Abstract
Acrylamide (AA) is a common chemical, produced during food processing and widely used in various industries and laboratory processes. Thus, AA causes a significant risk for human and animal health. Recently published studies have suggested that reproductive toxicity of AA and glycidamide (GA) was mainly due to the oxidative stress which can lead to cell apoptosis. The present experiment was conducted to investigate the effect of oxidative stress on the apoptosis of mouse Leydig (TM3) and Sertoli (TM4) cells induced by AA and its metabolite GA. TM3 and TM4 cells were exposed to AA (10 µM and 1 mM) and GA (1 µM and 0.5 mM) for 24 h. Following the exposure time, the Leydig and Sertoli cells were evaluated for measurement of cell viability, lactate dehydrogenase activity, lipid peroxidation and hydrogen peroxide levels, apoptosis/necrosis rate, and mRNA expression levels of apoptotic genes (caspase3, Bcl-2, Bax, and p53). The present study showed that AA and GA exposure caused decrease in cell viability and increase in excessive oxidative stress and apoptosis in both cell types. In conclusion, our in vitro results demonstrate that oxidative stress probably plays a major role in AA- and GA-induced apoptosis of Leydig and Sertoli cells.
Keywords
Introduction
Acrylamide (AA) is used as an industrial material in polyacrylamide production, treatment of industrial waste waters, construction of drinking water reservoirs and sewage systems, paper and textile industries, laboratories for use in gel electrophoresis, and cosmetics. 1 –4 AA is a component of cigarette smoke, and it has been calculated that cigarette smoke contains 1.1–2.34 µg AA on average. 5 As a result of frying/roasting/grilling above 120°C of carbohydrate-rich food products, such as potato chips, bread, and biscuits, AA is formed through the Maillard reaction between the carbonyl group of glucose and asparagine amino acid. 1,6 Low molecular weight and high solubility of AA enable to pass through various biological membranes in humans and animals. Following the ingestion, AA is metabolized into a more reactive form, glycidamide (GA), through the action of CYP2E1. 3,7
AA is known to have toxic effects on various tissues/organs in different organisms. These toxic effects can be classified as neurotoxicity, carcinogenicity, genotoxicity, and the effects on reproductive systems. 3,4,8 Various animal studies have investigated the effects of AA on reproductive functions, and these studies have suggested that AA has a detrimental effect on the male reproductive capacity. 9,10 Recent reports on rats showed that AA treatment leads to atrophy in seminiferous tubules, formation of giant cells with multiple nuclei, vacuolization, apoptotic cell formation, reduced sperm viability, and abnormal spermatozoa development. 11,12 Several previous studies have reported that AA decreases spermatogenesis capacity by reducing Leydig cell viability and serum testosterone levels. 11
The increase in intracellular reactive oxygen species (ROS) plays a role in various disease pathogenesis and toxicity of compound. 13 The formation of ROS, including hydroxyl radical, hydrogen peroxide (H2O2), and superoxide anion, after AA exposure causes oxidative stress and DNA damage. 14 Moreover, studies have demonstrated that metabolism of AA leads to the formation of oxidative stress-induced apoptosis. This effect is mediated by increasing oxidative stress-related lipid peroxidation, and causing mitochondrial dysfunction. 13,15 In vitro studies on human astrocytoma cells, 16 human liver embryo L-02, 17 and BV-2 microglial cells 18 and in vivo studies on the testes of Sprague-Dawley 11,19 and Wistar Albino 20 rats have demonstrated that AA treatment might result in oxidative stress and apoptosis. Previous studies have shown that apoptotic pathway is regulated by several genes. 10,14 To clarify the AA- and GA-induced apoptosis, mRNA expression of the antiapoptotic Bcl-2, executioner protein caspase3, proapoptotic Bax, and tumor suppression factor p53 was evaluated in the current study.
In spite of several studies, the exact effects of AA and GA on the male reproductive system remain unknown. In the present study, we used Leydig and Sertoli cells, which play crucial roles in the male reproductive system, as an in vitro model. Based on the previous studies, we hypothesized that low-dose AA and GA could cause oxidative stress-related toxicity in the reproductive system. The aim of this study is to investigate that AA and GA may cause oxidative damage in Leydig and Sertoli cells and apoptosis could induce by oxidative stress.
