Abstract
Acrylamide is oxidized by cytochrome P450 2E1 (CYP2E1) to its epoxide form, glycidamide, which is believed to be responsible for the mutagenic and carcinogenic activities. This study was carried out to investigate the early changes that may be related to the carcinogenic activity of acrylamide in thyroid, adrenal glands and testis in male rats. Forty adult Sprague Dawley male rats were divided equally into four groups, rats of Group I served as control, and rats of Groups II, III and IV were treated orally with acrylamide with doses 5, 10, 15 mg/kg/day body weight for 8 weeks. The results indicated that the plasma carcino embryonic antigen (CEA) and malondialdehyde (MDA) levels are higher, but free and total testosterone, triiodothyronine (T3) and thyroxine, or 3,5,3′,5′-tetraiodothyronine (T4) and corticosterone levels are lower in rats treated with acrylamide than that in control rats. This study provides evidence of endocrine disturbance to the testis, thyroid and adrenal glands, which are also the organs in which acrylamide has been shown to cause tumors in experimental animals.
Introduction
Acrylamide does not occur naturally. It was found in many human foodstuffs which include fried, deep-fried, oven-baked foods, chips, bread, biscuits, crackers and breakfast cereals.1–3 The major pathway in the formation of acrylamide in food cooked at high temperature involves the Maillard browning reaction between reducing sugars and asparagine, an amino acid found in potatoes and cereals.4–6 Acrylamide has been found to cross the placenta in humans and it has been found in human breast milk. Thus, dietary acrylamide exposure is likely begun in utero and continue throughout the life span. 7
Acrylamide is chemically reactive toward nucleophiles, including amino and thiol groups in amino acids and proteins, by Michael additions to the carbon–carbon double bond. This kind of reaction generates adducts of acrylamide with the N-terminal valine residue in hemoglobin. 8
Acrylamide is weakly reactive with DNA in vitro, generating adducts with the ring nitrogen atoms and the extranuclear amino groups of adenine and guanine. 9 The biological significance of these purine adducts remains to be clarified. In vivo, acrylamide is biotransformed to its epoxide, glycidamide (GA). 10 This biotransformation of acrylamide to GA is mediated by cytochrome P450 2E1 (CYP2E1). 11 GA has been reported to be 100–1000 times more reactive with DNA than acrylamide, and GA is considered as genotoxic in a variety of test systems. 12 DNA adducts formation in adult mice from acrylamide treatment showed a supralinear dose–response relationship, which was consistent with the saturation of oxidative biotransformation of acrylamide at higher doses. 13
There is evidence that the genotoxicity of acrylamide predominantly results from metabolic conversion into its epoxide derivative GA,14,15 due to the formation of GA-DNA adducts following the administration of acrylamide.13,16,17 Acrylamide was a clear tumor initiator in several classic tumor ‘initiation-promotion’ studies in mice.18–20 Several previous studies of the carcinogenicity of acrylamide 21 indicated that acrylamide caused tumors at multiple sites in mice18–20 and rats 22 when given systemically by various routes.
The aim of this study is to investigate that acrylamide is a multiorgan carcinogen in experimental rat model which presents a potential carcinogenic hazard to humans and also to study the possible mechanism of acrylamide induction of some hormonal disturbance.
Materials and methods
Experimental animals
Forty male Sprague Dawley rats weighing 100–130 g (from the Animal House Colony of the National Research Centre, Cairo, Egypt) were maintained on standard laboratory diet and water ad libitum. After an acclimation period of 1 week, the rats were randomly divided into 4 groups (10 rats/group) and housed in a room maintained at 23 ± 1°C, 40–60% relative humidity and artificially illuminated (12-h dark/light cycle). Group I rats served as control, rats of Groups II, III and IV were given acrylamide orally by stomach tube at doses of 5, 10 and 15 mg/kg/day, respectively (Merck-Schuchardt Chemical Company, Hohenbrunn, Germany), dissolved in water for 8 weeks.
Sample collection
At the end of the experimental period, the animals were kept fasting for 12 hours and the blood samples were collected from the retro-orbital venous plexus under diethyl ether anesthesia. Blood samples from each animal were collected in tubes containing EDTA and plasma was separated after centrifugation at 3000 rpm for 15 minutes at 4°C. Plasma was used for the determination of carcino embryonic antigen (CEA), malondialdehyde (MDA), total antioxidant, free and total testosterone, triiodothyronine (T3), 3,5,3′,5′-tetraiodothyronine (T4) and corticosterone levels. After blood collection, the rats were killed; the whole testis, thyroid and adrenal glands of each animal were washed with isotonic saline for histopathological investigation.
