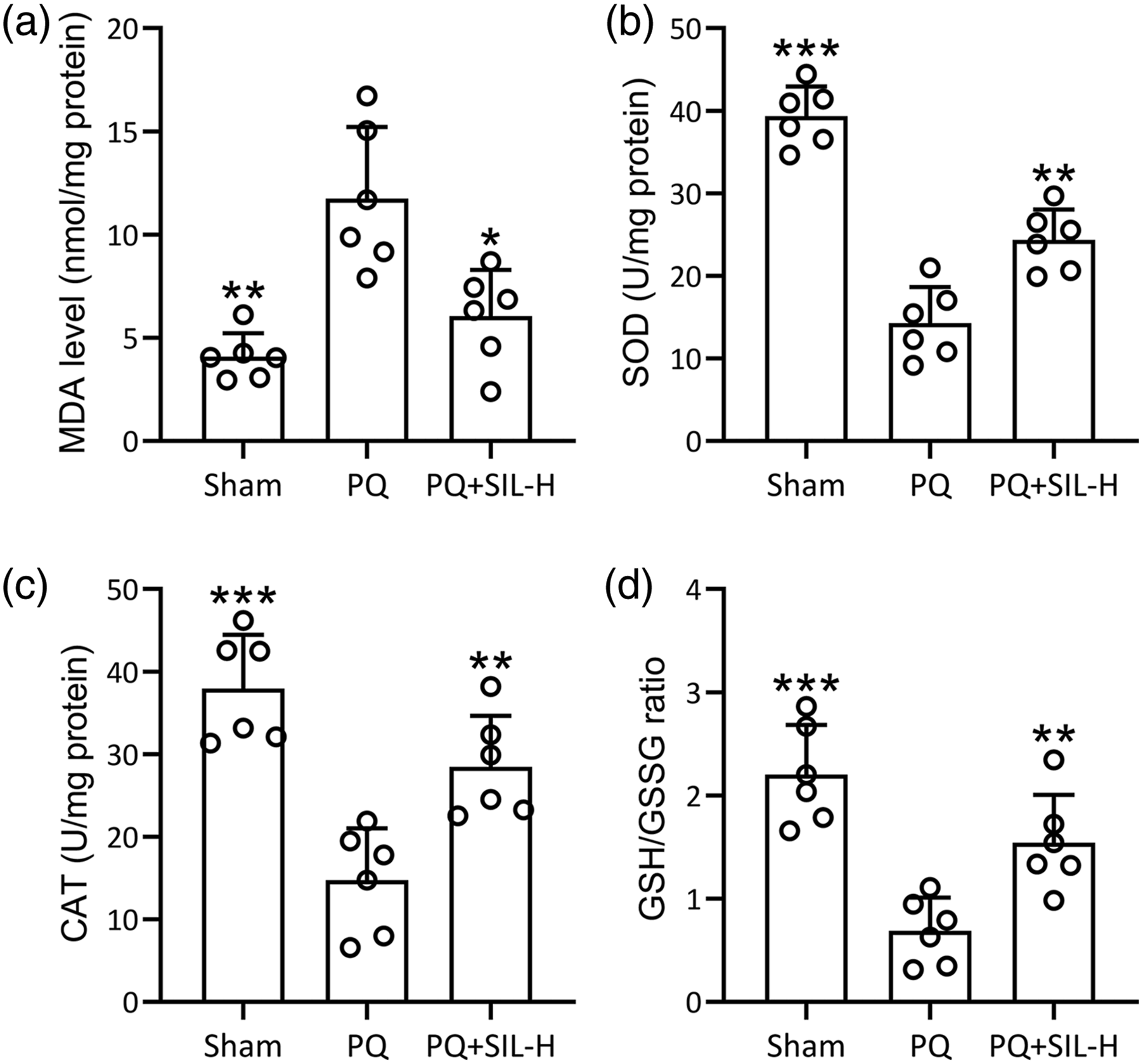
Silymarin activates Nrf2 signaling pathway in the renal tissues
Nrf-2 is an antioxidant transcription factor that involves in the oxidative stress processes through its nuclear translocation and the up-regulation of its downstream genes, such as NQO1, HO-1, and SOD1. In comparison with Sham and PQ rats, silymarin co-treatment significantly increased the nuclear translocation of Nrf-2 in renal tissues (Figure 6(a) and (b)). Gene expression analysis further confirmed the protective effects of silymarin in acute kidney injury, as evidenced by up-regulated expression of Nrf-2 targets, NQO1, HO-1, and SOD1 (Figure 6(c)–(e)). All these data indicated that silymarin had important protective effect on PQ-induced kidney injury via inhibiting oxidative stress in PQ poisoning rats. Silymarin activated the Nrf2 signaling pathway in the renal tissues. Western blotting was used to measure nuclear and cytoplasmic Nrf2 protein expression (a) and the nuclear translocation of Nrf2 presented as Nrf2 nuclear/cytoplasmic ratio (b). The mRNA expressions of NQO1 (c), HO-1 (d) and SOD1 (e) in the renal tissues were measured by qRT-PCR. 
