Abstract
Puerarin, a potent free radicals scavenger, has been demonstrated to have protective efficacy in oxidative damage induced by nephrotoxins. In the present study, the attenuating effect of puerarin (PU) on lead (Pb)-induced apoptosis and oxidative stress was investigated in cultured primary rat proximal tubular (rPT) cells. Results showed that exposure to 0.5 µM Pb induced a decrease in cell viability accompanied with obvious cellular morphological alterations and caused an increase in apoptotic rate and apoptotic morphological changes. Simultaneously, depletion of mitochondrial membrane potential (ΔΨ) and intracellular glutathione (GSH); elevation of caspase-3 activity, intracellular reactive oxygen species, and malondialdehyde levels; and inhibition of GSH peroxidase (GSH-Px) activity were revealed in the cells exposed to Pb alone. However, simultaneous supplementation with PU (50 and 100 µM) protected rPT cells from Pb-induced cytotoxicity through inhibiting apoptosis, attenuating lipid peroxidation, renewing mitochondrial function, and elevating the intracellular antioxidants (nonenzymatic and enzymic) levels. In conclusion, these findings suggested that PU, as a widely distributed dietary antioxidant, contributes potentially to inhibition of Pb-induced cytotoxicity in rPT cells.
Introduction
Lead (Pb) is a known occupational hazard and an environmental pollutant. Unlike complex organic pollutants, Pb cannot be degraded by microorganisms; instead, it accumulates in ecosystems and enters the food chain for an extremely long biological half-life. The exposed population may get exposed to Pb due to food and water contamination, occupational exposure, and air pollution caused by industrial emission. 1 Long-term exposure to Pb exerts multiorgan toxic effects in mammals, and kidney is the critical target organ for Pb toxicity. 2,3 Pb nephrotoxicity has been extensively investigated by our research group, with growing evidence suggesting that oxidative stress and mitochondrial damage are among the fundamental molecular mechanisms of it. 4 –6 Moreover, apoptotic death formed in the presence of Pb could be responsible for its toxic effects. 7
In recent years, numerous studies explored the therapeutic properties of extracts from different parts of various medicinal plants. The positive effects of isoflavones on human health have been given much attention. Puerarin (C21H20O9, PU) is an isoflavone isolated from a traditional Chinese medicine ge-gen (Radix Puerariae). 8 PU is widely used in clinical treatment of myocardial infarction, cerebral ischemia, and diabetic nephropathy in China, which possesses a lot of activities including antioxidative activity, anti-inflammation, and antiapoptosis. 9,10 In addition, some evidence shows that PU can alleviate renal damage induced by nephrotoxins. 11 In view of these considerations, PU was chosen to evaluate its protective effect on Pb-induced cytotoxicity in this study.
However, concerning the protective effect of PU on Pb-induced nephrotoxicity, only few experiments concentrated on in vivo studies and primary cultures of tubular cells were minimally applied. 6 The reason is that primary cultures can better represent the live tissue than cultures of permanent cell lines; moreover, the renal tubule is one major site of Pb toxicity; 3 a very pure preparation of rat proximal tubular (rPT) cells was used for culture in this study. It has been confirmed that apoptotic cell death mediated by oxidative stress plays a chief role in renal damage induced by Pb exposure in vitro at lower exposure levels. 7 Hereby, a series of indices related to oxidative stress and apoptosis were chosen to elucidate the protective effect of PU on Pb-induced cytotoxicity in rPT cells. These results would provide a protective means against environmental Pb-induced renal damage and generate more comprehensive and reliable data for toxicological risk evaluation.
Materials and methods
Chemicals
All chemicals were of highest grade purity available. Fetal bovine serum (FBS) was purchased from Sijiqing Biological Engineering Material (Hangzhou, China). Malondialdehyde (MDA) assay kit and glutathione peroxidase (GSH-Px) assay kit were obtained from Keygen Biotech Co. Ltd (Nanjing, Jiangsu, China). Caspase-3 activity kit and Annexin V–fluorescein isothiocyanate (FITC) apoptosis detection kit were from Beyotime Institute of Biotechnology (Haimen, Jiangsu, China). Dulbecco’s modified Eagle medium–F12 (1:1), lead nitrate, 3-(4, 5-dimethylthiazol-2-yl)-2, 5-diphenyl- tetrazolium bromide (MTT), 5′-chloromethylfluorescein diacetate (CMF-DA), 2′,7′-dichlorofluorescein diacetate (DCFH-DA), Hoechst 33258 staining, antibiotic–antimycotic solution, rhodamine 123 (Rh 123), collagenase, trypsin, PU, ethylenediaminetetraacetic acid (EDTA)/ethylene glycol tetraacetic acid (EGTA) and all other chemicals were purchased from Sigma-Aldrich (St Louis, Missouri, USA).
