Abstract
A novel galacto-oligosaccharide (GOS) manufactured by a two-step enzyme reaction of lactose was examined in a comet assay for its potential to induce DNA damage in vivo by estimating the DNA fragmentation level in the cellular nuclei of the glandular stomach, colon, and peripheral blood. GOS was orally administered at doses of 0 (vehicle alone), 500, 1000, and 2000 mg/kg/day to five male Crl: CD(Sprague Dawley) rats per group three times (48, 24, and 3 h before the animals were terminated). The specimens were prepared in accordance with the standard protocol (version 14.2) of the “International Validation of the In Vivo Rodent Alkaline Comet Assay for the Detection of Genotoxic Carcinogens” organized by the Japanese Center for the Validation of Alternative Methods. No significant differences in the percentage of DNA in the tail were observed between the GOS-treated groups and vehicle controls in any of the organs evaluated. Additionally, no GOS-related clinical signs or effects on body weight were seen. Based on these results, the comet assay of GOS in the glandular stomach, colon, and peripheral blood using rats was judged negative. Therefore, it is concluded that GOS did not induce DNA damage in vivo under the conditions employed in this study.
Introduction
Human breast milk contains approximately 100 times more galacto-oligosaccharides (GOSs) than cow’s milk. Breast milk-fed infants are reported to have an intestinal microflora dominated by bifidobacteria, whereas the number of bifidobacteria is much smaller in formula-fed infants. 1 –4 In addition to adding GOSs to infant formulas to reinforce some of the functional attributes associated with breast milk, GOSs are added to a variety of commercial food commodities. 5,6 Prebiotics, including GOSs, are added to a variety of foods in the Western diet because of their safety, resistance to digestion in the upper bowel, and fermentability in the colon. 7 –14 GOSs promote the growth of beneficial bacteria (bifidobacteria and lactobacilli) while inhibit the growth of certain enteropathogenic Escherichia coli in the human gut. 1,2,8,11,12
The GOS in this report is manufactured by a two-step reaction from lactose and is assimilated by enteric bacteria, especially bifidobacteria in vitro. GOS also has been shown to increase bifidobacteria in the human intestine after oral administration. 15 GOS, an indigestible oligosaccharide, 16 increases both stool frequency and softens feces in individuals with mild constipation. Our GOS has been shown to be safe in humans following repeated intake. 17 The safety of this novel GOS also has been demonstrated in a repeated dose of 90-day toxicity study in rats and by the absence of genotoxicity in the bacterial reverse mutation assay (Ames test) in Salmonella typhimurium TA98, TA100, TA1535, and TA1537 strains and in E. coli WP2uvrA strain, in the chromosome aberration assay in cultured Chinese hamster lung (CHL/IU) cells, and in the micronucleus assay with mouse peripheral blood erythrocytes. 18
The comet assay (single cell gel electrophoresis assay) is increasingly used to investigate the genotoxicity of industrial chemicals, agrochemicals, food additives, and pharmaceuticals. Among the versions of the assay, the alkaline (pH of the unwinding and electrophoresis buffer >13) 19 method enables detection of the broadest spectrum of DNA damage; double- and single-strand breaks, alkali-labile sites that are expressed as single-strand breaks, and single-strand breaks associated with incomplete excision repair. Unlike the chromosomal aberration or the micronucleus assay, the comet assay can be applied to any organ regardless of their cell proliferation status. In particular, this test is considered a good assay for investigation of suspected tissue-specific genotoxic activity, which includes ‘site of contact’ genotoxicity.
Because of its resistance to degradation in the bowel, only limited GOS reaches the systemic circulation. Therefore, the genotoxicity of this novel GOS was evaluated using the comet assay in glandular stomach and colon, which would be in direct contact with the GOS, and in peripheral blood, considering the possibility of its systemic transfer into the blood.