Biochemical analysis of plasma
Assay of CEA
Enzyme-linked immunosorbent assay (ELISA) was used for quantitative determination of CEA in plasma using kit purchased from Medical Technology Promedt Consulting (GmbH), Germany. 23
MDA measurement
Plasma MDA was assayed by thiobarbituric acid method as described by Esterbauer and Cheeseman.24,25
Assay of total antioxidant
Quantitative determination of total antioxidant in plasma was carried out by colorimetric method using the kit purchased from Biodiagnostic, Egypt, by the reaction of antioxidants in the sample with a defined amount of exogenously provided hydrogen peroxide (H2O2). The antioxidants in the sample eliminate a certain amount of the provided H2O2. The residual H2O2 was determined colorimetrically by an enzymatic reaction that involved the conversion of 3,5,dichloro-2-hydroxybenzensulphonate into a colored product. 26
Hormonal assays
ELISA procedure was used for quantitative determination of plasma-free testosterone 27 (Diagnostics Biochem, Canada), total testosterone 28 (Dima Gesellschaft Fur Diagnostika [GmbH], Germany), T3 29 , T4 30 (International immuno Diagnostics Co., CA, USA) and corticosterone 31 (kit purchased from Calbiotech Inc).
Histopathological investigation
The testis, thyroid and adrenal gland samples of five rats in each group were fixed in 10% buffered formalin solution, dehydrated, cleared and embedded in paraffin wax. Paraffin sections of 5-μm thickness were prepared and stained with hemotoxylin–eosin. 32 All stained slides were viewed using Olympus microscope (Olympus BX51, Korea) and the images were captured by a digital camera (Cannon 620, Korea). Brightness and contrast were adjusted using Adobe Photoshop software (version 4.0.1; Adobe Systems, Mountain View, CA, USA).
Statistical analysis
Data are presented as mean ± standard deviation (SD) and mean ± standard error (SE). One-way analysis of variance (ANOVA) followed by Tukey multiple comparison method was carried out to compare the mean value of different groups using SPSS 7.5 student version. Comparisons were made between Group I versus Groups II, III and IV, and between Group II versus Groups III and IV. p < 0.05 was considered statistically significant.
Results
There was significant decrease in body weight in rats treated with 5, 10 and 15 mg/kg of acrylamide (Groups II–IV) compared to control rat (Group I), with p value < 0.001, with a percentage change of −10.35%, −11.07% and −12.06%, respectively (Table 1 and Figure 1). On the other hand, plasma CEA level was 2.7-, 3.3- and 4.2-fold higher in rats of Groups II, III and IV compared to rats in the control group, with p value < 0.001 (Table 2). Plasma MDA was significantly increased in Groups II, III and IV compared to control (Group I) with p value < 0.001, with a percentage change of 13.24%, 15.05% and 18.04%, respectively, but there was no significant difference between groups. Plasma total antioxidant had significant decrease in Groups II (p value < 0.05), III and IV (p value < 0.01) when compared to the control Group I with a percentage change of −18.5%, −29.53% and −37.01%, respectively. On the other hand, there was no significant difference in plasma total antioxidant between the groups (Table 2). There was significant decrease in each of plasma-free and total testosterone, T3, T4 and corticosterone in all groups in comparison with the control rats (Table 3 ). On the other hand, there was no significant difference in plasma total testosterone and T3 between Group II and Group III and between Group III and Group IV. There was no significant difference in plasma-free testosterone and T4 between Group III and Group IV, but there was significant decrease in Group III in comparison with Group II, with p value < 0.01. There was significant decrease in plasma corticosterone in Group III in comparison with Group II and in Group IV in comparison with Group III, with p value < 0.001 and 0.01, respectively.
Mean ± S.D. of rat body weights before and after experiment in the different groups a

a p ≤ 0.05 is considered significant, p > 0.05 is considered nonsignificant (NS). Groups II, III, and IV compared with control group I.
b p < 0.001.

The mean ± standard error (SE) weights (mg) of rat groups before and after the experiment.