Cell culture and Pb treatment
Isolation, identification, and culture of rPT cells have been described in our previous study. 7 Cell purity of the isolated proximal tubular cells was routinely higher than 95%. The primary cells were subcultured by the trypsin–EDTA digestive method. The first passage was used to perform the experimental design when cultured for 120 h, which was in its highest cell viability (according to the growth curve, data not shown). Based on the doses of Pb in our previous study, 12 0.5 µM Pb were applied in this study. Regarding the optimal concentration of PU chosen for this experiment, cells were treated with a range of PU concentrations (25, 50, 100, and 200 μM) and/or 0.5 μM Pb for 12 h (Figure 1), and cell viabilities were tested using MTT assay. The stock solution of lead nitrate and PU was dissolved in sterile ultrapure water and methanol, respectively. The concentration of methanol in the medium was ≤0.1%. Based on an initial screening, cell cultures undergoing exponential growth were incubated with PU (50 and 100 μM) and/or 0.5 μM Pb, in a serum-free medium at 37°C for 12 h. After 12 h of treatment, cell culture photos were taken under phase-control microscopy (Olympus, Tokyo, Japan) to check cell morphology.
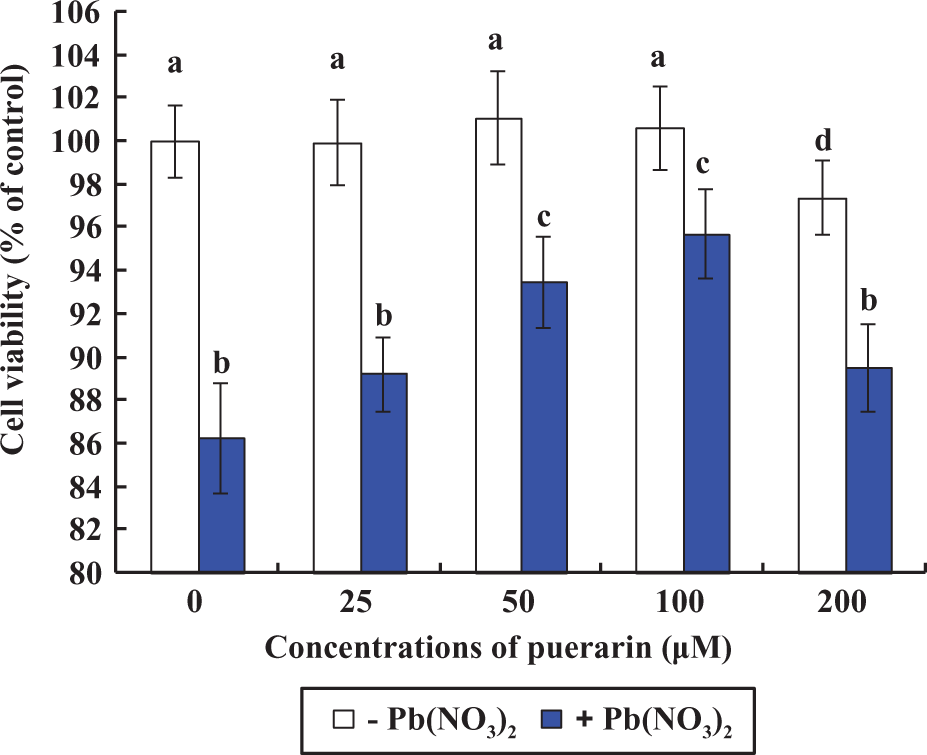
Effects of Pb and/or PU on cell viabilities in rPT cells after 12 h exposure. The cells were incubated with a range of PU concentrations (0, 25, 50, 100, and 200 µM) and/or 0.5 µM Pb for 12 h to determine the cell survival. Two different colors were chosen to point out which cells were treated with Pb(NO3)2 (+) and which were not (–); that is, white columns for no Pb treatment and blue for Pb treatment. Data are presented as mean ± SD (n = 6). Bars with different superscripts are statistically different (p < 0.05). Pb: lead; PU: puerarin; rPT: rat proximal tubular.
Hoechst 33258 staining
Morphology of apoptotic cell nuclei was detected by staining with the DNA-binding fluorochrome Hoechst 33258 stain (bisbenzimide). rPT cells were seeded on sterile cover glasses placed in the 24-well plates. After various treatments (0 and 0.5 µM Pb, 50 µM PU + 0.5 µM Pb, and 100 µM PU + 0.5 µM Pb) for 12 h, cells were washed with phosphate-buffered saline (PBS) and fixed with 4% paraformaldehyde for 10 min and then incubated with Hoechst 33258 staining solution (50 µM in PBS) for 10 min in the dark at room temperature. After three washes with PBS, the cells were viewed under an inverted fluorescence microscope (Olympus) at an excitation wavelength of 352 nm through FITC filter (blue fluorescence). For assessing the extent of the apoptosis induced by Pb and/or PU, 200 cells were randomly selected to count those apoptotic cells within every batch of experiment, each one performed in triplicate.
