Abstract
Xenobiotics make their way into organisms from diverse sources including diet, medication, and pollution. Our understanding of ocular toxicities from xenobiotics in humans, livestock, and wildlife is growing thanks to laboratory animal models. Anatomy and physiology are conserved among vertebrate eyes, and studies with common mammalian preclinical species (rodent, dog) can predict human ocular toxicity. However, since the eye is susceptible to toxicities that may not involve a histological correlate, and these species rely heavily on smell and hearing to navigate their world, discovering visual deficits can be challenging with traditional animal models. Alternative models capable of identifying functional impacts on vision and requiring minimal amounts of chemical are valuable assets to toxicology. Human and zebrafish eyes are anatomically and functionally similar, and it has been reported that several common human ocular toxicants cause comparable toxicity in zebrafish. Vision develops rapidly in zebrafish; the tiny larvae rely on visual cues as early as 4 days, and behavioral responses to those cues can be monitored in high-throughput fashion. This article describes the comparative anatomy of the zebrafish eye, the notable differences from the mammalian eye, and presents practical applications of this underutilized model for assessment of ocular toxicity.
Introduction
Nonclinical toxicology studies typically involve high or repeat doses of the test article in 2 mammalian species (ICH M3 (R2) (2009)). Pharmacokinetics, toxicology, and histopathology from those studies are used in conjunction with efficacious plasma levels to generate a therapeutic safety index which helps guide initial human dose levels. Provided sufficient exposure and no gross toxicological findings from late-stage toxicology studies, histopathology is relied upon to expose cell or tissue lesions that would have otherwise gone undetected. During conventional drug development programs, such unwelcomed discoveries were made after significant investment in the test article, including efficacy studies, structure–activity optimization, lead molecule selection, and relatively large-scale production. 1 Investigative toxicology studies that can be conducted concurrently with discovery efforts are being increasingly relied upon to deliver earlier prediction of tissue-specific adverse effects, 2,3 thereby allowing prioritization of the safest drug candidates to proceed into later stage animal testing.
Many platforms used in investigative toxicology are considered alternatives to traditional animal testing paradigms, in line with the 3Rs (reduction, refinement, and replacement) philosophy toward using animals in research. 4,5 If they are conducted in the early-life stages, studies with embryonic and larval zebrafish fall into this category. Currently, according to the European Commission Directive from 2010 (Directive 2010/63/EU), research conducted with zebrafish before they become independent from the maternally derived yolk (around 120 hours post-fertilization, hpf) is considered an alternative to animal testing and thus is a replacement of animal research. 6 The Food and Drug Administration (FDA) accepts supporting data from nonmammalian toxicology models in regulatory submissions, and the Center for Drug Evaluation and Research encourages dialog with sponsors regarding the use of new approaches, such as research with early-life stages of zebrafish, that may contribute to more efficient drug development. 7
Toxicities that impact vision can go unnoticed during the in-life portion of nonclinical studies as the standard species used (rat, dog) rely heavily on the senses of smell and hearing to navigate their world, and quantitative behavioral experiments aimed at testing for functional vision are not a routine part of toxicology studies. Moreover, the eye is susceptible to operative changes that may not involve a histological correlate, so some effects on vision may be detected only through functional evaluation. 8 Investigative toxicology studies using zebrafish can deliver tissue-specific ocular toxicity data, as well as functional analysis, early in the drug development process using small amounts of test article. 9 –11 Examples of this capability are collected from the literature into this review along with an overview of zebrafish as an ocular toxicology platform, discussion of comparative anatomy and physiology of the zebrafish visual system, and a description of functional platforms for assessing vision.
General Advantages and Challenges of Zebrafish as an Animal Model
One of the traits of zebrafish that empowers vertebrate research is that they are small. Their small size (5-mm long when larvae) allows for in-vivo investigation on an in-vitro scale, which takes less space, less test article, and less time than mammalian studies. However, their small size also presents some of the main challenges of the model. Typical toxicology studies are relied upon to deliver information on a chemical’s absorption, distribution, metabolism, excretion (ADME), as well as on the therapeutic window that might be expected between a chemical’s efficacious and toxic plasma levels. Due to the small size of zebrafish larvae, these studies are challenging given today’s technology; however, key advances in bioanalytical techniques supporting zebrafish ADME are being made in the field. 12 –14 Zebrafish require relatively simple husbandry skills, although regular monitoring for common fish pathogens, whether parasitic or commensalistic, is recommended, as some infections may interfere with study results. 15 Two additional basic traits that have made zebrafish a popular model are that they have high fecundity (a single breeding session typically yields 100-300 fertilized eggs), and the eggs develop externally into free swimming larvae within 5 days post-fertilization (dpf). 16 –18
Since laboratories use different food and incubation temperatures for their fish, there are discrepancies regarding the ages associated with developmental life stages. Based on variable growth rates, anatomical hallmarks are increasingly being used to define these stages. 19 Zebrafish life stages can be roughly defined by age, with the following morphological descriptions. The embryonic stage ends around 72 hpf, with the appearance of a protruding mouth and, typically, hatching. Morphological hallmarks during zebrafish embryonic development have been defined. 20 The larval stage follows the embryonic stage and lasts to around 30 dpf, at which time, typically, adult characteristics, aside from sexual characteristics, appear, including a complete pattern of scales and complete loss of the larval fin fold. Zebrafish are considered juvenile after this time until they reach adulthood with the production of viable gametes, which typically occurs around 3 months. 19 In this article, the post-fertilization times associated with various anatomical structures of the eye are taken from multiple reports (laboratories), and the reader is advised to read the cited work for the husbandry practice of the reporting laboratory.
Zebrafish embryos are amenable to genetic engineering and have been extensively used as a vertebrate proving ground for novel genome altering tools. 21 –24 The application of those tools in zebrafish has vastly broadened our understanding of basic vertebrate biology and human diseases in diverse areas such as metabolic, muscular, skeletal, neuronal, cardiovascular, and cancer. 25 –31 Larvae are transparent, facilitating in vivo examination of internal structures, and can survive for several days post-fertilization in just a drop of water. These advantages have made possible the development of several high-throughput assays for discovering genes related to disease, treatment, toxins, and toxic mechanisms. 9,32 –40 However, due to the greater phylogenetic distance between fish and human, compared to that of mammalian models, translatable results from zebrafish studies are more dependent on careful selection of the systems and end points examined. Investigators using zebrafish for translation will want to be well versed in current literature so that efforts are focused on systems having conserved biology. Research conducted over the past 20 years has greatly improved our understanding of the similarities and differences between zebrafish and human in gene expression, cell signaling, tissue architecture, and organ function. 41 –43 Teleosts experienced a whole-genome duplication event near the root of their lineage, around 350 million years ago, and zebrafish often have a and b forms of mammalian gene orthologs. 44 Another major distinction of the model for consideration is the capacity for zebrafish to regenerate many tissue types that are considered postmitotic/nonregenerative in mammals, including retina. 45,46 Although this brings value for understanding regeneration mechanisms, 47 it requires careful consideration of study design, as well as data interpretation, when zebrafish are used to model toxicity because of the comparably poor regenerative capacity of mammalian tissues. For example, it was demonstrated that regenerative signaling pathways contribute to partial protection of retinal neurons in zebrafish exposed to CoCl2. 48
Studies of chemical toxicology using zebrafish typically expose the fish (larvae, juvenile, or adult) to the test article by dissolving it in the ambient water. Although this is convenient and is amenable to building high-throughput assays (which typically use larvae), it relies on common solubility and absorption among chemicals being compared. One way this gap is being addressed is to relate biological, behavioral, or toxicological end points to actual absorption values, versus using the chemical’s nominal concentration in the water. 49 –51 In-water dosing may bring about different effects from those that arise from oral or other dosing routes (as used in mammalian models) as the chemical can be absorbed by multiple routes (gills, topical, oral), and the fish are continuously exposed.
Processing Zebrafish Eyes for Histological Evaluation
Zebrafish should be humanely euthanized by immersion in 4 °C water or in an overdose of an anesthetic such as tricaine methanesulfonate. 52 Immediately following euthanasia, tissues should be collected and placed in an appropriate fixative, such as 10% neutral buffered formalin, or modified Davidson’s. Processing techniques and type of fixative used to prepare zebrafish for histological examination can vary, depending on the age and desired end points. However, just as size is a benefit of working with zebrafish, it also serves as a challenge when it comes to histological processing. Quality ocular sections are dependent upon the technical expertise of a skilled histologist and require anatomical knowledge, attention to detail, and careful handling to minimize artifacts. Despite their small size, zebrafish eyes are still subject to artifacts similar to those that are observed in other species, in part due to the complex tissues and differing matrices present in the eye. The fully developed adult eye is somewhat more challenging than larval eyes in which the anterior chamber and vitreous are not yet developed. Although quality sections can be obtained through paraffin embedding, plastic embedding may produce sections with fewer artifacts, with the added advantage of enabling thinner sections, providing enhanced microscopic detail. The techniques described below using paraffin embedding were used to obtain sections for the representative images provided in this article.
