Abstract
In this study, the removal of methylene green (MG) from aqueous solution based on two new adsorbents including silver nanoparticles and zinc oxide nanorods loaded on activated carbon (Ag-NP-AC and ZnO-NR-AC, respectively) has been carried out. The dependency of removal process to variables such as contact time, pH, amount of adsorbents, and initial MG concentration were examined and optimized. It was found that the maximum MG removal percentage was achieved at pH = 7.0 following stirring at 400 r min−1 for 7 and 6 min for Ag-NP-AC and ZnO-NR-AC, respectively. Equilibrium data were well fitted with the Langmuir model having maximum adsorption capacity of 166.7 and 200 mg g−1 for Ag-NP-AC and ZnO-NR-AC, respectively. Thermodynamic parameters of MG adsorption on Ag-NP-AC such as enthalpy and entropy changes, activation energy, sticking probability, and Gibbs free energy changes show the spontaneous and endothermic nature of the removal process. Among different conventional kinetic models, the pseudo second-order kinetics in addition to particle diffusion mechanism is the best and efficient model for the prediction and explanation of experimental data of MG adsorption onto both adsorbents.
Keywords
Introduction
Most commercial applied dyes have complex structure and are chemically stable and difficult to remove from waste water. 1 Moreover, treatment procedures of all general and conventional dyes are insufficient for removal and elimination of the azo dyes especially at high concentration. 2 As problem emerge during treatment of dyes via biological degradation, 3 the preferred treatment techniques for removal of the azo dyes must be based on the combination of pretreatment and biological methods that significantly improve the efficiency and amount of biodegradability of the azo dyes. 4 –8 One of the most prominent dye treatment procedures is the adsorption procedure, especially based on the application of nanotoxic and green adsorbents with high surface area and adsorption capacity. The nanoscale-based adsorbents with high amount of surface reactive atoms and high surface area have been proven to possess great potential applicability to remove various types of environmental pollutants. 7 –13 Methylene green (MG; 7-(dimethylamino)-4-nitrophenothiazin-3-ylidene-dimethylazanium chloride; Figure 1) 14,15 is a heterocyclic aromatic dye compound. It stains nuclei green and is sometimes used as a counterstain to red or purple primary stains. Therefore, there is great emphasis to prepare and design new nanoscale-based adsorbent suitable for efficient and low-cost treatment and removal of dyes from aqueous media.
The present study focuses on the utilization of silver (Ag) and zinc oxide (ZnO) nanostructures loaded on activated carbon (AC) for the removal of MG from aqueous solution. 15 The figures of merit and usability of both adsorbents for MG removal has been calculated and compared. Ag nanoparticles are widely utilized as photosensitive components, as catalysts, in surface-enhanced Raman spectroscopy as well as in chemical analysis due to their antibacterial properties, high electrical conductivity, and unique optical properties and have unique adsorption peak known as surface plasmon resonance (SPR). This material similar to antibiotics has antimicrobial activity and superior to the latter in terms of its usability for most of the bacteria. Ag nanoparticles have high specific surface area at nanometer scale and have more reactivity and active center that make it possible for dye removal and extraction procedures. 16 –25 On the other hand, preparation of nanoparticle-based adsorbent is associated with unique advantages such as high number of reactive surface atoms and area that makes them very interesting for many applications such as dyes and organic pollutants removal. This work reported the characterization and application of two green, nontoxic, and high-adsorption capacity adsorbents that can easily be synthesized in a routine and simple procedure. Then, the effect of various experimental parameters on their removal ability was investigated and optimized. Following the optimization of variables, the data were fitted to different kinetic and isotherm models and their constant was calculated.
Experimental studies
Instruments and reagents
MG stock solution (100 mg L−1) was prepared by dissolving 10 mg of solid MG in 100 mL double distilled water, and the working solution was prepared by subsequent dilution of the stock solution. The MG content was determined at its maximum wavelength using ultraviolet–visible (UV-Vis) spectrophotometer (model V-570; JASCO International Co. Ltd, Tokyo, Japan). The pH measurements were carried out using pH/ion meter (model-682; Metrohm, Herisau, Switzerland). Potassium hydroxide (KOH), hydrochloric acid (HCl), and potassium chloride and other compounds with the analytical reagent grade are purchased from Merck Chemicals (Darmstadt, Germany). X-Ray diffraction (XRD) patterns was collected using an automated X’Pert x-ray diffractometer (Philips) with copper-Kα radiation (40 kV and 30 mA) for 2θ values over 10–80°. The shape and surface morphology of proposed nanostructure materials were investigated using field-emission scanning electron microscopy (FE-SEM, model S4160; Hitachi, Tokyo, Japan) under an acceleration voltage of 15 kV.