Mean ± S.E. for rat plasma CEA, MDA and total antioxidants levels in the different groups a
CEA: carcino embryonic antigen, MDA: malondialdehyde.
a p ≤ 0.05 is considered significant; p > 0.05 is considered nonsignificant (NS).
b Groups compared with control Group I.
c Group III compared with Group II.
d Group IV compared with Group III.
e p < 0.001.
f p < 0.05.
g p < 0.01.
Mean ± S.E. for rat plasma-free and total testosterone, T3, T4 and corticosterone level in the different groups a

T3: triiodothyronine, T4: 3,5,3′,5′-tetraiodothyronine.
a p ≤ 0.05 is considered significant; p > 0.05 is considered nonsignificant (NS).
b Groups compared with control Group I.
c Group III compared with Group II.
d Group IV compared with Group III.
e p < 0.001.
f p < 0.01.
Morphologically, the testis of the control rats showed normal arrangement of seminiferous tubules (Figure 1A). Figure 2B showed damages with degenerated spermatogonia in the seminiferous tubules in Group II; while in Group III, the seminiferous tubules showed damages associated with absence of cells and the lumen was empty without sperms (Figure 2C). Testes in Group IV showed damages with the arrested spermatogenesis at the level of primary spermatocytes in the seminiferous tubules; while it was absent in the rest of the cells (Figure 3D ).
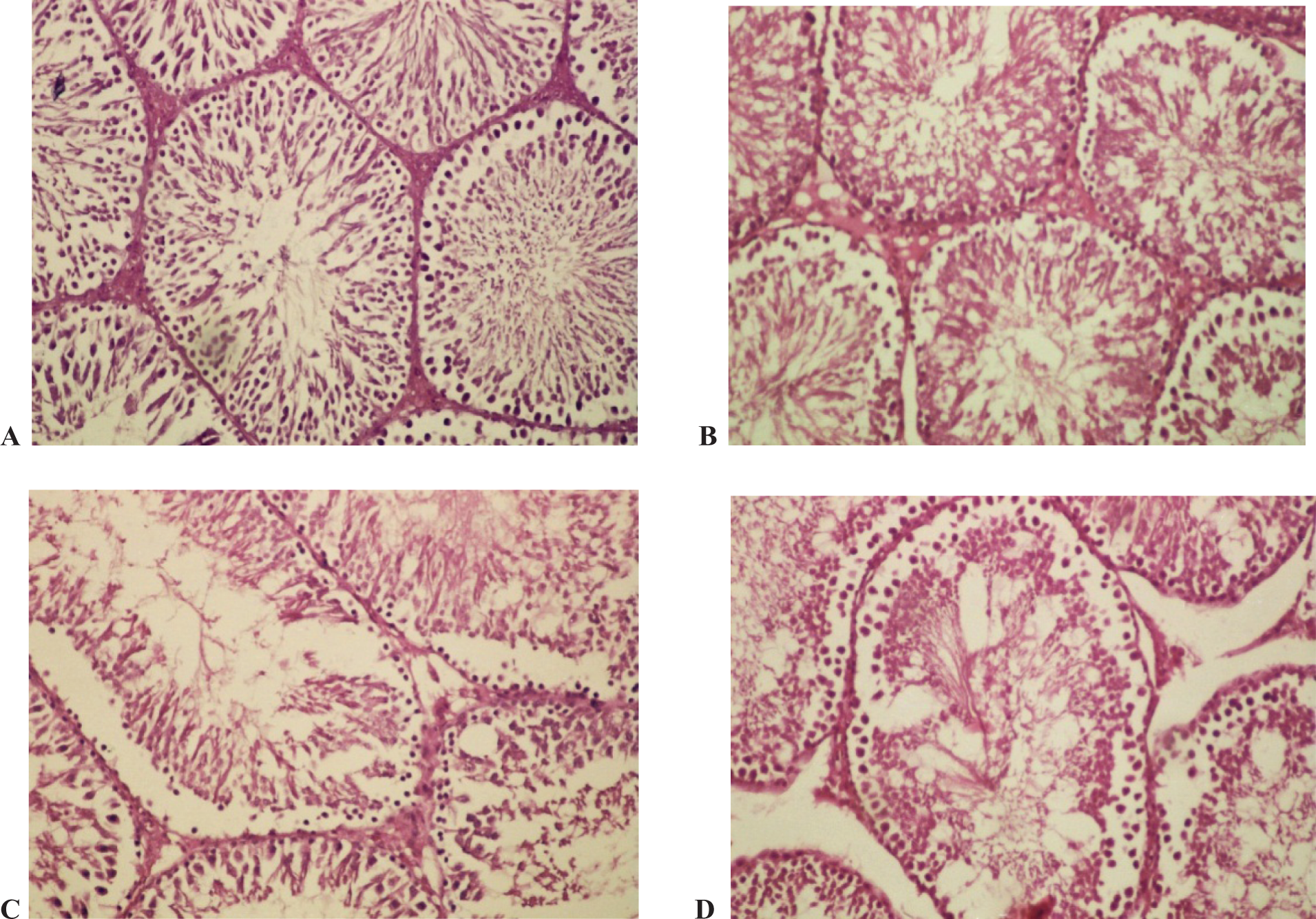
Photomicrographs of transverse section of testis stained with hemotoxylin–eosin (H&E). (A) Control rat. (B) Rats treated with 5 mg/kg/day. (C) Rats treated with 10 mg/kg/day of acrylamide. (D) Rat treated with 15 mg/kg/day of acrylamide (×100).