Larvae are particularly amenable to whole-body processing. Immediately following euthanasia, they should be transferred to 10% neutral buffered formalin and stored a minimum of 24 hours at room temperature prior to further processing. Difficulties associated with handling and orientation of larvae can be mitigated by embedding each larva in agar prior to processing, enabling proper orientation while they are embedded in paraffin. When examination of eyes is the primary goal, whole larvae should be embedded cranial side down in a 2% agarose matrix until the agar has set, followed by standard processing. After processing, the agarose block containing the whole larva can then be embedded cranial side down in paraffin. Maintaining the whole fish in a perpendicular orientation enables microtomy of the head in the transverse plane. Serial sections taken from rostral to caudal, 4-µm thick, allows for all features of the eye to be captured. As an added benefit, serial sections can be readily obtained through the entire length of the larva permitting evaluation of other organ systems in conjunction with the eyes, particularly important in toxicological studies involving larvae.
Adult zebrafish eyes should be excised and embedded separately from other tissues. Eyes should be immediately excised following euthanasia and placed into modified Davidson’s fixative. Eyes should be stored at room temperature at least overnight but not longer than 48 hours prior to processing. There are marked anatomical differences between the dorsal and ventral regions of zebrafish eyes, and they need to be embedded in a manner that enables transverse sectioning so that both regions are captured in section. Following fixation, orientation is aided by use of a dissecting microscope to identify the ventral floor of the posterior segment. In the adult eye, the ventral floor is characterized by a silvery reflective layer, composed of iridophores that contain quinine-based crystals, which is an extension of a similar layer of iridophores present in the iris. 53 This reflective layer, termed the tapetum lucidum, is analogous in function to that observed in the posterior segment of some mammals, such as dog. A marking dye on the exterior surface (eg, lateral border) can then be used to ensure embedding is done in a dorsal-ventral plane. Following embedding, 5-µm transverse sections can be obtained for staining and examination.
Comparative Ocular Anatomy and Physiology
The lineage that led to ray-finned (teleost) fish split from that of mammals about 400 million years ago. However, the camera-style eye that today’s vertebrates inherited evolved 100 million years prior to that separation. 54,55 This common origin is reflected in anatomical and physiological similarities among the visual systems of zebrafish and mammals. Although there are several important differences of which investigators need be aware, the zebrafish visual system offers abundant opportunity for translational toxicology. Although larvae are typically used in ocular assays, adult zebrafish are utilized for investigating potential eye toxicity in wild fish in environmental toxicity studies. 56,57 As such, this review includes descriptions of embryonic, larval, and adult eyes.
During zebrafish and human embryogenesis, eye structures develop from 3 distinct embryonic layers. For example, the optic stalk, pigmented epithelium, retina, and ciliary body originate from the neuroectoderm. The lens, conjunctival epithelium, corneal epithelium, and corneal stroma originate from the surface ectoderm. The corneal endothelium, iris stroma, and sclera are derived from the mesenchyme that originates from neural crest cells. 58 Eye development that closely resembles that of human in a model with relatively large eyes and rapid, external, development have made zebrafish popular for studying developmental ocular abnormalities and teratogenicities. 59 –64
The timing and pattern of cellular differentiation in zebrafish eye tissues have been studied in great detail, 65 –71 as well as how these tissues become vascularized. 72 By the end of the third day post-fertilization, zebrafish eyes are mature enough to enable a light-evoked startle response, and to saccade (quick movement of the eyes) in response to a visual stimulus. 73 Structures readily apparent in larvae include the cornea, lens, iris, all layers of the retina including retinal pigment epithelium (RPE), optic nerve, choroid, and sclera. The vitreous and anterior chamber, and structures in the anterior segment are not yet developed. As described below, zebrafish eye tissues have similar gene expression, cellular and extracellular constituents, and architecture to those of human eyes. 65,74
The zebrafish cornea consists of all 5 layers found in the human counterpart: epithelium, Bowman’s layer, stroma, Descemet membrane, and endothelium. 71,75 The adult zebrafish cornea is around 20-µm thick. The epithelium, like that of human cornea, is composed of about 5 layers of squamous cells, the membranes of the outermost cells have microvilli and microplicae and are joined with tight junctions and desmosomes. These characteristics are key elements of the structural stability of the cornea. The epithelium is separated from the stroma by Bowman’s layer, a layer of collagen fibers that may contribute to the healing process after corneal injury in humans. 76 The zebrafish corneal stroma is approximately 6-µm thick and consists of about 30 layers of collagen fibers. Within each layer, the fibers are parallel to each other but perpendicular to that of adjacent layers. The stroma is bordered posteriorly by Descemet’s membrane, the collagenous basement membrane of the single cell endothelium layer. The corneal epithelium and stroma begin as thin tissues with relatively few layers and gradually thicken over time, obtaining mature thickness approximately 1-month post fertilization. The human cornea is 30× thicker than the zebrafish cornea. A major difference in corneal constitution between the 2 species is that the stroma makes up 90% of the thickness in human but only 40% in zebrafish. 71
Although the cellular and extracellular components and the general role of the zebrafish lens are like those of mammals, there are specific differences that need to be considered while interrogating translational toxicology. The key differences are in development, shape, and accommodation (change in lens shape that changes focal point). During development, the early mammalian lens placode invaginates to form a fluid filled vesicle, the walls of which are derived from ectoderm that will become the lens epithelium. After they differentiate, the posterior epithelial cells elongate to fill the vesicle with cells that will further differentiate into lens fibers. During zebrafish lens development, there is no vesicle. Instead, a small group of ectoderm cells develops posteriorly to the presumptive cornea and as this mass of cells grows, the internal cells differentiate into lens fibers while the external cells differentiate into the lens epithelium. 77 The mature zebrafish lens is spherical, not ellipsoid, and does not accommodate to change the focal point; the ciliary zone (CZ) is devoid of the muscles found in the human counterpart (cilary body) that control such changes. 70,75 Like in mammals, the lens fibers of the zebrafish are long cells, devoid of membranous organelles, arranged parallel to each other in concentric spheres. The membranes of fiber cells adjacent to one another are interlocked by regularly spaced digitations resulting in membrane enfolding by adjacent cells. 70,77 Posterior epithelial cells positioned along the equatorial region differentiate into new fiber cells as they do in the human lens. This margin of epithelial covering is however not positioned near the middle of the lens as in humans, but is located more posterior and medial, so that the zebrafish lens is covered to a greater extent with epithelium. The entire lens is covered externally by a thin basement membrane forming the lens capsule. 70,77
As the epithelial cells continually differentiate into fiber cells throughout the life of the fish, they lose their nuclei and other organelles. This takes place in a temporal and, importantly, spatial fashion. The area of organelle loss is restricted to the outer 30 to 40 µm (1/17 of the diameter) of the lens, and degrading nuclei are moved toward the equator of the lens as new fiber cells mature; as such, material with high potential to scatter light is moved out of the primary path of light. 58,65,70,73,77 Zebrafish lens fibers contain alpha, beta, and gamma crystallins. 78 The crystallin content increases with fiber age; mature fibers found in deeper layers of the lens are abundant in crystallins that contribute significantly to the refractive index of the lens. Thus, like humans, the zebrafish lens has a gradient of refractive indices; however, since zebrafish are unable to accommodate by other mechanisms, they are dependent upon this gradient to focus different wavelengths on the retina. 79,80
The zebrafish lens affords almost all light refraction to the eye, whereas in the eyes of most land-based mammals, including humans, corneal refraction plays a large role. The refractive index of water is very similar to that of the cornea making refraction at the water–cornea interface much lower than that of air–cornea interface. 65,70,77 The lens is suspended in place by zonular fibers that are connected to the ciliary body; in terrestrial mammals, dynamic changes to the shape of lens allowing for accommodation of focal distance is accomplished through contraction of the smooth muscle embedded in the ciliary body that circumscribes the anterior segment. Accommodation does not occur in zebrafish as neither the iris nor the ciliary body (ciliary zone in zebrafish, referred to as the CZ) contain smooth muscle. 70,75
Unlike human lenses, the anterior section of the zebrafish lens partially protrudes through the pupil into the anterior chamber. Light is blocked from the outer, most immature, layers of the lens by the iris, which serves reflective and camouflage functions. 53 The reflective nature is chiefly supplied from iridophore cells that contain guanine crystals, and the camouflage capability is from melanophores which, although located in a thin (5 µm) layer throughout the iris, are predominantly visible on the top of the eye so that light from above is absorbed by the melanin, and not reflected, which makes the fish less conspicuous from above. 53 Iridophores are arranged into 2 layers within the iris, and the arrangement and density of crystals within those 2 layers produces a silvery reflection termed the “iris argenta.” 53 The outer layer is more highly ordered than the inner layer and is continuous with a similarly ordered layer of iridophores located on the ventral floor of the posterior segment, external to the anterior retina. 53 As the zebrafish iris lacks muscle, contraction or dilation of the pupil does not occur. The pigmented cells of the zebrafish iris lie on the interior side, closest to the lens, as they do in mammalian eyes (Figures 1 and 2).