Measurements of MG uptake
Quantification of MG concentrations was carried out based on calibration curve obtained by plotting absorbance versus MG concentration. Examination dependency of adsorption and MG removal percentage by both adsorbents (range of 0–10 min) at room temperatures show that the equilibrium was established after 7 min for Ag nanoparticles loaded on AC (Ag-NP-AC) and 6 min for ZnO nanorods loaded on AC (ZnO-NR-AC). The effect of initial pH at 15 mg L−1 of MG on its adsorptions by both adsorbents was studied in the pH range of 1–9 (adjusted by addition of HCl and/or KOH). Maximum MG adsorption was achieved at pH 7 and isotherms studies were undertaken at room temperatures in the MG concentrations range of 5–45 mg L−1. The amount of adsorbed MG (q
e; in milligram per gram) was calculated by the following mass–balance relationship
where C 0 and C e are the initial and equilibrium dye concentrations in solution, respectively (in milligram per liter), V is the volume of the solution (in liter), and W is the mass (in grams) of the adsorbent.
Synthesis of Ag-NP-AC
The Ag nanoparticles were synthesized in a one-step reduction process in an aqueous solution. All reactions were carried out in oxygen-free water under nitrogen. In a typical synthesis, 600 µL aliquot of 0.1 M silver nitrate aqueous solution was added into 100 mL of 0.15 wt% of the soluble starch and vigorously stirred for 1 h, while the pH was adjusted to 8.0. The initial reaction mixture was colorless, and the time dependency of the Ag-NPs growth was monitored using UV-Vis absorption spectroscopy. The solution developed light yellow color after about 1 h, showing the formation of Ag-NPs. After maintaining the reaction mixture at 50°C for 24 h, a yellow color appeared. 12,15 The procedure for the preparation of Ag-NP-AC was applied as follows: 200 mL of the freshly prepared Ag-NPs solution was mixed with 10 g of AC in a 500-mL flask under magnetic stirring for up to 12 h, resulting in the deposition of the Ag-NPs on AC. The Ag-NP-AC were then filtered and washed with double distilled water. The Ag-NP-AC was dried at 110°C in an oven for 10 h. A mortar was used to homogeneously ground the Ag-NP-AC powders.
Synthesis of ZnO-NR-AC
The synthesis of the ZnO-NRs was based on the reaction of zinc acetate (Zn(CH3COO)2) with hexamethylenetetramine ((CH2)6N4) on the surface of the AC. For synthesizing ZnO-NRs, 10 mL of (CH2)6N4 solution (1 mol L−1) that acts as a stabilizing and copping agent was added into another solution containing Zn(CH3COO)2 at a pH of 8.0 with vigorous stirring and then were uniformly mixed with 10 g of the AC in an ultrasonic bath. The size and morphology of ZnO nanostructure significantly depend to the concentration and rate of their mixing. It was found that the lowest possible size with maximum removal percentage in short time that can be easily synthesized was Zn(CH3COO)2 and (CH2)6N4with a concentration of 0.05 and 0.1 mol L−1, respectively. The resulting mixture was heated to 70°C, and the growth of the ZnO-NRs began slowly on the surface of the AC. The ZnO-NR-AC were separated and washed with distilled water and ethanol to remove the impurities and (CH2)6N4. Finally, the ZnO-NR-AC was dried at 70°C for 6 h in an oven prior to characterization. 15,26
Results and discussion
Structural properties of Ag-NP-AC
Figure 2(a) shows the UV-Vis absorption spectra corresponding to SPR of the Ag-NP. It is known that SPR of Ag-NP significantly depends on time, pH, and concentration of starch and temperature. The maximum SPR was achieved with 0.15% w/v of starch after 24 h with pH 8 at 50°C. It seems that at higher temperature due to higher kinetic energy following agglomeration, crystal growth was predominated and the size of Ag-NP was increased. The broadband indicates a relatively high polydispersity, both in size and shape of the Ag particles. XRD pattern of Ag-NPs (Figure 2(b)) have distinguished and unique peaks at 2θ of around 38.17, 44.21, 64.32, and 77.12 that strongly support its cubic lattice structure. 27 According to effective bandwidth of these peak and Debye–Scherrer equation, Ag-NPs have maximum abundant average size around 55 nm. 28 The FE-SEM image of the Ag-NPs (Figure 2(c)) shows semi-spherical shape and quite uniform in size distribution (15–80 nm), which strongly confirmed the results obtained by the XRD. At lower temperature, the agglomeration leads to an increase in the size of NP. Another important parameter is the concentration and order of addition of reagent that significantly influence on the size and its absorption. It seems that at higher concentration and super saturation ratio, the coagulation lead to increase in the size of material. The starch acts as both a reducing and a stabilizing agent, probably at higher concentration, due to significant change in its surface properties.
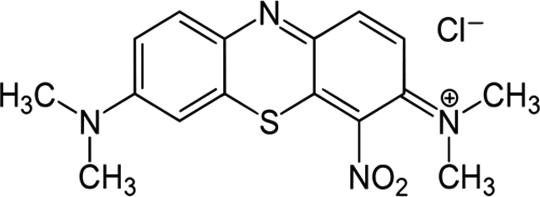
Molecular structure of methylene green.
