Abstract
Puerarin (PU), a natural flavonoid, has been reported to possess anti-oxidative and anti-inflammatory activities. In the present study, female Sprague-Dawley rats received lead (Pb) nitrate (300 mg/L, via drinking water) and/or PU (400 mg/kg/day, orally) to investigate the protective effects of PU on Pb-induced renal damage. Renal toxicity was evaluated by detecting urinary proteins excretion as well as levels of serum urea nitrogen and serum creatinine. Ultrastructural observations and real-time quantitative polymerase chain reaction analyses were performed on kidney cortex tissues to identify the mitochondrial damage and quantify gene expression levels of cytochrome oxidase submits (COX-I/II/III), respectively. Renal cell damage was assessed by light microscopic examination. Lipid peroxidation (LPO) levels and antioxidant status in kidney were also evaluated. Animals that received both Pb and PU showed a better renal function than those that received Pb alone, with minor pathological damage. Moreover, PU significantly reduced LPO and markedly restored the enzymatic and non-enzymatic antioxidants levels in kidney of Pb-treated rats, which may be related to its restoring mitochondrial function. Furthermore, PU administration significantly increased urinary Pb excretion and decreased its level in the serum and kidney. In conclusion, these results suggested that PU reduces renal damage induced by chronic Pb administration through its antioxidant properties and chelating ability.
Introduction
Lead (Pb) is one of the most common toxic metals present in the atmosphere, water and soil and exposure to Pb is still a major medical problem in both environmental and occupational settings. As Pb cannot be rendered harmless by chemical or biological remediation processes, it is particularly worrisome among the environmental toxins. 1 It has been known that Pb can induce a wide range of physiological, biochemical and behavioral dysfunctions both in animals and in human beings. 2 The persistence of Pb in animals and humans and the associated health risk is a topic of current concern.
Pb is a multi-targeted toxicant that causes alterations in different organs of the body, especially the kidney as a target site for chronic Pb toxicity. 3 –5 Although the precise mechanism of chronic Pb-induced renal damage is not completely clear, various experimental models indicate that administration of Pb induces the production of reactive oxygen species (ROS) that result in lipid peroxidation (LPO), DNA damage and depletion of cell antioxidant defense systems, that is oxidative stress formed in the presence of Pb may be responsible for its nephrotoxicity. 6 –9 Thus, the association of oxidative stress with Pb intoxication suggests that an antioxidant may enhance the efficacy of therapeutic agents used in the treatment of Pb poisoning.
Puerarin (C21H20O9, PU) is an isoflavone isolated from a traditional Chinese medicine Ge-gen (Radix puerariae). 10 PU is widely used in the clinical treatment of myocardial infarction, cerebral ischemia and diabetic nephropathy in China. 11 Many reports have revealed that PU possesses substantial biological activity, such as anti-oxidative properties, anti-inflammation and anti-apoptosis. 12–14 In addition, some evidence shows that PU can alleviate renal damage induced by nephrotoxins. 15 A recent study suggests that PU has protective effect on Pb-induced nephrotoxicity by the modulation of PI3K/Akt/eNOS pathway. 16 However, this article did not concentrate on urinary excretion kinetics of proteins, ultrastructural modifications of mitochondria and Pb excretion. 16 Therefore, this study was designed to further investigate the protective effect of PU on Pb-induced nephrotoxicity. As ingestion is the most important route of human exposure, an experimental model of rats treated with Pb at relatively high levels (300 mg Pb(NO3)2/L) via drinking water for 8 weeks was used in this study. In this model, we have evaluated the effect of the antioxidant PU given 4 weeks after starting Pb administration on Pb-induced alteration in the structure and function of kidney. Furthermore, we have assessed if the protective effect of PU on Pb-induced renal damage is based on its antioxidant properties and/or in preventing renal Pb uptake.
Materials and methods
Animals and treatment
This study was approved by the Institutional Ethical Committee on Animal Experimentation, Shandong University Hospital, Faculty of Medicine, Jinan, China. It was carried out on 30-day-old inbred female Sprague-Dawley (SD) rats of initial body weight between 90 and 95 g, which were purchased from the Branch of National Breeder Center of Rodents (Shanghai, China). A total of 32 rats were housed individually in conventional conditions at a temperature of 22 ± 1°C, with a relative humidity of 50 ± 10% and a 12/12 h light/dark cycle (lights on from 07:30 to 19:30 h). They had free access to drinking water and commercial standard pellet diet (metal contents, in mg/(kg dry weight), Cu 13.1, Mn 83.5, Fe 212.3, Zn 90.1, Se 0.30, Pb 0.008 and Cd 0.00).
After 1-week acclimatization to the laboratory conditions, the animals were randomly divided into four groups (eight rats in each group), maintained under the supervision of a licensed veterinarian in accordance with the principles set forth in the U.S. National Institute of Health (NIH) Guide for the Care and Use of Laboratory Animals. 17 The dose that rats consumed a solution of Pb via drinking water was selected on the basis of previous studies. 18 Moreover, the dose of PU was selected on the basis of the previous studies 16 and our preliminary experiment. The experimental period was 8 weeks. Rats were divided into 4 groups: (1) control group: rats consumed redistilled drinking water; (2) PU-treated group: rats received a daily oral gavage administration of PU (Sigma-Aldrich, Missouri, USA) at dose of 400 mg/(kg body weight)/day beginning from the fifth week; (3) Pb-treated group: rats received an aqueous solution of Pb(NO3)2 (300 mg/L, Sigma-Aldrich, Missouri, USA) as the only drinking fluid for 8 weeks and (4) Pb + PU-treated group: these animals received both Pb and PU at the doses, periods and routes of administration described above. PU and Pb solutions were prepared fresh in redistilled water. PU solution was prepared daily by filtration sterilization and its concentration was adjusted such that a rat would receive 0.3 mL per day. Pb solution was replaced daily to minimize the precipitation, which was analyzed by inductively coupled plasma mass spectrometry (ICP-MS) method (ELAN-6000 model, Perkin–Elmer, Sciex, Toronto, Canada) to verify its concentration. Water consumed by the control group and the PU-treated group was also analyzed to determine the absence of Pb. During the experimental period, water consumption and weight gain were measured every day.
