Abstract
The main purpose of this study was to assess the role of S100B protein, neuron-specific enolase (NSE), and glial fibrillary acidic protein (GFAP) in the evaluation of hypoxic brain injury in acute carbon monoxide (CO)-poisoned patients. This cross-sectional study was conducted among the patients with acute CO poisoning who referred to the emergency department in a 1-year period. Serum levels of S100B protein, NSE, and GFAP were determined on admission. A total of 55 CO-poisoned patients (mean age ± standard deviation, 45 ± 20.3 years; 60% women) were included in the study. The control group consisted of 25 healthy adults. The patients were divided into two groups according to whether they were conscious or unconscious. The serum levels of S100B, NSE, and GFAP were higher in patients than that in the control group. There was no significant difference between unconscious and conscious patients with respect to these markers. There was a statistically significant difference between the conscious and unconscious patients and the control group in terms of S100B and NSE levels. There was also a statistically significant difference between the unconscious patients and the control group in terms of GFAP levels. Increased serum S100B, NSE, and GFAP levels are associated with acute CO poisoning. These biomarkers can be useful in assessing the clinical status of patients with CO poisoning.
Keywords
Introduction
Carbon monoxide (CO) poisoning is the cause of more than one half of the fatal poisonings in the world. 1,2 In severe CO poisoning, cardiovascular and central nervous system (CNS) findings become prominent. Neuropsychiatric symptoms may be seen in approximately 67% of the patients. 2 Brain injury (BI) occurs at varying degrees depending on the severity of the clinical picture. In patients with CO poisoning, the diagnosis and prognosis is still controversial. In the literature, the use of BI markers in poisoning has been investigated in several studies. 1 –4 In different studies, markers such as S100B, neuron-specific enolase (NSE), and glial fibrillary acidic protein (GFAP) have been used to determine the prognosis after BI. 1 –4 S100B is mainly found in astroglia, bone marrow, adipose tissue, and skeletal muscle; it can be determined immediately after injury, and its half-life ranges from 30 min to 2 h. 5 NSE is found in neuron cytoplasm, erythrocytes, and blood platelets and can be detected within 6 h of poisoning and had a half-life of about 24 h. GFAP is an intermediate filament of skeleton astroglia and its level in body fluids is the key tool in detecting astrogliosis. 6 Although the role of astrogliosis in CNS injury is controversial, GFAP increases after reactive astrogliosis. 7
In their study, Yardan et al. 1 demonstrated increased serum S100B and NSE levels in CO poisoning, and this increase is associated with the loss of consciousness. In addition, they indicated that serum S100B and NSE levels may be useful markers in clinical status assessment of CO poisoning. 1 In another study, it was reported that serum S100B levels parallel the severity of cerebral response to CO poisoning and can be used as an early useful biochemical marker in deciding whether to apply hyperbaric oxygen therapy (HBOT). 2 In an experimental study, it was reported that GFAP level, an astrocytic marker, increases after prenatal exposure to CO and this is associated with severe toxicity. 3 However, in the literature, there is no study investigating the serum levels of GFAP in patients with acute CO poisoning.
In the diagnosis of CO poisoning, the carboxyhemoglobin (COHb) levels and anamnesis are measure. 1 The measurement of COHb levels which is helpful in the diagnosis is insufficient to determine the clinical features and prognosis. 1,2 Thus, new biochemical markers are needed to predict clinical symptoms and severity and prognosis. 8 For this purpose, the present study evaluated the possible use of biomarkers S100B protein, NSE, and GFAP known to be in the brain in patients with acute CO poisoning. The main purpose of this study was to assess the role of S100B protein, NSE, and GFAP in the evaluation of hypoxic BI in acute CO-poisoned patients.
Methods
Study design and setting
Fifty-five patients with acute CO poisoning who were referred to the emergency department (ED) of Ondokuz Mayıs University, Faculty of Medicine, were examined prospectively. After obtaining approval from local ethics committee, this prospective clinical study was conducted between November 2011 and November 2012. Written informed consent was obtained from patients who were conscious and relatives of patients who were unconscious.