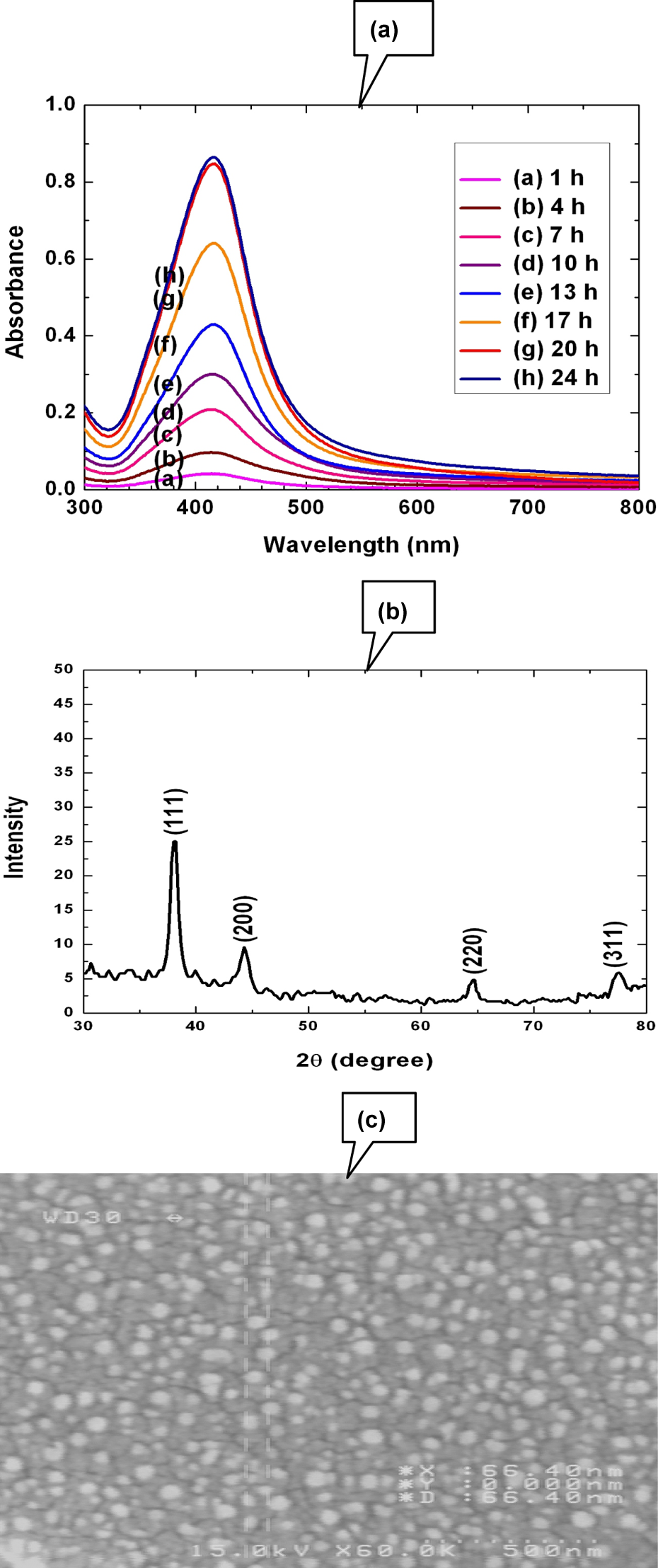
(a) Temporal evolution of UV-Vis absorption spectra after addition of AgNO3 solution into soluble starch solution at 50°C. (b) XRD pattern of the starch-stabilized Ag-NPs. (c) FE-SEM image of the Ag-NP-AC. UV-Vis: ultraviolet–visible; AgNO3: silver nitrate; XRD: x-ray diffraction; Ag-NPs: silver nanoparticles; Ag-NP-AC: silver nanoparticles loaded on activated carbon; FE-SEM: field-effect scanning electron microscopy.
Structural properties of ZnO-NR-AC
In this research, glycol solution probably acts as an auxiliary agent and hinders from crystal growth and further precipitation. In the absence of glycol, it is expected that both stoichiometry and size of the produced NP significantly get affected. Maximum and constant absorbance correspond to ZnO-NRs dispersed in the glycol solution (Figure 3(a)) has shifts to the shorter wavelengths. This behavior strongly supports the decrease in the size of the NRs and emerged from quantum confinement. The energy band gap is a unique and distinguished property of semiconductor-type material. This property can generally be calculated based on the following well-known equation.
28
(a) Evolution of absorption spectra of the ZnO-NRs taken at 2 h intervals following the initiation of the reaction for the first 12 h, (b) FE-SEM images of the carbonic and (c) ZnO-NR-AC. ZnO-NRs: zinc oxide nanorods; FE-SEM: field-effect scanning electron microscopy; ZnO-NR-AC: zinc oxide nanorods loaded on activated carbon.
The energy band gap of the ZnO-NRs according to equation (2) was estimated and found to be in the range of 3.44–3.55 eV, which is somewhat larger than the typical value of the bulk ZnO (around 3.32 eV). This difference is assigned to quantum size effect and reduction in the size of compound showing lower reactivity of this compound for photo degradation and its possible application in adsorption study. The XRD pattern of ZnO-NR shows peak around 31.75°, 34.45°, 36.32°, 47.52°, 56.60°, 62.85°, 66.45°, 67.95°, and 69.15° that emerged and belong to its (hexagonal structure). 29 The FE-SEM images of the bare and ZnO-NRs loaded on AC (Figure 3(b) and (c)) show formation of hexagonal structural ZnO-NRs loaded on AC with growth along the wurtzite c axis direction (diameter between 50 and 200 nm and length between 250 and 500 nm).