Methods
Compliance statement
This study was performed in compliance with “Organisation for Economic Co-operation and Development (OECD) Principles of Good Laboratory Practice” (OECD Council: November 26, 1997). The study protocol was approved by the Institutional Animal Care and Use Committee of the test facility prior to the start of this study.
Test article
The GOS used in this study is a sugar manufactured by a two-step enzyme reaction of lactose with Sporobolomyces singularis cells (first step) and β-galactosidase derived from Kluyveromyces lactis (second step). The composition of GOS complied with the manufacturer’s specifications. The composition of the test article, GOS (oligomate 55N, Lot no. 110412AA, Yakult Pharmaceutical Industry Co. Ltd, Tokyo, Japan), used in this study was 56.9% GOS and 43.1% monosaccharides or lactose. The GOS was used within the expiration period of the product. The stability of the test article in the dosing solution over the period used was verified analytically using a high-performance liquid chromatography (HPLC) system. Water for injection with specification of the Japanese Pharmacopoeia (Lot no. 0J92N, Otsuka Pharmaceutical Factory, Inc., Tokushima, Japan) was used as a negative control (vehicle). The positive control, ethyl methanesulfonate (EMS, Lot no. 1423147), was purchased from Sigma-Aldrich Corporation (St Louis, Missouri, USA).
Preparation of test article formulations
A requisite amount of the test article (GOS) was weighed into a measuring glass and dissolved in the vehicle (water for injection) by shaking and mixing. The mixture was filled up to the final volume with the vehicle to prepare 50, 100, and 200 mg/mL formulations. Each formulation was divided into one-day aliquots and stored in brown glass bottles in a refrigerator until used.
Concentrations of GOS in the test article formulations were analyzed using an HPLC system (LC 10 Series, Shimadzu Corporation, Kyoto, Japan) using both an exclusive ion-exchange column (Shodex SUGAR KS-802, Showa Denko K. K., Tokyo, Japan) and a normal phase column (Shodex Asahipak NH2P-50 4E, Showa Denko K. K., Tokyo, Japan). The measured values of each test article formulation were 93.5, 93.5, and 90.7% of the nominal concentrations at 50, 100, and 200 mg/mL, respectively. The analytical results satisfied the acceptance criteria (within 100.0 ± 10.0% of the nominal concentration); therefore, the test article formulations prepared were judged to have been appropriate.
Animals
Male Crl: CD(Sprague Dawley; SD) specific pathogen-free rats were purchased at 7 weeks of age from Charles River Laboratories Japan, Inc. (Yokohama, Japan). After 5 days of quarantine and acclimatization, animals were assigned to treatment groups (five rats/group) by the stratified-by-weight randomization method based on their body weights measured at the end of the quarantine period and dose administration of GOS was started at 8 weeks of age (body weights: 281–315 g). Animals were housed, two to three per cage, in polycarbonate cages (265(width) × 426 (diameter) × 200 (height) mm3) in environmental-controlled rooms (temperature: 19–25°C, humidity: 35–75%, ventilation frequency: 6–20 times/h with filtered fresh air, and lightning: 7:00–19:00 h). Pellet diet (mixed feed, Oriental Yeast Co. Ltd, Tokyo, Japan) and tap water were given to animals ad libitum. Analytical results of the feed, water, and bedding (Beta-Chip®, Charles River Laboratories Japan, Inc., Tokyo, Japan) satisfied the criteria prescribed in the standard operating procedure of the test facility.
Dosage and administration
The experimental design conformed to the standard protocol (version 14.2) of “International Validation of the In Vivo Rodent Alkaline Comet Assay for the Detection of Genotoxic Carcinogens” organized by the Japanese Center for the Validation of Alternative Methods (JaCVAM). 20 The highest dose of the test article, 2000 mg/kg/day, was set at the upper limit dose specified by the JaCVAM protocol. Lower doses were set at 1000 and 500 mg/kg/day. Animals were administered the test articles or the negative control (vehicle alone) three times (48, 24, and 3 h before sampling), or the positive control (EMS; 200 mg/kg/day) twice (24 and 3 h before sampling) using a gastric feeding tube (10 mL/kg body weight volumes). Sampling of cells was performed 3 h after the final administration according to the method described in the JaCVAM protocol.
