Abstract
Background
Neuron-specific enolase (NSE) is released into serum when nerve cells are damaged, and the levels thereof are used to determine neurological prognosis in patients who have suffered cardiac arrest or stroke. Delayed neuropsychiatric sequelae (DNS), a major complication of carbon monoxide poisoning (COP), can be caused by inflammatory response which is a mechanism of neuronal injury in cardiac arrest and stroke. NSE is known as a predictor of neurological prognosis in ischemic brain injury after cardiac arrest, and it is also reported as a predictor of DNS in acute COP. When serum NSE is measured serially in cardiac arrest patients, the best time to predict neurological prognosis is known at 48–72 h, but there are no studies analyzing serial serum NSE in acute COP. Thus, we explored whether serum NSE levels measured three times at 24 h intervals after COP predicted the development of DNS.
Methods
This prospective observational study was conducted on patients treated for COP from May 2018 to April 2020 in a tertiary care hospital in Korea. Neuron-specific enolase levels were assessed 24, 48, and 72 h after presentation at hospital. We used logistic regression to explore the association between NSE levels and DNS development.
Results
The NSE level was highest at 48 h, and the difference between the DNS group and the non-DNS group was greatest on the same time point. On multivariable logistic regression analysis, the NSE level at 48 h of >20.98 ng/mL (odds ratio [OR], 3.570; 95% confidence interval [CI], 1.412–9.026; P = .007) and the initial Glasgow Coma Scale (GCS) score of <9 (OR, 4.559; 95% CI, 1.658–0.12.540; P = .003) was statistically significant for DNS development.
Conclusion
Early identification of those who will experience DNS in acute COP patients is clinically important for deciding treatment. In this study, we revealed that NSE level of >20.98 ng/mL at 48 h time point can be used as an independent predictor of DNS (OR, 3.570; 95% CI, 1.412–9.026; P = .007; AUC, 0.648).
Introduction
Carbon monoxide (CO) is a colorless, tasteless, odorless toxic gas produced from incomplete combustion of carbon-based fuels and materials. 1 Carbon monoxide binds to hemoglobin with an affinity approximately 250-fold greater than that of oxygen. Even a low amount of inhaled CO can trigger hypoxia via the formation of carboxyhemoglobin (COHb), thus shifting the oxyhemoglobin dissociation curve to the left. 2 Due to these characteristics, carbon monoxide poisoning (COP) is one of the leading causes of mortality and morbidity from poisoning, with incidence and mortality rates of 137 and 4.6 per million worldwide, respectively.3,4 Even a small amount of CO can easily damage the brain, which is particularly vulnerable to hypoxia if oxygen-carrying capacity is reduced. 5 Also, CO can cause inflammation through multiple pathways which are independent to hypoxia, resulting in cardiac and neurologic injury. 6
Delayed neuropsychiatric sequelae (DNS) caused by COP are defined as a set of brain injuries that develop after a lucid interval of several days to 6 weeks after recovery from hypoxic injury. 7 Signs of DNS may include memory and movement disorders, psychosis, and Parkinsonism.6,8 The etiology of DNS remains incompletely understood, but it is assumed that the brain is injured by hypoxia with inflammatory reactions mediated by CO. 6 Because of these lethal properties of DNS, previous studies suggested factors such as Glasgow Coma Scale (GCS) score, S100B protein, lactic acid, and acute brain lesion on magnetic resonance imaging (MRI), as predictors.9–12
Neuron-specific enolase (NSE), one of the several isoenzymes of enolase, is a glycolytic enzyme; an isoform is found in the neuronal cytoplasm of the central nervous system. Also, NSE has attracted attention as an adjunct test for the small cell lung carcinoma, Creutzfeldt–Jakob disease and neuroendocrine tumors. 13 The enzyme is thought to be released from neuronal and glial tissues into the blood when cell membrane integrity is lost, so diagnosis or prognosis of brain damage can be predicted by measuring the serum NSE level. 14 Thus, in patients with ischemic brain injury developing after cardiac arrest, or ischemic stroke caused by ischemic–reperfusion injury and inflammation, the enzyme level serves as a marker for neurological damage.13–15 Delayed neuropsychiatric sequelae may occur not only by similar mechanism of cardiac arrest and stroke, but also by intracellular oxidative stress, impaired mitochondrial function, lipid peroxidation, and apoptosis which are caused by CO. 6 We thus hypothesized that the NSE level would predict DNS development. Previous studies on the relationship between the NSE level and DNS checked the enzyme levels only on initial presentation.10,16 When NSE levels were measured at 24 h intervals to predict neurological prognosis after cardiac arrest, the most useful value was measured at 48–72 h time point.17,18 Therefore, we explored whether any serum NSE level measured three times at 24 h intervals to 72 h after presentation at hospital, or any other clinical parameter, was prognostic of DNS.