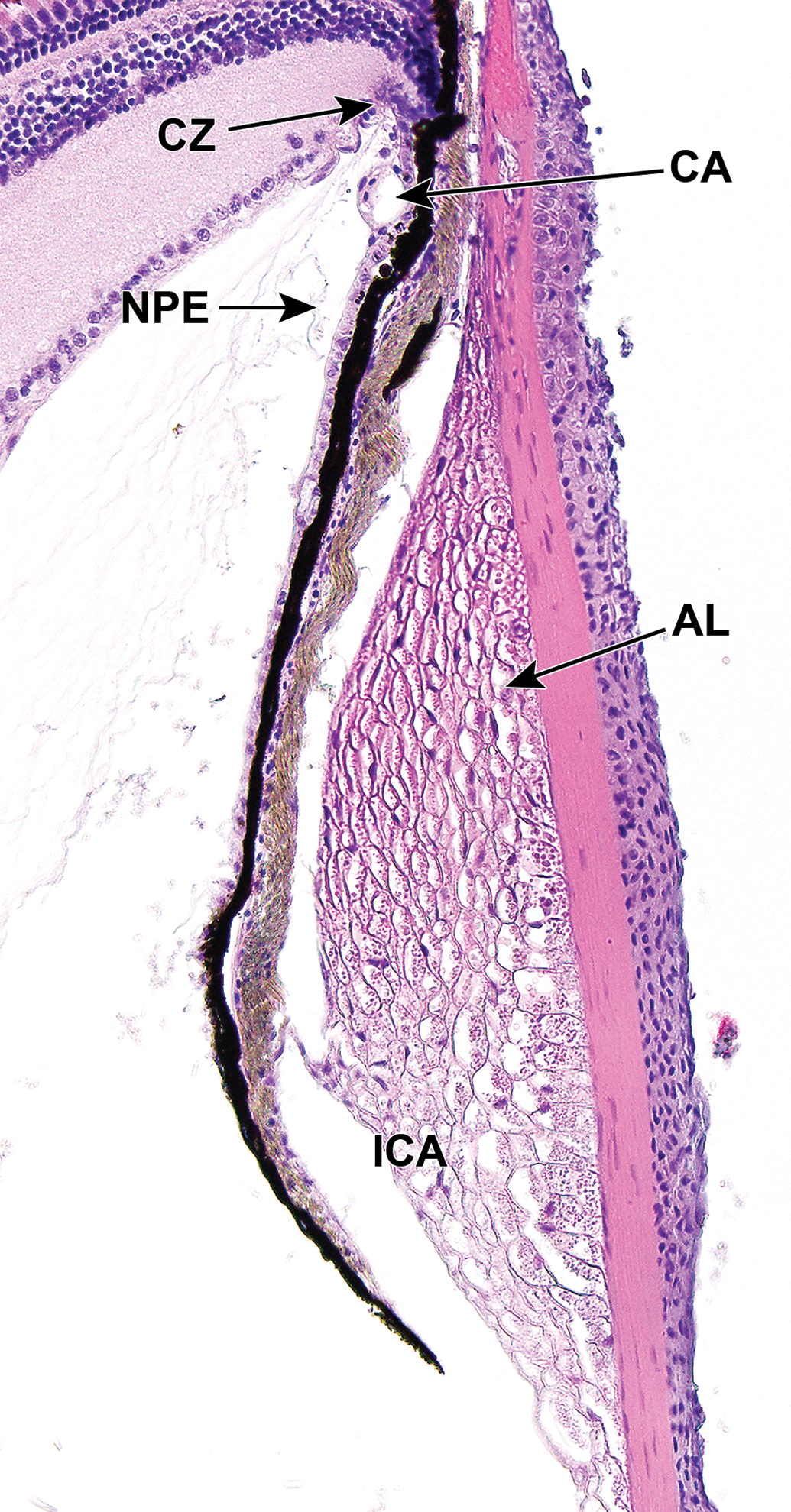
Dorsal iridocorneal angle (ICA) of an adult zebrafish. The ICA contains a circumferential meshwork of mesenchymal cells known as the annular ligament (AL), thought to provide stability to the eye. The AL is separated from the iris by a band of connective tissue (pigmented tissue; intervening gap is an artifact of processing). Note the layer of nonpigmented epithelium (NPE) on the posterior aspect of the iris, and a layer of dark pigment immediately posterior to the iris vasculature. There is no ciliary body; instead, a ciliary zone (CZ) is present at the base of the iris. CA indicates ciliary artery.
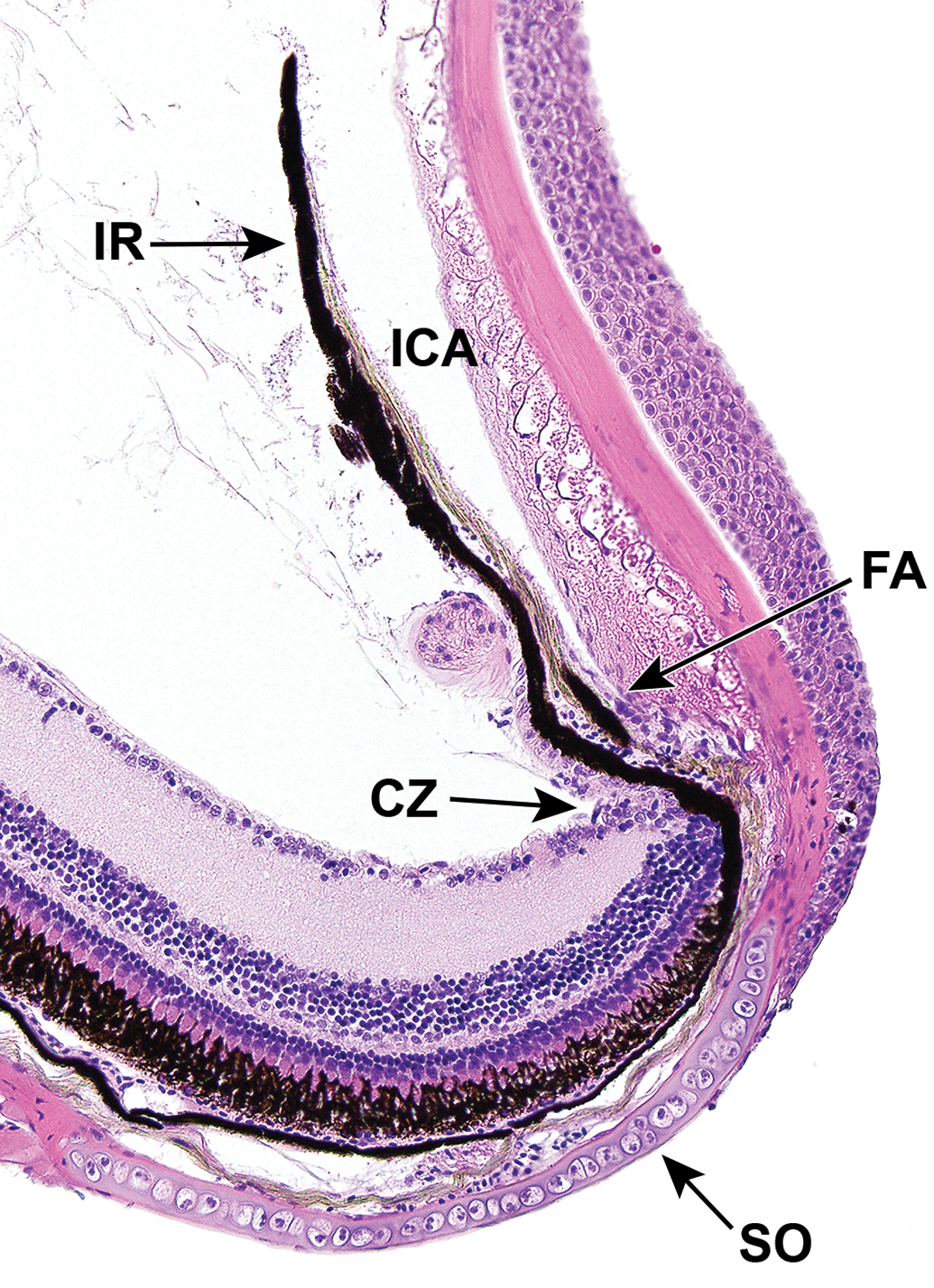
Ventral iridocorneal angle (ICA) of an adult zebrafish. The ventral ICA is narrower than the dorsal ICA, and houses the filtration angle (FA). The anterior aspect of the posterior segment is surrounded by a rim or cartilage known as the scleral ossicle (SO), which is embedded in the sclera. CZ indicates ciliary zone; IR, iris.