Materials and methods
Cell culture
Mouse Leydig (TM3) and Sertoli (TM4) cell lines derived from the testis of immature BALB/c mice were originally characterized based on their morphology, hormone responsiveness, and metabolism of steroids. TM3 and TM4 are non-tumorigenic cell lines derived from 11- to 13-day-old mice and were purchased from American Type Culture Collection (Manassas, USA). Cell lines were maintained in DMEM/F12 medium with 5% horse serum, 2.5% fetal bovine serum, and 1% antibiotics (penicillin–streptomycin–amphoterin). The cells were grown at 37°C in a humidified incubator with 5% carbon dioxide (CO2) in the air.
Treatment of cells
Approximate value of the AA and GA concentrations in our experiments was calculated by reference level which could be expected in human serum after estimated realistic exposure. 21 TM3 Leydig and TM4 Sertoli cells were exposed to AA (10 µM and 1 mM) and GA (1 µM and 0.5 mM) for 24 h. Following the exposure time, the Leydig and Sertoli cells were evaluated for measurement of cell viability, lipid peroxidation, H2O2, apoptosis/necrosis rate, and mRNA levels of apoptotic genes.
Cell viability assay
Cell viability was evaluated using a 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT) assay. The MTT assay based on the transformation of the yellow tetrazolium salt MTT to blue formazan crystals by mitochondrial dehydrogenases. Leydig and Sertoli cells were seeded into each well of a 96-well plate culture at 5000 cells with test medium, and then, the plates were incubated at 37°C with CO2 for 24 h. After 24 h incubation, 10 µl MTT I solution was added into each well and cells were incubated for 4 h. Immediately after incubation, 100 µl MTT II solution (SDS) was added to each well and stored in CO2 incubator for one night. The optical density of the each well was measured at 540 nm with an ELISA reader.
Lactate dehydrogenase activity
Lactate dehydrogenase (LDH) is an intracellular enzyme existing in almost all tissues and cells. The leakage of LDH enzyme from the intracellular environment to extracellular environment shows cell cytotoxicity. LDH was measured using the microplate-based Cytotoxicity Detection Kit (LDH; Roche Molecular Biochemicals, Mannheim, Germany). The cells were seeded in 96-well plates (1 × 104 cells/well) with test medium and incubated at 37°C with CO2 for 24 h. After incubation, 100 μl of the kit mixture consisting of stain and enzyme solutions was added to each well. Following 30-min incubation, the results were measured at 492 nm wavelength in the spectrophotometer.
Measurement of lipid peroxidation and H2O2
After treatments, cells (5 × 105) were harvested from six-well plates and transferred into Tris-HCl buffer (pH: 7.2). Then, the cells were sonicated using an ultrasonicator, and the resulting cell suspension was centrifuged at 14,000 ×
Lipid peroxidation was measured according to the malondialdehyde (MDA) content as in the method of Devasagayam and Tarachand. 22 This method was based on the reaction with thiobarbituric acid in an acidic pH at boiling water for 20 min. After cooling, the absorbance was measured at 532 nm wavelength on a spectrophotometer.
H2O2 determination was measured according to the method of Holland and Storey. 23 This method is based on the increasing deviation, resulted from the oxidation of acidified ferrocytochrome c. The level of H2O2 was quantitated at 550 nm wavelength on a spectrophotometer.
Detection of apoptosis and necrosis
Hoechst 33342, a kind of blue-fluorescence dye, stains the condensed chromatin in apoptotic cells, and propidium iodide (PI), a red-fluorescence dye, is permeant to dead cells. The staining pattern resulted from the simultaneous use of these dyes makes it possible to distinguish normal, apoptotic, and dead cell populations by fluorescence microscopy.
Leydig and Sertoli cells were cultured in 24-well plates with 104 cells per well and treated with AA and GA as indicated. After the experimental periods, the cells were washed with PBS (1 M) solution. Then, 20 μl PI (1 mg/ml PI) and 20 μl HO342 (1 mg/ml Hoechst 33342) solutions were prepared by dissolving in 3960 μl PBS. From the prepared solution, 0.2 ml was added to each well and incubated for 15–30 min at 37°C. After the incubation, the cells were washed with PBS for one to two times and examined under the fluorescent microscope. Cells were examined with UV filter via Olympus IX71 (NewYork, USA) fluorescent attachment microscope and photographed in series with equal intervals by Olympus DP72 (NewYork, USA) video camera. The ratio of viable, early apoptotic, apoptotic, and necrotic cells was calculated via a total of 1000 cell counting for each experimental group in series of photographs.