Clinical signs and body weight
Animals were observed for clinical signs before each administration and intermittently for about 1 h after each administration. Body weight was measured before each administration.
Comet assay
The experimental procedure basically conformed to the JaCVAM protocol for international validation study. 20 All animals were anesthetized with sodium thiopental 3 h after the final administration. Approximately 0.2 mL of blood sample was collected from the abdominal vena cava using a syringe rinsed with sodium heparin (1000 unit/mL, Ajinomoto Pharmaceuticals Co. Ltd, Tokyo, Japan). Collected blood sample was diluted 10-fold with a cold mincing buffer (20 mM ethylenediaminetetraacetic acid–disodium salt (EDTA-Na2) and 10% dimethyl sulfoxide (DMSO) in Hank’s balanced salt solution, pH 7.5) to prepare the blood samples. Animals were euthanized via exsanguination by cutting the abdominal aorta, and then, the stomach and colon were removed.
The stomach was cut along its greater curvature and washed twice with cold phosphate-buffered saline (PBS), then, washed with a cold mincing buffer, and the forestomach was removed and discarded. The colon was cut longitudinally and washed twice with cold PBS, then, washed with cold mincing buffer. The glandular stomach and colon were immersed in fresh cold mincing buffer for 15 to 30 min. The mucosal epithelia were gently scraped (glandular stomach: a few times, colon: once) with a cell scraper (3.0 cm highly pliable thermoplastic elastomer blade, Becton Dickinson and Co., Franklin Lakes, New Jersey, USA), and this layer was exfoliated and washed with a cold mincing buffer. The mucosal epithelia were scraped five or more times with a spatula (stainless steel 180 mm, AS ONE Corporation, Osaka, Japan) to release cells. Cells were suspended in 3 mL of the cold mincing buffer by pipetting gently for about 15 times, and passed through a cell strainer (pore size: 40 μm, BD Falcon). The cell suspensions were diluted with a cold mincing buffer to give a cell density of about 2.0 × 105 cells/mL, that is, glandular stomach or colon samples.
Each sample and 0.5% w/v low-melting agarose gel (NuSieve GTG Agarose, Lonza Rockland Inc., Basel, Switzerland) were mixed at 1:10 ratio. Each mixture was placed into a separate well of a slide (CometSlide HT, 20-well slide, Trevigen Inc., Gaithersburg, Maryland, USA) at 40 μL/well. The slide specimens were immersed in a cold lysing solution (100 mM EDTA-Na2, 2.5 M sodium chloride, 10 mM tris (hydroxymethyl) aminomethane (Tris), 1% Triton X-100, 10% DMSO, pH 10.0) overnight (≥14 h) in a refrigerator, shielded from light. After overnight lysing, the specimens were rinsed twice with cold purified water and immersed in an alkaline solution (300 mM sodium hydroxide, 1 mM EDTA-Na2, pH ≥ 13) for 20 min to unwind double-stranded DNA and then electrophoresed at a constant voltage of 0.7 V/cm (about 0.3 A) for 20 min at 10°C or below. After electrophoresis, the slide specimens were immersed in a neutralization buffer (400 mM Tris, pH 7.5) for 10 min and then dehydrated in absolute ethanol and air-dried. For each sample, four slides were assigned to blood, and three slides each were allocated to the glandular stomach and colon.