In most vertebrates, the cornea is separated from the lens by aqueous humor contained within the anterior segment. In human eyes, aqueous humor is produced circumferentially by epithelial cells of the ciliary body processes. The aqueous humor reaches the anterior segment by flowing through the pupil of the iris and leaves the eye through the ring-shaped trabecular network at the iridocorneal junction. After passing through the trabecular network, the aqueous humor is circumferentially collected in Schlemm’s canal which transports it into venous circulation. 81 The anatomy and physiology of the zebrafish anterior chamber is similar with some notable exceptions. The CZ of zebrafish does not have processes, and instead, aqueous humor is produced primarily by nonpigmented epithelial cells of the CZ located within the dorsal aspect of the eye. 70
Additionally, in zebrafish, an annular ligament (AL) resides within the iridocorneal angle (ICA; Figure 1). This is a circumferential meshwork of mesenchymal cells that is thought to have a structural role in the anterior segment of teleosts. 70,75 In zebrafish, the anterior surface is covered by a layer of endothelium that extends from the inner corneal surface; it’s postulated that the endothelium may have roles in absorption of ions, proteins, and metabolic byproducts. 70 Structural differences are present in the ventral aspect of the AL, which is narrower and funnel shaped, relative to the dorsal aspect. Within a narrow region of the ventral aspect of the ICA, a canalicular network that appears to be homologous to Schlemm’s canal is housed between the AL and the base of the iris on the anterior surface (Figure 2). Aqueous humor drains through this region which is continuous with the episcleral vasculature. 70,82 Another endothelium-lined canicular system, observable by electron microscopy, is located on the posterior aspect of the iris base and also feeds into the aqueous plexus. 70 The AL develops comparatively late, becoming apparent ∼17 dpf. Temporal changes in the anterior segment of the zebrafish eye during development from embryonic to adult stages have been studied in great detail. 70
The vitreous cavity is comparatively small owing in part to the larger space requirements of a spherical lens. The vitreous volume of the adult zebrafish eye has been estimated at 200 to 500 nL. 83 To the authors’ knowledge, the embryology of the zebrafish vitreous has not been described.
Ocular vascularization in zebrafish resembles that in mammals from embryonic origin to adult anatomy and physiology including cell signaling molecules, vacuolar-mediated transfer of nutrients, and genetic control. 72,84,85 In zebrafish eyes, vascularization is derived from an intraocular hyaloid system that is similar to humans, and a ciliary vascular system composed of superficial ocular vasculature and the choroidal vascular plexus. Although independently derived, live imaging of developing larvae indicates these systems become rapidly integrated through anastomoses and/or remodeling during development. 86 The choroidal vasculature supplies the iris, the ciliary body, and the RPE. 70,85 It is thought that the photoreceptors also depend upon the choroidal vascular system in zebrafish, which is not the case for mammalian eyes. 72 In mammals, these cells are nourished by the retinal vasculature, which consists of capillaries that branch into the inner and outer plexiform layers, following the regression of the embryonic hyaloid vasculature. 72,84,85,87 In zebrafish, the hyaloid system develops from the optic artery 24 to 29 hpf, initially as a single loop that enters the optic fissure as the hyaloid artery, extending to the posterior surface of the lens, and exits as the hyaloid vein. Unlike mammals, the hyaloid vessel does not regress in zebrafish, but gradually retracts from the lens, and develops into a plexus that comes to reside on the inner retinal surface. Through remodeling, the hyaloid system develops into retinal vasculature residing in the ganglion cell layer. 58,75 It is thought that since the zebrafish retina is comparatively thin, diffusion from the retinal surface and choroidal vasculature is sufficient to reach all retinal layers. 72
The zebrafish retina is morphologically (Figure 3) and functionally similar to the mammalian retina. 88 Neurogenesis is initiated in the central region of the retina and is replete with all layers and neuronal subtypes by 3 dpf. 89 The retina develops rapidly and is capable of generating measurable electrophysiological potentials by 72 hpf, at which time the embryo is visually responsive. 58 After 5 dpf, the retina is fully functional. 75,90 Morphological and functional similarities to mammalian visual system, coupled with rapid development positions zebrafish as a reasonable high-throughput ocular model for small-molecule screening of retinal toxicity.
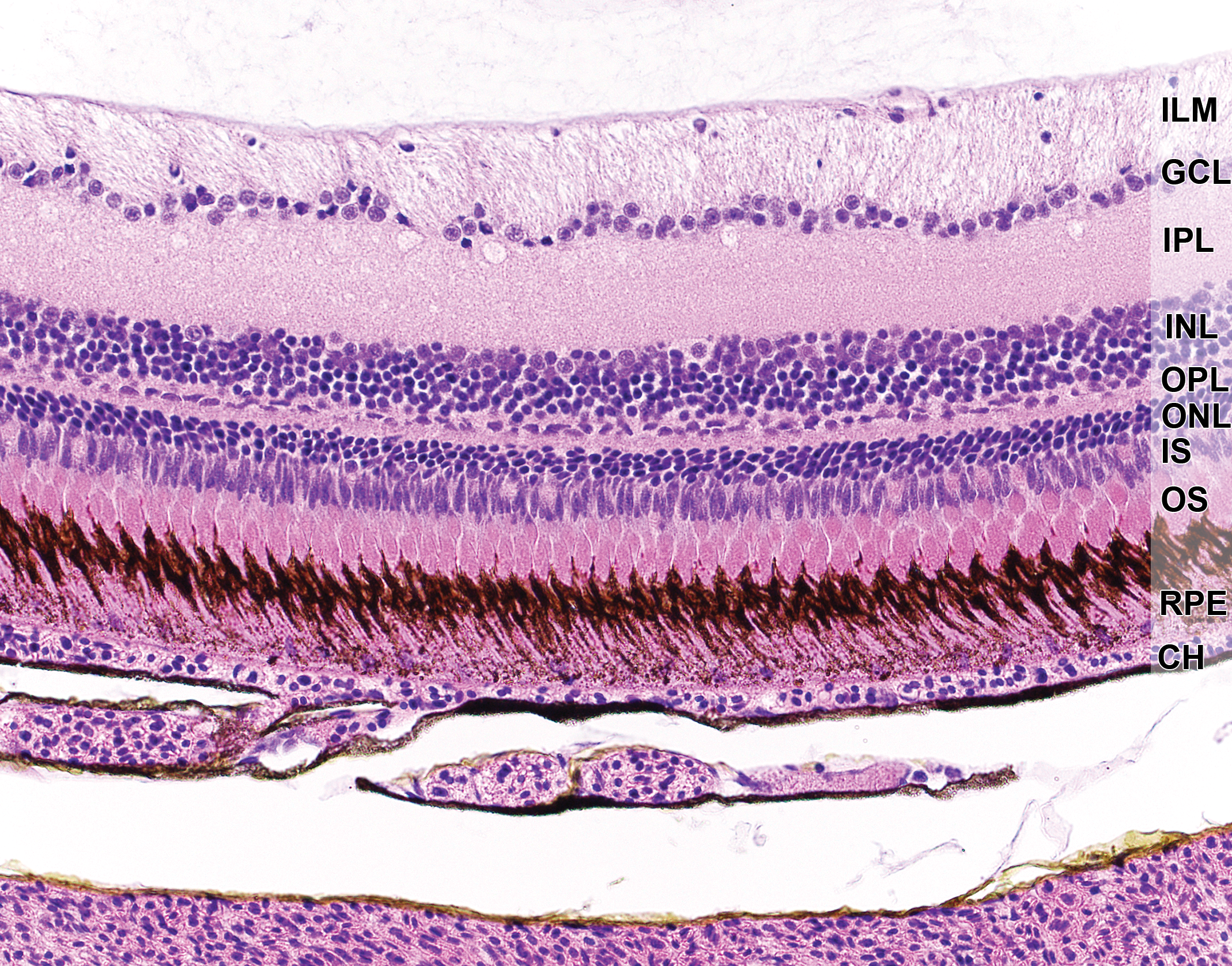
Retina, RPE, and choroid of an adult zebrafish. Note the long, pigmented microvilli of the RPE, which interdigitate with the outer segments (OS) of the photoreceptors. CH indicates choroid capillaris; GCL, ganglion cell layer; ILM, inner limiting membrane; INL, inner nuclear layer; IPL, inner plexiform layer; IS, inner segments; ONL, outer nuclear layer; OS, outer segments; RPE, retinal pigment epithelium.