Methods
Study design and setting
This prospective observational study was conducted using a CO registry that we established for all COP patients who visited the emergency department (ED) of a tertiary care hospital located in Gyeonggi-do, Korea; our hospital treats approximately 70,000 patients each year. This study was approved by the Institutional Review Board of Soonchunhyang University Hospital and permission was also granted by the Medical Ethics Committee.
Participant selection
All COP patients visiting our ED were enrolled in the CO registry. Patients who described appropriate histories of CO exposure, exhibited the expected signs on physical examination, and exhibited COHb levels >5% for non-smokers (COHb levels >10% for smokers) on presentation to the ED were thought to be suffering from COP. We enrolled all COP patients treated from May 2018 to April 2020. The exclusion criteria were age <18 years, the lack of serum NSE data due to unintentionally omitted blood sampling (more than one time), discharge against medical advice, loss to follow-up, persistent neurological symptoms on discharge, and presentation more than 24 h after CO exposure.
Clinical and laboratory assessments
Data on vital signs, demographic data, medical comorbidities, GCS scores, whether COP was accidental or intentional, accompanying symptoms, and laboratory results were collected. Venous blood was collected on presentation at our ED. And for determining changes in serial serum NSE levels, venous blood was also drawn three times at 24 h intervals from 24 h after presentation at the hospital. Serum NSE levels (reference range, 0–16.33 ng/mL) were determined using a Cobas 8000 analyzer (Roche Diagnosis, Indianapolis, IN, USA). All patients were scheduled for MRI within 2 days of entry into the ED.
DNS were defined as neurological abnormalities developing within 3 months after discharge and included symptoms such as depression, psychosis, urinary or fecal incontinence, disturbed consciousness, mutism, gait disturbance, memory disorder, and Parkinson-like syndrome.8,19 To evaluate cognitive ability, neurologist-administered Mini-mental state examination was performed and a score lower than 24 was defined as cognitive disorder. 20 And patient who have problem in recalling a word list was defined to have a memory disorder. 20 Psychiatric disease was defined by the criteria of the Diagnostic and Statistical Manual of Mental Disorders-5. And Parkinsonism was defined as having two or more of the symptoms like bradykinesia, resting tremor, impairment of postural reflexes, and rigidity. Neurological symptoms and signs were prospectively estimated at discharge. At 1 month after discharge, patients visited the ED and saw emergency physicians. Telephone interviews exploring DNS development were also conducted with all patients through cross-checking with their families at 2 weeks, 6 weeks, and 3 months after discharge. Patients and their families were also educated to revisit hospital if DNS symptoms were suspected to develop within 3 months after discharge. Re-evaluating DNS was based on additional examinations, including a mini-mental state examination, brain MRI, and consultations with neurologists and psychiatrists. Since some of the symptoms of DNS can be mistaken for psychiatric diseases, we checked if there was any history of psychiatric disease such as a depression by consulting a psychiatrist at the time of arrival at the ED or early in hospital admission. And, if patients complained of DNS suspicious symptoms within 3 months, the development of DNS was decided under the assessment of the same psychiatrist who had previously met the patient.
