Abstract
A novel galacto-oligosaccharide (GOS) was administered by gavage to groups (10 males and 10 females) of Sprague-Dawley specific pathogen-free rats for 6 weeks from day 4 after birth at doses of 0, 500, 1000, or 2000 mg/kg/day. Each pup was subjected to a variety of observations to examine for development effects/changes after birth: general condition, clinical signs, functional examinations, grip strength and spontaneous movement, body weight and feed consumption, external differentiation, ophthalmological examination, urinalysis (including water consumption), hematology, blood chemistry, necropsy, organ weight, and histopathology. During the study period, no deaths occurred in any group and there were no observed effects from administration of GOS. Therefore, it was concluded that GOS had no effects on the development of animals 4 days after birth. Since, there were no abnormalities due to administration of GOS in the macroscopic examination, organ weight or histopathology of the reproductive organs or differentiation (incisor eruption and eyelid opening) of males or females, it was concluded that repeated oral administration of GOS at 2000 mg/kg/day for 6 weeks from day 4 after birth had
Introduction
Prebiotics, including materials like the indigestible carbohydrate galacto-oligosaccharides (GOSs), are often included in Western diets because of safety, resistance to digestion in the upper bowel, and their fermentability in the colon as well as their ability to increase beneficial enteric bacteria. 1 –12 In addition, GOSs promote the growth of beneficial bacteria such as bifidobacteria and lactobacilli while inhibiting the growth of virulent bacteria (enteropathogenic Escherichia coli) in the gut. 2 –5,8,9,11,13,14 Bifidobacteria and lactobacilli also help maintain a balance of intestinal organisms by producing lactic acid and acetic acid, which increase the acidity of the intestine, inhibit growth of harmful bacteria, and as a result, improve symptoms of diarrhea and constipation.
Infants fed with human breast milk, which contains approximately 100 times more GOSs than cow’s milk, generally have an intestinal microflora dominated by bifidobacteria, while the number of these organisms was much lower in infants fed with infant formula. 11,13 –18 In addition to adding GOSs to infant formulas to reinforce some of the functional attributes associated with breast milk, GOSs are added to a variety of adult foods including dairy products, soups, breakfast cereals, beverages, snack bars, ice creams, and bakery products.
GOS is assimilated by enteric bacteria, especially bifidobacteria in vitro and increases bifidobacteria in the human intestine. 18 GOS has been reported to be safe in humans following repeated intake. 19
In this report, the safety of this novel GOS was demonstrated further in a 6-week oral gavage toxicity study in juvenile Sprague-Dawley specific pathogen-free (SPF) rats of both sexes.
Methods
Compliance statement
These studies were performed in compliance with “OECD Principles of Good Laboratory Practice” (Organisation for Economic Co-operation and Development (OECD) Council: November 26, 1997). The Ethics Committee for Animal Experiments at Bozo Research Center approved this study based on the following: (1) “Law Concerning the Protection and Control of Animals” Law No. 105, October 1, 1973, Finally Revised on June 2, 2006 as Law No. 50; (2) “Standards Relating to the Care and Management of Laboratory Animals and Relief of Pain” Notification No. 88 of the Ministry of the Environment, Japan, April 28, 2006; and (3) “Guidelines for Proper Conduct of Animal Experiments” (Science Council of Japan, June 1, 2006).
Test article
The GOS used in this study is a sugar manufactured by a two-step enzyme reaction of lactose with Sporobolomyces singularis (first step) and β-galactosidase derived from Kluyveromyces lactis (second step) and was supplied by Yakult Central Institute for Microbiological Research, Yakult Honsha Co, Ltd, Japan. The brix sugar content of the GOS was 75%, and the sugar composition of the GOS used in this study was 56.9% GOS and 43.1% monosaccharides plus lactose. Stability analysis showed that the test article (GOS) was stable at room temperature over the administration period of the study.
Preparation of dosing solutions
Distilled water suitable for injection according to The Japanese Pharmacopoeia (D.W., Lot No. 1A76 and 1B72; Otsuka Pharmaceutical Factory, Inc, Japan) was used as the control article.
A requisite amount of the test article (GOS) was weighed in a beaker and dissolved in distilled water to a specified volume. After mixing, the test solution was divided into 1-day aliquots and stored in brown glass bottles in a refrigerator until used. The concentration of GOSs in the test solution was verified by high-performance liquid chromatography using both an exclusion-type ion exchange column and a normal phase column (LC 10 Series, Shimadzu Corporation, Japan).