Retina cell differentiation is observable first in ganglion cells, which appear by 32 hpf, with first axons reaching the optic tectum 32 to 40 hpf. 58,75,91 Ganglion cell production is rapidly followed by development of cells within the inner nuclear layer. Molecular profiling of the teleost retina has led to the identification of at least 17 types of bipolar cells, 4 types of horizontal cells, and up to 70 amacrine cell types. 75 Connections between the cells of the inner nuclear layer and connections to photoreceptors and ganglion cells are complex and beyond the scope of this review; the reader is referred to more comprehensive coverage. 92,93
The zebrafish retina contains 5 types of photoreceptors consisting of one rod, sensitive in low light conditions, and 4 cone types, sensitive across the light spectra in red, green, blue, and ultraviolet zones. Red and green cones form “double cones” through fusion of their inner segments, while cones responsive to “long” blue and “short” ultraviolet wavelengths are single. 75 The highest density of cones is found within a specialized region of the temporal retina known as the “area temporalis,” postulated to provide increased visual acuity in the anterior visual field. 58 In this regard, the area temporalis appears analogous to the fovea centralis of dogs, visual streak of rabbits, and the primate fovea. Cone development in neurogenesis precedes that of rods, and thus larval vision is predominated by cone function. In the dark, functional retinal responses rapidly and progressively diminish, and larvae become nonresponsive to visual stimuli. 94 Diminished visual response is attributed to disassembly of ribbon synapses with on-center (ON; light-active) bipolar cells, and loss of neurotransmitter secretion. 94 In low light conditions, the larval integument darkens in a manner that provides camouflage, referred to as visual background adaptation, caused by the dispersal of melanosomes (melanin-filled vesicles) throughout the dorsum (see Figure 6). 95 The ability of larval fish to camouflage under low light conditions is suggestive of a coevolutionary compensatory survival mechanism to diminished vision.
As in the mammalian retina, Müller cells are the primary glia cells and provide architectural support to the retina. Although the cell body resides in the inner nuclear layer, processes extend to the outer limiting membrane, inserting between, and surrounding groups of photoreceptor processes in the outer plexiform layer. They extend to the internal limiting membrane where the endplates form a basement membrane. 96 Müller cells are accorded numerous critical roles to retina function 97 including homeostatic and metabolic support to photoreceptors, regulation of extracellular fluid and maintenance of blood ocular barriers, regulation of potassium distribution and synaptic activity of the inner nuclear layer (that contributes to the b-wave on electroretinography [ERG]), retinoic acid synthesis, glutamate uptake and recycling, release of neurotransmitters, and phagocytosis of retinal debris.
Müller cells display plasticity and regenerative potential during retinal injury. Retinal injury in mammalian eyes often results in Müller cell gliosis. However, activated Müller cells produce and secrete growth factors that can result in cellular transdifferentiation, proliferation, and migration, resulting in formation of membranous or fibroblastic profile of tissue. 98 To the authors’ knowledge, this has not been described in zebrafish; however, a well-described phenomenon in zebrafish is that Müller cells maintain stem cell capabilities and have an additional role in continuous retinal neurogenesis and retinal replacement (see below).
A remarkable feature of the teleost retina is the persistence of neurogenesis throughout life which is thought to be related to the continuous growth of the fish and enlargement of the eye.
58,75,89
Progenitor cells of retinal neurons have been identified through their expression of homeobox
The optic nerve is composed of axons from retinal ganglion cells that cross at the optic chiasm and project contralateral to the optic tectum similar to mammalian eyes. Axons project topographically and terminate into one of 4 laminae of the optic tectum. 91 Throughout life, retinal ganglion cells are replenished from the peripheral retina. In models of cell injury or optic nerve transection, both retinal ganglion cells and the optic nerve exhibit robust regeneration with functional recovery. 91
The RPE is derived from the neuroectoderm and forms a single mosaic layer posterior to the photoreceptors (Figure 3). Microvilli, which interdigitate with the outer segments of the photoreceptors, are remarkably long and densely packed with melanosomes in zebrafish. Zebrafish RPE contain 2 types of melanosomes, round and cylindrical, that appear at 2 dpf. Cylindrical melanosomes reside predominantly within the RPE microvilli. Their shape and location are postulated to maximize photoreceptor light access, reduce backscatter, and provide structural support to the microvilli. 110 The zebrafish RPE also demonstrates regenerative potential, evidenced in ablation models. 111 The peripheral RPE serves as the source of replacement of those cells that are lost, and proliferation proceeds centrally until restoration is complete. In extensive injury, morphological and functional RPE replenishment occurs within 1 to 2 weeks or 1-month postinjury in larvae and adult zebrafish, respectively. 111 In other respects, the zebrafish RPE is similar to that of other vertebrates, including having a primary role in maintenance of the blood–ocular barrier, phagocytosis of shed outer photoreceptor segments, retinoic acid recycling, and metabolic and nutritional support to the retina.
The sclera is a fibrous outer tunic that originates at the limbus and extends to cover the posterior region of the eye. Composed primarily of interwoven bundles of collagen, the sclera gives the eye shape, provides protection from injury, and has a role in maintaining ocular pressure. A rim of cartilaginous tissue (scleral ring) is embedded in the stroma and encircles the eye anteriorly (Figure 2); the interior side of the stroma is covered by membranous tissue containing pigmented cells and elastin. 75 The outer surface of the sclera (episclera) is populated with microplicae that contribute to the production of a glycocalyx that lubricates the eye. 75
Functional Vision Testing
The benefits brought to ocular toxicology by a tiny vertebrate with clinically translatable anatomy and physiology are augmented by the amenability of zebrafish for visually based behavioral analyses. 88,112 Quantifying behavioral responses to visual stimuli can noninvasively expose a potential ocular lesion or reveal toxicities impacting vision that do not have obvious histological correlates. 88 This can be done quantitatively and in high-throughput fashion using larval zebrafish. 113,114 Several behavioral responses to visual stimuli, described below, have been characterized, including optomotor response (OMR), optokinetic response (OKR), visual startle, and visual motor response (VSR and VMR, respectively). These behavioral responses have been used in toxicology as well as in studies designed to discover what types of visual cues play a role in driving behavior, 115,116 genes involved in vision and vision disorders, 117 –119 and by using in vivo functional imaging, which neurons/circuits are involved in perceiving visual stimuli and eliciting the behavioral response. 120,121
The OMR assay takes advantage of the instinct of larval zebrafish to swim in the direction of optic flow, thereby minimizing perceived displacement. This can be performed using freely swimming or immobilized larvae. 116,119,122 The heads of the immobilized larvae are typically embedded in agarose, while the trunk/tail is allowed to move. The OMR is typically stimulated from a video that displays moving stripes, or other patterns, that alternatively present light/dark settings viewable from below and/or alongside the larvae. Larvae instinctively swim in the same direction as the pattern moves. Response can be scored from video recordings of swimming behavior (typical for immobilized larvae) or by recording position with respect to stimulus direction. The latter technique can be conducted in high-throughput fashion using 384-well plates. 114 Using OMR, it was recently demonstrated for zebrafish, as has been shown for mouse, 123 that dynamic visual experience is required for the proper stratification of retinal layers in early eye maturation. 124 Studies have shown that the OMR displayed by larval zebrafish is lost around 16 dpf and that adult zebrafish rely mainly on mechanosensory stimuli via the lateral line for sensing movement, and on positive rheotaxis (defined as the tendency to turn to face into an oncoming current) for maintaining position in flowing water. 125 Larval zebrafish also display positive rheotaxis.
