Abstract
This study aimed to determine whether the coding (A4889G) and noncoding region (T6235C) polymorphisms of the gene coding for cytochrome P4501A1 (CYP1A1), a xenobiotic-metabolizing enzyme responsible for the metabolism of carcinogenic polycyclic aromatic hydrocarbons, are involved in the pathogenesis of ischemic stroke in Turkish population. Study group consisted of 226 ischemic stroke patients and 113 controls. Genotypes were attained by allele-specific polymerase chain reaction (PCR) for A4889G and PCR/restriction fragment length polymorphism analysis for T6235C. Frequency of 6235C allele was significantly lower in patients (0.151) compared with controls (0.226, P = 0.015). Prevalence of hypertension and hypertension-associated ischemic stroke risk was lower for 6235C allele carriers. This allele decreased ischemic stroke risk twofold (adjusted odds ratio = 0.48, P = 0.005). There was almost no difference in 4889G allele frequencies in patients (0.445) and controls (0.425). However, prevalence of hypertension was lower in 4889G allele carriers when compared with the wild-type genotypes. In addition, risk of ischemic stroke for smoker and hypertensive individuals was lower when they have at least one 4889G allele. The present study demonstrated that CYP1A1 genetic variants contribute to interindividual variability in smoking- and hypertension-induced ischemic stroke risk.
Keywords
Introduction
Stroke, the second leading cause of death worldwide, 1 occurs when a blood clot blocks an artery that carries blood from the heart to the brain or when a blood vessel breaks, interrupting blood supply to an area in the brain. Cytochrome P450s (CYPs) are a superfamily of oxygen-reacting hemoproteins, which are unique with their remarkable functions and widespread distribution in biological systems. The main function of CYPs is to activate the molecular oxygen for the oxidative metabolism of lipophilic organic chemicals. 2 Cytochrome P4501A1 (CYP1A1) is an important member of the CYP family present mainly in extrahepatic tissues. 3 CYP1A1 is responsible for the metabolism of a large number of xenobiotics as well as a small number of endogenous substrates. Particularly, it acts as the main enzyme in the metabolism of polycyclic aromatic hydrocarbons (PAHs), such as benzo[a]pyrene (B[a]P), 4 which are considered as important carcinogens found in cigarette smoke. 5 Apart from their well-recognized carcinogenic effects, these molecules are also implicated in chemical atherogenesis: PAHs were found to increase the development of atherosclerotic lesions in experimental animals. 6 CYP1A1, in addition to metabolizing B[a]P, is also induced by this chemical. 7 Thus, CYP1A1 might modulate the degree of smoking-induced atherogenesis. 8 –10 In addition to its role in the metabolism of PAHs, CYP1A1 is also involved in the production of arachidonic acid–derived vasoactive substances, epoxyeicosatrienoic acids (EETs) and hydroxyeicosatetraenoic acids (HETEs), 11 which represent an endogenous protective mechanism against atherosclerosis. 12 Moreover, CYP1A1 also functions in the oxidative metabolism of estradiol and estrone. 13 Estradiol is known to have cardiovascular protective effects that are mediated via its endogenous metabolites. 14 On the contrary to these potentially protective roles, CYP1A1 is known to contribute to the cellular production of reactive oxygen species (ROS), due to the consumption of NADPH during the mono-oxygenation reactions. As atherosclerosis, a major cause of ischemic stroke, may be associated with the disruption of vascular homeostasis due to the formation of ROS, CYP1A1 activities may be potentially related to this condition.