Dosing solutions were stable for 8 days at refrigeration temperature and for 24 h at room temperature.
Juvenile rat study
Sixteen pregnant female SPF Sprague-Dawley rats (Crl: CD(SD), Atsugi Breeding Center, Charles River Laboratories, Japan, Inc., Japan) were obtained on day 18 of gestation, quarantined, and acclimated to the facility. Dams were observed for general condition and behavior once every day until delivery. Dams were individually identified by ear tags. For live born pups, individual identification was by marking the number of animal on the dorsal skin using an oil-based ink on the day of grouping, and by ear tags on the day of weaning (day 21 after birth).
Animals were housed in an animal room in which the temperature was maintained at 22 ± 3°C, the relative humidity at 50 ± 20%, the air ventilation at 10 to 15 changes per hour, and 12-hour illumination per day (07:00 to 19:00). During lactation, pups were housed with dams as a litter in plastic Econ cages (CLEA, Japan, Inc., Japan) with bedding (Lot No. 2011-1, Charles River Laboratories, Japan, Inc.). After weaning, animals were housed individually in bracket-type metallic wire-mesh cages (Nihon Cage Co., Ltd, Japan). Animals were allowed free access to a pelleted CRF-1 diet (radiation sterilized, Oriental Yeast Co., Ltd., Tokyo, Japan, Lot Nos. 110106 and 110302) via stainless steel feeders and to tap water (Gotemba City, Japan) using water bottles during the lactation period and the automatic water supply system after weaning.
Each lot of feed and bedding was analyzed for contaminants by Eurofins Scientific Analytic (Japan). The water supplied to the animals was analyzed by Shibaura Semtek Co., Ltd (Japan) on a quarterly basis in accordance with the Japanese Waterworks Law and satisfied the criteria prescribed in the standard operating procedures (SOP) of the test facility. Analytical results of the feed and bedding including concentrations of contaminants such as residual pesticides were confirmed to satisfy the criteria prescribed in the SOP of the test facility.
The number of live born and stillborn pups was counted. Pups’ sex was noted and they were also observed for external abnormalities and mortality once a day until day 3 after birth. There were no stillborn pups or pups with external abnormalities. The number of live born pups per litter was 11–16, the number of pups that died between the day of birth and day 3 after birth was 3, and the sex ratio of male and female pups was approximately 1:1. A total of 40 male and 40 female pups were selected based on body weight for inclusion into the control group and into one of the three test groups. Administration of the test material (GOS) was initiated from day 4 after birth. The body weight range at the start of dose administration for all groups was within the mean ± 20% for both males and females. Remaining animals were euthanized by asphyxiation with carbon dioxide.
Pups were dosed daily for 6 weeks orally by gavage using flexible stomach tubes from day 4 after birth up to day 45 after birth at 0, 500, 1000, and 2000 mg/kg/day. Animals in the control group received the vehicle, that is, distilled water. Individual dose volume for all animals was calculated based on the animal’s most recently recorded body weight at the time of dosing.
Animals were weighed on days 1, 4, 7, 10, 14, 17, 21, 24, 28, 31, 35, 38, and 42. At necropsy, animals were weighed after fasting for approximately 16 h in order to calculate relative organ weight. Cumulative feed consumption was measured for days 21–24, 24–28, 28–31, 31–35, 35–38, and 38–42 of administration and mean daily feed consumption per animal was calculated. One-day water consumption from the previous day was measured using water bottles at the time of urinalysis in week 6 of the study. All animals were observed for incisor eruption on days 8 and 11 of administration and for eyelid opening on days 11 and 14 of administration. Ohthalmological examination was performed on day 42 with a direct ophthalmoscope (Neitz Instruments Co, Ltd, Japan) and with a binocular indirect opthalmoscope (Heine Optotechik, Germany) for mydriasis and for the anterior portion of the eye, optic media, and fundus oculi, respectively.
Dams were observed once daily. All animals (pups/weanlings) were observed for the presence or absence of abnormalities in clinical signs such as external appearance, nutritional condition, posture, behavior, and excrement before, immediately after and between 1 and 3 hours after dosing and on the day of necropsy.