Effect of contact time and initial MG concentration
The examination of MG removal percentage variation with contact time at two initial concentrations (10 and 20 mg L−1; Figure 4) shows that the maximum amount of MG adsorption on Ag-NP-AC and ZnO-NR-AC takes place within the contact time of 6–7 min and further addition of contact time has not significantly influenced on the MG removal percentage. At initial concentration of 10 mg L−1 up to 15 mg L−1, >98% of MG removal for Ag-NP-AC and 96% for ZnO-NR-AC adsorption has been occurred for first 5 min. At 15 mg L−1 of MG for Ag-NP-AC removal, it is 98% and 93% for ZnO-NR-AC, while at 20 mg L−1, the removal percentage is 40% and 75% for Ag-NP-AC and ZnO-NR-AC, respectively. This result may be attributed to the more reactive sites and higher tendency of zinc center toward binding MG through its functional group; on the other hand, it seems that oxygen of ZnO has more ability to bind with the functional group of MG molecule. As it can be seen, at low MG concentration due to high vacant adsorbent sites, the amount and rate of MG diffusion to adsorbent surface is high. This fact makes it possible to achieve higher MG removal percentage at lower MG concentration that decreases with increase in initial MG concentration, while at higher concentration, it needs longer time to reach the equilibrium. On the other hand, with increase in MG concentration, due to high competition for the adsorption on vacant sites (low dye molecule to vacant site ratio), the rate of adsorption process slows down. 30

Effect of contact time on the removal of (a) Ag-NP-AC and (b) ZnO-NR-AC. Ag-NP-AC: silver nanoparticles loaded on activated carbon; ZnO-NR-AC: zinc oxide nanorods loaded on activated carbon.
Effect of pH
A view glance to MG structure shows the presence of various reactive atoms and functional groups. The pH is also known to affect the structural stability and MG color intensity. It seems that MG protonated in acidic medium and deprotonated in basic medium, and consequently, it has high positive charge density at low pH. 31 Therefore, the solution pH affects the dye structure, and the charge density of adsorbent affects the efficiency of removal process. The pH dependency of removal process clearly indicates that MG