Discussion
Paraquat is a highly toxic herbicide that can cause severe damage to organs, including liver, lung, heart, brain, kidney, and gastrointestinal tract. In PQ intoxicated patients, acute kidney injury occupied more than 45% cases, which had higher mortality rate than non-acute kidney injury patients. 25 Oxidative stress is one of the major characters of PQ toxicity. 12 In this study, we investigated the protective effect of silymarin, a polyphenolic flavonoid extracted from seeds and fruits of milk thistle, 15 on PQ-induced acute kidney injury in rat model, and found that silymarin treatment (100 and 200 mg/kg body weight) for 3 days could effectively reduce PQ poisoning caused death. At the physiological level, silymarin treatment was able to alleviate PQ-induced elevation of serum creatinine, BUN, and urine NGAL. Moreover, silymarin treatment significantly improved the renal function through inhibiting the apoptosis and oxidative stress induced by PQ exposure. Mechanistically, silymarin activates the Nrf2 signaling pathway via promoting the nuclear translocation of Nrf2. Our findings suggest that silymarin has antioxidant activity with therapeutic potential for the treatment of PQ-induced acute kidney damage.
The interrelationship between acute kidney injury and oxidative stress has been illustrated by several studies recently. Oxidative stress is characterized by an increased generation of ROS or reactive nitrogen species, which are largely involved in the development of acute kidney injury. 26 Various reports have indicated that the natural compounds could exert antioxidant activities.27,28 Recently, Lin et al. and Chen et al. reported that targeting on the oxidative stress processes showed protective effect on kidney damage, respectively.29,30 Betanin, a scavenger of ROS, 31 could protect the kidney against the PQ-induced acute damage via suppression of ROS generation. 21 Similarly, high dose of vitamin C administration was able to significantly protect kidney from PQ toxicity. 13 Silymarin has been reported to have the antioxidant activity and display the protective effect in multiple tissues.32,33 In line with the above findings, the MDA concentration, a marker of oxidative stress, was significantly decreased with the treatment of silymarin. In contrast, the activities of SOD and CAT, and the GSH/GSSG ratio in kidney tissues of PQ-poisoned rats were significantly increased after 3 days silymarin administration. In addition, the PQ-induced cell apoptosis of renal tissues was reduced significantly. All these results establish the protective role of silymarin in acute chronic kidney injury in animal models.
Paraquat can cause acute kidney damage and various complications, and greatly increase the probability of the individual death.6,25,34 As a polyphenolic flavonoid, silymarin has been confirmed to have the anti-inflammatory and antioxidant activity.35,36 Upon the silymarin administration, the BUN and NGAL, blood and urine indicators that related to kidney damage, were improved significantly. Moreover, renal injury score suggested that silymarin administration reduces the PQ-induced histopathological anomalies and relieves the structural damage of kidney tissue markedly. Furthermore, silymarin treatment significantly reduced the PQ-induced mortality of rats (19-day observation) in a dose-dependent pattern. All these findings indicated that silymarin suppressed PQ-induced apoptosis and oxidative responses in kidney tissues, and reduced kidney damage and mortality effectively.
Nrf2 is a basic region leucine-zipper transcription factor that works as a key mediator in oxidative stress, 37 which involves in the oxidative stress processes through its nuclear translocation and the up-regulation of its downstream genes, such as NQO1, HO-1, and SOD1. In glomerulosclerosis mouse model, antroquinonol, a natural ubiquinone derivative, could decrease the oxidative stress and inflammation levels in kidney tissues through enhancing Nrf2 signaling pathway. 38 In this study, activation of the Nrf2 signaling pathway after silymarin treatment was observed. Importantly, we further explored that the increased nuclear translocation of Nrf2 might be the major driver of the activation of this signaling pathway in PQ-induced injury kidney tissues. However, the underlying mechanism of silymarin mediated nuclear translocation of Nrf2 and whether silymarin treatment involve in the inflammatory response of acute kidney injury are still unknown, which need to be addressed in the future studies.
Conclusion
This study demonstrates that silymarin exerted the protective effects against PQ-induced kidney injury through activating the Nrf2 signaling pathway. Hence, this flavonoid might be a potential therapeutic drug for the treatment of acute kidney injury.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethics approval
All animal experiments were carried out according to the guidelines created by the University Committee on Use and Care of Animals at Xingtai People’s Hospital.