Management of carbon monoxide poisoning
All COP patients received 15 L of oxygen/min through a non-rebreather mask in the ED prior to hyperbaric oxygen (HBO) therapy. According to the Undersea and hyperbaric medical society indications, HBO therapy was delivered if the initial COHb was ≥25% (COHb ≥15% for pregnant women), if there was any history of loss of consciousness or neurological abnormalities (including decreased consciousness) regardless of COHb levels, and/or if acute cardiac injury was obvious. 21 Hyperbaric oxygen therapy was delivered within 30 min of arrival at the ED considering contraindications (three sessions at intervals of 6–12 h within a single day). The first session lasted for 150 min, with a target pressure of 3atmosphere absolute (ATA). The subsequent sessions lasted for 120 min at 2 ATA. 22 Compression was conducted over the first 30 min of each session and decompression from 30 min before the end of each session.
Statistical analysis
Data are expressed as medians with interquartile ranges (IQRs) for continuous variables and as absolute numbers or relative frequencies for categorical variables. Patients were categorized into two groups (non-DNS and DNS). Continuous variables that were normally distributed were compared using Student’s t test and those that were not normally distributed using the Mann–Whitney U test. Pearson’s χ2 test and Fisher’s exact test were used to compare categorical variables. To obtain the optimal cutoff value of the serum NSE level to predict DNS, the receiver operating characteristic (ROC) curve was determined for each 24 h serial serum NSE levels, respectively, and the results were obtained using the Youden index. The results showed area under curve (AUC), sensitivity (Se) and specificity (Sp). Multivariable logistic regression analysis was performed to identify predictive factors and a statistical significance of P-value <.05 was set for variables between the two groups in the univariable analysis and a factor divided by the cutoff value of serum NSE. Adjusted odds ratios (ORs) with 95% confidence intervals (CIs) were calculated using logistic regression analysis. And P-value <.05 was taken to indicate statistical significance. All statistical analyses were performed with the aid of SPSS for Windows software ver. 26.0 (IBM Corp., Armonk, NY, USA).
Results
From May 2018 to April 2020, a total of 236 patients visited our ED with COP. Of these, 49 were excluded for the following reasons: five were <18 years of age, seven lacked serum NSE data due to unintentionally omitted blood sampling, 12 were discharged against medical advice, 14 were lost to follow-up, seven exhibited persistent neurological symptoms at discharge, and four presented more than 24 h after COP (Figure 1). Therefore, 187 patients were finally included. Flow chart of the study subjects. Note: COP: carbon monoxide poisoning; NSE: neuron-specific enolase; DNS: delayed neuropsychiatric sequelae.
General characteristics of the study subjects.

Note: Values are expressed as median [interquartile range], or number (proportion).
Note: BMI: body mass index; GCS: Glasgow Coma Scale; CO: carbon monoxide; HBO: hyperbaric oxygen; DNS: delayed neuropsychiatric sequelae.
Comparison of clinical variables between the non-DNS and DNS groups.
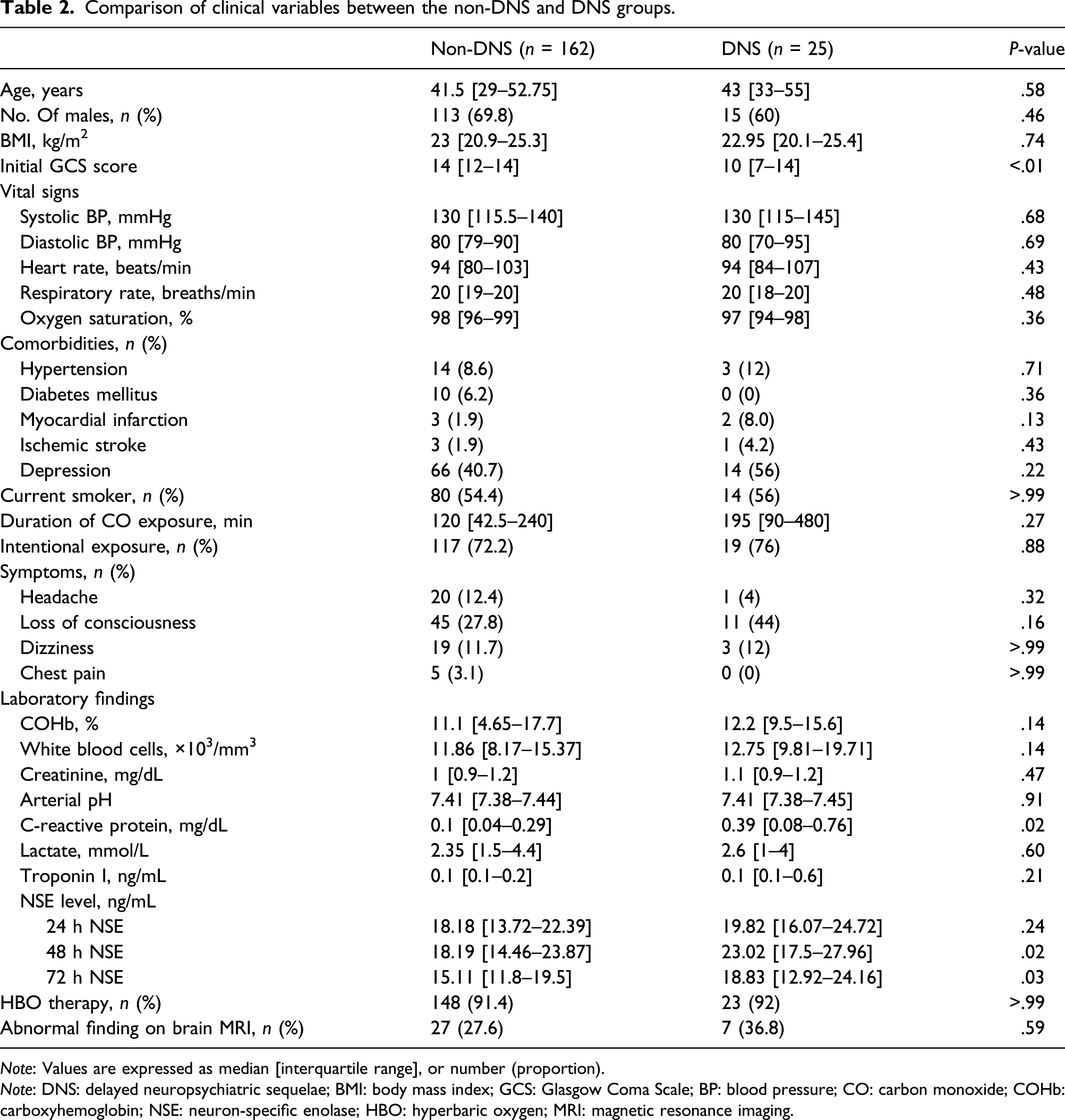
Note: Values are expressed as median [interquartile range], or number (proportion).
Note: DNS: delayed neuropsychiatric sequelae; BMI: body mass index; GCS: Glasgow Coma Scale; BP: blood pressure; CO: carbon monoxide; COHb: carboxyhemoglobin; NSE: neuron-specific enolase; HBO: hyperbaric oxygen; MRI: magnetic resonance imaging.

A line plot of neuron-specific enolase levels by neurological outcome. Note: DNS: delayed neuropsychiatric sequelae. Dots indicate median values.