Collection of rat urine
On the day before the experiment (W0) and at the end of 2, 4, 6 and 8 weeks of treatment (W2, W4, W6 and W8, respectively), rats were kept in individual metabolic cages for 24 h urine collection. The rats were provided with water but no feed was given. The urine collected on ice was free from fecal contamination. Urine samples were centrifuged at 1000×g for 10 min and aliquots were separated. One portion was used for the determination of various proteins and Pb levels immediately.
Collection of rat tissues
After the exposure period (8 weeks), rats were fasted overnight and killed by cervical decapitation under ether anesthesia. Blood was collected from the heart to obtain the serum by centrifugation at 2000×g for 15 min. Then the kidneys were removed, weighted, dissected out and washed in ice-cold saline for the isolation of the renal cortex tissues. Some pieces were immediately homogenized (10%, w/v, at 4°C) in Tris-HCl buffer (pH 7.4; Jiancheng Institute of Biotechnology, Nanjing, China) and centrifuged (1000×g for 10 min, at 4°C) and the clear fluid following supernatant was stored on ice for various assays. Approximately 0.2 g of renal cortex tissues from each sample was immediately frozen in liquid nitrogen for RNA extraction. Some tissues were immediately fixed for morphological studies as described later.
Serum and urinary markers of renal toxicity
The levels of serum urea nitrogen (SUN) and creatinine (Cr) in serum were determined with a Synchron LX20 analyser (Beckman, Massachusetts, USA). Urinary total protein concentration was estimated according to the method of Lowry et al. 19 with bovine albumin (Sigma, Missouri, USA) as a standard. Urinary β2-microglobulin (β2-MG) and α1-microglobulin (α1-MG) were determined by the radioimmunoassay.
Electron microscopic examination of the renal damage
Renal cortex tissues were cut into small blocks (1 mm3) and fixed by immersion in fixative (2.5% glutaraldehyde in 0.1 M cacodylate buffer, pH 7.4) for 12 h at 4°C. After washing in 0.1 M cacodylate buffer, the blocks were dehydrated in graded ethanol, embedded in gelatin capsules with Durcupan resin and polymerized in an oven at 60°C for 48 h. Ultrathin sections (400 nm) were cut, mounted on grids and stained with uranyl acetate and Pb citrate. They were examined with a PHILIPS CM-120 Transmission Electron microscope (Amsterdam, The Netherlands) at 80 KV by the same histologist in a ‘blind’ fashion.
Histological assessment of the renal damage
Some of the renal cortex tissues were fixed in 10% formalin solution, dehydrated in ethanol and xylene and embedded in paraffin. Paraffin sections of 5-μm thicknesses were stained with hematoxylin and eosin (HE) for the histological assessment. The slides were examined under a light microscope (Nikon Eclipse E600W, Suzhou, China) by the same pathologist blinded to the treatment.
Quantitative real-time polymerase chain reaction (qRT-PCR)
RNA isolation and complementary DNA (cDNA) synthesis
Total RNA was extracted from frozen renal cortex tissues using the Trizol reagent (Invitrogen life Technologies, USA). The RNA purity was verified by the optical density at 260 nm (OD260)/OD280 absorption ratio (>1.8). The isolated RNA was reverse transcribed using SuperScripTM III reverse transcriptase (Invitrogen life Technologies, California USA). Briefly, the RNA was denatured by heating for 5 min at 65°C, cooled on ice and incubated with reverse transcriptase reaction mixture. The standard mixture contained 2 µg of total RNA, 25 U of RNAase inhibitor, 0.5 mM each of dNTPs, 1.5 µM reverse primer and 200 U of SuperScripTM III reverse transcriptase in a total volume of 25 µL. For reverse transcription, tubes were incubated at 42°C for 60 min, followed by rapid cooling.
Quantitative RT-PCR
Quantitative RT-PCR was performed on the Rotor-Gene TM 6000 real-time rotary analyzer (Corbett Robotics Inc., Sydney, Australia) using the one-step SYBR® QRT-PCR kit (Takara, Dalian, China). The QRT-PCR was performed in a 20 µL reaction mixture. Relative quantification of QRT-PCR product was performed using the comparative ΔΔCT method and SYBR green fluorescent labeling. 20,21 Four pairs of specific primers (Table 1) were designed according to the alignments of the published cDNA sequences of GADPH, COX-I, COX-II and COX-III genes in rat. These four pairs of primers were tested for their specificity by conventional reverse transcription PCR (RT-PCR) before being used for the QRT-PCR studies. PCR amplification conditions were as follows: denaturation at 95°C for 2 min and 40 cycles at 95°C for 10 s, 60°C for 20 s and 72°C for 30 s. The reaction was then subjected to a melting protocol from 67°C to 95°C with a 0.5°C increment and 30 s holding at each increment to check the specificity of the amplified products. Quantitative RT-PCR reaction was performed in triplicate for each sample and a mean value used to calculate messenger RNA (mRNA) levels. Eight biological replicates were measured for each group.
Primer sequences with their corresponding PCR product size.

COX: cytochrome oxidase; GADPH: glyceraldehyde 3-phosphate dehydrogenase; PCR: polymerase chain reaction.