Flow cytometric analysis
All the following assays were carried out on fluorescence-activated cell sorter Aria flow cytometer (BD Biosciences, Franklin Lakes, New Jersey, USA) and the 488-nm laser was utilized. Cells were seeded at a density of 3 × 105 in 6-well plates and treated with Pb and/or PU (0 and 0.5 µM Pb, 50 µM PU + 0.5 µM Pb, 100 µM PU + 0.5 µM Pb) for 12 h. After the treatment, the adherent cells were collected with trypsin–EDTA solution (1.25 g/L trypsin and 0.2 g/L EDTA). The detached and adherent cells were pooled and harvested by centrifugation at 1500 r/min for 5 min. The harvested cells (at least 1 × 106 cells per group) were washed twice with PBS and incubated with various fluorescent dyes for the flow cytometric analysis. Cell debris characterized by a low forward/side scattering was excluded from analysis. The data were analyzed by Cell Quest software (Becton-Dickinson, NJ, USA), and mean fluorescence intensity was obtained by histogram statistics.
Quantitation of apoptosis
Detection of apoptotic cell distribution was performed using Annexin V/propidium iodide (PI) staining assay. The amount of early apoptotic cells, late apoptotic cells, living cells, and necrotic cells was determined as the percentage of Annexin V+/PI−, Annexin V+/PI+, Annexin V−/PI−, and Annexin V−/PI+ cells, respectively. 13 Therefore, the total apoptotic proportion included the percentage of cells with fluorescence Annexin V+/PI− and Annexin V+/PI+. After the exposure (12 h), the harvested cells were labeled with Annexin V–FITC and PI using an apoptosis detection kit according to the manufacturer’s protocol. The FITC and PI fluorescence was measured through FL-1 filter (530 nm) and FL-2 filter (585 nm), respectively, and 10,000 cells were analyzed.
ROS measurement
Intracellular reactive oxygen species (ROS) generation was detected using a well-characterized probe DCFH-DA. Following the treatment described above, 1.5 × 106 harvested cells per milliliter was incubated with DCFH-DA (100 µM final concentration) for 30 min in the dark at 37°C. The incubated cells were harvested and suspended in PBS, and ROS generation was measured by the fluorescence intensity (FL-1, 530 nm) of 10,000 cells on flow cytometer.
Mitochondrial ΔΨ assay
For the detection of mitochondrial membrane potential (ΔΨ
GSH measurement
The cellular level of GSH was monitored by CMF-DA. This fluorescent probe reacts with intracellular sulfhydryls and reflects predominantly GSH. 14 The harvested cells were incubated with CMF-DA (1 µM final concentration) for 30 min in the dark at 37°C. The incubated cells were harvested, suspended in PBS, and GSH was measured by the fluorescence intensity (FL-1, 530 nm) of 10,000 cells. 15
Caspase-3 activity assay
Caspase-3 activity was measured using the colorimetric caspase-3 assay kit as described previously by Qu et al. 16 The detailed analysis procedure is described in the manufacturer’s protocol. Caspase-3 activity was expressed as the change in enzyme activity relative to control cultures.
Measurement of intracellular GSH-Px activity and lipid oxidation (MDA)
Cells were seeded at a density of approximately 6–8 × 105 in 4-well plates, exposed to various treatments (0 and 0.5 µM Pb, 50 µM PU + 0.5 µM Pb, and 100 µM PU + 0.5 µM Pb) for 12 h. After the treatment, the detached and adherent cells were pooled and harvested by centrifugation. The protein concentrations of the samples were determined by the method described by Bradford 17 to normalize the GSH-Px activity and MDA level.
The harvested cells were lyzed in ice-cold physiological saline by sonication followed by centrifugation at 15,000g for 5 min at 4°C. The resulting supernatants were used immediately for measuring the activity of GSH-Px and MDA level. The activity of GSH-Px was assessed according to the kit’s instruction which makes use of the reaction: H2O2 + 2GSH → 2H2O + GSSG (oxidized glutathione). The absorbance was determined at 412 nm and the enzyme activity was expressed in micromoles per milligram protein. The quantification of MDA was based on measuring formation of thiobarbituric acid reactive substances according to the manufacturer’s protocol. The reaction mixture was incubated at 95°C for 40 min. After cooling, the chromogen was read spectrophotometrically at 532 nm and the MDA level was expressed in nanomoles per milligram protein.
Statistical analysis
Data are presented as mean ± SD from at least three independent experiments with different batches of cells, each one performed in duplicate or triplicate. Statistical comparisons were made using one-way analysis of variance (Scheffe’s F test) after ascertaining the homogeneity of variance between the treatments. All statistical analyses were carried out using Statistical package for Social Sciences version 13.0 (SPSS, Chicago, Illinois, USA). The critical value for statistical significance was p < 0.05.