Patients
Diagnosis of CO poisoning was established via medical history, clinical findings, and levels of COHb exceeding 3% (10% in smokers). Patients with a previous history of neuropsychiatric disorders were excluded. Patients’ demographic characteristics, symptoms and findings, poisoning sources, referral season, duration of exposure and loss of consciousness, initial Glasgow Coma Scale (GCS) score, and electrocardiography (ECG) findings, blood COHb, and troponin levels were recorded. Venous blood samples were obtained to determine serum S100B, NSE, and GFAP levels on admission and thereafter. The patients were divided into two groups—conscious and unconscious. The loss of consciousness was described as “the loss of consciousness at the scene of the accident,” “during the transfer to hospital,” and “on admission to ED.” The loss of consciousness at the scene of the accident was determined by obtaining anamnesis from patients’ relatives who witnessed the incident and the loss of consciousness during the transfer to hospital were determined by obtaining anamnesis from the ambulance personnel. The patients having loss of consciousness on admission to ED were objectively evaluated by the GCS. The loss of consciousness in the patients recruited in our study was determined by anamnesis in the prehospital period and by GCS on admission to ED. The control group consists of nonsmoking healthy volunteers without chronic illnesses or occupational exposure to CO. The control group was comparable with the patient group in terms of age and gender. The most common cause of CO poisoning was stove (78.2%). Also water heater, barbecue, exhaust, and fire were the other reasons (Table 1).
Characteristics of the patients (n = 55).

CO: carbon monoxide; ICU: intensive unit care.
Normobaric oxygen (NBO) therapy by face mask and other supportive treatments were applied to all patients in the ED. HBOT is not available in our hospital or nearby units; for this reason, all patients received NBO therapy.
Blood samples and measurements
The bloods samples were collected in test tubes. Whole blood was allowed to clot at room temperature for 30 min. The samples were then centrifuged at 3000g for 10 min at 4°C. Following centrifugation, the serum was removed and transferred into a clean tube. All samples were stored at −80°C until analysis. We stored the samples at 2–8°C for 24 h before working and helped them thaw automatically.
Human S100B was studied in accordance with the directions of the manufacturer (Cat. No. RD192090100R; BioVendor-Laboratorni medicina a.s., Brno, Czech Republic). The principle of the test is a sandwich enzyme immunoassay. The results were expressed as pg/ml. NSE levels were studied in accordance with the directions of the manufacturer (Ref. DKO073; DiaMetra, Milano, Italy). Human NSE concentrations in human serum were determined using the immunoenzymatic colorimetric method at 450 nm. The results were presented as ng/ml. GFAP levels were studied in accordance with the directions of the manufacturer (Cat. No. CSB-E08601h; Cusabio Biotech Co., Ltd., Wuhan, P. R. China). The principle of the test is a sandwich enzyme immunoassay. The results were presented as ng/ml.
Statistical analysis
Statistical analyses were performed using SPSS (version 21.0; SPSS, Chicago). For data analysis, the chi-square and Mann-Whitney U tests were used. The discrete variables were expressed as numbers and percentages and descriptive values as medians and interquartile ranges for variables with nonnormal distribution. A p value of <0.05 was considered statistically significant.
Results
Fifty-five patients were included in the study. Thirty-three (60%) were female and the mean age was 45.6 ± 20.3 (range, 19–92) years. All cases involved accidental poisoning. The mean initial COHb level was 19.74 ± 13.64%. The control group consisted of 25 healthy volunteers (16 female and 9 male). Demographic characteristics of the patients are shown in Table 1.
Stove was the major cause of poisoning (n = 43, 78.2%). Referrals to the hospitals occurred in winter between 22:00 and 10:00 h. In our study, in summer, there were no hospital referrals due to CO poisoning. Although 31 (56.4%) patients exhibited loss of consciousness at different levels, only 5 (9.1%) had loss of consciousness on admission. Neurological (N) symptoms other than loss of consciousness are headache, dizziness, weakness, and confusion. The most common symptoms in patients were syncope (52.7%), nausea (47.2%), dizziness (43.6%), and headache (41.8%). When the patients were analyzed with respect to the types of symptoms, concomitant neurologic and gastrointestinal symptoms (N + G) were the most common (30.9%), followed by isolated N symptoms (27.3%; Table 2).
Symptoms and clinical and laboratory findings of the patients, n (%).

GCS: Glascow coma scale; GIS: gastrointestinal system; ECG: electrocardiography; COHb: carboxyhemoglobin.