protonation takes place in highly acidic medium, while with decrease in acidity (increasing pH), adsorbent functional group gets deprotonated and receives negative charge. Due to the positive charge of MG and adsorbent in acidic media, the adsorption of MG on both adsorbents significantly decreased at low pH. At higher pH, via formation of an electrical double layer around the adsorbents and change in their polarity, 32 strong electrostatic interaction facilitates the adsorption of MG. The expectable mechanism for MG adsorption on both adsorbents are through soft–soft interaction (binding dye molecule to Ag or Zn atoms), through nitrogen or π–electrons interaction or hydrogen binding between functional group of AC and MG molecule (Figure 5). As it is known, various reactive centers such as nitrogen, sulfur, and strong conjugated π system and positive charge on dye molecule make its adsorption possible on various adsorbents through different pathways. All these mechanisms are facilitated at higher pH.
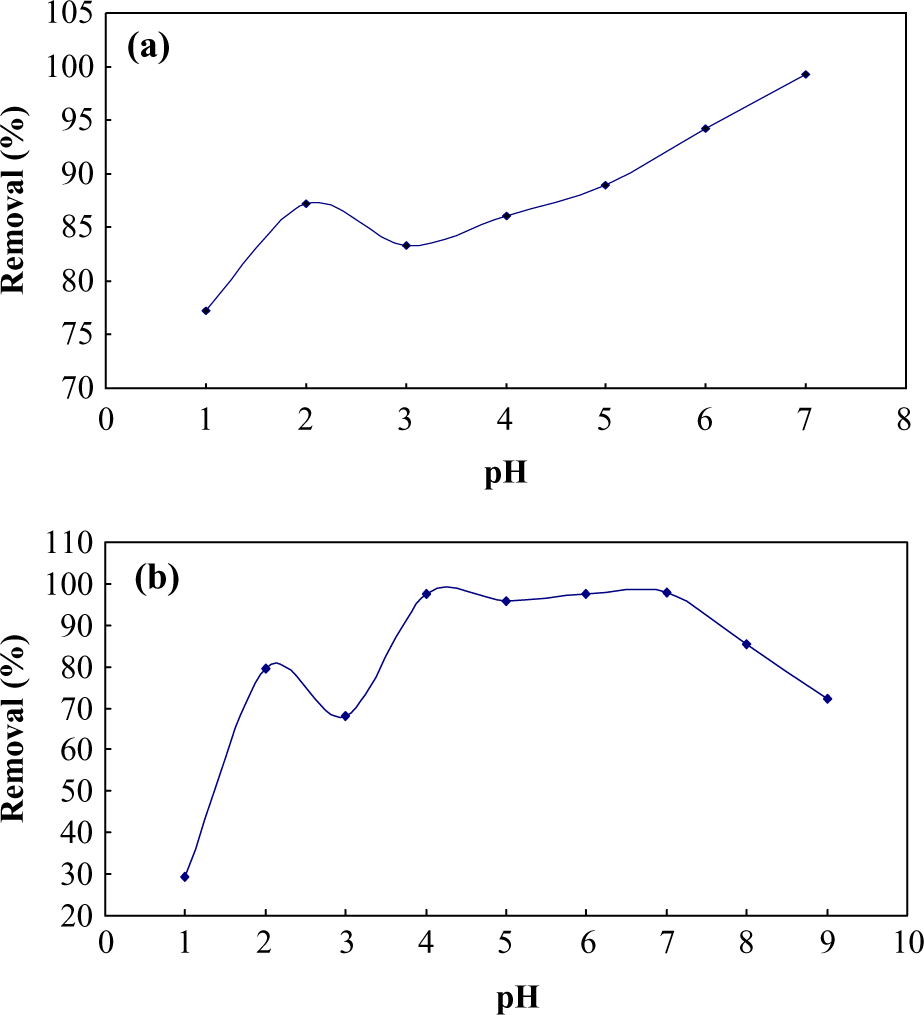
Effect of pH on adsorption of (a) Ag-NP-AC and (b) ZnO-NR-AC. Ag-NP-AC: silver nanoparticles loaded on activated carbon; ZnO-NR-AC: zinc oxide nanorods loaded on activated carbon.
Effect of amount of adsorbent
The effect of amount of both adsorbents on the MG removal was studied by varying the Ag-NP-AC from 0.026 to 0.4 g L−1 and ZnO-NR-AC amount in the interval of 0.026–0.28 g L−1 for pH 7 and at three different concentrations of MG over various contact time at room temperature. As it can be seen (Figure 6), by increasing the amount of adsorbents, the number of vacant sites and concentration gradient significantly improved. The high initial rate of adsorption with increasing adsorbent value can be attributed to higher driving force and larger surface area. Subsequently, following the approximate saturation of adsorbent surface, the adsorption rate slowly rises and due to increase in the number of pores and their volume, the possible and prominent mechanism is intraparticle diffusion. 33 –35
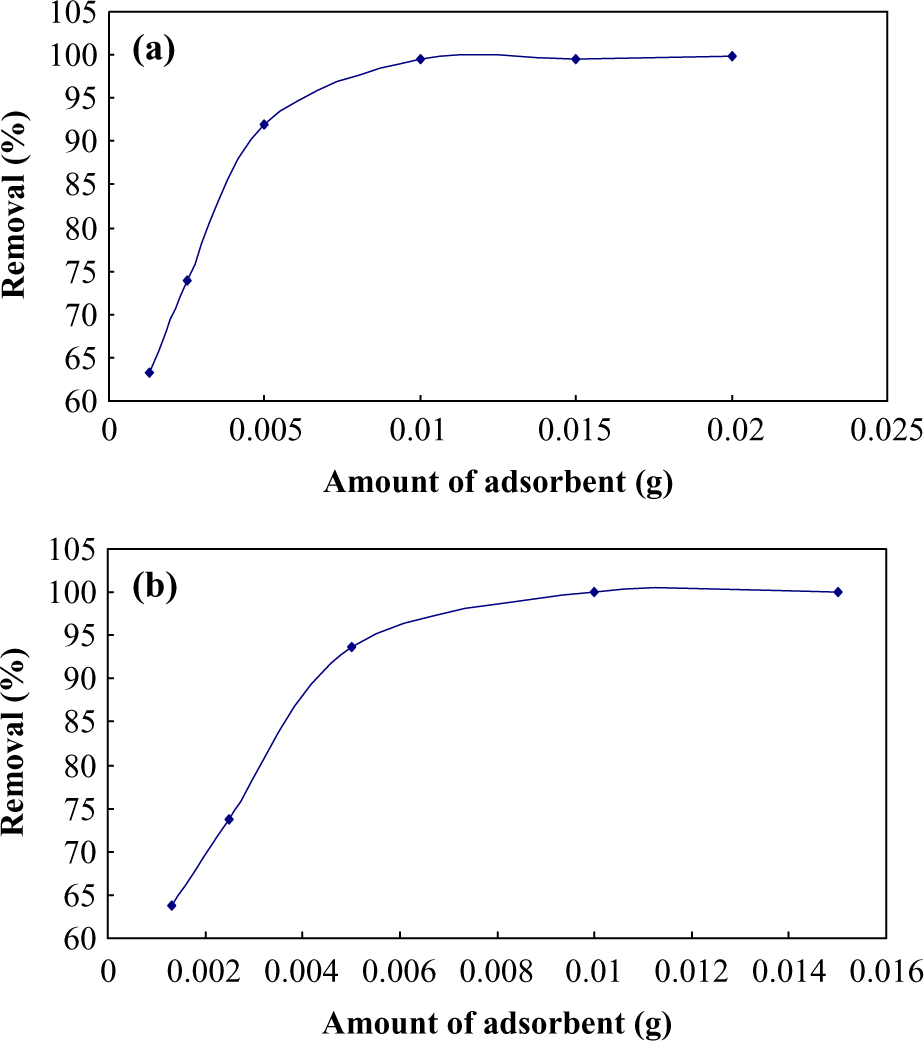
Effect of amount of adsorbent (a) Ag-NP-AC and (b) ZnO-NR-AC. Ag-NP-AC: silver nanoparticles loaded on activated carbon; ZnO-NR-AC: zinc oxide nanorods loaded on activated carbon.