Results
Protective effect of PU on cellular death induced by Pb
Compared with the control, cell viability manifested no significant change after treatment with 25–100 μM PU alone for 12 h (p > 0.05). However, treatment with 200 μM PU alone resulted in a significant decrease in cell viability (p < 0.05; Figure 1). In addition, incubation with 50 and 100 μM PU exhibited significant inhibition of cellular death against 0.5 µM Pb (p < 0.05). Hereby, 50 and 100 μM PU were chosen to evaluate its protective effect on cellular death induced by Pb exposure. As shown in Figure 2, morphological investigation associated with the protective effect of PU against Pb toxicity was assessed qualitatively using phase-contrast microscopy. Compared with the control, the Pb-treated group displayed low density, shrinkage, vacuolated cells, and rounding. However, the morphologic changes of cells were reversed in two combined groups (50 μM PU + 0.5 μM Pb and 100 μM PU + 0.5 μM Pb), demonstrating the significant protective effect of PU on Pb toxicity.

Effect of treatment with PU on Pb-induced morphological changes in rPT cells observed under phase-contrast microscopy. Cell morphological changes are marked in the figures. Representative images, magnification ×200. The groups shown are (a) control; (b) 0.5 µM Pb; (c) 50 µM PU + 0.5 µM Pb; and (d) 100 µM PU + 0.5 µM Pb. Pb: lead; PU: puerarin; rPT: rat proximal tubular.
Effect of PU on Pb-induced apoptosis
Because apoptosis plays an important role in Pb-induced cellular death in rPT cells, experiments were performed to explore whether PU mediated cytoprotective effect via apoptosis. Annexin V/PI staining was applied to determine apoptotic cells after 12 h exposure. Figure 3(b) shows that the number of apoptotic cells (Annexin V+/PI−(early) + Annexin V+/PI+(late)) enhanced significantly (p < 0.01), being 6.19-fold of that of the control after exposure to 0.5 μM Pb; while the apoptosis was significantly prevented by PU (p < 0.01), being 2.85-fold and 2.32-fold of that of the control after exposure to 50 μM PU + 0.5 μM Pb and 100 μM PU + 0.5 μM Pb, respectively.
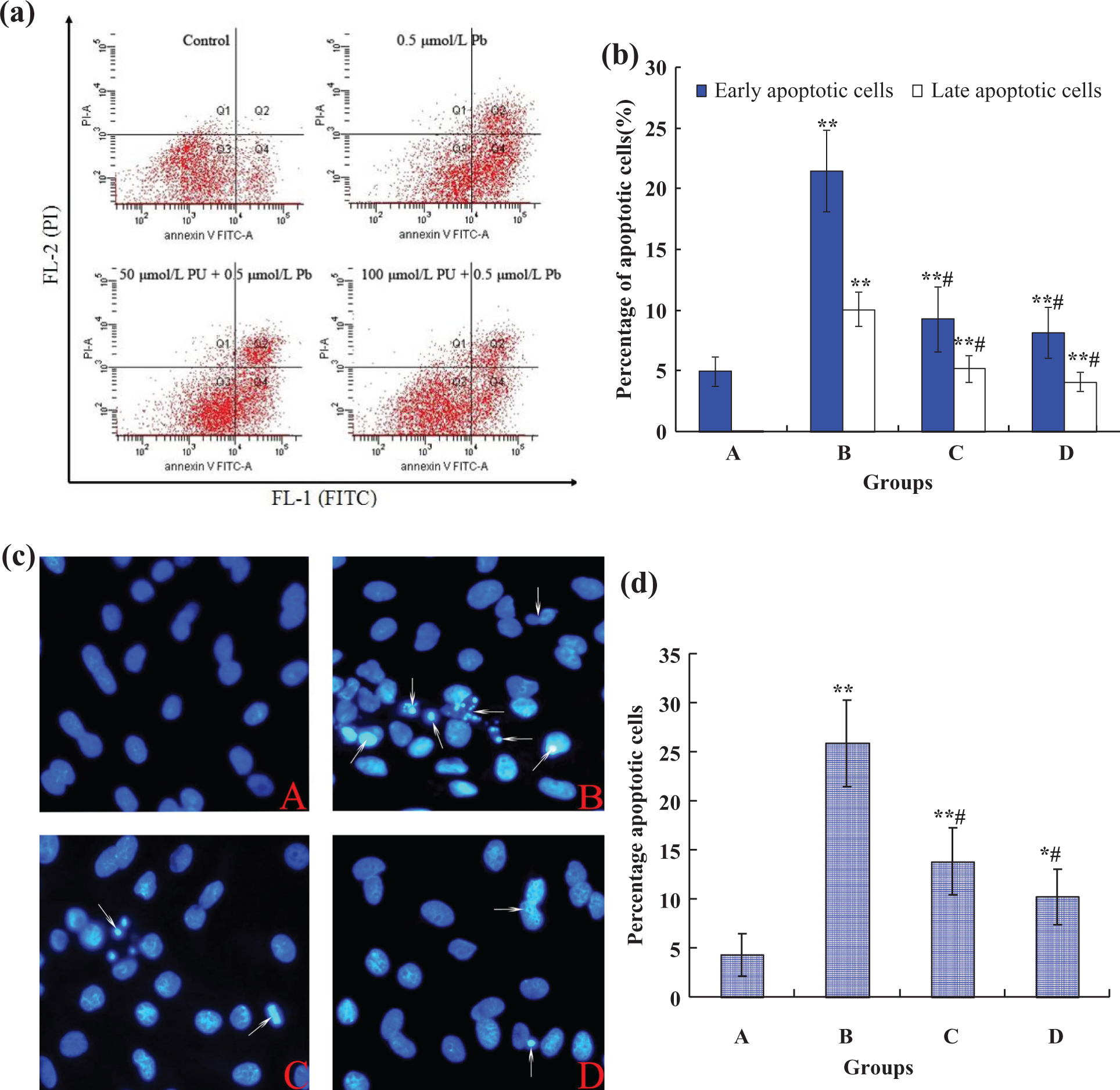
Effects of Pb and/or PU on apoptosis (a, b) and apoptotic morphological changes (c, d) in rPT cells, measured by flow cytometry and fluorescence microscopy. The groups shown are (a) control; (b) 0.5 µM Pb; (c) 50 µM PU + 0.5 µM Pb; and (d) 100 µM PU + 0.5 µM Pb. *p < 0.05; **p < 0.01: compared with control, #p < 0.01: statistical significance between cells cultured in single Pb treatment (group B) and presence of PU (groups C and D). (a) Cells were treated with 0.5 µM Pb and/or PU (50 and 100 μM) for 12 h and then processed for Annexin V/PI staining and analyzed by flow cytometry. (b) The percentage of early apoptotic cells and late apoptotic cells. Results are mean ± SD of three separate experiments and each