The AUC were 0.574, 0.648 and 0.634 for each 24 h serial serum NSE levels, respectively. And the optimal cutoff values were 33.92 (Se 0.240, Sp 0.963), 20.98 (Se 0.680, Sp 0.660) and 16.79 ng/mL (Se 0.720, Sp 0.623) for each 24 h serial serum NSE levels, respectively (Figure 3). ROC curves for the serum NSE level and development of DNS following acute COP. 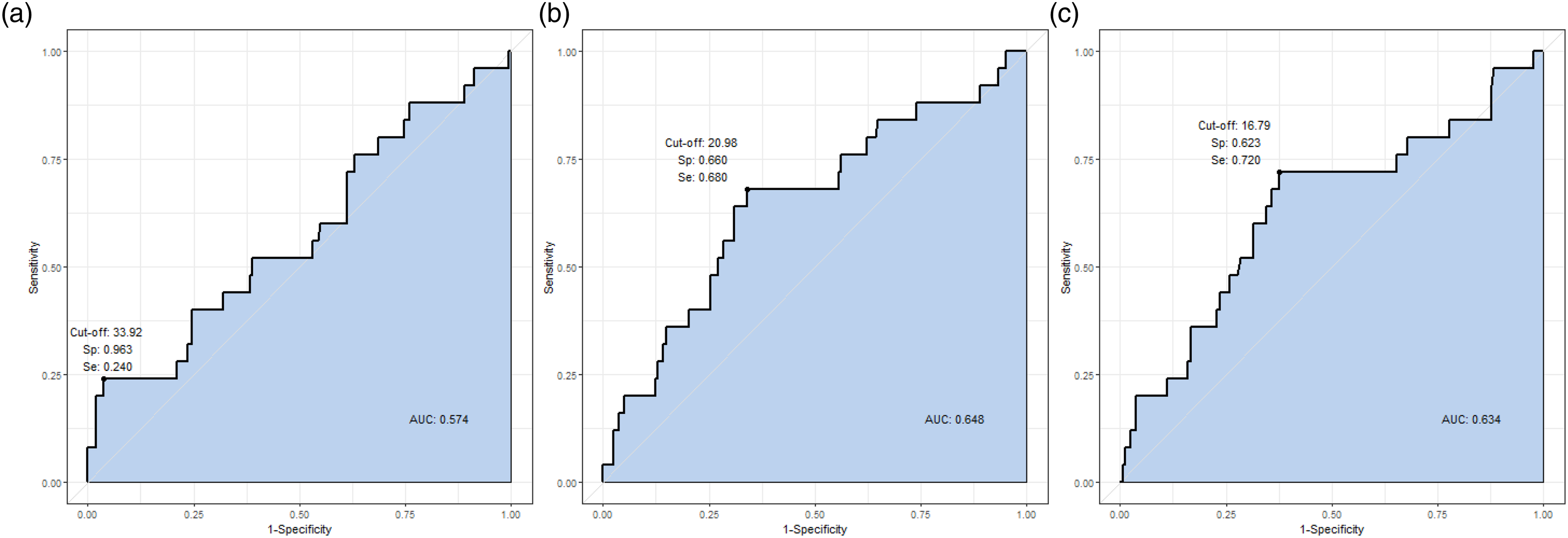
Multivariable logistic regression analysis of potential risk factors for DNS in patients with acute COP.

Note: OR: odds ratio; CI: confidence interval; GCS: Glasgow Coma Scale; NSE: neuron-specific enolase.
Discussion
We explored whether the serum NSE level, measured three times serially at 24 h intervals from 24 h after presentation, could predict DNS in patients with COP. From 24 h to 72 h time point, the NSE levels changed from 19.82 to 23.02 and 18.83 ng/mL in the DNS group and from 18.18 to 18.19 and 15.11 ng/mL in the non-DNS group. The levels and difference between the non-DNS and DNS groups were greatest at 48 h time point. Our study revealed that serum NSE level of >20.98 ng/mL at 48 h time point which was verified by ROC analysis can be used as an independent predictor of DNS (OR, 3.070). To the best of our knowledge, this is the first study to explore the relationship between 24 h serial serum NSE levels and DNS development.
Zhang et al. 23 found that both the GCS scores on site and in the ED independently predicted DNS. Ku et al. 24 also reported similar result that lower GCS scores could be a meaningful predictor for developing DNS. We found that the initial hospital GCS score was lower in the DNS group and predictive of DNS. A low GCS score reflects CO exposure for a rather long time, which causes severe brain injury. The cerebral hypoxia and inflammatory response induced by CO can injure brain cells and trigger decreased consciousness. 6 In our study, the rate of intentional exposure was 72.7%, which is more than the majority. Although the toxic co-ingestion rate was not obtained in our study, according to previous studies, toxic co-ingestion was relatively common in patients with intentional COP. 25 Also, patients who attempted suicide are usually uncooperative to the medical staff. 26 This can lead to clinical exaggeration of GCS scores which, while being statistically meaningful, could lead to bias in the study results.