For comet image analysis, specimens were stained with SYBR Gold (SYBR® Gold Nucleic Acid Gel Stain, Invitrogen™, Life Technologies Corporation, Carlsbad, California, USA). Images of cell nuclei were captured with a fluorescent microscope (at 200 × magnification with B excitation) and subsequently analyzed using a comet image analyzer (comet assay IV, Perceptive Instruments Ltd, Suffolk, UK). For each sample, 100 cells (50 cells/mL) were analyzed to calculate the percentage of DNA in the tail (% tail DNA). Heavily damaged cells, commonly referred to as “hedgehogs” consisting of small or nonexistent head and largely diffused tails, were excluded from the comet data collection. Hedgehog cells were separately scored for 100 cells/sample (50 cells/slide), and the frequency of hedgehogs was calculated.
Necropsy
The stomach and colon, removed from each animal, were macroscopically examined.
Statistical analysis
Body weights with the exception of the positive control group were initially analyzed by Bartlett’s test for homogeneity of variance among the groups (significance level: 5%). Since the variances were homogeneous at all the measurement points, one-way analysis of variance was applied to compare each of the test article groups with the negative control group (two-tailed significance level: 5%). The positive control group was compared with the negative control group by the F test for homogeneity of variance between the two groups (significance level: 5%). As the variances were homogeneous at all the time points, Student’s t test was applied to compare the means of the two groups (two-tailed significance level: 5% and 1%).
Mean values of % tail DNA of the test article groups were statistically analyzed with those of the negative control group by Dunnett’s test (two-tailed significance level: 5 and 1%). The positive control group was analyzed with those from the negative control group by Student’s t test to compare the means of the two groups (two-tailed significance level: 5 and 1%).
Results
No abnormal clinical signs or mortality was observed in any treated group at any time point. Body weight changes in all the test article groups were similar to the negative control group throughout the experimental period. In the positive control group, body weights before the second dosing were slightly lower than those before the first dosing; however, no statistical significances were detected at any time points.
Macroscopic observation of the stomach and colon revealed no abnormal findings in any of the treated groups.
The result of the comet image analysis of the glandular stomach is shown in Table 1. The mean values of % tail DNA were 6.48 ± 0.68, 5.18 ± 1.03%, 5.22 ± 1.01, and 6.12 ± 1.51% for 0, 500, 1000, and 2000 mg/kg/day, respectively. No statistically significant differences in the % tail DNA were detected in the test article groups compared to the negative control group. In the negative control group, individual data (mean value) of the % tail DNA ranged from 5.80% to 7.59%, well within the historical control data for the test facility (mean ± 3SD: 6.58 ± 4.98%). The mean value of the % tail DNA for the positive control group was 22.82 ± 2.46% and was significantly higher than the value for the negative control group. Frequencies of hedgehogs ranged from 0.0 to 0.4% indicating relatively low values in all the treated groups; there were no statistically significant differences among the groups.
Results of the comet assay in the glandular stomach of rats treated with GOS.

GOS: galacto-oligosaccharide; EMS: ethyl methanesulfonate; % Tail DNA: percentage of DNA in the tail.
aCalculated from 100 nuclei per animal.
bAdministered at 48, 24, and 3 h before sampling.
cAdministered at 24 and 3 h before sampling.
d p < 0.01: significantly different from the negative control group by Student’s t test.
The result of the comet image analysis of the colon is shown in Table 2. The mean values of % tail DNA were 6.04 ± 1.58, 5.62 ± 1.58, 4.41 ± 0.71, and 5.12 ± 1.57% for 0, 500, 1000, and 2000 mg/kg/day, respectively. No statistically significant differences in the % tail DNA were detected in the test article groups compared with the negative control group. In the negative control group, individual data (mean value) of the % tail DNA ranged from 4.05 to 7.79% and were within the historical control data at the test facility (mean ± 3SD: 5.27 ± 7.83%). The mean value of the % tail DNA for the positive control group was 21.16 ± 2.69% and was significantly higher than the value for the negative control group. Frequencies of hedgehogs ranged from 0.2 to 1.0% indicating relatively low values in all the treated groups, and there were no statistically significant differences among the groups.