one performed in triplicate. (c) Cells were incubated with PU (50 and 100 μM) and/or 0.5 µM Pb for 12 h and nuclear chromatin changes (apoptosis) were assessed by Hoechst 33258 staining; changes in nuclei fragmentation with condensed chromatin are evident (thin arrows); A–D: original magnification ×400. (d) The statistical results of apoptotic cells obtained from counting are expressed as mean ± SD of three separate experiments. Pb: lead; PU: puerarin; rPT: rat proximal tubular.
Effect of PU on Pb-induced apoptotic morphological changes
Apoptotic morphological changes in the nuclear chromatin were detected by staining with the DNA-binding fluorochrome Hoechst 33258 (Figure 3(c)). In control group, the majority of cells appeared normal with uniformly stained nuclei and the chromatin of normal nuclei was unaltered and spread uniformly throughout the entire nucleus. Whereas Pb-treated cells exhibited morphological changes typical of apoptosis, that is, fragmented chromatin was characterized by a scattered, drop-like structure, and condensed chromatin was located at the periphery of the nuclear membrane. The nuclei of apoptotic cells appeared smaller and shrunken compared with intact cells. However, combined treatment with PU (50 and 100 μM) restored all these parameters to similar levels with that of the control (Figure 3(c)). In addition, the statistical result (Figure 3(d)) of cells with apoptotic characteristics shows that the number of apoptotic cells enhanced significantly when exposed to Pb alone. Regarding the apoptosis, two combined groups (50 μM PU + 0.5 μM Pb and 100 μM PU + 0.5 μM Pb) caused less lesion (p < 0.01) than the Pb treatment alone, respectively.
PU inhibited the loss of ΔΨ and caspase-3 activation in Pb-treated cells
Loss of ΔΨ is an early event in apoptosis. Thus, the impact of Pb and/or PU on ΔΨ in rPT cells was determined by flow cytometry using Rh 123 fluorescence dye. As shown in Figure 4(a), a significant decrease in mitochondrial ΔΨ occurred in rPT cells after exposure to 0.5 μM Pb for 12 h (p < 0.01). Likewise, PU (50 and 100 μM) exhibited a significant (p < 0.01) increase in the loss of ΔΨ induced by Pb exposure. During activation of mitochondrial dysfunction, a number of signals can cause the activation of caspase-3. Therefore, the influence of PU on Pb-induced caspase-3 activation was examined (Figure 4(b)) by a spectrophotometric assay in cell lysates from control cultures, Pb-treated cultures, and cultures treated with Pb + PU. rPT cells treated with 0.5 μM Pb for 12 h showed a significant increase in caspase-3 activity (p < 0.01). In contrast, co-incubuation with PU (50 and 100 μM) significantly inhibited the Pb-induced activation of caspase-3 and 100 μM PU can completely inhibit its activation (p < 0.01), suggesting that PU reduced Pb-induced apoptosis by blocking caspase-3-dependent apoptosis.