The gene coding for CYP1A1 enzyme is polymorphic and two single-nucleotide polymorphisms (T6235C and A4889G) attracted far more attention than others. T6235C polymorphism, which involves the transition of a thymine nucleotide into a cytosine in the 3′ noncoding region, downstream from exon 7 of the CYP1A1 structural gene, causes an increase in the inducibility of the enzyme. 15 A4889G polymorphism, also known as Ile462Val polymorphism due to the amino acid change near the heme-binding region of the CYP1A1 protein, modulates B[a]P metabolism; mutant valine protein demonstrates increased enzyme activity when compared with the wild-type isoleucine protein. 16 These polymorphisms have been studied extensively in relation to cancer risk. 17 –20 Several researchers have also investigated the role of CYP1A1 genetic variants in coronary artery disease (CAD), myocardial infarction or angina. 8,21 –23 However, to date, only two studies have directly investigated whether these polymorphisms have an association with stroke risk, 24,25 and such an association study has not yet been carried out in Turkish population. Thus, in the present study, we addressed the question whether the coding region (A4889G) and noncoding region (T6235C) polymorphisms of the CYP1A1 gene are associated with ischemic stroke risk in Turkish population, paying special attention to the effects of hypertension and smoking.
Methods
Study population
The study population was composed of 226 adult Caucasian patients with the diagnosis of acute hemispheric ischemic stroke and 113 symptom-free Caucasian controls from the same geographic region (Central Anatolia, Turkey). All participants were informed about the procedure, and their informed consents were obtained. The study was approved by the ethical committee of the medical faculty and was carried out in accordance with the principles of the Declaration of Helsinki. As explained before by Can Demirdöğen et al., 26 the ischemic stroke patients enrolled in the study had an anterior circulation stroke that resulted from carotid artery atherosclerotic disease. The cerebral infarction was initially diagnosed on the basis of neurological examination and brain computer tomography (CT) scan and then transthoracic echocardiographic examination, Holter study and Transcranial Doppler emboli detection procedure to rule out emboli source. In order to be considered eligible, the patients should have met the following criteria: having anterior circulation stroke, no other major illnesses, including autoimmune diseases, neoplasms, coagulopathies, hepatic or renal failure, no known embolic source (aortic arch, cardiac or carotid), no family history of hematological, autoimmune or chronic inflammatory diseases, no history of myocardial infarction within the past 3 weeks or no history of transient ischemic attack at any time. We excluded the subjects with ischemic stroke resulting from cardioembolism, lacunary mechanism, coagulation abnormality, vasculitis, etc. Our classification system is similar to the Trial of ORG 10172 in Acute Stroke Treatment (TOAST). We included TOAST “large-vessel disease” group. We reduced the sample size further by excluding other TOAST groups with anterior circulation stroke and including only Oxfordshire Community Stroke Project “total anterior” and “partial anterior circulation infarcts” groups into the study. The control group was selected randomly from the neurology outpatient clinics. All exclusion criteria were applied to the controls exactly plus not having carotid stenosis (lumen narrowing) >50% or ulcerated carotid plaque. All subjects underwent bilateral carotid Doppler ultrasound (CUSG) and transthoracic echocardiographic studies. A detailed history of conventional vascular risk factors and conditions from each participant were obtained. Hypertension was defined as systolic blood pressure >140 mm Hg and/or diastolic blood pressure >90 mm Hg and/or use of antihypertensive drugs. Diabetes was defined as fasting glucose ≥6.99 mmol/L and/or use of pharmacological treatment. Obesity was assigned when the body mass index was ≥30. Smoking status of an individual was assigned “yes” if the individual was currently smoking or had quit smoking <3 months earlier. Routine laboratory tests, including electrocardiogram, chest x-ray, complete blood count, leukocyte differential count, erythrocyte sedimentation rate, and routine biochemistry tests, including fasting glucose, lipid profile (triglycerides, total cholesterol, low-density lipoprotein (LDL)-cholesterol and high-density lipoprotein (HDL)-cholesterol), creatinine, sodium, potassium, bilirubin, liver function tests, routine urine tests and rheumatologic screening tests, were performed for all participants.