Results of the comet assay in the colon of rats treated with GOS.

GOS: galacto-oligosaccharide; EMS: ethyl methanesulfonate; % Tail DNA: percentage of DNA in the tail.
aCalculated from 100 nuclei per animal.
bAdministered at 48, 24, and 3 h before sampling.
cAdministered at 24 and 3 h before sampling.
d p < 0.01: significantly different from the negative control group by Student’s t test.
The result of the comet image analysis of the peripheral blood is shown in Table 3. The mean values of % tail DNA were 0.88 ± 0.14, 0.97 ± 0.18, 0.87 ± 0.07, and 0.80 ± 0.15% for doses of 0, 500, 1000, and 2000 mg/kg/day, respectively. No statistically significant differences in the % tail DNA were detected in the test article groups compared to the negative control group. In the negative control group, individual data (mean value) of the % tail DNA ranged from 0.66 to 1.01% and were within the historical control data at the test facility (mean ± 3SD: 0.66 ± 0.66%). In the positive control group, the mean value of the % tail DNA was 25.60 ± 2.68%, and it was significantly higher than that in the negative control group. Frequencies of hedgehogs were 0.0% in the negative control group and all the test article groups. In contrast, the frequency was 5.6% in the positive control group.
Results of the comet assay in the peripheral blood of rats treated with GOS.
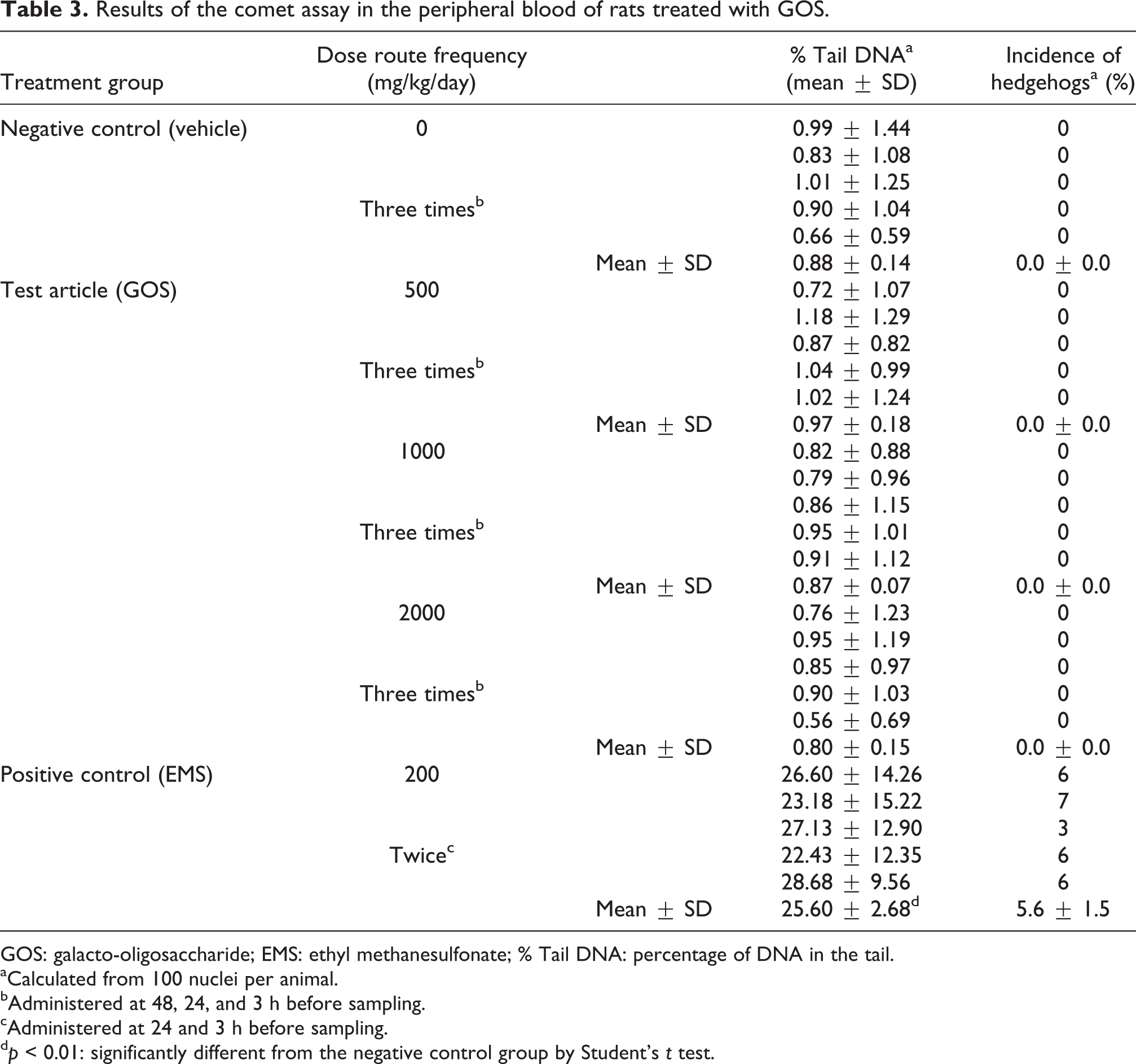
GOS: galacto-oligosaccharide; EMS: ethyl methanesulfonate; % Tail DNA: percentage of DNA in the tail.
aCalculated from 100 nuclei per animal.
bAdministered at 48, 24, and 3 h before sampling.
cAdministered at 24 and 3 h before sampling.
d p < 0.01: significantly different from the negative control group by Student’s t test.
Discussion
Orally taken GOS can hardly reach the systemic circulation because of its resistance to degradation in the bowel. Therefore, the potential induction of DNA damage in vivo of this novel GOS was evaluated using the comet assay in the glandular stomach and colon, which would be in direct contact with the GOS, and in peripheral blood, considering the possibility of its systemic transfer into the blood.