Effects of Pb and/or PU on mitochondrial membrane potential (a) and caspase-3 activity (b) in rPT cells. (a) Cells were treated with 0.5 µM Pb and/or PU (50 and 100 μM) for 12 h. Rh 123 was added, and the harvested cells were incubated for 30 min. The fluorescence was measured using flow cytometer with FL-1 filter. Fluorescence results were expressed as mean fluorescence intensity. (b) Caspase-3 activity was detected using a commercial kit as described in the Materials and Methods section. Data are presented as fold-change from the control group. The groups shown are (a) control; (b) 0.5 µM Pb; (c) 50 µM PU + 0.5 µM Pb; (d) 100 µM PU + 0.5 µM Pb. Each bar represents mean ± SD (n = 6). **p < 0.01: compared with control, #p < 0.01: statistical significance between cells cultured in single Pb treatment (group B) and presence of PU (groups C and D). Pb: lead; PU: puerarin; rPT: rat proximal tubular; Rh 123: rhodamine 123.
PU suppressed ROS generation and intracellular MDA levels in Pb-treated cells
The generation of intracellular ROS was detected by the fluorescent probe DCFH-DA. As shown in Figure 5(a), the level of ROS was increased by the Pb treatment alone, being 3.20-fold of that of the control after exposure to 0.5 µM Pb. Exposure to the combined groups led to a significant (p < 0.01) decrease in ROS generation compared with the single Pb exposure, being 1.74-fold and 1.38-fold of that of the control after exposure to 50 μM PU + 0.5 μM Pb and 100 μM PU + 0.5 μM Pb, respectively. Simultaneously, the level of intracellular MDA (a common end product of lipid peroxidation) was measured to study the protective effect of PU on Pb-induced oxidative lesions (Figure 5(b)). Single Pb exposure resulted in the significant generation of MDA levels in rPT cells, compared with the control group. When PU (50 and 100 μM) was coadministered with Pb for 12 h, it showed that Pb-induced intracellular MDA levels could be significantly (p < 0.01) attenuated by PU.
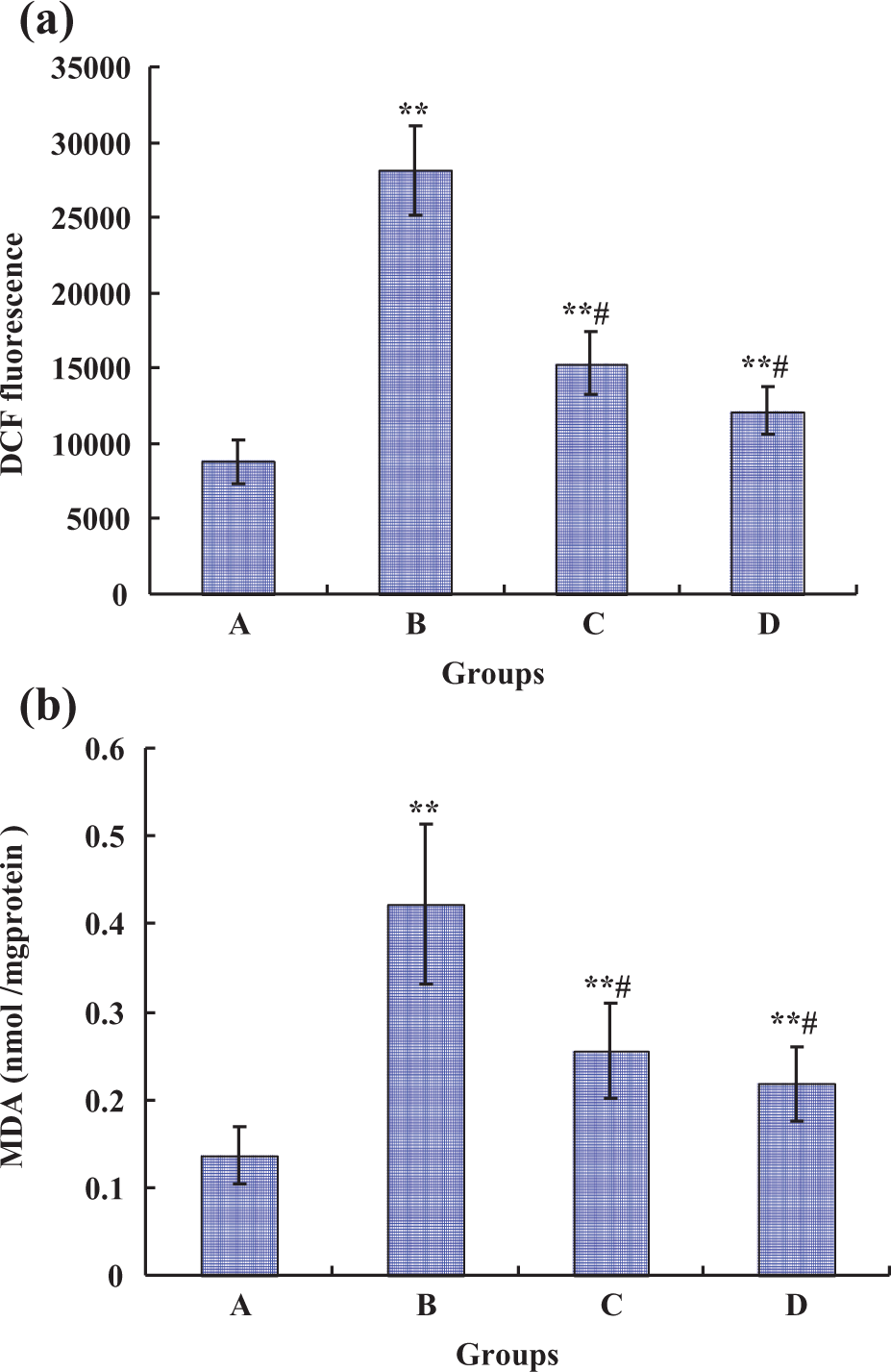
Effects of Pb and/or PU on generation of ROS (a) and intracellular MDA levels (b) in rPT cells. Cells were treated with 0.5 µM Pb and/or PU (50 and 100 μM) for 12 h. Then, the cells were collected. (a) The harvested cells were incubated with 100 µM DCFH-DA for 30 min at 37°C. DCF fluorescence was measured using flow cytometer with FL-1 filter. Fluorescence results were expressed as mean fluorescence intensity. Each bar represents mean ± SD (n = 6). (b) The harvested cells were used to detect the MDA levels using a commercial kit. Data are mean ± SD of three experiments from cells of different cultures, each one performed in triplicate. **p < 0.01: compared with control, #p < 0.01: statistical significance between cells cultured in single Pb treatment (group B) and presence of PU (groups C and D). Pb: lead; PU: puerarin; rPT: rat proximal tubular; DCFH-DA: dichlorofluorescein diacetate; DCF: dichlorofluorescein; MDA: malondialdehyde; ROS: reactive oxygen species.