Genotype determination
DNA was isolated as described by Lahiri and Schnabel. 27 Allele-specific polymerase chain reaction (PCR) method described by Hayashi et al. 16 was used for CYP1A1 A4889G (rs1048943), and standard PCR/restriction fragment length polymorphism protocols were used for genotyping the CYP1A1 T6235C polymorphism (rs4646903). 16
Statistical analyses
Continuous variables were expressed as mean ± SD. Normality of the sample distribution of each continuous variable was tested with the Kolmogorov–Smirnov test. Differences in continuous variables were evaluated either by the independent samples t test or by the Mann–Whitney U test, depending on the shape of the distribution curves. Categorical variables were expressed as proportions and compared using χ 2 test or Fisher’s exact test. Logistic regression with backward stepwise (likelihood ratio) was used to determine the ability of vascular risk factors, lipid parameters and CYP1A1 genotypes in the prediction of ischemic stroke. Two-tailed probability values with 95% confidence intervals (CIs) were estimated for each odds ratio (OR). The Hosmer–Lemeshow goodness-of-fit test was used for the calibration of the regression analysis. Statistical Package for Social Sciences version 16.0 (Chicago, Illinois, USA) was used for these statistical analyses. A P value of <0.05 was evaluated as statistically significant. Power calculation to test the sufficiency of the sample size was carried out using G-Power 3.1 software. A power value >50% was considered adequate.
Results
The results of the clinical laboratory tests, demographic features and the prevalence of conventional risk factors of patients and control subjects are given in Table 1. The age of the study population varied between 20 and 85 years in patients and 41 and 90 in the control group. The prevalance of the conventional risk factors (hypertension, diabetes, smoking and obesity) were significantly higher in the patient group when compared with the control group. Level of LDL-cholesterol was significantly higher, whereas that of HDL-cholesterol was significantly lower in patients (Table 1).
Clinical characteristics and prevalence of conventional risk factors in patients with ischemic stroke and controls.a

CI: confidence interval; HDL: high-density lipoprotein; LDL: low-density lipoprotein; OR: odds ratio.
a Values are either number of subjects, percentage or mean ± SD.
b Mann–Whitney U test is applied.
c χ 2 test is applied.
d Independent samples t test is applied.
The distribution of CYP1A1 genotypes and allele frequencies for the T6235C and A4889G single-nucleotide polymorphisms within the ischemic stroke patient and control groups are presented in Table 2. Distribution of genotypes for the T6235C polymorphism was as follows: TT: 71.7%, TC: 26.5% and CC: 1.8% in stroke patients and TT: 58.4%, TC: 38.1% and CC: 3.5% in controls. The frequency of the minor allele (6235C) was significantly lower in patients (0.151) when compared with controls (0.226, P = 0.015). The minor allele frequency for the A4889G polymorphism was 0.445 in patients and 0.425 in controls (P = 0.622).
Distribution of genotype and allele frequencies for CYP1A1 T6235C and A4889G polymorphisms in stroke patients and controls.

CYP1A1: cytochrome P4501A1.
a TC + CC vs. TT.
b C vs. T.
c AG + GG vs. AA.
d G vs. A.
Tables 3 and 4 show the relationship between minor alleles of CYP1A1, hypertension and ischemic stroke risk. To examine an overall effect of CYP1A1 mutant alleles on hypertension, the data from both patients and controls have been pooled (Table 3). A lower prevalence of hypertension was observed in 4889G and 6235C mutant allele carriers when compared with the wild-type genotypes. In detail, hypertension was observed in 70% of 4889AA individuals, whereas this frequency decreased to 56% in 4889G allele carriers (P = 0.05). For the 6235C allele, a similar trend was observed. Hypertensives accounted for 77% of wild-type 6235TT individuals, whereas only 41% of 6235C allele carriers were hypertensive (P = 0.000).