Indices related to oxidative stress in kidney
The levels of reduced glutathione (GSH) and activities of total superoxide dismutase (SOD), catalase (CAT) and glutathione peroxidase (GPx) in renal homogenate were measured spectrophotometrically, in accordance with the analysis kits detection protocol (Jiancheng Bioengineering Institute, Nanjing, China). LPO levels in renal homogenate were estimated by the method of Ledwozwy et al. 22 In brief, malondialdehyde (MDA) content was assessed in optical density at 532 nm, which is undertaken as an index of LPO. Results were expressed as nanomole of MDA per milligram of protein. The protein concentration in kidney homogenate was determined by the method of Lowry et al. 19 using bovine serum albumin as a standard.
Determination of Pb levels in the serum, urine and renal cortex
The collected serum and fresh urine samples were digested using HNO3 and H2O2 (Guaranteed reagents) in Teflon PFA microwave digestion vessels. Samples of the renal cortex (about 400 mg wet weight) were dried to constant weight (dry weight) at 80°C for 48 h. Then the dried samples were weighted and digested using HNO3 and H2O2 in Teflon PFA microwave digestion vessels. All the digested samples were diluted with de-ionized water so that the analyte was within the calibration range. Concentrations of Pb in the serum, urine and renal cortex were determined by ICP-MS method according to the manufacturer’s recommendation. Quality control was strictly carried out using standard reference materials (SRM 1598, NIST).
Statistical analysis
Experimental groups were compared using a two-ways analysis of variance (ANOVA) followed by Scheffe’s test when the data were normally distributed and by the Kruskal–Wallis test when they were not normally distributed. All data were expressed as mean ± SD (n = 8). The statistical significance was evaluated using SPSS Version 13.0 (SPSS, Cary, North Carolina, USA) and p < 0.05 was considered significant.
Results
Effect of Pb and/or PU on the performance of rats
Daily Pb dose ingested by rats during the exposure was calculated using the formula mL water consumed/day × metal concentration in the drinking water (mg/mL)/rat weight (kg), as shown in Table 2. As expected, there was no significant difference in the amounts of Pb consumed by rats between the Pb group and the Pb + PU group. Moreover, Pb intoxication had no effect on weight gain (Table 3) or on water and feed consumption (data not shown).Similarly, there were no specific signs attributable to the treatment with Pb and/or PU.
Pb consumption in treated rats and Pb levels in the serum and renal cortex of the experimental rats.a

Pb: lead; PU: puerarin.
aAmong the groups, control group (Control), PU-treated group (PU), Pb-treated group (Pb) and Pb + PU-treated group (Pb + PU). Data are mean ± SD (n = 8).
b p < 0.01 versus control group at the same time.
c p < 0.05, statistical significance between Pb and Pb + PU treatment at the same time.
Results of BW, UF and absolute and relative weights of kidneys in rats (n = 8).a
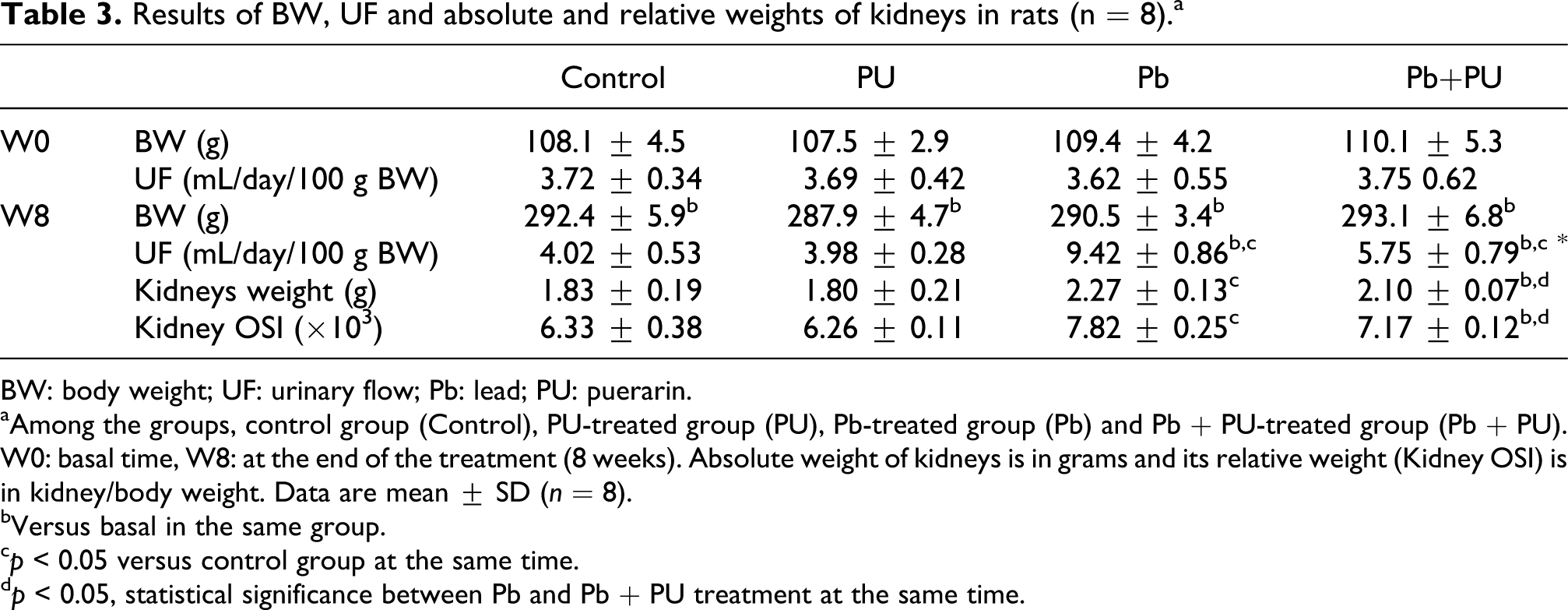
BW: body weight; UF: urinary flow; Pb: lead; PU: puerarin.
aAmong the groups, control group (Control), PU-treated group (PU), Pb-treated group (Pb) and Pb + PU-treated group (Pb + PU). W0: basal time, W8: at the end of the treatment (8 weeks). Absolute weight of kidneys is in grams and its relative weight (Kidney OSI) is in kidney/body weight. Data are mean ± SD (n = 8).
bVersus basal in the same group.
c p < 0.05 versus control group at the same time.
d p < 0.05, statistical significance between Pb and Pb + PU treatment at the same time.