In all, 12 (21.8%) patients had increased troponin levels due to myocardial damage. All of the patients with myocardial injury underwent echocardiography (ECHO). Of the patients with myocardial damage, only one underwent coronary angiography with stent implantation. Except for one stent-implanted patient, ECHO findings of all patients were within normal limits. In the follow-up, decreases in the cardiac markers of these patients were observed. The great majority of patients (n = 37, 67.3%) were discharged from the hospital after follow-up in ED. Of the 16 (29.1%) patients with cardiopulmonary arrest, 2 (3.6%) died in the ED.
Serum S100B, NSE, and GFAP levels were higher in the patient group than in the control group. When the patient and control groups were compared with respect to serum S100B, NSE, and GFAP levels, the results obtained were significantly higher in the patient group (p < 0.05; Table 3).
Biochemical markers in the patient and control groups.a
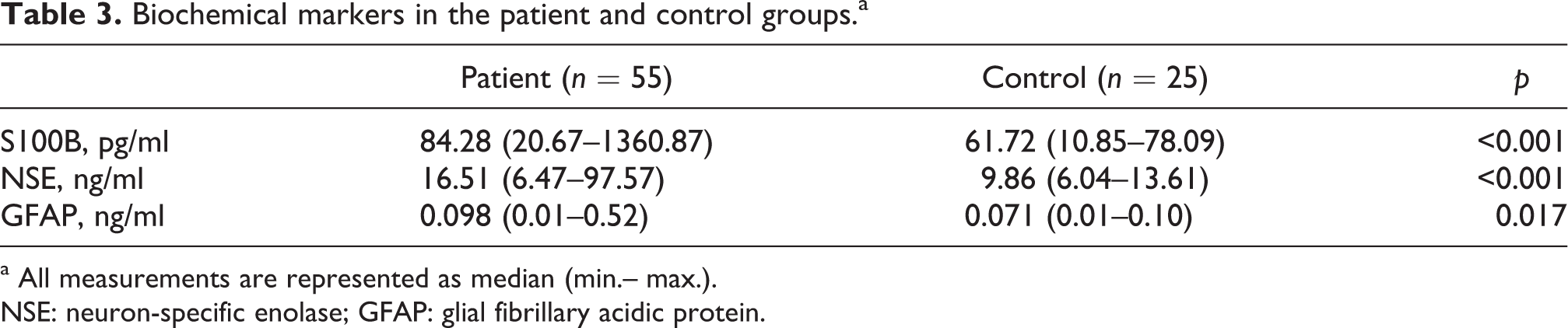
a All measurements are represented as median (min.– max.).
NSE: neuron-specific enolase; GFAP: glial fibrillary acidic protein.
While serum S100B and GFAP levels were higher in patients with loss of consciousness than in patients without loss of consciousness, NSE levels were lower. However, there was no statistically significant difference between the groups of unconscious and conscious patients in terms of serum levels of S100B, NSE, and GFAP (p = 0.993, p = 0.161, and p = 0.225, respectively). A statically significant difference was observed between the unconscious and control groups in terms of serum S100B, NSE, and GFAP levels (p < 0.05). While a statistically significant difference was observed between the conscious and control groups in terms of serum S100B and NSE levels (p < 0.05), GFAP levels were not statistically different (p > 0.05; Table 4; Figures 1, 2, 3).
Comparison of S100B, NSE, and GFAP levels in all study groups.a

aAll measurements are represented as median (min.–max.).
NSE: neuron-specific enolase; GFAP: glial fibrillary acidic protein.
b p < 0.001 compared to control group.
c p < 0.001 compared to control group.
d p < 0.001 compared to control group.
e p < 0.001 compared to control group.
f p = 0.001 compared to control group.

Levels of neuron-specific enolase (NSE) between unconscious, conscious, and control groups.

Levels of S100B between unconscious, conscious, and control groups.