Effects of the minor alleles of CYP1A1 on the prevalence of hypertension.a
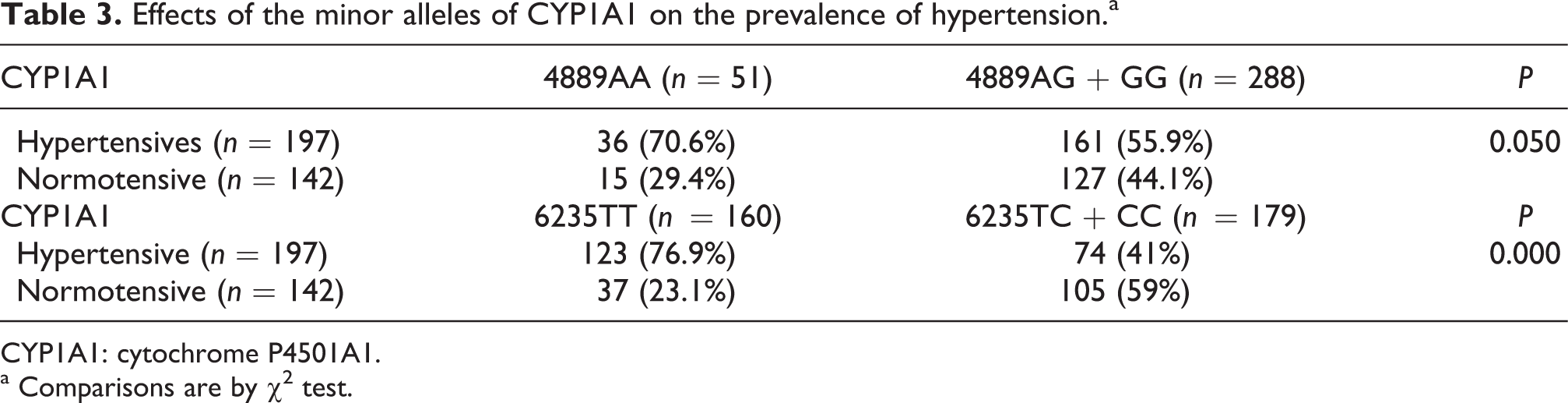
CYP1A1: cytochrome P4501A1.
a Comparisons are by χ 2 test.
Effects of the minor alleles of CYP1A1 on the risk of ischemic stroke with respect to hypertension.a

CI: confidence interval; OR: odds ratio; CYP1A1: cytochrome P4501A1.
a Comparisons are by χ 2 test.
In order to see whether these alleles have modified the risk of having ischemic stroke in hypertensive subjects, the stroke patients’ and controls’ data were presented separately in Table 4. Risk of having ischemic stroke for hypertensives was 3.428 times higher compared with normotensives when their genotype is homozygote wild type (4889AA) for the A4889G polymorphism. The stroke risk decreased to 2.385 when at least one mutant allele (4889AG or GG) was present. Likewise, ischemic stroke risk of hypertensives with respect to normotensives was 3.015 in subjects with 6235TT genotype and decreased to 2.423 in 6235TC + CC individuals.
We had sufficient statistical power to analyze all the genotype subgroups shown in Tables 3 and 4 because the power values of the χ 2 tests were 99.9% for 4889AG + GG subgroup, 57% for 4889AA, 99.5% for 6235TT and 88.5% for 6235TC + CC subgroup.
A similar subgroup analysis was also carried out with smokers, and the results are summarized in Table 5. The risk of having ischemic stroke for individuals with CYP1A1 4889AA genotype was 2.7 times higher for smokers when compared with nonsmokers. If these subjects have at least one G allele at 4889 position, risk of ischemic stroke decreases to 2.435 (P = 0.012). When T6235C genotypes were considered, different results were observed: the risk of ischemic stroke for smokers relative to nonsmokers was 1.716 when they had 6235TT genotype and increased to 5.304 when they had at least one 6235C allele. Since the number of subjects for each genotype group was the same, the power analysis results obtained from Table 5 were the same as those obtained from Tables 3 and 4; and therefore, the sample size in each subgroup in Table 5 was sufficient to make a reliable statistical analysis.
Effects of the minor alleles of CYP1A1 on the risk of ischemic stroke with respect to smoking.a

CI: confidence interval; OR: odds ratio; CYP1A1: cytochrome P4501A1.
Comparisons are by aFisher's exact test; bchi-square test.