Effects of PU on Pb-induced renal damage
Exposure to Pb for 8 weeks induced a significant increase in urinary flow (UF) that was significantly reduced by PU administration (p < 0.05; Table 3). Simultaneously, significant increase in absolute and relative weight of kidneys was observed in rats treated with Pb and with Pb + PU (p < 0.05; Table 3).
Exposure to Pb induced a time-dependent increase in urinary total protein (TP) excretion that reached statistical significance after 2 weeks of treatment with an increase of 1.68-fold over basal values after 8 weeks of administration (Figure 1(a)). In the Pb + PU group, the increase of proteinuria was markedly lower (p < 0.01) when compared with rats that received Pb alone. Similarly, Pb treatment resulted in the increased excretion of low-molecular-weight protein in urine, such as β2-MG; while this increase was markedly lower in rats that also received PU (Figure 1(b)). Microalbuminuria (Alb) was also increased with increase in exposure time as shown in rats treated with Pb, while this increase was markedly decreased by administration with PU (Figure 1(c)). No significant changes in urinary TP, β2-MG and Alb excretion occurred in rats that received PU alone (Figure 1). In addition, a significantly (p < 0.05) increased levels of SUN and Cr in the serum was observed in Pb-treated rats, while this increase was significantly decreased by treatment with PU (Figure 2). The above results indicate that PU treatment decreased the Pb-induced renal damage in rats.
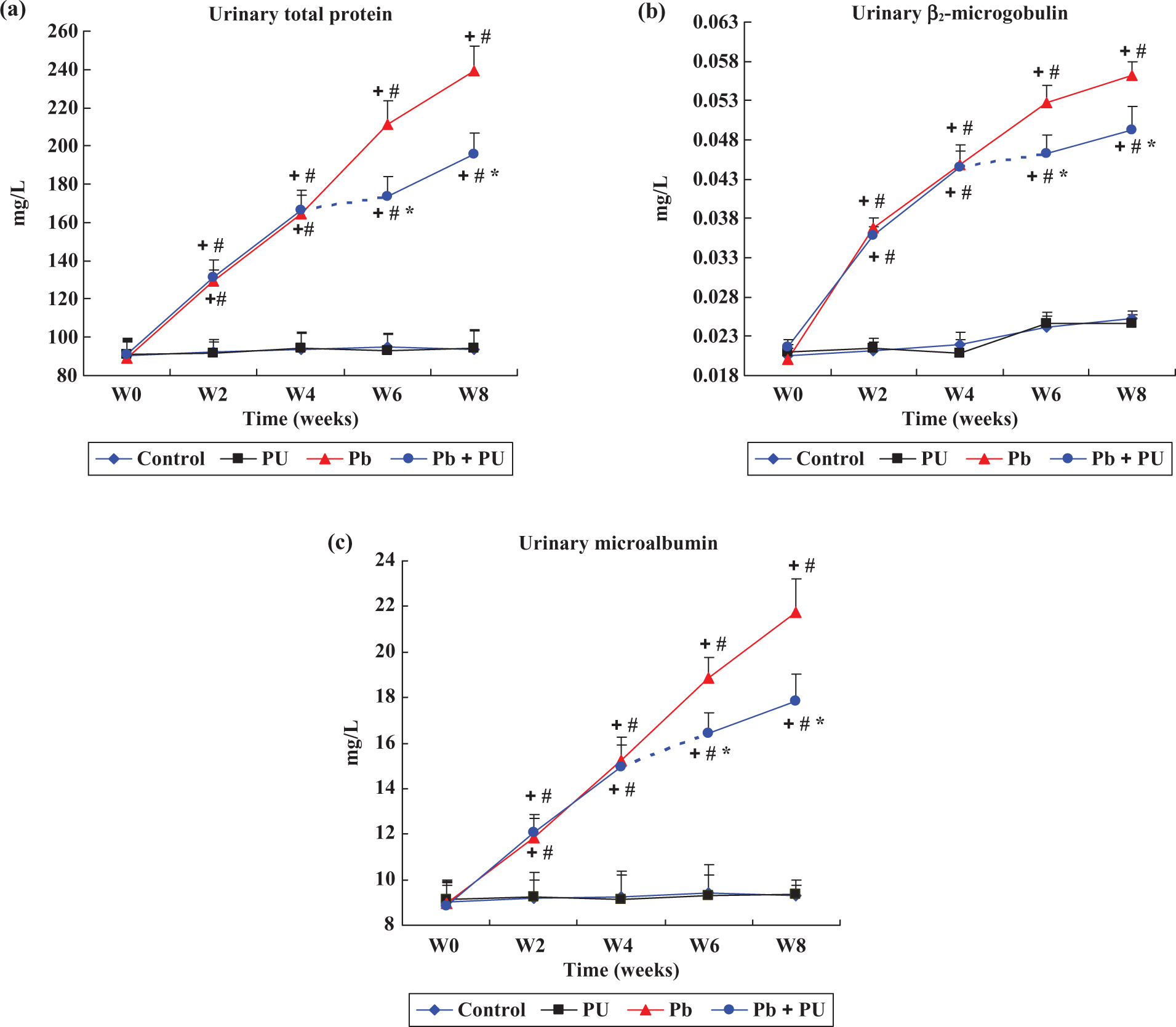
Changes in the contents of total protein (a), β2-MG (b) and Alb-U (c) in urine during the experiment. Among the groups, control group (Control), PU-treated group (PU), Pb-treated group (Pb) and Pb + PU-treated group (Pb + PU). W0: Basal time, W2, W4, W6, W8: at the end of 2, 4, 6, 8 weeks treatment. Dash line between W4 and W6 in the Pb + PU group indicated the onset of the PU treatment. Data are mean ± SD (n = 8), + p < 0.05 versus W0 in the same group, # p < 0.05 versus control group at the same time; * p < 0.05, statistical significance between Pb and Pb + PU treatment at the same time. Alb-U: protein albumin; β2-MG: urinary β2-microglobulin; Pb: lead; PU: puerarin

Effect of PU on Pb-induced changes in renal functional markers: (a) serum urea nitrogen, (b) serum Cr in rats at the end of treatment (8 weeks). Among the groups, control group (Control), PU-treated group (PU), Pb-treated group (Pb) and Pb + PU-treated group (Pb + PU). All the values are expressed as mean ± SD (n = 8), # p < 0.05 compared with control group; * p < 0.01, statistical significance between Pb and Pb + PU treatment. Pb: lead; PU: puerarin.