Effect of the ionic strength
The concentration of sodium chloride (NaCl) has little influence on the adsorption capacity and removal percentage. At higher NaCl concentration, due to increase in ionic strength, the MG adsorption increases because of partial neutralization of the adsorbents charge and compression of the electrical double layer by the chloride (Cl−) anion. The Cl− ion also enhances adsorptions of MG via formations of ion pair that neutralizes its charge and enhances its molecular diffusion to adsorbent surface. Therefore, by reducing the repulsion between the MG molecules adsorbed on the surface and adsorbents surface, the removal percentage significantly improved. 36 –38
Effect of temperature on adsorption process
Temperature has important effects on the adsorption process. An increase in the diffusion coefficient subsequently increases the rate of diffusion of adsorbate molecules to the external boundary layer and mesopores of the adsorbent particle. 39 All these stages lead to changes in the value of equilibrium capacity of the adsorbent. 39 –41 The results show that adsorption of the MG onto Ag-NP-AC and ZnO-NR-AC reached the maximum value at 323.15 K. It seems that similar to previously reported results, elevating the temperature leads to the dislodging of the solvent molecule (water) from interfacial region and providing and exposing more number of adsorption sites. This behavior enhances the possibility of interaction between MG and adsorbents. In addition, at elevated temperatures, due to increase in free volume of adsorbent, the adsorption percentage improved. The increase in the equilibrium uptakes with further increase in temperature means that the dye sorption process is endothermic.
Adsorption kinetics
The examination of adsorption kinetics indicates the efficiency of each new adsorbent for efficient removal of dye in short time. The rate-limiting step of adsorption process was examined by fitting the experiment (removal percentage) to three conventional kinetic models including pseudo-first- and second-order and Elovich kinetic models.
Pseudo-first-order kinetic model
The values of q
e and k
1 as constant parameter of the pseudo-first-order kinetic model were obtained from the intercepts and the slopes of the plots of
Kinetic parameters for the adsorption of MG onto Ag-NP-AC and ZnO-NR-AC adsorbents.
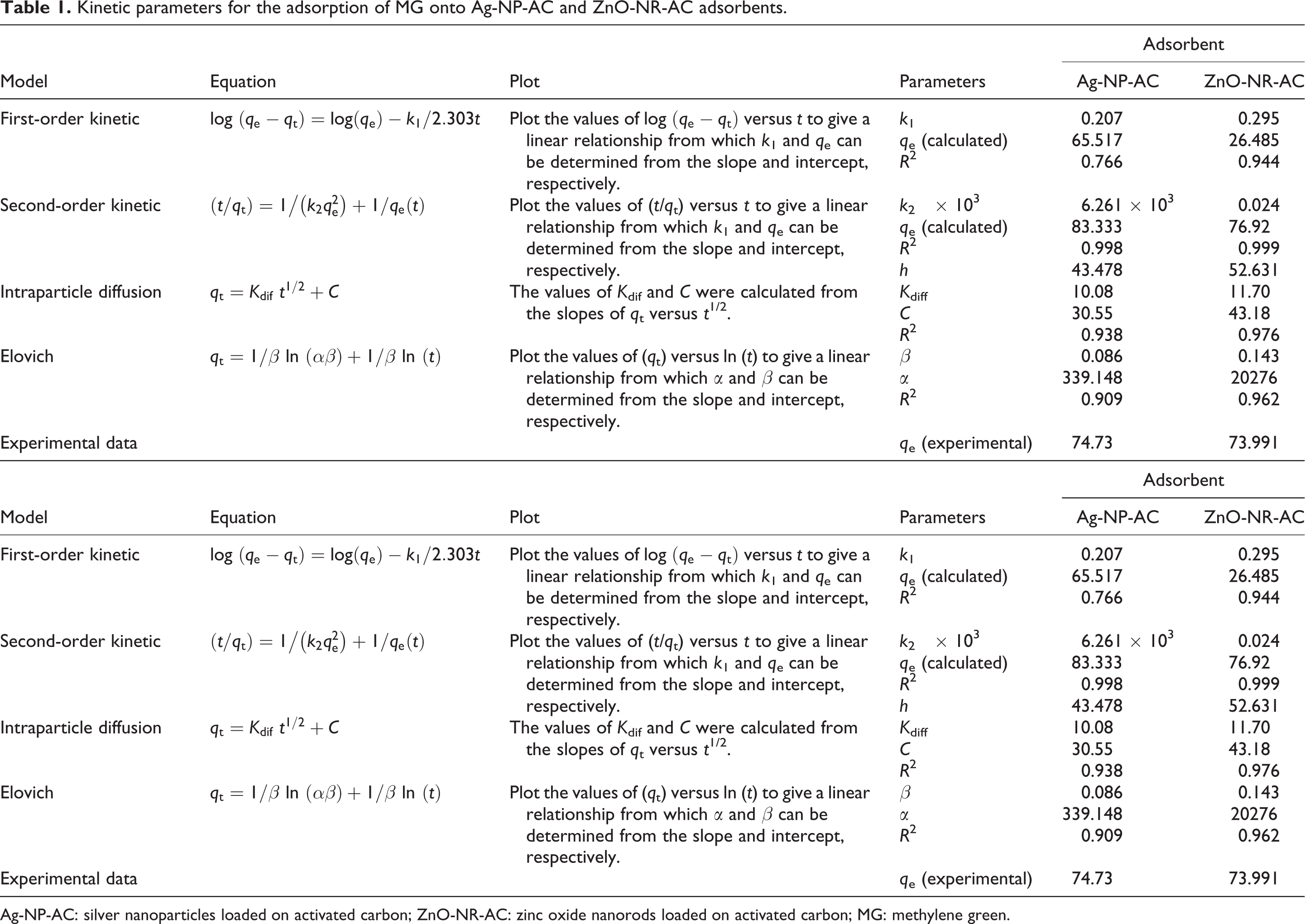
Ag-NP-AC: silver nanoparticles loaded on activated carbon; ZnO-NR-AC: zinc oxide nanorods loaded on activated carbon; MG: methylene green.
Pseudo-second-order kinetic model