There were no significant differences in the % tail DNA between the GOS-treated groups and the negative control group in any of the organs evaluated in the in vivo comet assay. Moreover, there were no evident differences in the frequencies of hedgehogs among any of the groups. In the negative control group, all individual % tail DNA of all organs were within historical control data of the test facility. In the positive control group (EMS), the % tail DNA was significantly higher than those in the negative control group for all the organs evaluated. Thus, this comet assay study was judged to have been conducted under appropriate conditions.
Because GOS did not induce significant DNA fragmentation levels in the cellular nuclei of the glandular stomach, colon, or peripheral blood in rats, GOS was considered to be negative in the comet assay at doses up to 2000 mg/kg. Additionally, no GOS treatment-related changes were observed in clinical signs, body weight, body weight changes, or mortality. Furthermore, there were no necropsy findings suggestive of cytotoxicity in the glandular stomach or colon.
The safety of this novel GOS has been demonstrated in a repeated dose 90-day-toxicity study in rats and by the absence of genotoxicity in the bacterial reverse mutation assay (Ames test) in S. typhimurium TA98, TA100, TA1535, and TA1537 strains and in E. coli WP2uvrA strain, in a chromosome aberration assay in cultured Chinese hamster lung (CHL/IU) cells, and in the micronucleus assay with mouse peripheral blood erythrocytes. 18 As our novel GOS was negative in the comet assay as well as in the above cited studies and has been showed to have beneficial effects in human, 15 –17 it was concluded that the GOS manufactured by a two-step enzyme reaction of lactose with P. singularis cells (first step) and β-galactosidase derived from K. lactis (second step) continues to be safe as a food ingredient.
Footnotes
Acknowledgment
The authors would like to acknowledge Ms Yoko Watanabe for her valuable technical advise.
Conflict of interest
The authors declared no conflicts of interest.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