Photomicrographs of transverse section of adrenal cortex stained with hemotoxylin–eosin (H&E). (A) Control rat. (B) Rats treated with 5 mg/kg/day. (C) Rats treated with 10 mg/kg/day of acrylamide. (D) Rats treated with 15 mg/kg/day of acrylamide (×100).
Photomicrograph of transverse section of thyroid gland of control rat showed thyroid follicles lined with tall cuboidal vacuolated cells and their lumen of the follicles is filled with acidophilic colloid with serrated edge (Figure 4A ); while Group II showed collapsed colloid in some thyroid follicles as shown in Figure 4B. While in Group III the small-sized thyroid follicles had colloid with less serrated edge, the follicles were lined with flat epithelial cells (Figure 4C); while the thyroid gland in Group IV had small-sized thyroid follicles that were lined with flat epithelial cells as shown in Figure 4D.

Photomicrographs of transverse section of thyroid gland stained with hemotoxylin–eosin (H&E). (A) Control rat (×100). (B) Rats treated with 5 mg/kg/day of acrylamide (×20). (C) Rats treated with 10 mg/kg/day of acrylamide (×10). (D) Rats treated with 15 mg/kg/day of acrylamide (×10).
Photomicrograph of transverse section of adrenal cortex of control rat showed zona glomerulosa, zona fasiculata and zona reticulata as shown in Figure 3A; while the adrenal cortex in Group II showed vacuolation of the cytoplasm of fasciculata cells. Many of acidophilic cells can be observed as shown in Figure 3B; while Group III showed separation of the cells of zona fasciculata and empty spaces between it. Notice the disappearance of its nuclei as shown in Figure 3C; while Group IV showed vacuolation in the cytoplasm of fasciculate cells. Many of it showed acidophilic stain as shown in Figure 3D.
Discussion
The metabolism of acrylamide in the body may result in the generation of reactive oxygen species (ROS) which plays a role in the oxidative stress of acrylamide and causes oxidative DNA damage, which may in turn play a role in its carcinogenicity. 33 Moreover, lipid peroxidation (LP) is one of the main manifestations of oxidative damage and has been found to play an important role in the toxicity and carcinogenicity of many carcinogens. 33
Wang et al. 34 and Mei et al. 35 used 5 and 10 mg/kg/day of acrylamide on male Sprague Dawley and big blue rats, respectively, their results were consistent with acrylamide (AA) being a gene mutagen in the rats via metabolism of glycidamide (GA). In this study, the doses of acrylamide treatment were 5, 10 and 15 mg/kg/day and it has been found that the current study deals with the carcinogenicity of acrylamide and interaction between acrylamide and hormonal disorders in male rats. CEA 36 is a cell surface glycoprotein. It was elevated in patients with many cancers and the elevation of plasma levels of CEA are related to the stage and the extent of the tumor. Animals that are orally treated differently with acrylamide showed increase in CEA compared with control animals; those rats treated with 5 mg/kg/day of acrylamide showed increase in the CEA levels than the control; while rats that were treated with 10 mg/kg/day of acrylamide showed an increase in CEA than those treated with 5 mg/kg/day of acrylamide. The rats that were treated with 15 mg/kg/day of acrylamide showed an increase in CEA levels over rats that were treated with 10 mg/kg/day of acrylamide, and this improves the carcinogenicity of acrylamide and thus the damage caused by acrylamide takes place in dose-dependant manner.