In this model, plotting the t/q t of experimental data versus t is used to show its suitability for explanation of whole adsorption data. 43 It was found that the plot of t/q t versus t is a straight line. The k 2 and q e values (the slopes and intercepts of the plot) and the corresponding correlation coefficients of this model are shown in Table 1. The k 2 and equilibrium adsorption capacity (q e) values as main parameters of this model was evaluated from the intercept and slope of this line and the respective value is presented in Table 1. The high and reasonable values of correlation coefficients (R 2 ) and closeness of experimental and theoretical q e value indicate this ability of this model for explanation of adsorption process. This model is applicable for prediction of the behavior of MG adsorption on both adsorbents over the whole concentration range. The experimental and the calculated q e values (Table 1) agreement in addition to high value of correlation coefficient show the usefulness of this model for fitting experimental adsorption data.
Elovich equation
In this model, with known equations presented in literature, α is the initial adsorption rate (in milligram per per minute) and β is the desorption constant related to the extent of surface coverage and activation energy for chemisorption (in grams per milligram). The parameters
Isotherm analysis
The equilibrium sorption isotherm is required to design an efficient sorption system and evaluate adsorbent capacity. On the other hand, follow-up of adsorption study allows attaining useful information on the surface properties and affinity of the adsorbent toward each adsorbate molecule. Equilibrium relationships usually show the ratio between adsorbed and remained adsorbates in solution at fixed temperature that show application and describe the nature of adsorbate–adsorbent interaction. The analysis of equilibrium data with theoretical or empirical equations is necessary to design an applicable experimental equation suitable for interpretation of adsorption data. 47
The theoretical Langmuir sorption isotherm is valid for adsorption of solute from liquid solution as monolayer adsorption on a surface containing a finite number of identical sites. The model is based on the adsorption of target compound onto homogenous and identical adsorbent sites as monolayer through uniform energies of adsorption onto the surface without transmigration of adsorbate in the plane of the surface. Therefore, the Langmuir isotherm model was chosen for the estimation of maximum adsorption capacity corresponding to complete monolayer coverage on the sorbent surface. The linear equation of Langmuir isotherm model can be written using the following equation
Isotherm constant parameters and correlation coefficients calculated for various adsorption MG onto Ag-NP-AC and ZnO-NR-AC adsorbent.

Ag-NP-AC: silver nanoparticles loaded on activated carbon; ZnO-NR-AC: zinc oxide nanorods loaded on activated carbon; MG: methylene green.
Thermodynamic parameters for the adsorption of MG onto Ag-NP-AC.