Logistic regression analysis with age, sex, hypertension, smoking status, diabetes, obesity, lipid parameters (total cholesterol, triglycerides, LDL-cholesterol and HDL-cholesterol), and CYP1A1 A4889G and T6235C genotypes as covariates revealed that hypertension, smoking, levels of LDL-cholesterol and HDL-cholesterol and the presence of 6235C allele were significant predictors of stroke (Table 6). The adjusted OR for the CYP1A1 6235C allele was 0.48 (95% CI = 0.288–0.802, P = 0.005), which implies that having mutant 6235C allele decreased the risk of ischemic stroke twofold. The model predicted 90.3% of cases correctly, and the Hosmer–Lemeshow goodness-of-fit test pointed out that the calibration of the model was satisfactory (χ 2 = 8.049; 8 degrees of freedom; P = 0.429).
Logistic regression analysis of vascular risk factors (age, sex, hypertension, smoking, diabetes and obesity), lipid parameters (total cholesterol, triglycerides, LDL-cholesterol and HDL-cholesterol) and CYP1A1 4889G and 6235C alleles.a
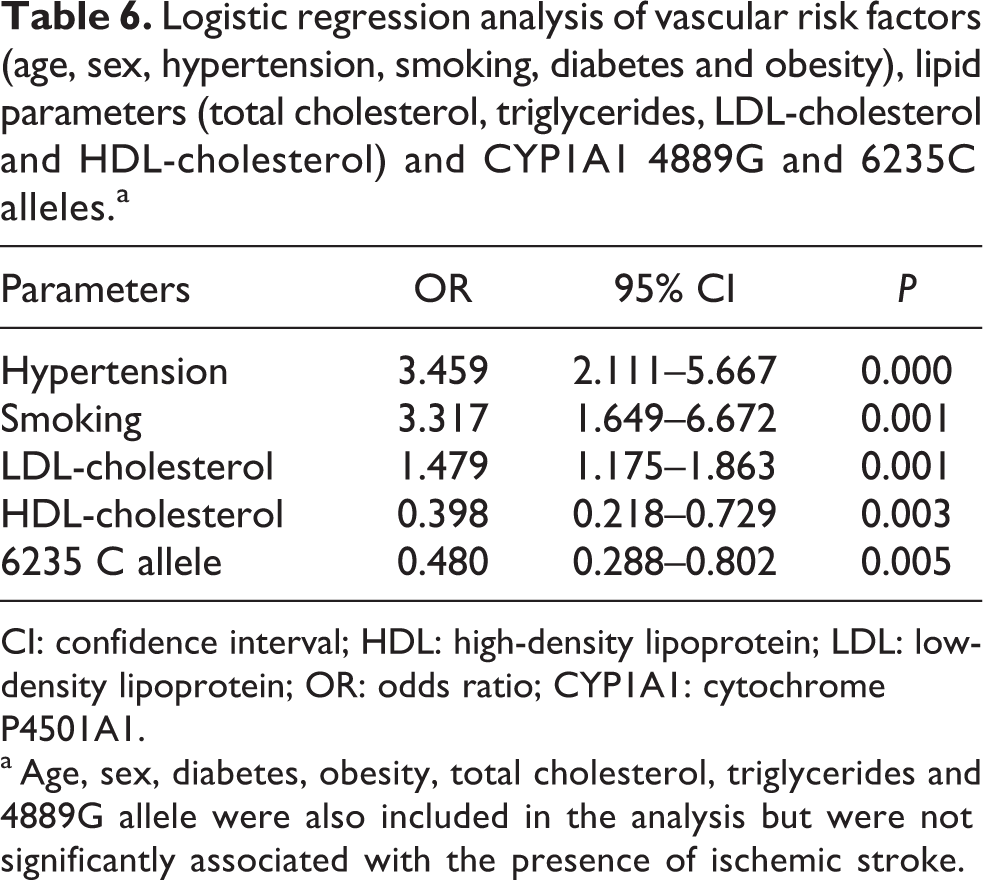
CI: confidence interval; HDL: high-density lipoprotein; LDL: low-density lipoprotein; OR: odds ratio; CYP1A1: cytochrome P4501A1.
a Age, sex, diabetes, obesity, total cholesterol, triglycerides and 4889G allele were also included in the analysis but were not significantly associated with the presence of ischemic stroke.