PU administration restored the antioxidant levels in Pb-treated cells
The GSH level and GSH-Px activity were determined to reflect intracellular antioxidant status. After incubation with 0.5 µM Pb for 12 h, a significant (p < 0.01) decrease in the level of nonenzymic antioxidant (GSH) and significantly decreased activity of enzymic antioxidant (GSH-Px; p < 0.01) were observed in Figure 6, compared with the control group. However, compared with single Pb exposure, the GSH level and GSH-Px activity displayed a significant increase in the presence of PU (p < 0.01), especially supplementation with 100 μM PU completely restored the intracellular antioxidant levels in rPT cells.

Effects of Pb and/or PU on intracellular GSH levels (a) and GSH-Px activity (b) in rPT cells. Cells were treated with 0.5 µM Pb and/or PU (50 and 100 μM) for 12 h. Then, the cells were collected. (a) The harvested cells were incubated with 1 µM CMF-DA for 30 min in dark at 37°C. CMF fluorescence was measured using flow cytometer with FL-1 filter. Fluorescence results were expressed as mean fluorescence intensity. Each bar represents mean ± SD (n = 6). (b) The collected cells were used to determine the intracellular GSH-Px activity. Data are mean ± SD of three experiments from cells of different cultures, each one performed in triplicate. **p < 0.01: compared with control, #p < 0.01: statistical significance between cells cultured in single Pb treatment (group B) and presence of PU (groups C and D). Pb: lead; PU: puerarin; rPT: rat proximal tubular; GSH: glutathione; GSH-Px: glutathione peroxidase; CMF-DA: 5′-chloromethylfluorescein diacetate
Discussion
Pb, a potent nephrotoxic toxicant, causes damage to the proximal tubular epithelium. 18 Now many investigations concentrated on the therapeutic effects of some extracts from medicinal plants on Pb toxicity. As a widely distributed dietary antioxidant, little is known about the protective action of PU on Pb-induced cytotoxicity in vitro, although it contributes potentially to prevent Pb toxicity in vivo. 6,10,19 –21 Several epidemiologic studies found a higher prevalence of chronic kidney disease at blood Pb levels of 10 µg/dL (0.48 µM) to 80 µg/dL (3.85 µM). 1,22,23 Based on this, combined with our previous results, 7 0.5 µM Pb was chosen in this study. In addition, 50 and 100 µM PU were selected to be the optimal concentrations used in this study through an initial screening (Figure 1). Herein, this study was designed to investigate the protective effect of PU on Pb-induced cytotoxicity in cultured primary rPT cells and events of 12 h exposure were chosen primarily.