Isolation of RNA and RT-PCR for targeted gene expression
TM3 cells and TM4 cells were plated at 1 × 106 per well in six-well plates with a volume of 3 ml culture medium and treated with indicated AA and GA doses for 24 h. Total RNA was extracted from the treated cells using the RNeasy Mini Kits (Qiagene, Hilden, Germany) according to manufacturer’s instructions. RNA quality and quantity were determined with spectrophotometry (ND-2000c NanoDrop, Thermo Scientific, Darmstadt, Germany). First-strand complementary DNA was synthesized from RNA using Turbo 1 reverse transcriptase (Biomatic, Ontario, Canada) and random primer (Integrated DNA Technologies, Leuven, Belgium) as described by the manufacturer. The primer sequences used for real-time PCR are provided in Table 1. Reverse transcription was achieved by first preincubation for 10 min at 25°C after incubation for 50 min at 42°C, and the reaction was terminated by heating the samples at 85°C for 5 min. Real-time PCR reactions were performed in triplicate for each sample and carried out on Light Cycler 480 system (Roche). LightCycler® 480 SYBR Green 1 Master (Roche Applied Science, Mannheim, Germany) kit was used according to the manufacturer’s instructions for the quantification of gene expression. The housekeeping gene β-actin was used as an internal control. Thermal cycling steps were as follows: 95°C for 5 min, followed by 45 cycles of denaturation for 10 s at 95°C, primer annealing for 30 s at Tm degrees, and extension for 25 s at 72°C. The 2−▵▵ Ct method of Livak and Schmittgen 24 was used to analyze the data, and the changes in gene expression levels were reported as the fold changes of the AA- and GA-treated groups to the respective control groups.
Primer sequences used for gene expression studies.

Statistical analysis