Discussion
In the current work, we analyzed the effects of T6235C and A4889G polymorphic forms of CYP1A1 gene in ischemic stroke risk, with special attention on smoking and hypertension. In this study, the C allele of the T6235C polymorphism of CYP1A1 was found to be a twofold protective factor against ischemic stroke in Turkish population. Similarly, Yeh et al. 10 reported a statistically non-significant decreased risk of coronary artery disease (OR = 0.69; 95% CI = 0.36–1.35) for the homozygote mutant genotype (6235CC). However, this observation is contradictory to some of the previous reports, 24,25 which stated that the 6235C allele increased the risk of cerebral infarction, although the number of subjects were very low (nine patients with stroke and 23 controls) in one of these studies. 24 Similarly, this allele was found to be associated with greater mortality following onset of acute coronary syndromes, independent of ethnicity and clinical risk factors, but related to smoking history. 22 In addition, Wang et al. 8 reported that the C allele carriers had an increased risk for triple vessel disease in light smokers but not in heavy smokers or nonsmokers. On the contrary, some other authors have found no significant association of this polymorphism with coronary artery disease, myocardial infarction or angina. 9,10,21,23
In the present work, it was also found that 6235C variant carriers have a reduced prevalence of hypertension. Furthermore, hypertension-associated ischemic stroke risk was lowered for 6235C mutant allele carriers when compared with 6235TT subjects. A similar observation was also made, although not discussed, by Moon et al. 25 who reported that 55.4% of 6235TT subjects have hypertension, whereas 49.7% of CT/CC subjects have hypertension. Lança et al. 28 reported that C allele of CYP1A1 T6325C polymorphism is a risk factor for hypertension in postmenopausal women. A relationship between hypertension and this polymorphism was not detected in other studies. 24,29
T6235C polymorphism is in the 3′ noncoding region of the CYP1A1 gene, which is important for messenger RNA stability and expression of gene products. Functional consequence of this polymorphism was reported to be an increase in inducibility of the enzyme. 15 Our finding of lower prevalence of hypertension and lower hypertension-associated ischemic stroke risk in 6235C allele carriers might has resulted in higher frequency of variant 6235C allele in the control group. These findings may be due to the role of CYP1A1 in the arachidonic acid metabolic cascade to produce metabolites that are the promising therapeutic agents for both treatment and prevention of ischemic stroke. 16 As mentioned earlier, CYP1A1 also has an important role in estrogen metabolism. It is well known that estrogen metabolites have protective effects for the arteries. 14
In addition to these roles of CYP1A1 that may be related to atherosclerosis and ischemic stroke, this enzyme is also implicated in the metabolism of PAHs. These molecules are widely known for their carcinogenic effects. 5 The atherogenic properties of PAHs are, however, less known. Although there are several studies in which PAHs were directly injected to experimental animals and atherosclerotic plaque development was observed, 6,30,31 the mechanism of chemical atherogenesis is less clear compared with that of chemical carcinogenesis. Atherosclerotic plaques were considered as benign smooth muscle cell tumors of the aortic vessel wall, and chemical carcinogenic processes (i.e., DNA binding, mutagenesis and proliferation) were thought to be linked to atherogenesis. However, Curfs et al. 32 found that PAHs induce an inflammatory atherosclerotic plaque phenotype irrespective of their DNA-binding properties. Furthermore, both the non-DNA binding, noncarcinogenic PAH benzo[e]pyrene, as well as its DNA binding, carcinogenic structural isomer B[a]P caused a significant and comparable increase in atherosclerotic plaque progression. 32