Home cage (posture, convulsion, and behavior), in hand activity (ease of removal from cage, fur/skin condition, secretions from eyes/nose, exophthalmos, palpebral closure, mucosal membrane, autonomic nerve function (lacrimation, salivation, piloerection, pupil size, and abnormal respiration), reactivity to handling, and vocalization at handling), and open field observations (arousal, convulsion, abnormal behavior, stereotypy, gait, posture, grooming, rearing count, and excretion (defecation count and urination)) were observed or measured on days 36 and 37. Functional observations (auditory response, approach response, touch response, tail pinch response, pupillary reflex, aerial righting reflex, and landing foot splay) were also recorded on days 36 and 37. Additional test measured on days 36 and 37 included: (1) grip strength using a CPU gauge Model RX-5 (Aikoh Engineering Co. Ltd, Japan) and (2) motor activity using a motor activity sensor NS-ASO1 for 1 h (NeuroScience, Japan).
Urinalysis
On days 39 and 40 of administration, animals were placed in metabolism cages and 4-h and 20-h urine samples were collected. Total protein, pH, ketones, glucose, occult blood, bilrubin, urobilinogen, color, and sediment were measured by Multistick Test Paper (Siemens Healthcare Diagnostic KK, Japan) using the 4-h urine sample. A 4-h urine volume was also measured. Urine volume, osmotic pressure (cryoscopy), sodium (ion selective electrode), potassium (ion selective electrode), and chloride (colometric) were measured in the 20-h urine sample.
Hematology
At the scheduled necropsy, after fasting overnight, all animals were subjected to a laparotomy under isoflurane anesthesia and blood (1 mL) was collected via the abdominal aorta into blood-collecting tubes containing EDTA-2K (SB-41: Sysmex Corporation, Japan). Red blood cell count, hemoglobin, hematocrit, mean corpuscular hemoglobin volume, mean corpuscular, mean corpuscular hemoglobin concentration, reticulocyte percentage, platelet count, white blood cell count, and differential white blood count were determined by one of the following methods: dual-laser flow cytometry, cyanmethemoglobin method, hematocrit, or by calculation. In addition, blood samples collected with test tubes containing 3.8% sodium citrate were centrifuged (3100 r/min, i.e. 1690g for 12 min), and prothrombin time, activated partial thromboplastin time, and fibrinogen using either a Adiva 120 Hematology System (Siemans Healthcare Diagnostics, Illinois, USA) or a coagulometer (ACL Elite Pro, Instrumentation Laboratory, Miami, USA) were determined.
Blood chemistry
Blood samples (3 mL) were collected in test tubes containing a coagulation promoting agent (Venoject II-Autosep, Terumo Corporation, Tokyo, Japan) and centrifuged at 3100 r/min, that is, 1690g for 12 min. The resulting sera were examined for alkaline phosphatase, total cholesterol, triglyceride, phospholipids, total bilirubin, glucose, blood urea nitrogen, creatinine, sodium, potassium, chloride, calcium, inorganic phosphorus, total protein, albumin, and A/G ratio (A/G) in a Clinical Laboratory System TBA-120FR (Toshiba Medical System, Japan).
Blood samples (2 mL) that were collected into test tubes containing heparin sodium (20 units of heparin per 1-mL blood) were centrifuged at 3100 r/min, that is, 1690g for 12 min, and the plasma obtained was examined for aspartate aminotransferase, alanine aminotransferase, and lactate dehydrogenases in a Clinical Laboratory System TBA-120FR (Toshiba Medical System, Japan).
Gross examination
At the scheduled necropsy, all animals were killed by exsanguination via the abdominal aorta after blood collection and were carefully examined externally. The following organs/tissues were removed and weighed: brain (cerebrum and cerebellum), pituitary, thyroid glands (including parathyroid glands), adrenal, thymus, spleen, heart, lungs (including bronchus), salivary glands, (submandibular and sublingual), liver, kidney, testis, epididymis, prostate, seminal vesicle, ovary, and uterus. The weight per 100 g body weight (relative weight) of each organ/tissue was calculated from the animal’s terminal body weight and the absolute organ weight. For paired organs, organ weight was measured separately; however, evaluation was carried out on the total values of the right and left organs.
The thyroid gland (left) was not present (aplasia) in one male in the control group and the thyroid gland (left) in one male in the 1000 mg/kg group was lost, and thus there are no organ weight values for these two organs from these two animals.