Statistical analysis was performed using GraphPad Prism 6 software (GraphPad Software, San Diego, California, USA) and analysis of variance (ANOVA) was used for comparisons among all groups. The data was calculated by a one-way ANOVA with Tukey’s multiple comparisons test. The results were expressed as mean ± standard error and values of
Results
Effects of AA and GA on cell viability and LDH activity
Cell viability ratios in the experimental and control groups were presented in Figure 1. When control and experimental groups were compared in Leydig and Sertoli cells, it is observed that only high AA and GA doses have significantly decreased Leydig cell viability; on the other hand, high AA and both GA doses caused significantly decrease in Sertoli cell viability (

Dose-dependent effects of AA and GA on cell viability of (a) Leydig and (b) Sertoli cells in vitro. Each bar denotes mean (±SEM) of three independent experiments carried out in triplicates. Significance at **
As shown in Figure 2, the LDH activities of Leydig and Sertoli cells were induced at the low and high doses of AA and GA. When the groups were compared, as in the LDH activity of Leydig and Sertoli cells, a significant dose-dependent increase was observed in all AA- and GA-treated groups (

Dose-dependent effects of AA and GA on LDH activity of (a) Leydig and (b) Sertoli cells in vitro. Each bar denotes mean (±SEM) of three independent experiments carried out in triplicates. Significance at *
Effects of AA and GA on MDA levels
MDA level is calculated by the spectrophotometric method in control and experimental groups of Leydig and Sertoli cells at 24 h. There was a significant increase in MDA levels in the all AA- and GA-treated groups compared to the control group in Leydig cells. However, in Sertoli cells, only high AA and GA doses caused a significant increase in MDA levels (