The OKR assay assesses the movement of zebrafish eyes as they follow a moving stimulus (typically alternating dark and light stripes) that rotates horizontally into and then out of the field of vision. 126,127 This is typically performed on fish immobilized in body (by embedding the fish in methylcellulose), but not in eye, movement, and results in a series of slow tracking movements followed by fast repositioning movements. The repositioning is called a nystagmus and relies on communication between the retina and the pretectum. 128 Thus, like the OMR, the OKR relies on functional vision, central nervous, and musculoskeletal systems. The OKR can be scored manually by direct observation, or from video recordings, and recently an automated scoring tool was released. 129 In zebrafish, the optical system is fully capable of OKR after 96 hours of development. 127 Among others, zebrafish OKR studies have revealed neuronal signaling pathways, 130 –133 pharmacological and environmental impact, 134 –137 and genetic players and mechanistic pathways 130,132,138 –145 involved in vertebrate vision.
The VSR assay is typically performed in multi-well plates in high-throughput fashion, monitoring one embryo or larva per well. 73,146 The movement of the fish within 1 to 2 seconds of a dark/light transition is quantified from video using commercially available software capable of processing data from all wells simultaneously. 113 The responses are often compared to responses invoked by an auditory or other stimulus with the aim of confirming a change in visual acuity over gross motor skill defects as the reason for a loss of response to light (Figure 4). 50 A similar assay, the VMR, measures activity over multiple periods of light and dark, yielding information about startle response as well as circadian behavior. 147 Since the VSR and VMR are suited for high throughput, they have been used in experiments designed to discover promising therapeutic compounds, 147 toxicities, 148 as well as genes associated with vision 62,117 and ocular diseases. 149
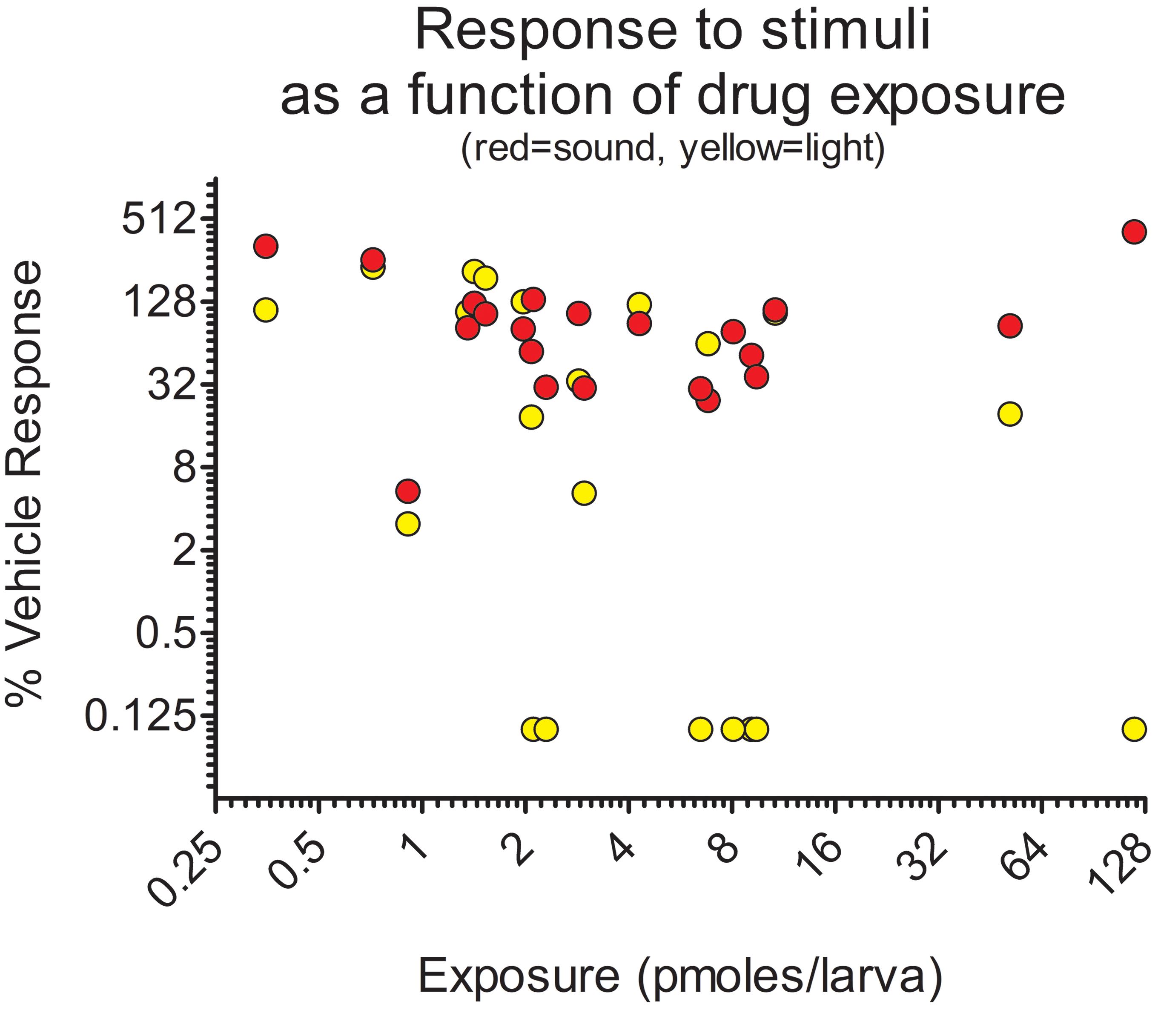
Relative startle responses of zebrafish larvae to light (yellow) or sound (red) as a function of exposure to various dose levels of exploratory drug candidates (small molecule inhibitors of nicotinamide phosphoribosyltransferase [NAMPT]) which were associated with retinal histopathology. As chemical exposure increases, the relative response to light tends to decrease whereas the response to sound does not, indicating an ocular-specific toxicity. Copied with permission from reference 50 and Oxford University Press, copyright 2017.
Chemical Toxicity Research
Studies using zebrafish have exposed and characterized ocular toxicities associated with chemicals of various sources impacting different tissues (Table 1). The literature has many examples, detailed below; however, 70% of those studies have been reported only during the past 5 years, indicating that the use of the model in this field is still in its larval stage. Outside of chemical toxicology, zebrafish studies focused on eyes are also adding to our understanding of radiation toxicity, 150,151 ocular diseases, 152 –154 retinal genetics and regeneration, 155 –158 as well as aiding therapeutic discovery. 159 –161
Published Ocular Toxicity Studies That Used Zebrafish as an Animal Model, Sorted According to Life Stage(s) Used.