It is known that CYP1A1 enzyme is induced at the transcriptional level by PAHs, including B[a]P, the major mutagens found in tobacco smoke. 7 CYP1A1 6235C variant has increased inducibility. 15 What happens in the body when the total CYP1A1 activity increases due to induction by cigarette smoking might be very complicated due to various functions of this enzyme, which may be related to both atherosclerosis and cancer. This induction was considered as an adaptive and predominantly protective response to chemical exposure. Indeed, it was shown that PAH-mediated enzyme induction reduced chemical carcinogenesis. 33 However, as indicated above, the mechanisms for chemical carcinogenesis and chemical atherogenesis may differ. For this reason, increased production of ROS in reactions catalyzed by the PAH-induced CYP1A1 enzyme, leading to higher levels of lipid peroxidation and atherosclerotic plaque formation in the vessel wall, might be an explanation for our finding of higher risk of ischemic stroke for 6235C allele-carrying smokers. Wang et al. 8 made a similar observation and reported that the presence of the rare C allele of the CYP1A1 gene in smokers may enhance predisposition to severe CAD and type 2 diabetes. However, Manfredi et al. 9 observed no significant differences in CYP1A1 T6235C genotype frequencies between CAD and non-CAD smokers. It is known that the degree of cigarette smoking-induced atherosclerosis is variable from person to person. Our findings here indicate that part of this variability may be due to the genetic polymorphism found in CYP1A1 gene, at least in the studied Turkish population. The frequency of C allele in the control population of this study was slightly higher than that found in a previous study on Turkish population. 34 It was also higher when compared with other Caucasian populations, 18,19,35 but lower than that reported for Indian, 36 Chinese 20 and Japanese 22 populations.
CYP1A1 4889G allele might have an effect on hypertension due to its increased catalytic activity in the production of vasoprotective estrogens and vasoactive EETs and HETEs, which modulate hypertensive response. Indeed, consistent with our findings, Yeh et al. 10 also found a significantly decreased risk for coronary artery disease associated with the 4889GG genotype (OR = 0.32; 95% CI = 0.15–0.70) and no association with the wild-type 4889AA genotype. 10 Lança et al. 24 also studied the relationship between A4889G polymorphism of CYP1A1 and essential hypertension-related stroke risk, but no conclusion could be drawn due to the small number of subjects (32 hypertensive subjects, nine of whom had a history of stroke). Furthermore, stroke patients were not specified as being ischemic or hemorrhagic stroke patients in that work. 24 In the present study, 4889G allele also decreased the cigarette smoking-associated ischemic stroke risk, most probably in the same manner as it decreased hypertension-associated ischemic stroke risk.
Regression analysis performed in the present study confirmed LDL-cholesterol to be a significant risk factor and HDL-cholesterol to be a significant protector for ischemic stroke. Lipid peroxidation of LDL-cholesterol and HDL-cholesterol is a key event in the development of atherosclerosis, which is a risk factor for ischemic stroke. HDL-cholesterol has been long known to protect LDL-cholesterol against lipid peroxidation and thus prevent vascular plaque accumulation. Thus, it was not surprising to find these lipid parameters as significant predictors of stroke. Diabetes and obesity are also associated with increased lipid peroxidation and oxidative stress. 37 Obesity increases blood pressure, which is considered the strongest risk factor for stroke. 38 Accordingly, diabetes and obesity were found to be at least twice more common among stroke patients in this study.
Conclusions
To the best of our knowledge, this is the first study to evaluate whether polymorphisms in CYP1A1 gene are associated with the risk of ischemic stroke in Turkish population. Taken together, we conclude that in Turkish population, CYP1A1 6235C allele is associated with a decreased risk of ischemic stroke. This finding might be at least in part due to the association of this allele with decreased risk of hypertension and hypertension-associated ischemic stroke. Similar to previous studies, the 6235C allele significantly increased the susceptibility of smokers to ischemic stroke. In addition, we showed for the first time that the A4889G polymorphism lowered the hypertension- and smoking-related ischemic stroke risk. The current study suggests that interindividual variations in CYP1A1 gene may modify susceptibility of individuals to hypertension- and smoking-induced ischemic stroke.
Footnotes
Funding
This work was supported by the Middle East Technical University Research Project Fund [METU-BAP-2008-R-08-11-06].