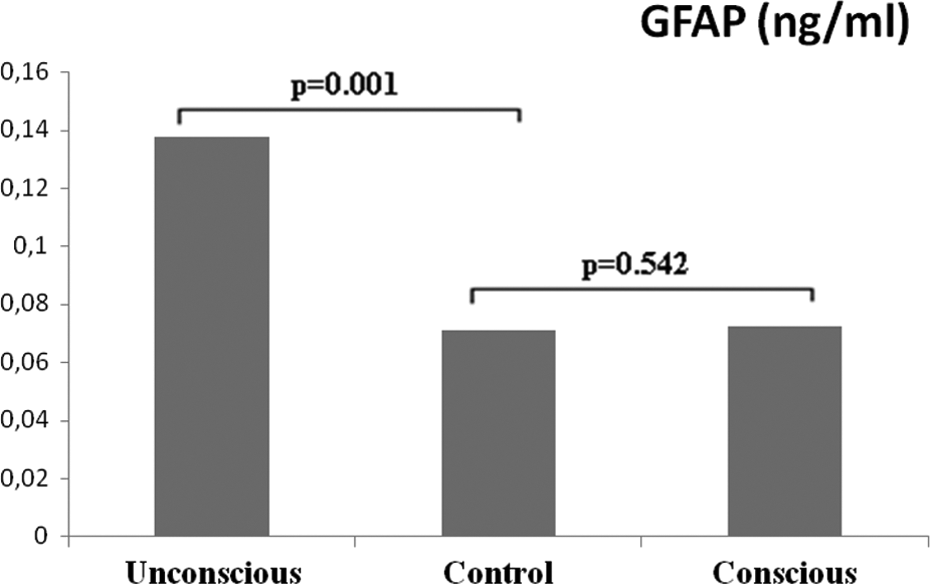
Levels of glial fibrillary acidic protein (GFAP) between unconscious, conscious, and control groups.
Serum NSE levels were higher in the high-troponin group than in the normal-troponin group. On the other hand, S100B and GFAP levels were lower. Intergroup comparisons were not statistically significant (p > 0.05; Table 5).
Biochemical markers in the high- and normal-troponin groups.a

a All measurements are represented as median (min.–max.)
NSE: neuron-specific enolase; GFAP, glial fibrillary acidic protein.
Serum S100B and NSE levels in the group with isolated gastrointestinal (G) symptoms and GFAP levels in the group with N + G symptoms were higher than those in other groups. Intergroup comparisons were statistically insignificant (p > 0.05).
When the patients were compared for serum S100B, NSE, and GFAP levels and duration of exposure, it was observed that serum S100B and NSE levels were higher in the 3- to 6-h exposure group, whereas GFAP levels were higher in the 6- to 12-h exposure group. Intergroup comparisons were not statistically significant (p > 0.05).
The duration of loss of consciousness was found to be longer in groups with high S100B and NSE levels, but there was no relation in the GFAP group. Intergroup comparisons were statistically insignificant (p > 0.05).
S100B, NSE, and GFAP levels were tested by the Shapiro-Wilk test. These marker levels did not show normal distribution (p < 0.001). As the data did not follow a normal distribution, the relationship between S100B, NSE, and GFAP levels were analyzed by the Spearman correlation. It yielded no significant correlation between S100B-NSE, S100B-GFAP, and NSE-GFAP (the correlation coefficients and p values were r = 0.245, r = −0.007, and r = −0.034 and p = 0.071, p = 0.960, and p = 0.806, respectively. There was no significant correlation between S100B, NSE, and GFAP and CoHb levels. The correlation coefficients and p values were r = 0.186, r = −0.007, and r = −0201 and p = 0.182, p = 0.958, and p = 0.149, respectively.
Discussion
Determination of serum COHb levels may be helpful in establishing a diagnosis in patients with suspected CO poisoning. However, COHb levels and the clinical findings may not be correlated. In addition, certain COHb levels do not suggest symptoms or clinical outcomes. 9 CO poisoning may result in inflammation of the brain parenchyma. 10 The lack of correlation between COHb levels and clinical findings suggest hypoxia-independent inflammation. 10,11 Therefore, even in the absence of cerebral hypoxia, CO-associated inflammation may be damaging. Low or moderate COHb levels trigger multiple biochemical cascades that may result in brain damage and morbidity. Therefore, in CO poisoning, new biochemical markers are needed in the determination of clinical severity and prognosis.
Clinical symptoms of CO poisoning are highly variable and nonspecific. Cakir et al. 2 reported that concomitant N + G symptoms were more common, whereas the presence of isolated N symptoms was less frequent. In our study, concomitant N + G symptoms were most frequent, followed by isolated N symptoms.