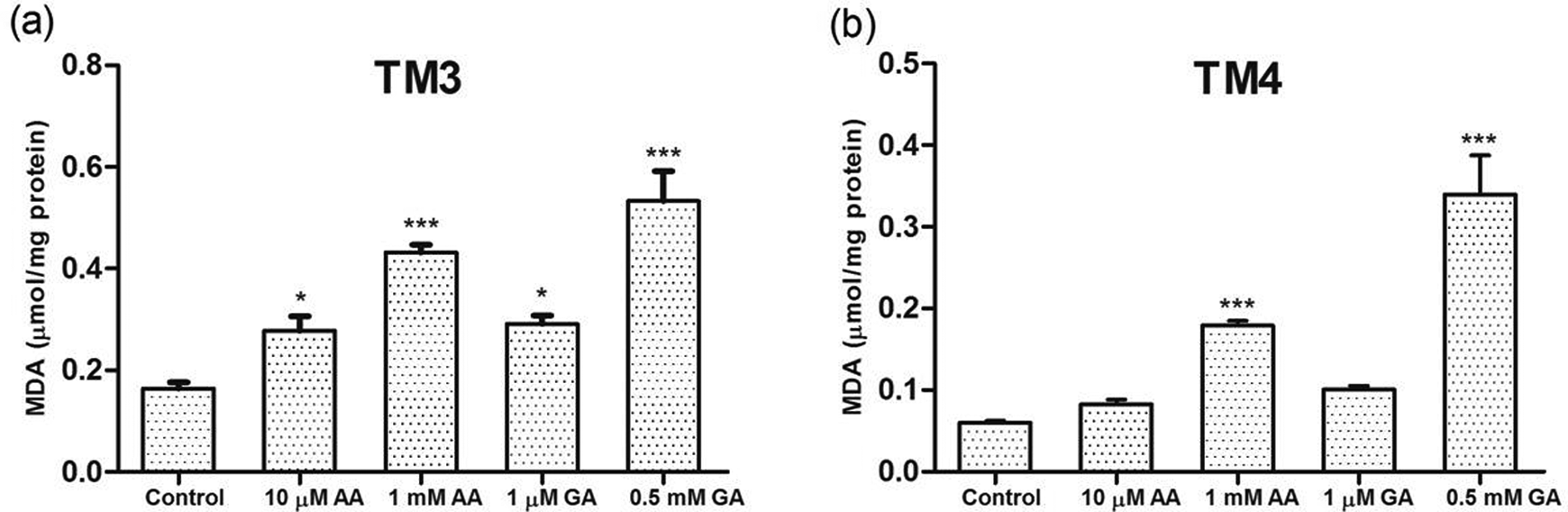
Dose-dependent effects of AA and GA on MDA activities of (a) Leydig and (b) Sertoli cells in vitro. Each bar denotes mean (±SEM) of three independent experiments carried out in triplicates. Significance at *
Effect of AA and GA on H2O2 levels
As can be seen in Figure 4, the exposure to AA and GA induced H2O2 levels of Leydig cells as revealed by a significant dose-dependent increase in H2O2 levels (
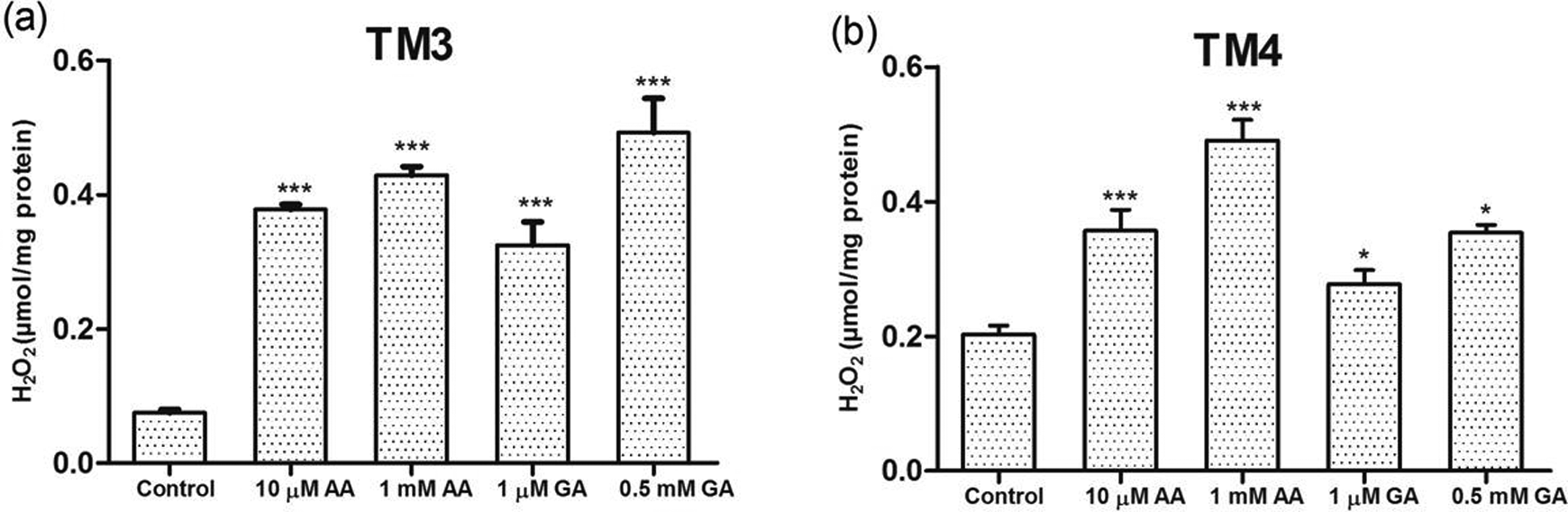
Dose-dependent effects of AA and GA on H2O2 activities of (a) Leydig and (b) Sertoli cells in vitro. Each bar denotes mean (±SEM) of three independent experiments carried out in triplicates. Significance at *
Apoptosis and necrosis rate in Leydig and Sertoli cell exposed to AA and GA
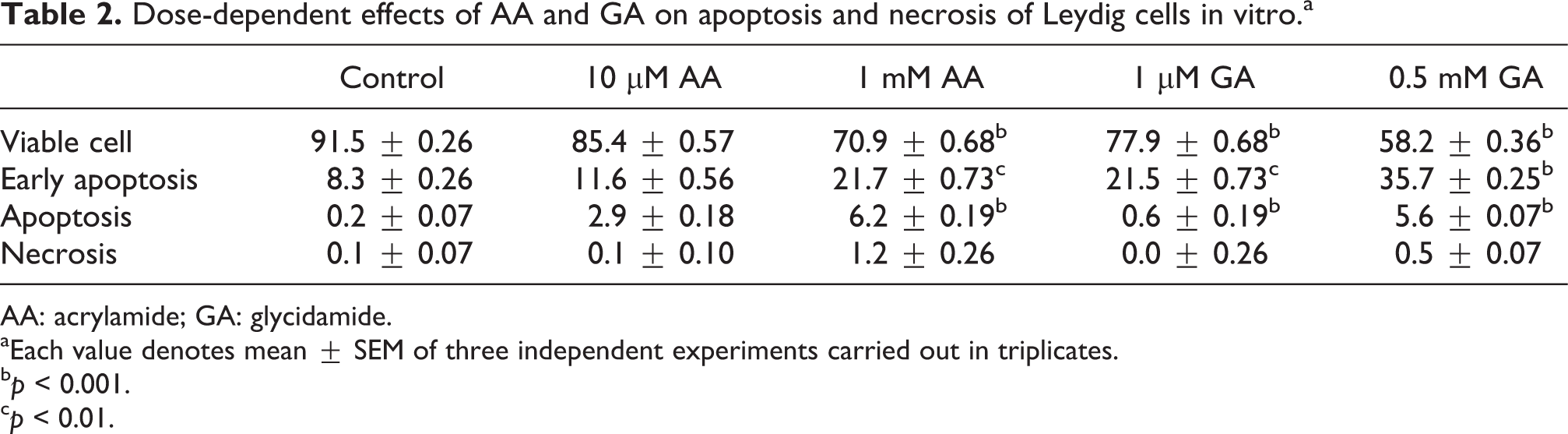
The percentages (Tables 2 and 3) and images (Figures 5 and 6) of viable, early apoptotic, apoptotic, and necrotic cells had a significant alterations, and the effect was compared in a dose-dependent manner with the control group (
Dose-dependent effects of AA and GA on apoptosis and necrosis of Leydig cells in vitro.a
AA: acrylamide; GA: glycidamide.
aEach value denotes mean ± SEM of three independent experiments carried out in triplicates.
b
c
Dose-dependent effects of AA and GA on apoptosis and necrosis of Sertoli cells in vitro.a

AA: acrylamide; GA: glycidamide.
aEach value denotes mean ± SEM of three independent experiments carried out in triplicates.
b
c
d

The effects of AA and GA on apoptosis–necrosis rate of TM3 Leydig cells after 24 h exposure: (a) control, (b) 10 μM AA, (c) 1 mM AA, (d) 1 μM GA, and (e) 0.5 mM GA; →: viable cell, ⇒: early apoptosis, ∇: necrosis, and 〉: apoptosis. AA: acrylamide; GA: glycidamide.