Several studies have explored the relationship between COP and serum NSE levels. Yarden et al. found that unconscious patients with acute COP had higher serum NSE levels than did conscious patients. 9 We found that the GCS score was lower in the group with a high serum NSE level, suggesting that the neuronal injury caused by hypoxia and inflammation was sufficiently severe to cause a change in consciousness. Cha et al. 10 reported that the initial serum NSE measured in the ED served as an early predictor of DNS development. Cha et al. measured the serum NSE level only once at the initial presentation. However, according to previous studies, the best time to measure serum NSE for predicting neurological prognosis in ischemic brain injury after cardiac arrest was 48–72 h.17,18 Therefore, we measured the serial serum NSE level in acute COP patients to confirm which time point is the best to check the neurological prognosis, reflecting previous results. The etiology of DNS remains unclear, but brain hypoxia, lipid peroxidation, increased levels of reactive oxygen species, and the inflammatory response to COP can injure neuronal cells.6,27 In previous studies, the DNS group experienced more severe neuronal injury compared to the non-DNS group as evidenced by the higher serum NSE levels.10,28 In another study, the serum NSE level was elevated in children with hypoxic ischemic encephalopathy caused by COP. 29
In our study, the NSE level at 48 h after initial presentation was higher than that measured 24 h after presentation, and the difference between the DNS and non-DNS groups was also greatest at 48 h time point. An earlier study investigated the association between the NSE level and neurological prognosis after cardiac arrest; an elevated serum NSE level at 48 h after arrest predicted a poor outcome. 17 Although the pathophysiological mechanisms underlying NSE release when cerebral ischemia develops after COP or cardiac arrest remain unclear, several studies on focal cerebral ischemia indicated that the NSE release profile may reflect the several steps of ischemic brain injury evolution. The first peak may reflect rapid NSE release from initially injured tissue, followed by a second peak reflecting secondary brain injury caused by the inflammatory response that triggers neuronal cell death or persistent disturbance of the blood–brain barrier.30–32 The NSE release pattern after COP is thought to be similar; the fact that the NSE level was highest at 48 h time point might indicate that brain injury caused by inflammation (via multiple pathways) developed after initial ischemia. 6
Moon et al. reported that the serum NSE level at presentation was not associated with the risk of DNS in 236 patients with acute COP. However, only the initial NSE level was measured. 16 We sought to identify a time point at which serum NSE levels might differ markedly; we thus took three serial 24 h measurements. The 48 h time point values exhibited the greatest difference. Rasmussen et al. measured serum NSE levels at 12-h intervals in 20 COP patients. The level was approximately 10 ng/mL regardless of the COHb level and did not change over time. 33 However, the sample size was very small, perhaps explaining the absence of a significant relationship between the NSE level and DNS development.
Our work had several limitations. First, this was a single-center study; representativeness might be insufficient due to small sample size, so a larger multicenter study is required to obtain more reliable results. Second, although we investigated the relationship between serial NSE levels and DNS, we did not consider any biomarker other than NSE (such as the s100b protein, the level of which is predictsive of brain injury). Third, the cerebrospinal fluid NSE level is also elevated after neuronal cell injury. 9 However, we did not analyze cerebrospinal fluid. Fourth, the AUC values range between 0.574 and 0.648, which are of inadequate predictive value of DNS occurrence. Fifth, GCS score was also found to be a predictor of DNS in our study. However, most of the patients enrolled in our study were intentionally exposed to CO for suicide, which could lead to bias in clinical examination or GCS score related to sedating drugs, alcohol, or patients’ uncooperative attitude. But, unlike clinical examination, concentration of biomarker such as NSE is unlikely to be affected by other factors and is easy to assess blindly. A well-designed large-scale study will be helpful in validating the findings of this study.
Conclusion
In patient with acute COP, serum NSE level of >20.98 ng/mL at 48 h time point can be used as an independent predictor of DNS (OR, 3.570; 95% CI, 1.412–9.026; P = .007; AUC, 0.648). The serum NSE levels, measured three times at 24 h intervals from 24 h after presentation at the hospital, were highest at 48 h time point, and the difference in levels between the DNS and non-DNS groups was greatest at the same time point.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Soonchunhyang University Research Fund (grant no. 10210035).
Ethical approval
The study protocol was approved by the Institutional Review Board of Soonchunhyang university hospital and conducted in accordance with the provisions of the Declaration of Helsinki.