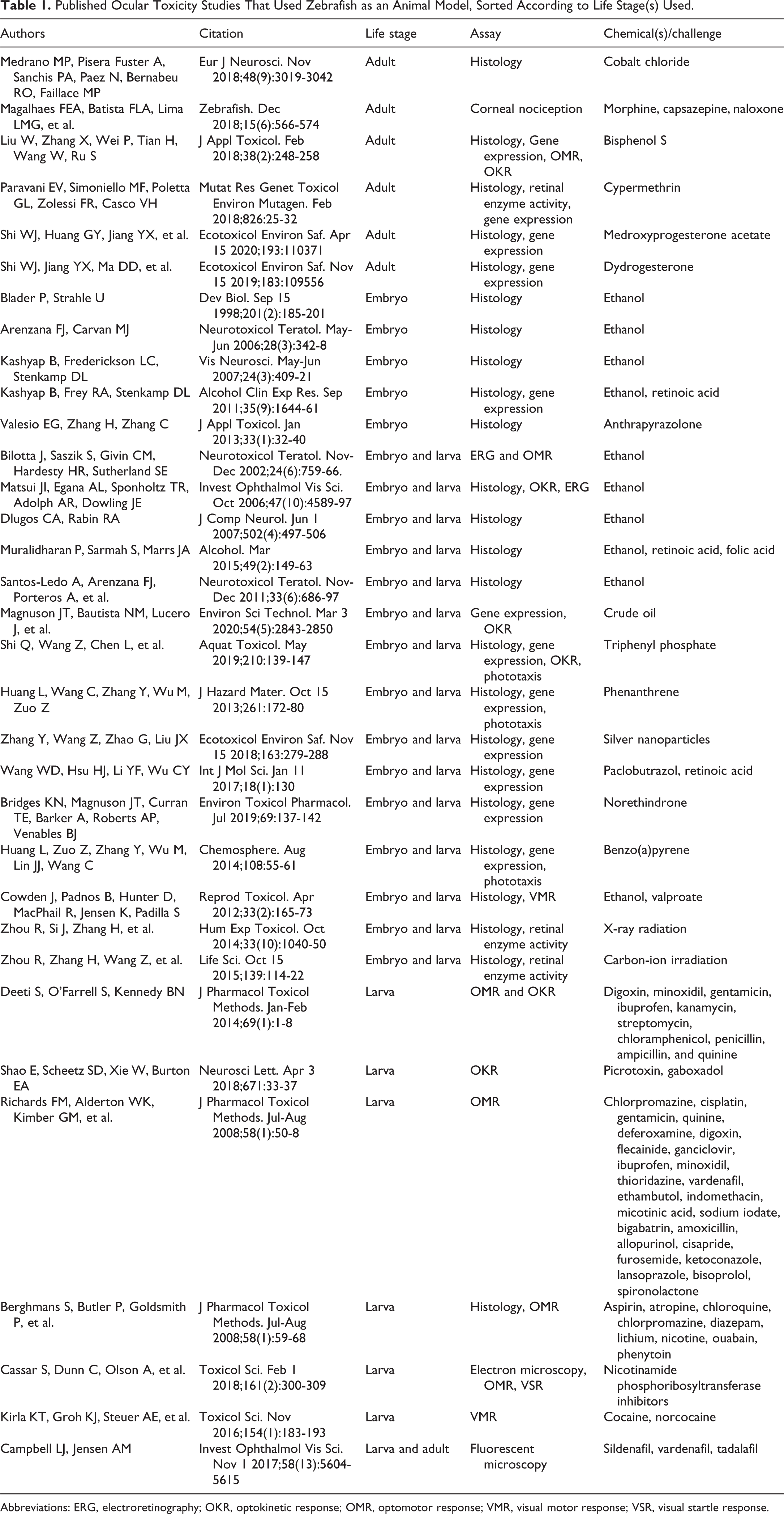
Abbreviations: ERG, electroretinography; OKR, optokinetic response; OMR, optomotor response; VMR, visual motor response; VSR, visual startle response.
An exception among areas of ocular toxicology studied using zebrafish is how ethanol effects the developing embryo; this has been studied for more than 40 years. 162,163 The eye is a primary system impacted by ethanol during development, and ocular issues are common among children with fetal alcohol syndrome. 164 Early studies indicated translatable toxic end points from ethanol, such as microphthalmia, altered retinal physiology, loss of retinal function (electroretinogram), and visual acuity (OMR). 162,165 –168 Other studies have revealed details of the physiological effects and mechanisms of ethanol embryotoxicity on the zebrafish eye, 169 –171 how exposure time and timing impact the toxicity, 172 –174 and the cell signaling pathways and genetics involved. 175 –178
Human activities impact all life and environments. We are under-researching the effects of our pollution on the planet. 179 Research on the effects of pollution on eyes and visual systems is no exception. 10 Recently, zebrafish studies have contributed to our understanding of the potential effects of pollutants on wildlife and humans. 180 –182 Some of these studies have focused on ocular toxicities caused by industrial pollution on the developed eye, 56,183 –185 while others report on pollutants impacting eye development. 64,186,187 Pharmaceutical pollutants are also being investigated for ocular-specific effects using zebrafish. 188 –190 Additionally, there are recent reports of the use of toxicogenomics to elucidate mechanisms of ocular toxicity from industrial and pharmaceutical pollutants. 191,192
New drug discovery currently depends on safety testing that uses animals. Efforts to refine procedures on, reduce the number of, or replace animals in this research are continually being pursued. 193 Testing on larval zebrafish is one of these efforts because it can reduce the amount of research that uses adult mammalian models and, due to their transparency and amenability to transgenic expression, zebrafish offer opportunities for refined research that uses intact and living organisms to acquire knowledge on cellular, tissue, and organ toxicities. 9 The eye is no exception; it has been demonstrated that drugs with established ocular toxicities, such as chlorpromazine, cisplatin, gentamicin, quinine, deferoxamine, minoxidil, thioridazine, and vardenafil, among others, impact zebrafish vision. 134,135,194 Collectively, the results of those studies indicate that zebrafish assays are sensitive 68% to 83% of the time and specific 75% to 100% of the time. Accordingly, the potential for mammalian ocular toxicity for drugs and drug candidates may be predicted from zebrafish studies. Indeed, there are some examples, also demonstrating that toxic mechanisms might be disclosed from the same studies. Cowden et al 195 investigated the embryonic neurotoxicity of valproate, an anticonvulsant, and an established teratogen. They demonstrated that subteratogenic levels of valproate impact larval zebrafish locomotor activity and decrease the retinotectal projection area in the optic tectum. Valesio et al 196 demonstrated eye-specific toxicities from embryonic exposures to SP600125 (anthrapyrazolone), an exploratory therapeutic immune modulator designed to interfere with c-jun N-terminal kinase. An off-target interaction between SP600125 and the aryl hydrocarbon receptor, discovered in cell culture by other groups, 197,198 compelled the zebrafish study which reported microphthalmia and ectopic, multiple lenses in exposed embryos. In a mechanistic study, Campell and Jenson 199 demonstrated that 2 common drugs used for erectile dysfunction, sildenafil and vardenafil impact rod outer segment (ROS) renewal in the zebrafish retina. Both drugs inhibit the activity of phosphodiesterase (PDE) 6; sildenafil is a cosuppressor of PDE5/6 and vardenafil inhibits only PDE6. The authors report that tadalafil, a PDE5-specific inhibitor, had no effect on ROS renewal. They used zebrafish expressing an inducible fluorescent marker in the ROS to allow tracking of tissue turnover. Cassar et al 50 reported that larval zebrafish studies predicted rat and dog retinal toxicities associated with several inhibitors of nicotinamide phosphoribosyltransferase (NAMPT; Figure 5). These compounds were discovered as potential chemotherapies from cell culture experiments with human cancer cells and were identified as NAMPT inhibitors by affinity capture. 