The effects of AA and GA on apoptosis–necrosis rate of TM4 Sertoli cells after 24 h exposure: (a) control, (b) 10 μM AA, (c) 1 mM AA, (d) 1 μM GA, and (e) 0.5 mM GA; →: viable cell, ⇒: early apoptosis, ∇: necrosis, and 〉: apoptosis. AA: acrylamide; GA: glycidamide.
Effects of AA and GA on mRNA expression levels of key apoptotic genes
In this study, quantitative real-time PCR was performed to analyze the mRNA levels of apoptotic markers (caspase3, Bcl-2, Bax, and p53) in TM3 and TM4 cells exposed to AA and GA. The mRNA levels (caspase3, Bcl-2, Bax, and p53) in the control cells were set at 1.00-fold. The result indicates that 10 μM AA exposure of TM3 cell line significantly increased the expression of caspase3 (2.6-fold), Bax (1.3-fold), and p53 (1.1-fold) and decreased the expression of Bcl-2 (0.5-fold) compared to control. Among the higher dose of AA in TM3 cell line, compared with control group, the expression of caspase3 (2.5-fold), Bax (2.3-fold), and p53 (2.5-fold) significantly increased and Bcl-2 (0.3-fold) decreased. According to GA groups in TM3 cell line, mRNA levels were markedly increased caspase3 (2.1-fold), Bax (2.2-fold), and p53 (1.4-fold) and decreased the expression of Bcl-2 (0.5-fold) by treatment at 1 μM; on the other hand, caspase3 (3.6-fold), Bax (4.6-fold), and p53 (4.9-fold) increased and Bcl-2 (0.2-fold) decreased by treatment at 0.5 mM compared to untreated group (Figure 7). The mRNA levels of selected apoptotic genes in Sertoli cells after exposure to AA and GA are shown in Figure 8. There were significant upregulation on expression levels in 10 μM and 1 mM AA concentration of caspase3 (0.6- and 2.2-fold), Bax (1.4- and 1.2-fold), and p53 (1.1- and 1.2-fold) and downregulation in Bcl-2 (0.7- and 0.6-fold), compared to control values, respectively. The relative mRNA levels of apoptotic genes: caspase3, Bax, and p53 were enhanced in a dose-dependent manner 0.7- and 1.7-fold, 1.3- and 1.4-fold, and 1.3- and 1.4-fold following exposure of GA treatment, whereas Bcl-2 mRNA was decreased 0.5-fold in 1 μM and 0.4-fold in 0.5 mM GA concentration.

Dose-dependent effects of AA and GA on the mRNA expression of (a) caspase3, (b) Bcl-2, (c) Bax, and (d) p53 in Leydig cells. Each bar denotes mean (±SEM) of three independent experiments carried out in triplicates. Significance at **

Dose-dependent effects of AA and GA on the mRNA expression of (a) caspase3, (b) Bcl-2, (c) Bax, and (d) p53 in Sertoli cells. Each bar denotes mean (±SEM) of three independent experiments carried out in triplicates. Significance at **
Discussion
The harmful effects of AA and GA have been studied on various testicular cells and male animal models. 9,10,13,25 Recent studies have shown that AA and GA resulted in male infertility through inducing changes in sperm count and morphology, causing a decrease in testosterone levels and histopathological changes in testis tissue. 9,10 In the study by Camacho et al., 10 high doses of AA caused testicular and epididymal lesions in male rats. In another study, AA was administered orally in male rats and shown a significant reduction in testosterone levels. 25 Song et al. 13 reported a reduction in sperm viability and an increase in the number of abnormal sperm with the administration of AA. In spite of many in vivo studies, there is no in vitro study performed to demonstrate the detrimental effects of AA on the male reproductive system. The present study evaluated the toxicity of AA and its epoxide GA on Sertoli and Leydig cells, which have an important role in the male reproductive system.