Effects of PU on histology changes in Pb-induced nephrotoxicity
Ultrastructural modifications of mitochondria in the renal cortex of rats are shown in Figure 3. Changes in mitochondrial ultrastructure included swelling, disruption of cristae, flocculent density inclusions and loss of cristae when rats were exposed to Pb alone (Figure 3(c)). While rats treated with the combination Pb + PU showed some minor alterations in damaged mitochondria, with the alterations always less pronounced than that in rats treated with Pb alone (Figure 3(d)). No alterations in mitochondrial ultrastructure were observed in control and PU-treated rats (Figure 3(a) and (b)).

Representative electron microscopy images of mitochondria from rat kidneys of control rats (a) and rats treated with PU alone (b), rats treated with Pb (c) and rats treated with Pb + PU (d). (a–d) Original magnification 18,500×; Disruption of mitochondrion cristae and loss of mitochondrion cristae (thin arrows) are marked in the figures. Pb: lead; PU: puerarin.
Light microscopic examination of kidney tissues of control rats showed normal structure of renal tubules and glomeruli (Figure 4(a)). Rats treated with PU alone also indicated normal structure of glomeruli and tubules (Figure 4(b)). Necrosis of tubular epithelial cells marked by contracted and pycnotic nuclei with condensed chromatin, loss of regular tubular lumen, intraluminal casts and irregularity of the tubular shape as well as slight glomerular swelling and decreased cellularity of the glomeruli were evident in kidneys of all Pb-exposed rats (Figure 4(c)). Rats treated with Pb + PU showed more or less normal renal tubules and glomerulus, but some pathological changes in the renal tubules and glomeruli still presents (Figure 4(d)). It showed that administration with PU partly alleviated the renal damage in rats exposed to Pb.
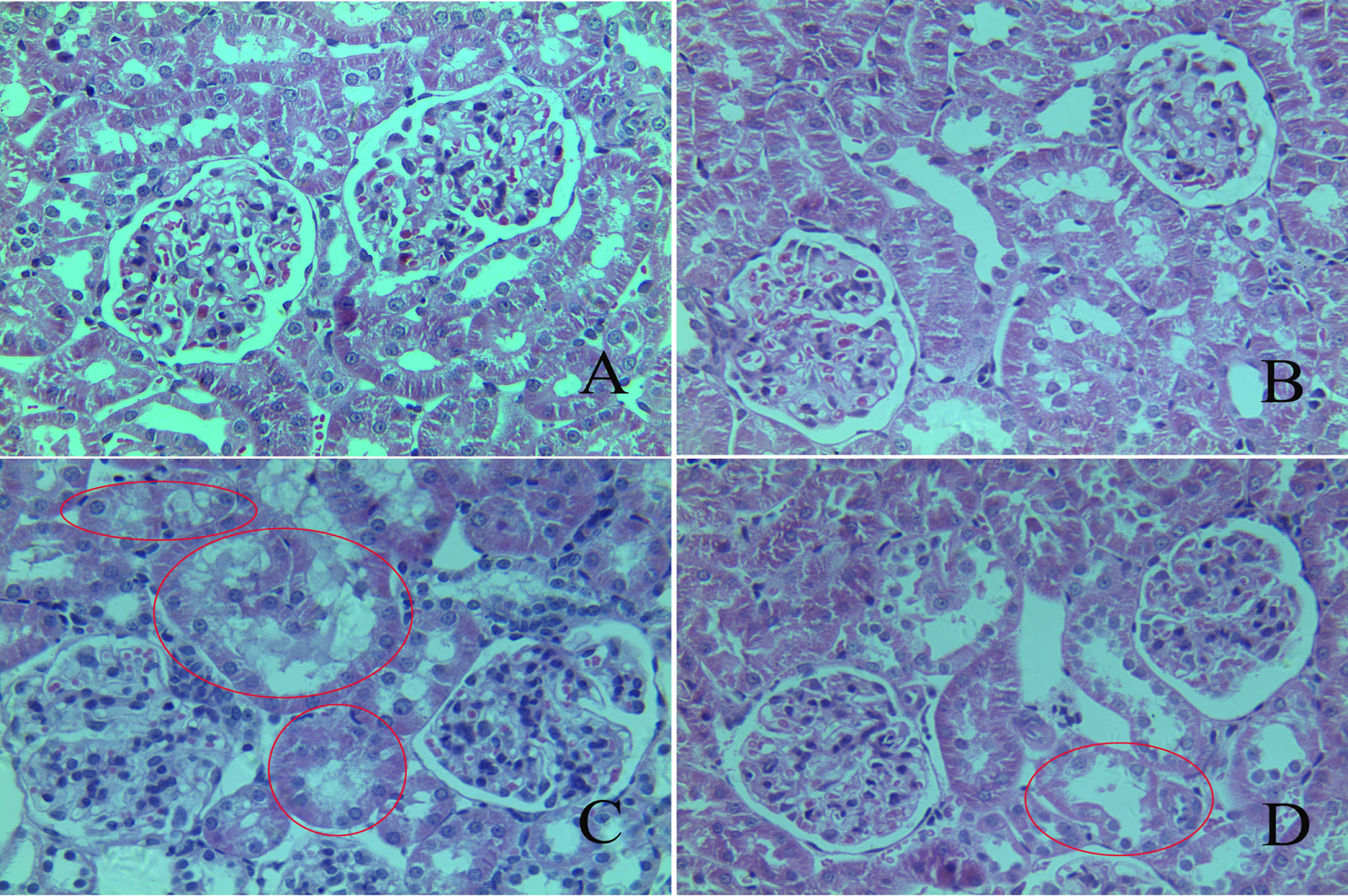
Hematoxylin and eosin-stained kidney slices from treated rats. Control group (a) and PU-treated group (b) with normal structure of tubules and glomeruli in renal cortex. Pb-treated rats (c) with damaged kidney structure. Pb-treated rats fed with PU (d) with reduced effects of Pb toxicity on renal cortex. Pathological changes are marked in the figures. Original magnification: 400×. Pb: lead; PU: puerarin.