As shown in Figure 1, 0.5 μM Pb markedly decreased the cell viability of rPT cells, which was further demonstrated by cell morphology observation, that is, lower density, rounded cells, and vacuolar (Figure 2). It showed that the cell death was significantly prevented by PU, that is, significantly increased cell viabilities and altered cell morphology, whereas no evidence of cytotoxicity of PU was found at the dose of 50–100 μM. Our previous investigation demonstrated that apoptosis played a chief role in the cellular death induced by low-dose Pb (0.25–1.0 µM) exposure and oxidative stress is implicated in the apoptotic death of rPT cells mediated by Pb exposure. 7 Thus, indices related to oxidative stress and apoptosis were chosen to investigate the protective effect of antioxidant PU on apoptotic death induced by Pb exposure in this study.
Concerning the apoptotic rates measured by flow cytometry, the apoptosis induced by Pb was prevented by PU significantly (Figure 3(a) and (b)). Also, apoptotic morphological changes mediated by Pb exposure, assessed by Hoechst 33258 staining, could be obviously reversed by simultaneous supplementation with antioxidant PU (50 and 100 µM; Figure 3(c)), which further shows that the antagonistic effect of PU on Pb-induced apoptotic death in rPT cells. The change tendency in the percent of apoptotic cells (Figure 3(d)) is consistent with the result obtained by flow cytometric analysis (Figure 3(b)). However, PU (50 and 100 µM) alone caused no significant change in apoptotic rates and apoptotic morphological changes in rPT cells compared with the control (data not shown), which indicated that PU represents a low-toxic antioxidant and may play a role in attenuating the Pb-induced cytotoxicity.
It is well known that mitochondria play a central role in the execution of apoptosis via disruption of the mitochondrial ΔΨ and activation of effector caspase 3. 16,24,25 Loss of mitochondrial ΔΨ renders the mitochondrial membrane permeable, leading to release of pro-apoptotic factors and further ROS production. 25,26 Caspase 3 is a major executioner protease, capable of cleaving or degrading many key proteins and responsible for initiating in the apoptotic program. 16 As shown in Figure 4, Pb exposure produced a breakdown of mitochondrial ΔΨ and activation of caspase 3 in rPT cells, which indicated that mitochondrial dysfunction plays a key role in apoptosis induced by Pb exposure. However, PU (50 and 100 µM) could significantly renew mitochondrial dysfunction, which contributed to inhibiting Pb-induced apoptosis.
Meanwhile, mitochondria are the major source of intracellular ROS generation, and mitochondrial function is highly sensitive to disruption by excess ROS. 27 Lipid peroxidation is involved in mitochondrial dysfunction, and alterations in mitochondrial function are largely attributed to the damaging effects of ROS. 28 Moreover, several studies confirmed that Pb-induced nephrotoxicity was associated with oxidative stress that included lipid peroxidation and production of ROS. 6,7 In order to determine whether PU can attenuate lipid peroxidation to renew mitochondrial dysfunction, intracellular ROS and MDA levels (markers of oxidative stress) were assessed. Results from our study (Figure 5) are in accordance with these reports, demonstrating further that oxidative stress played a critical role in the cytotoxicity induced by Pb. However, simultaneous supplementation of Pb and antioxidant PU (50 and 100 µM) significantly alleviated the production of ROS as well as the decreased generation of intracellular MDA levels. Therefore, we can see that PU could renew mitochondrial dysfunction due to its ability to alleviate the lipid peroxidation in rPT cells.
In addition, our in vivo studies showed that PU could significantly restore the inherent antioxidants levels in kidney of Pb-treated rats. 6 In this study, GSH level and GSH-Px activity were chosen to evaluate the effect of PU on antioxidant status in rPT cells. We have observed that simultaneous supplementation with 100 µM PU and Pb significantly alleviated the damaged antioxidant status to near the normal level of intracellular GSH and GSH-Px activity (Figure 6), which suggests that PU represents a potent antioxidant that possesses cytoprotective effects in significant restoration of the nonenzymatic and enzymic antioxidants levels in rPT cells exposed to Pb.
In summary, Pb exposure induced cytotoxicity in cultured primary rPT cells involving mitochondrial oxidative damage with breakdown of mitochondrial ΔΨ and ROS overproduction, which promoted the development of apoptosis. However, as a potent antioxidant, PU manifested a protective effect against Pb-induced cytotoxicity in rPT cells through inhibiting apoptosis, attenuating lipid peroxidation, renewing mitochondrial function, followed by elevation of the inherent antioxidants levels. These observations suggest that PU could potentially alleviate environmental Pb-induced nephrotoxicity and improve kidney health.
Footnotes
Conflict of interest
The authors declared no conflicts of interest.
Funding
This work was supported by the National Nature Science Foundation of China (No. 31101870), a project of Shandong province higher educational science and technology program (No. J13LF09), and a foundation for the author of national excellent doctoral dissertation of People’s Republic of China (No. 201266).