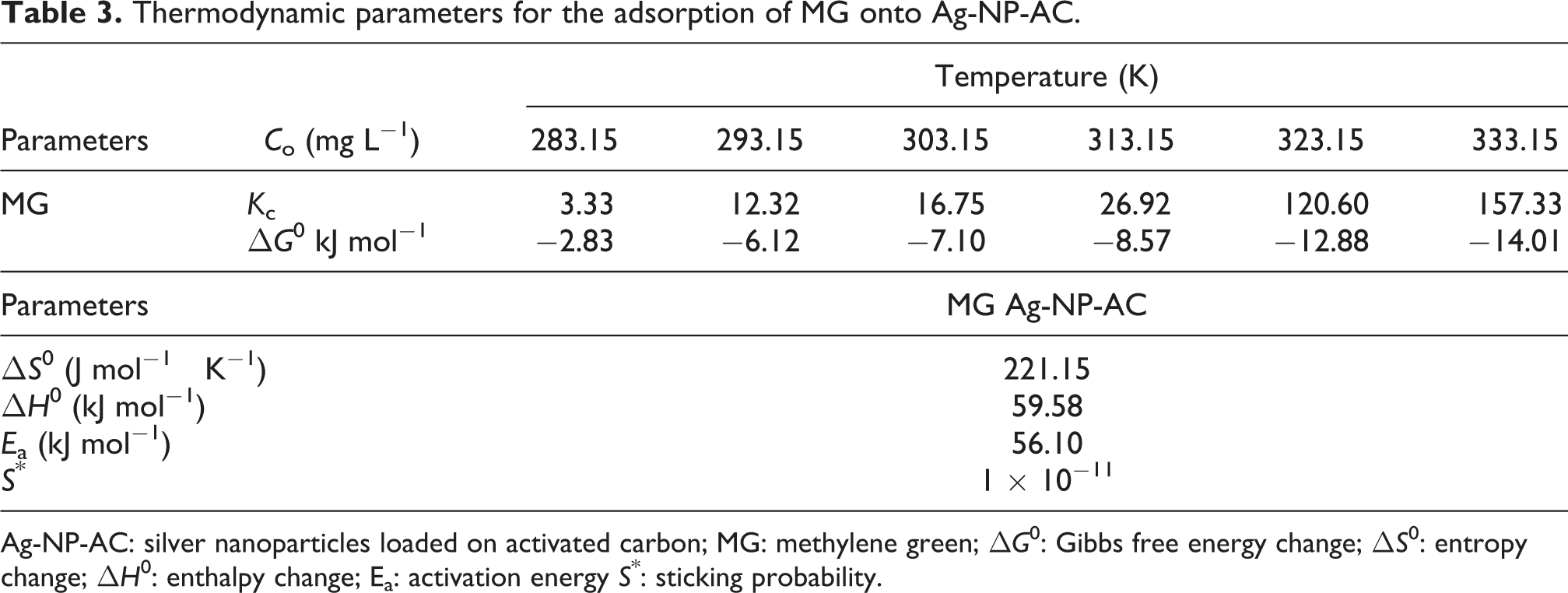
Ag-NP-AC: silver nanoparticles loaded on activated carbon; MG: methylene green; ΔG 0: Gibbs free energy change; ΔS 0: entropy change; ΔH 0: enthalpy change; Ea: activation energy S *: sticking probability.

where q
e is the equilibrium dye concentration on the adsorbent (in milligram per gram), C
e is the equilibrium dye concentration in solution (in milligram per liter), K
F (in milligram per gram) and n are isotherm constants that indicate the capacity and the intensity of the adsorption, respectively. The value of n varies with the heterogeneity of the adsorbent, and for favorable adsorption process, the value of n should be less than 10 and higher than unity.
24,25,36
The slope of plot (1/n) in the range of 0 and 1 is a measure of adsorption intensity or surface heterogeneity, becoming more heterogeneous as its value gets closer to zero. The values of K
f and 1/n were determined from the intercept and slope of linear plot of
Thermodynamic study
Thermodynamic parameters are required to be aware from the adsorption nature of the present study. This study is based on following equations
Values of K
c may be calculated from the relation 
Also ΔG
0 can be obtained from ΔH
0 and ΔS
0 evaluated from the van’t Hoff equation at each temperature range using the following equation:
The increase in randomness at the solid–liquid interface during adsorption show physical nature of adsorption. The values of activation energy (E
a) and sticking probability (S*) were estimated from the experimental data based on the modified Arrhenius equation related to surface coverage (θ)
48
as the linear form. The surface coverage (θ) is generally evaluated from the following equation
The E
a value according to the slope of plot of
Conclusion
The major interest of this study was to investigate sorption of basic dye (MG) from aqueous solution using Ag and ZnO nanostructures loaded on AC as two new nontoxic and green adsorbents with high adsorption capacity. Adsorption of MG on these adsorbents was studied in batch method, and influence of effective variables such as pH, amount of adsorbents, contact time, and rate on removal percentage of MG on both adsorbents was investigated. The present nanostructures loaded on AC have high adsorption capacity and removal percentage in short time (<8 min). Ag-NP-AC and ZnO-NR-AC, as high surface area adsorbents (high adsorption capacity), need short extraction times (<30 min) with low consumption of adsorbent (0.005 g of both adsorbents). Although Ag-NP-AC has a higher adsorption capacity (166.667 mg g−1) compared with ZnO-NR-AC (200 mg g−1). Finally, among the proposed adsorbents oxidized, ZnO-NR-AC was superior to other adsorbents in terms of adsorption capacity to environment. The main removal process in this study is the adsorption. We think due to the absence of strong light such as UV-Vis or mercury lamp, the photochemical decomposition is very low in contribution. On the other hand, for most of the time in various studies, the product of photochemical decomposition is vey toxic than the original compound and can be followed using spectrophotometric method. In our study, such cases were not observed.
Footnotes
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.