Relative quantification of mitochondrial COX subunit I/II/III mRNA expression levels
As shown in Figure 5, mitochondrial COX subunit I/II/III expression levels were all significantly (p < 0.01) down-regulated in rats exposed to Pb. After 8 weeks of exposure, the expression level of the Pb group was significantly decreased for COX-I (factor 0.54), while COX-II and COX-III was decreased with a factor of 0.63 and 0.50, respectively, compared with the control group. Similarly, there is a significant difference in the relative expression of COX-I, COX-II and COX-III between the Pb group and the Pb + PU group. However, no significant changes in the relative expression level of COX-I/II/III occurred in rats that received PU alone.

The graphs represent the relative transcription levels of genes in kidney of rats exposed to Pb and/or PU for 8 weeks. The transcription levels are quantified in a relative way to the control group, whose expression level is set at one. (a) COX subunit I (COX-I), (b) COX subunit II (COX-II), (c) COX subunit III (COX-III). Among the groups, control group (Control), PU-treated group (PU), Pb-treated group (Pb) and Pb + PU-treated group (Pb + PU). Data represent mean ± confidence interval determined by the Fieller method (n = 8). # p < 0.01 compared with control group; * p < 0.01, statistical significance between Pb and Pb + PU treatment. COX: cytochrome oxidase; Pb: lead; PU: puerarin.
Effects of PU on Pb-induced LPO and antioxidant status in kidney
Information on oxidative stress status in kidneys after the treatment is shown in Figure 6. Renal MDA levels in rats exposed to Pb were increased to 1.66-fold compared with that of the control (p < 0.01), while PU significantly (p < 0.01) decreased Pb-induced renal MDA levels (Figure 6(a)). In rats administrated PU and Pb, a significant (p < 0.05) increase in the non-enzymatic antioxidant (GSH) level and increased activities of enzymatic antioxidants (SOD, CAT and GPx) were observed. In contrast, Pb alone-treated rats showed significantly (p < 0.01) decreased GSH level and activities of SOD, CAT and GPx when compared with control values (Figure 6(b) to (e)). Animals that received PU alone did not show significant changes in kidney MDA and GSH levels and activities of SOD, CAT and GPx compared with the control group.