CO poisoning generally results from inhaling gases from any combustion reaction, car exhaust, or an improper chimney structure. 12,13 In different studies conducted in Turkey, stoves were found to be the most common cause of CO poisoning, with an incidence ranging from 33.3 to 61.1%, followed by water heaters and fires. 13 In our study, the most common source of CO was coal burning stoves (78.2%).
In a study conducted in Turkey, it was reported that CO poisoning is more common during winter, with an incidence ranging from 40.9 to 98.5%. 14 The great majority of the patients in our study group were referred to the hospital during winter. CO poisoning usually occurs while sleeping at night. 15 For this reason, CO is also called a silent killer. The great majority of our patients presented in the ED between 22:00 and 10:00, which is defined as a time of resting and sleeping. This may be associated with the use of the sleep mode in coal burning stoves at night to maintain in-house temperature.
Myocardial injury is one of the well-known major complications of CO poisoning. This may result from COHb-associated hypoxia or direct damage to myocardial cellular respiration and coronary arteries. 16 In the literature, there are studies on ECG changes, myocardial dysfunction, and myocardial infarction occurring due to CO poisoning. 16,17 In their study, Cha et al. 17 reported that 50 (20%) out of 250 patients with CO poisoning developed myocardial injury associated with poor prognosis. Cakir et al. 2 reported an increased cardiac biomarker results in 4 (13.3%) and sinus tachycardia in 8 (26.7%) patients of their study contents. In our study, we detected elevated troponin-I levels in 12 patients (21.8%), sinus tachycardia in 13 patients (23.6%), ischemic ECG changes in 12 patients (21.8%), and arrhythmia in 6 patients (10.9%).
In different studies, the association between S100B, NSE, and GFAP levels and prognosis was investigated in patients who developed brain damage due to different factors. 1 –3,8 S100B is a calcium-binding protein produced by astroglia in the brain and has been used as a marker of neuronal damage after brain trauma. 18 It is released into the peripheral circulation as a result of brain damage due to impairment in the blood–brain barrier, ischemia, increased intracranial pressure, and edema. 1,19 In some cases, S100B has been reported in serum even after a short period of anoxia. The low molecular weight and high solubility of S100B facilitate this condition. 1 Because the half-life of S100B is around 60 min on average, S100B exhibits a rapid and transient increase after BI. 8 NSE, whose function has not been clearly defined, is a soluble cytoplasmic protein released by neurons, neuroendocrine cells, and tumors. S100B and NSE reflect ischemic neuronal damage triggered by hypoxia through an indirect mechanism. The efficacy of S100B and NSE in demonstrating neuronal damage in cases such as head trauma, cerebral infarction, intracranial hemorrhage, space-occupying lesions, cardiac arrest, and cardiac surgery has been demonstrated in many studies. 1,2,20,21
GFAP is the major protein of glial intermediate filaments in differentiated astrocytes. 22 Its level in body fluids is an important tool in detecting astrogliosis. 6 The role of reactive reagent astrogliosis in CNS injury is controversial, but increased GFAP levels after reactive astrogliosis have been clearly observed. 7 In a study conducted by Lopez et al., 3 it was reported that GFAP levels increased after prenatal exposure to CO, and this was associated with severe toxicity. Increased GFAP immunoreactivity after prenatal exposure to CO suggests a glial-mediated response to the constant presence of CO. 3 In the literature, there is a very limited number of studies on GFAP in CO poisoning. In addition, no study in the literature has used these three biomarkers together.
In the present study, we evaluated the roles of S100B, NSE, and GFAP in hypoxic BI in CO-poisoned patients. In their study, Yardan et al. 1 reported that serum S100B and NSE levels were high in unconsciousness patients, whereas these levels were not high in patients with consciousness. Brvar et al. 19 reported high S100B levels in patients with CO poisoning and this was found in association with the degree of change in consciousness. Rasmussen et al. 23 reported a high S100B and NSE levels in 2 patients who died among the 20 patients with CO poisoning. They did not find a significant difference between patient and control groups regarding S100B, NSE levels, and they reported that there was not any correlation between serum biomarker levels and consciousness of their patients. Cakir et al. 2 found high S100B levels in all 8 patients with loss of consciousness, whereas they observed elevated NSE levels in only 3 patients. Our results are similar to those of Brvar et al. 19 and Cakir et al. 2 but different from those obtained in Rasmussen et al.’s study. 23 Our results are similar to those obtained by Yardan et al., 1 with respect to S100B levels but the NSE levels are different. This difference may be due to the hospital admission duration, sampling time, or different etiological factors leading to CO poisoning in our study group.