The toxic effects of AA and GA were investigated on cell viability and LDH leakage in various cell lines and Leydig cells isolated from the testis. 26 –29 GA has been found to significantly decrease cell viability when administered to human breast epithelial cell lines at doses of 1 mM, 2 mM, 3 mM, and 4 mM for 24 h. 30 The administration of AA at doses of 0.1 mM, 0.5 mM, 1 mM, and 2 mM has been found to have remarkably reduced cell viability in human astrocytoma cells and rat astrocytes. 16,27 Another study in male rats that administered AA at a dose of 50 mg/kg/day for 14 days demonstrated a significant reduction in the number of Leydig cells. 10 Furthermore, LDH, which is an intracellular enzyme existing in almost all tissues and cells, is an indicator of cell cytotoxicity. Several in vivo and in vitro studies reported AA and GA markedly suppressed LDH activity. 11,26,31 According to our study, the results of significantly decreased cell viability and LDH leakage in Leydig and Sertoli cells after administration of AA at doses of 10 µM and 1 mM and administration of GA at doses of 1 µM and 0.5 mM show high correlation, and these results are parallel to the findings in studies on other cell lines and tissues.
AA affects glutathione levels and causes toxicity that results in excessive production of ROS at the mitochondrial level, which induces damage of cellular components. 32,33 In addition, lipid peroxidation products generated as the result of the oxidation of membrane lipids by these ROS impair membrane integrity. 25 Although in vitro studies on various cell lines have shown toxicity resulting in excessive ROS production, studies demonstrating ROS production and associated lipid peroxidation in the male reproductive system are only limited to in vivo studies. The previous study that used astrocytoma cells as an in vitro model found a significant increase in the production of ROS after 2 mM AA administration. 16 In vivo studies on male rats reported a significant increase in MDA levels in testis tissue and plasma compared to control groups after AA administration. 19,20,25 The present in vitro study showed that AA and GA administration increased the level of H2O2 in both Leydig and Sertoli cells and resulted in lipid peroxidation.
In general, the previous studies have revealed that apoptosis is induced by oxidative stress. This effect is mediated by the increase in lipid peroxidation associated with oxidative stress and mitochondrial dysfunction. 27 In vitro studies have shown that AA induces apoptosis in various cell lines and various tissues of the experimental animals. 18,19,27,28 Yang et al. 29 observed that AA administration increased Leydig cell death, and the study showed various histopathological abnormalities including apoptosis in the testes of male rats. Evidence emerged from several in vivo studies on male mice indicated apoptosis of germ cells after AA administration. 10,34 –36 The present in vitro results clearly showed that AA and GA treatment increased apoptosis in a dose-dependent manner, and additionally, there was a significant increase in the number of cells at the early phases of apoptosis. Similar results also have been reported in in vitro studies conducted on various cell lines. 16 In previous studies, it has been reported that apoptotic pathway is stimulated by expression of several genes. 37 To clarify the AA- and GA-induced apoptosis, mRNA expression of the antiapoptotic Bcl-2, executioner protein caspase3, pro-apoptotic Bax, and tumor suppression factor p53 was evaluated in Leydig and Sertoli cells. The study conducted by Li et al. 37 has shown that injection of low-dose AA into rat nervous system reduced the expression of Bcl-2 and increased the expression of caspase3 and Bax. Another study on cerebral cortex of rat that administered AA at a dose of 30 mg/kg/day for 30 days demonstrated a significant reduction in Bcl-2 mRNA level and increased Bax mRNA level. 14 In vivo study on male rats reported a significant higher expression level of caspase3 in testicular tissue compared to control groups after AA treatment. 10 Accordingly, Liu et al. 18 have also shown that AA induced apoptosis in microglial cells significantly repressed Bcl-2 protein and overexpressed Bax and caspase3 proteins. The current study demonstrated that exposure of AA and GA downregulated Bcl-2 mRNA level and upregulated Bax, caspase3, and p53 mRNA levels in a dose-dependent manner in both cell lines.
In summary, on the basis of the results obtained, we concluded that oxidative stress plays a major role in AA- and GA-induced apoptosis, even at low concentrations, in Leydig and Sertoli cells. When comparing the experimental data of AA and GA gained from the current study, GA has more effective toxicity than AA in all endpoints.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by Istanbul University Scientific Research Projects (Project Nos. 45010 and 45799).