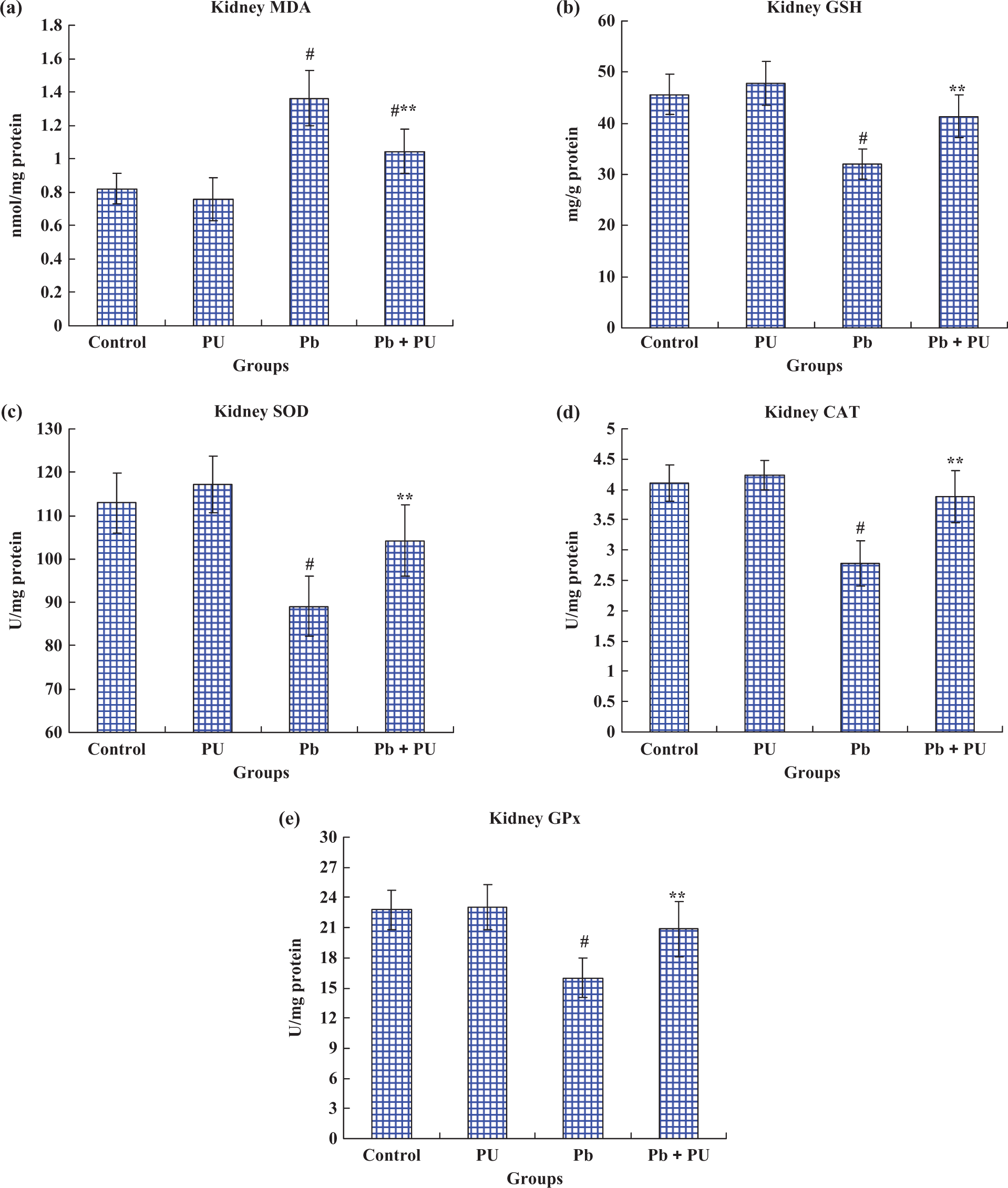
Effects of Pb and/or PU on the levels of lipid peroxidation, enzymatic antioxidants and non-enzymatic antioxidant status in kidney of rats at the end of the treatment (8 weeks). Among the groups, control group (Control), PU-treated group (PU), Pb-treated group (Pb) and Pb + PU-treated group (Pb + PU). (a) MDA content. It is expressed as nanomole per milligram of protein. (b) GSH level. It is expressed as milligram per gram of protein. (c) SOD activity in the kidney. It is expressed as follows: one unit of activity was taken as the enzyme reaction, which gave 50% inhibition of nitroblue tetrazolium reduction in 1 min/(mg protein). (d) CAT activity in the kidney. It is expressed as micromoles of hydrogen peroxide consumed per minute per milligram of protein, that is, U/(mg protein). (e) GPx activity in the kidney. It is expressed as microgram of glutathione consumed in minute per milligram of protein, that is, U/(mg protein). All values are expressed as mean ± SD (n = 8), # p < 0.01 as compared with control group; ** p < 0.01, statistical significance between Pb and Pb + PU treatment. CAT: catalase; GSH: reduced glutathione; GPx: glutathione peroxidase; MDA: malondialdehyde; Pb: lead; PU: puerarin; SOD: superoxide dismutase.
Distribution of Pb in the serum, kidney and urine
Exposure to Pb for 8 weeks resulted in a significant increase in its serum and renal concentrations (Table 2) and also a time-dependent increase in its urinary excretion (Figure 7). Treatment with PU significantly decreased the levels of Pb in the serum and kidneys and increased its excretion in the urine during treatment (p < 0.05). Urinary Pb excretion was undetectable in the control group and the PU-treated group.

Changes of Pb concentration in urine during the experiment. Among the groups, control group (Control), PU-treated group (PU), Pb-treated group (Pb) and Pb + PU-treated group (Pb + PU). W0: basal time, W2, W4, W6, W8: at the end of 2, 4, 6, 8 weeks treatment. Dash line between W4 and W6 in the Pb + PU group indicated the onset of the PU treatment. Pb was not detected in the control and PU-treated group, so their contents were expressed as ‘0.’ Data are mean ± SD (n = 8), + p < 0.05 versus W0 in the same group, # p < 0.05 versus control group at the same time; * p < 0.05, statistical significance between Pb and Pb + PU treatment at the same time. Pb: lead; PU: puerarin.
Discussion
PU, a natural flavonoid, is widely available in common foods and used in the treatment of many diseases by oral or intravascular administration owing to its wide spectrum of biological activities and its beneficial effects on human health, particularly its antioxidant properties and potential anti-inflammatory effects. 23,24 Meanwhile, many studies have shown that flavanoids could decrease accumulation of heavy metal in the body. 25 –27 Given these features, PU was chosen to investigate its protective efficacy on chronic Pb-induced nephrotoxicity. Furthermore, similar studies have been reported, 16 but different treatment modalities and biochemical indicators were applied in this article. The design of our study included the treatment with PU several weeks after starting the administration of Pb, in order to assess if this antioxidant agent could have an effect on Pb-renal toxicity when intoxication is already occurring.