Histopathology
The cerebrum, cerebellum, spinal cord (thoracic), sciatic nerve, glandulae ceruminosae, harderian gland, pituitary, thyroid, parathyroid, adrenal, thymus, spleen, mandibular lymph node, mesenteric lymph node, heart, thoracic aorta, trachea, lung (including bronchus), tongue, esophagus, stomach, duodenum, jejunum, ileum (including Peyer’s patch), cecum, colon, rectum, submandibular gland, sublingual gland, liver, pancreas, kidney, urinary bladder, prostate, seminal vesicle, ovary, uterus, vagina, oviduct, mammary gland (inguinal region), sternum (including bone marrow), femur (including bone marrow), femoral skeletal muscle, skin (inguinal region), larynx, and nasal cavity were fixed in phosphate-buffered 10 vol% formalin. The eyeballs and optic nerves were fixed in 3 vol% glutaraldehyde/2.5 vol% formalin, and the testes and epididymides were fixed in Bouin’s solution and preserved in phosphate-buffered 10 vol% formalin.
For histopathologic examination, all organs/tissues from all animals were embedded in paraffin, thin sections were prepared, and stained with hematoxylin and eosin (H&E). However, only the organs/tissues from control and high-dose groups were examined microscopically.
Statistical analysis
The percentage of external differentiation was calculated for each group using the following formula.
The difference between the control group and each dose group was analyzed for statistical significance. For body weight, body weight gain (from days 1 to 42 of administration), feed consumption, open field observation, functional examination, grip strength and motor activity, quantitative items in urinalysis (including water consumption), hematological examination, blood chemistry examination, and organ weights and mean with SD was calculated for each group and the homogeneity of variance in each group examined by Bartlett’s test (level of significance: 1% bilaterally). Then, the difference in the mean values between the control group and each dose group was analyzed by Dunnett’s test for homogeneous data and by Steel’s test for heterogeneous data. 20
The data in the observation of external differentiation and auditory response, approach response, touch response, tail pinch response, pupillary reflex, and aerial righting reflex in the functional examination were subjected to Fisher’s exact test, since there were cells where the expected frequency was not more than 5. Differences were considered significant when the levels of significance were 0.05 and 0.01, bilaterally.
Results
No deaths occurred in any group. Crust formation in the cervical area was observed in one or two animals (males and females) at each dose level beginning on day 38. These changes were incidental to treatment since their incidence and the period of their expression were not dose related and there were no differences between the control group and any test article group. Otherwise, there were no other external observations in any animal.
There were no differences observed in males or females in any treatment group in home cage activity (hand and open-field observations). In particular, there were no differences among the groups in rearing count. Motor activity following 6 weeks of GOS at 0, 500, 1000, and 2000 mg/kg/day for males and females is shown in Figures 1 and 2, respectively. There was no difference among the groups.
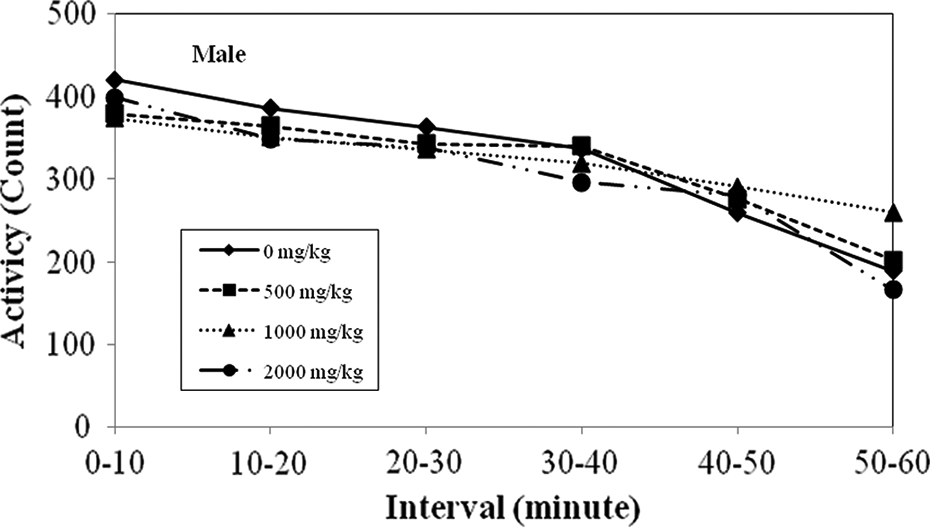
A 6-week oral gavage toxicity study of GOS in juvenile rats. Motor activity (week 6 of administration period): male.

A 6-week oral gavage toxicity study of GOS in juvenile rats. Motor activity (week 6 of administration period): female.