In this study, S100B and NSE levels were higher in the 3- to 6-h exposure group and GFAP levels were found to be significantly higher in the 6- to 12-h exposure group. The half-life of S100B varies between 30 min and 2 h. 5 NSE has a longer half-life of 24 h. 23 Therefore, time of sampling is extremely important. S100B can be used as a more appropriate marker in patients with CO poisoning whose blood samples are obtained and studied as early as possible. Due to its half-life and differences in the molecular structure, NSE may be used as a superior marker in patients with neurological damage. In our study group, GFAP levels were significantly higher in patients with loss of consciousness. In an experimental study, Ghorbani et al. 4 found significantly higher GFAP levels after exposure to CO in comparison to their control group. Lopez et al. 3 determined that prenatal exposure to CO increased the level of GFAP in association with severe toxicity.
In the present study, NSE levels in the group with high troponin levels were higher than the group with normal troponin levels. On the other hand, S100B and GFAP levels were significantly lower. Cakir et al. 2 compared the changes in cardiac markers with S100B and NSE levels and reported no significant difference. The results obtained from the study group may be associated with the time and number of samples. Due to the differences in the half-life of these biomarkers, the increase in S100B before troponin elevation in the early period and its decrease in the late period can be attributed to the increase in NSE levels. Since GFAP levels increase in the later period, elevated GFAP could not be detected in the period when troponin was studied.
Symptoms of CO poisoning generally occur when the COHb concentration exceeds 10%. 24 There is a weak correlation between clinical findings and blood COHb levels. 25 In our study, the S100B and NSE levels in the group with isolated G symptoms and the GFAP levels in the group with N + G symptoms were higher than those in other groups. This may be associated with the half-life of the biomarkers and the time of emergence and course of intoxication symptoms. G symptoms appearing in the early poisoning period are likely to become more prominent in shorter half-life biomarkers. On the other hand, N + G symptoms in ongoing CO exposure are likely to be associated with biomarkers with a longer half-life and the natural course of poisoning.
The most important limitation of our study is the small number of patients with CO poisoning. Prospective and controlled studies involving more patients with CO poisoning and evaluation of the presence of late neurological sequelae are needed.
In this study, all the patients except for 2—who were brought to the ED with cardiopulmonary arrest and died despite cardiopulmonary resuscitation—were discharged from the hospital with complete neurological recovery. In these 2 patients who died, the S100B and NSE levels were significantly higher than those in other patients. However, no correlation was detected between these 2 patients and the patients who were discharged with full neurological recovery with respect to GFAP levels. When the results of our study were evaluated in terms of loss of consciousness in CO-poisoned patients, although S100B and GFAP were higher in patients with loss of consciousness, there was no significant correlation between NSE and loss of consciousness. In their experimental study, Brvar et al. 26 reported that S100B is more valuable than the level of consciousness in assessing mortality.
Increased serum S100B, NSE, and GFAP levels are associated with CO poisoning. In CO poisoning, increased serum S100B and NSE levels may be associated with mortality. However, in CO-poisoned patients with loss of consciousness, serum S100B and GFAP levels also increase. Serum S100B, NSE, and GFAP levels can be employed as useful markers for predicting clinical severity, hypoxic brain damage, and mortality in patients with CO poisoning. Studying markers with blood samples obtained at the optimal time will contribute to the reliability of the results.
Footnotes
Acknowledgments
The authors thank Naci Murat for providing statistical advice.
Authors’ Note
This study was presented as an oral presentation at the 7th Asian Conference on Emergency Medicine (ACEM 2013), October 23-25, 2013, Tokyo, Japan.
Conflict of interest
The authors declared no conflicts of interest.
Funding
This study was supported by the Scientific Research Council of Ondokuz Mayis University with the number of PYO.TIP.1901.12.036.