As a sensitive indicator for renal damage, proteinuria most often reflects loss of the normal glomerular impermeability to filtration of plasma proteins and/or diminished reabsorptive capacity of the proximal tubules. 28,29 Combination of several urinary protein measurements may provide a better sensitivity and/or detection of the site of action of the toxic material. 30 For example, increased excretion of low-molecular-weight protein β2-MG has been used as an indicator of impaired tubular capacity for the absorption of proteins. Increased excretion of high-molecular-weight protein albumin (Alb-U) is commonly used as a marker of glomerular dysfunction. 31 Results from this study (Figure 1) suggest that Pb-intoxicated rats developed heavy proteinuria accompanied with low-molecular-weight proteinuria and high-molecular-weight proteinuria, indicating a mixed glomerular and tubular damage. Moreover, light microscopic examination of the kidney tissues demonstrated this idea (Figure 4(c)). In addition, significant increase in the serum Cr and SUN levels further indicated the decrease of glomerular filtration in rats exposed to Pb (Figure 2). Combined with the changes of urinary enzymes and proteins, it can be seen that Pb exposition induces the renal damage through the impaired reabsorptive capacity of the proximal tubules and decrease of glomerular filtration. As a result, UF was increased in rats exposed to Pb (Table 3). Based on the results shown in Figures 1, 2 and 4(d) and Table 3, it is proved that PU has a significant protective effect on the impaired tubular reabsorptive capacity and glomerular filtration in rats.
In our previous studies, it was determined that the nephrotoxic effect of Pb was related to LPO. 9,32 LPO is involved in mitochondrial dysfunction. Alterations in mitochondrial function are largely attributed to the damaging effects of ROS. 33 Furthermore, mitochondria are the major source of ROS and ROS production increases significantly when mitochondria suffered from pathological damage. 34,35 In this study, mitochondrial abnormalities were evident in the Pb-treated rats. Mainly the toxic effects involve disruption and loss of mitochondrial cristae together with mitochondrial swelling and disruption of mitochondrial membrane (Figure 3(c)). Moreover, minor alternation in mitochondrial structure in the Pb + PU group demonstrated the alleviated damage in kidney due to its protective effect (Figure 3(d)).
As the marker enzyme of mitochondrial function, COX is of special importance to the membrane potential and the redox state of electron transport chain (ETC), and to the regulation of mitochondrial respiration. 32,36,37 Any loss of COX activity can create a bottleneck in the activity of the ETC as a whole, which would not only attenuate the respiratory rate and potential for oxidative phosphorylation but also increase the production of ROS. 37 That is to say, COX dysfunction increases ROS, reduces energy stores and disturbs energy metabolism. 38 COX-I, II and III are the three largest mitochondrially encoded subunits, which constitute the catalytic functional core of the COX holoenzyme. 37 Subunit I is involved in the reduction of molecular oxygen to water and pumping protons from the mitochondrial matrix into the inter-membrane space. 39 Subunit II participates in providing sites for docking and transfers of electrons from cytochrome c and transferring electrons to heme α in subunit I. 39,40 Subunit III mainly modulates the rate of proton translocation and also facilitates a late step in the assembly of COX. It is thus apparent that due to the interdependent roles of COX subunits, differential losses of the mitochondrial DNA-encoded subunits (COX-I, II and III) could result in a decrease in enzymatic activity. 41 In addition, COX-I, II and III are quite vulnerable to oxidative damage, because both their mRNA as well as protein is in close proximity to the sites of ROS generation. 37 Accordingly, COX-I, II and III mRNA expression levels were quantified using quantitative RT-PCR to investigate the protective effect of PU on Pb-induced COX dysfunction. As shown in Figure 5, the expression levels of COX-I, II and III in the Pb-exposed group were all significantly lower than that in the control group, respectively; which is consistent with our previous study. 32 These results suggest that a decrease in the abundance of specific COX submits (I, II and III) is responsible for the COX dysfunction induced by Pb treatment, which may in turn affect mitochondrial function. However, significantly elevated gene expression levels of COX-I, II and III were observed after administration with PU, indicating that PU could effectively improve mitochondrial function by alleviating COX dysfunction.
In agreement with these features, significant elevation of MDA contents, depletion of GSH levels and decreased activities of SOD, CAT and GPx in kidney were seen in rats exposed to Pb, thus suggesting increased renal oxidative stress (Figure 6). As an active antioxidant, PU administration significantly blunted the Pb-induced increase of the MDA levels in kidney (Figure 6(a)); further suggesting that PU could reduce the production of oxygen free radicals. Furthermore, therapeutic efficacy of PU on Pb-induced oxidative damage was due to its restoring effect of the GSH levels and SOD, CAT and GPx activities (Figure 6(b) to (e)). Elevation of antioxidant levels may be related to its restoring mitochondrial function (Figures 4 and 5). In addition, oxidative stress-induced inflammation plays a major role in the renal damage produced by exposition to Pb. 42,43 This may be the cause of hypertrophy of the kidney in rats exposed to Pb (Table 3), while anti-inflammatory effects (decrease in kidney weight) observed after treatment with PU may also be related to its antioxidants properties.
Moreover, urinary Pb excretion was significantly increased in rats that received Pb + PU (Figure 7) compared with those treated with Pb alone. Moreover, we found that PU markedly decreased the levels of Pb in the serum and kidneys (Table 2), thus suggesting that the protective effect of PU is partly based on its chelating ability to reduce renal uptake and accumulation in Pb.
In summary, PU has beneficial actions against Pb-induced toxic effects in kidney that seems to be based on its strong antioxidant properties as well as its chelating ability to prevent renal Pb accumulation. It also demonstrates that administration of PU has a protective effect on Pb-mediated nephrotoxicity when Pb intoxication is already occurring.
Footnotes
Authors’ Note
L Wang and S Lin contributed equally to this work.
Conflict of interest
The authors declared no conflicts of interest.
Funding
This work was supported by the National Nature Science Foundation of China (No. 31101870), Shandong Provincial Natural Science Foundation of China (No.ZR2010CQ014) and Science & Technology Innovation foundation for young scholars of Shandong Agricultural University in China (No. 23690).