200 Larval fish exposed to the compounds failed the OMR assay and lost their VSR in a dose-dependent fashion. The retinal ganglion cell and inner nuclear layers were the most impacted by the treatments, and the larvae lost their capability for visual background adaptation (Figure 6), which, in healthy larvae, is thought to be driven primarily through signals from the ganglion cell layer. 201 The relative potencies of the compounds to impact zebrafish vision correlated with their potency to kill human cancer cells, implicating an on-target mechanism, and demonstrating translatable pharmacology. Additionally, translatable ADME was reported; as was observed in rat and dog, the compounds accumulated in the eye of zebrafish to a greater extent than in other tissues examined. This may be due to an interaction with melanin, as has been suggested for cocaine in a zebrafish model. 202
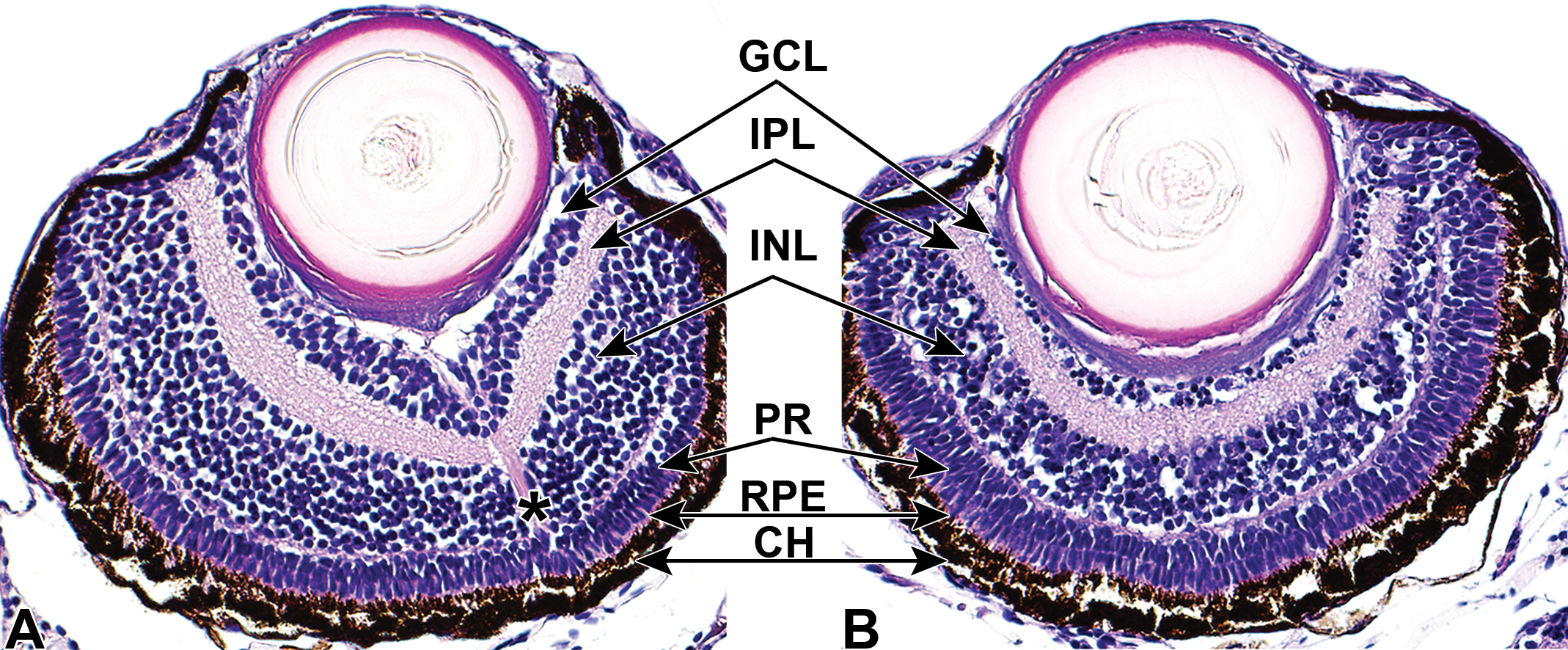
Representative ocular histology (A and B) of 7-day-old zebrafish larvae following treatment with vehicle control or a nicotinamide phosphoribosyltransferase (NAMPT) inhibitor. At this stage, the anterior chamber and vitreous are poorly developed. Vehicle control treated eye (A) depicts normal retinal features for stage of development. Decreased cell layers and numbers of nuclei are depicted in the nicotinamide phosphoribosyltransferase (NAMPT) inhibitor treated eye (B) in both the ganglion cell and inner nuclear layers. The inner plexiform layer has a disorganized, vacuolated appearance associated with cell loss in adjacent layers and their cell processes or axons. Optic nerve is designated with an asterisk, observed in section of control eye. Copied and adapted with permission from reference 50 and Oxford University Press, copyright 2017. CH indicates choroid; GCL, ganglion cell layer; INL, inner nuclear layer; IPL, inner plexiform layer; PR, photoreceptor layer; RPE, retinal pigment epithelium.
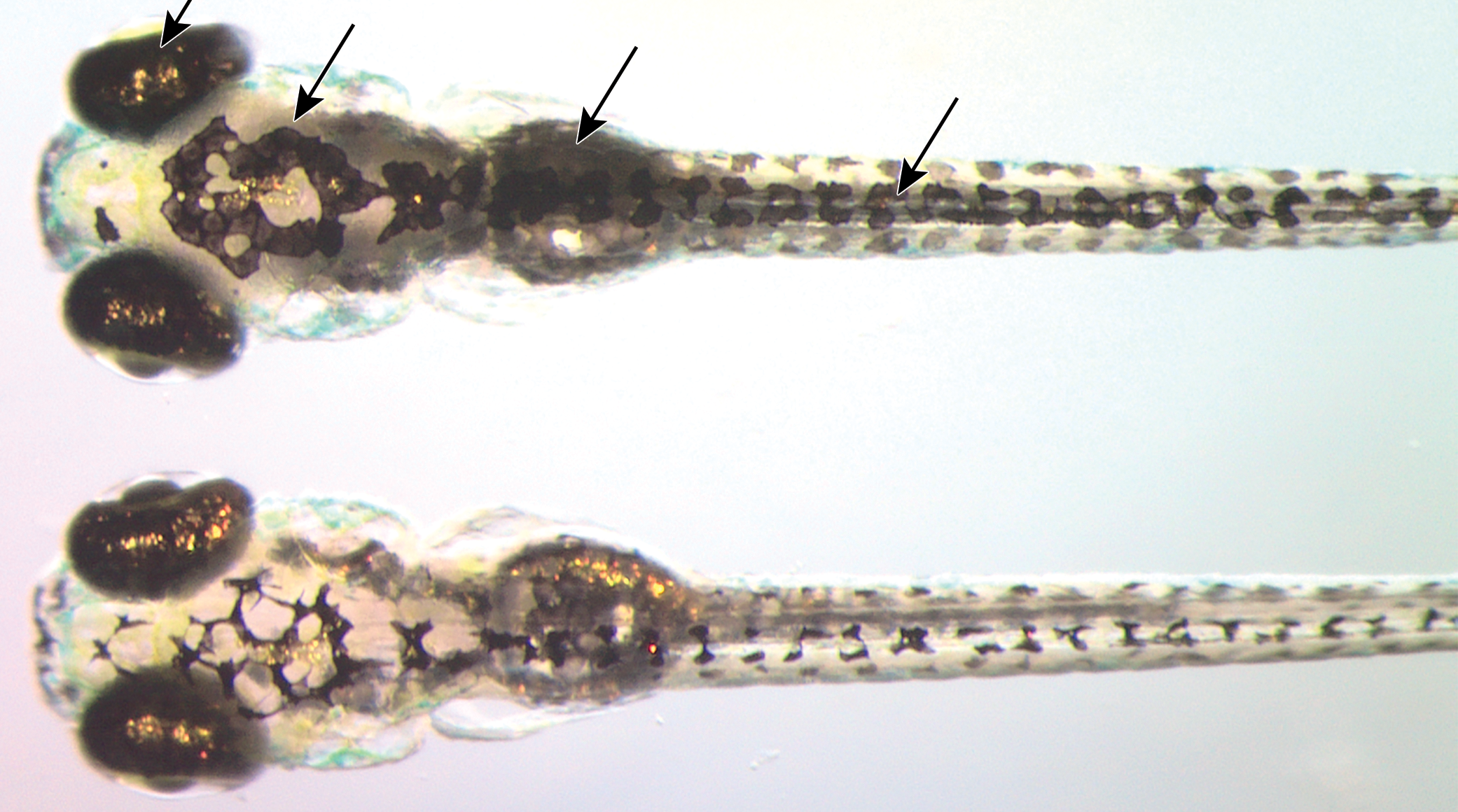
Representative dorsal images of 7 dpf zebrafish larvae following treatment with (top) a nicotinamide phosphoribosyltransferase (NAMPT) inhibitor associated with retinal histopathology, or (bottom) vehicle. Increased visible melanin can indicate a visual deficit (perceived low/no light). The image was captured during lights-on when melanosomes are typically contracted in response to retinal signals; this is manifested in a lighter appearance, as observed in vehicle-treated fish (bottom). Arrows mark increased visible melanin in skin. Copied with permission from reference 50 and Oxford University Press, copyright 2017.
Conclusion
Given the large amount of translatable biology and the high tractability of this model, research with zebrafish in the field of ocular toxicology is surprisingly low. Although many examples can be found, they are not as numerous as might be predicted. Only about 30 articles are retrieved from a PubMed search using the search phrase “zebrafish eye toxicity” for each of the last 2 years (2018, 2019), compared with over 800 using the phrase “eye toxicity.” However, the numbers are growing so that, at the time of this writing (August 2020), publications with zebra ish represent 5% of publications on eye toxicity for this year, compared to 1% from 10 years ago. This is an underutilized model that, if employed more extensively, can significantly contribute to our growing understanding of ocular toxicology in the years to come.
Animal Welfare
AbbVie is committed to the internationally accepted standard of the 3Rs (Reduction, Refinement, Replacement) and adhering to the highest standards of animal welfare in the company’s research and development programs. Animal studies were approved by AbbVie’s (Institutional Animal Care and Use Committee or Ethics Committee). Animal studies were conducted in an Association for Assessment and Accreditation of Laboratory Animal Care-accredited program where veterinary care and oversight was provided to ensure appropriate animal care.
Footnotes
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: All authors are employees of AbbVie and may own AbbVie stock. AbbVie sponsored and funded the study; contributed to the design; participated in the collection, analysis, and interpretation of data, and in writing, reviewing, and approval of the final publication.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