Similar results were seen in the auditory response, approach response, touch response, tail pinch response, pupillary reflex, righting reflex or in the landing foot test between the control group and the test article groups. A significantly low value in the grip strength of the hind limbs was observed in females in the 500 mg/kg group, but it was not a dose-related change. There were no significant differences in the motor activity between the control group and test groups in males or females. No differences were observed in incisor eruption or eyelid opening in any of the pups regardless of the treatment group. No ophthalmological finds were observed.
Feed and water consumption and body weight of the animals in the test article groups were comparable to that of the animals in the control group and there were no significant differences in body weight gain. Body weight for males and females are shown in Figures 3 and 4, respectively.

A 6-week oral gavage toxicity study of GOS in juvenile rats. Body weight: male.

A 6-week oral gavage toxicity study of GOS in juvenile rats. Body weight: female.
One male in the 1000 mg/kg group showed positive occult blood (2+) and positive red blood cells (2+) in the urine sediment, but these changes were judged to be of no toxicological significance since there were no remarkable changes in any other tests in this animal. All other urinalysis parameters measured in both the control and treated groups were within normal ranges and not different from each other. The females in the 1000 mg/kg group showed significantly high values in hematocrit and eosinophil count and a significantly low value in platelet count, but they were dose-unrelated changes and did not manifest themselves in the treated males leading to the conclusion that there were no hematological changes resulting from exposure to the test article in either males or females. Males in the 2000 mg/kg group showed a significantly low value in urea nitrogen, but it was judged to be of no toxicological significance since it was a minimal change and did not occur in the other two treatment groups. A significantly low value in chloride was observed in females in the 1000 mg/kg group, but it was not a dose-related change.
A significantly low value in the absolute weight of the epididymis was observed in the 500 mg/kg group, but it was not a dose-related change. Other than this change, there were no other changes in the absolute weights of any of the organs or tissues in either males or females in any dose group. No significant alterations were noted either at the macroscopic or at the microscopic level in the 2000 mg/kg/day dose group compared to the control group or at the macroscopic level in the 500 or 1000 mg/kg/day groups.
Discussion
GOS was administered orally by gavage to groups of Sprague-Dawley strain SPF rats (Crl: CD(SD)) for 6 weeks from day 4 after birth at doses of 0, 500, 1000, or 2000 mg/kg/day. Animals in the control group were given similar dose volume of orally injectable water over the same period of time as the test article groups. Each group consisted of 10 males and 10 females. Each animal was subjected to detailed observations and examinations including potential effects on development after birth such as: grip strength and spontaneous movement, body weight and feed consumption, external differentiation (incisor eruption and eyelid opening), ophthalmological examination, urinalysis hematology, blood chemistry, necropsy, organ weight, and histopathology. No deaths occurred in any group, including the control group, and there were no significant or dose-related effects from administration of the test article on any of the observations or examinations. Therefore, it was judged that GOS had no effects on the development (growth/progress) from day 4 to week 6 after birth. In a one-generation reproductive toxicity study in which the test article was administered at the same dose levels as this study to males from week 5 after birth and to females from week 12 after birth, there were no abnormalities caused by administration of the test article in the estrous cycle in females, mating ability in males or females, fertility, delivery/lactation of females, in developmental differentiation (pinna detachment, incisor eruption, and eyelid opening), sensory/reflex function examinations (righting reflex, free-fall reflex, pupillary reflex, Preyer’s reflex, and pain reflex), in male or female reproductive organs at necropsy or following histopathological examination. 21
A similar rodent study also reported negative results. 22 In this 90-day study, Sprague Dawley Crl: CD®(SD)IGS BR rats were fed by gavage GOS as Vivinal® GOS syrup (similar chemical profile to the GOS investigated in the current study) at 1125 and 2250 mg/kg/day. No toxicologically significant effects, including body weight, gross abnormalities, or test-related histopathology, were reported by the authors.
Based on the results of the current study, it was concluded that administration of GOS at 2000 mg/kg/day to rats from day 4 after birth to completion of maturation did not affect postnatal development (both physical and neurological). The no observed effect level of GOS by repeated oral administration for 6 weeks from day 4 after birth was at least 2000 mg/kg/day for both males and females.
Footnotes
Acknowledgments
The authors thank Ms Yoko Watanabe for her valuable technical advice.
Conflict of interest
The authors declared no conflicts of interest.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.