The levels of oxidative stress indices and LP (MDA) in the plasma of rats treated with acrylamide were significantly increased while total antioxidant activity was significantly decreased compared with their levels in the controls. These results clearly indicated that total antioxidant activity suppresses the formation of oxygen free radicals in the rat body. The decrease in total antioxidant activity in plasma of rats treated with acrylamide might indirectly lead to an increase in oxidative DNA damage. 37 Mannaa et al. 38 showed that treatment with acrylamide caused a significant decrease in total antioxidant and significant increase in LP in brain homogenate in female rats. There was a significant decrease in thiol groups in lungs, kidney, brain, testis and liver homogenates in male rats treated with acrylamide in a dose-dependent manner. 39 Glutathione is the principal thiol and redox buffer in mammalian cells, while serum albumin is the principal protein in the plasma fraction of blood. 40
The present study indicated that testis is a target organ of acrylamide action as it caused severe damage in seminiferous tubules and caused decreases in plasma-free and total testosterone in a dose-dependent manner and this is in agreement with Yang et al. 41 This study showed that the levels of free and total testosterone were significantly decreased in the plasma of rats treated with 5, 10 and 15 mg/kg/day of acrylamide compared to controls in contrast with the results of Wang et al. 34
The present results revealed that acrylamide induces severe stress on the endocrine function in rats, which included thyroid, adrenal cortex and adrenal medulla. Shelby 42 studied the effects of acrylamide on endocrine glands and concluded that acrylamide induced thyroid follicle adenomas and carcinomas in female rats. In agreement with our results, Khan et al. 43 reported that thyroxine (T4) was decreased in female rats as an early response to acrylamide toxicity, and the doses were 2 and 15 mg/kg/day. The decrease in levels of T3 and T4 in a dose-dependent manner clearly indicated that acrylamide caused hypothyroid gland activity. Friedman et al. 22 exhibited no evidence of dose-related thyroid follicular cell hyperplasia, a hallmark of the thyroid hormone disruption mechanism. Thus, the available data do not support a nongenotoxic, proliferative mechanism of thyroid tumor induction for acrylamide. Khan et al. 43 reported that the morphometry of the thyroid glands showed a significant decrease in the colloid area and a significant increase in cell height of the thyroid follicles in acrylamide-treated rats for 2 and 7 days, respectively. Mannaa et al. 38 showed that treatment with acrylamide 50 mg/kg body weight for 11 days caused a significant decrease in serum T3 and T4 in female rats, which was in agreement with our results which revealed that the levels of T3 and T4 was significantly decreased in the plasma of rats treated with 5, 10 and 15 mg/kg/day of acrylamide compared with the control. On the other hand, Bowyer et al. 44 studies showed virtually no evidence for systematic alteration of the hypothalamic–pituitary–thyroid axis and does not support hormone dysregulation as a plausible mechanism for AA-induced thyroid cancer in the Fischer 344 rat. Specifically, there were no significant differences in (1) mRNA levels in hypothalamus or pituitary for thyrotropin-releasing hormone (TRH), thyroid-stimulating hormone (TSH), thyroid hormone receptor alpha and beta, as well as 10 other hormones or releasing factors; (2) mRNA levels in thyroid for thyroglobulin, thyroid peroxidase, sodium iodide symporter, or type I deiodinases; (3) serum TSH or T3 levels (T4 was decreased at high dose only).
Mannaa et al. 38 showed that treatment with acrylamide caused a significant decrease in serum corticosterone in female rats. The present study revealed that acrylamide induces severe stress on the adrenal gland resulting in a significant decrease in plasma corticosterone in a dose-dependent manner, which suggested that adrenal cortex undergoes severe effects and/or acrylamide induces disturbances in the hypothalamic–pituitary–adrenal relationships. 45 Moreover, Erdreich and Friedman 46 stated that acrylamide administration increased the incidence of several tumor types in rats including adrenal pheochromocytomas.
In conclusion, this study indicated that acrylamide caused increase in LP and decrease in total antioxidant activity therefore may increase cancer risk. Acrylamide may cause endocrine disturbances to the testis, thyroid and adrenal glands. It also may cause tumor in experimental animals especially to endocrine systems and this may be a potential carcinogenic hazard to human.
Footnotes
Acknowledgement
We express sincere thanks to Prof. Dr Hanaa H Ahmed, Hormones Department, National Research Center and Dr Abdel Razik Hussein Farrag, Researcher of Pathology, Medical Research Division for their help in histological and histochemical investigations and their assistance in this manuscript.
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
