Abstract
It is known that lichens are utilized for the treatment of many diseases including ulcer, diabetes, and cancer for many years. Secondary metabolites in the structure of the lichens provide various activity properties for them. In the present study, cytotoxic and oxidative effects of main constituents of Pseudevernia furfuracea (L.) Zopf (Parmeliaceae), olivetoric acid (OA), and physodic acid (PA) were investigated on cultured human amnion fibroblasts (HAFs). OA and PA were isolated from P. furfuracea using column chromatography and their structures were determined by proton nuclear magnetic resonance and carbon-13 nuclear magnetic resonance. HAFs were incubated during 48 h in the presence of OA and PA, at different concentrations from 6.25 mg/L to 200 mg/L. OA showed higher cytotoxicity than PA. In fact, median inhibitory concentration values of OA and PA were 571.27 and 3373.69 mg/L, respectively. The lower concentrations (<50 mg/L) of OA and PA did not cause oxidative stress and genotoxicity; furthermore, they supported anti-oxidative capacity of HAFs. Therefore, all these data suggested that both tested metabolites, especially PA might be developed as natural health medicine to protect human body against oxidative stress and genotoxicity. As far as we know, this is the first report on the cytotoxic and anti-oxidative activities of OA and PA on HAFs.
Introduction
Fibroblasts are known to play an important role in wound healing. Protein and glycoproteins synthesized by fibroblasts essentially allow the completion of the critical point in wound healing creating collagen and intermediate. 1 Angiogenesis induced by fibroblast growth factor is the foundation of this event. 2 Considering growth and protection of fibroblasts were crucial for human health, many researchers carried out various studies on fibroblasts. Medicinal plants play a leading role in many treatments performed on fibroblasts. The main reason for focusing on the medicinal plants is that they contain natural and therapeutic products and due to these products, treatments are performed with low side effects. 3,4 Researches about the protective effects of some plants take attention on fibroblasts. Many scientists carried out studies related to growth of fibroblasts as well as their protection by using various natural products. 5 Ozgen et al. 6 revealed fibroblast growth stimulation by plant extracts and compounds on human amnion fibroblasts (HAFs). Many studies performed with plants on fibroblasts revealed that natural compounds in the structure of the plants decreased oxidative stress. 7,8
Lichens have been used for therapeutic purposes in many areas with their unique ingredients, namely secondary metabolites. On the other hand, cumulative effects of lichen metabolites may appear in extracts and so some researchers tested the activities of the lichen extracts. Anti-mutagenic, 9 anti-oxidant, 10 antibacterial, 11 and anti-viral 12 activities described as important for human health were elucidated with lichens. Likewise, curative effects of lichens on some diseases such as ulcer, arthritis, tuberculosis, 13 hemorrhoids, and dysentery 14 were reflected in many research results. Additionally, it was determined that combined treatment methods of several lichen compounds with chemotherapy and radiotherapy had a place in some cancer types such as lung, 15 breast, 11 pancreas, 16 colon, gastric, 17 skin, 18 and brain. 19 Lichen extracts which contain multiple secondary metabolites also play an important role in various cancer treatments. 20
Although several studies have shown the protective activities of some plants on human fibroblasts, no studies describing the protective properties of lichen secondary metabolites could be found in the literature. Thus, the aim of this work was to evaluate cytotoxic (by 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT) and lactate dehydrogenase (LDH) assays), oxidative (by measuring total antioxidant capacity (TAC) and total oxidant status (TOS) levels), and genotoxic (by measuring 8-hydroxy-2′-deoxyguanosine (8-OH-dG) level) effects of olivetoric acid (OA) and physodic acid (PA) isolated from Pseudevernia furfuracea (L.) Zopf (Parmeliaceae) on cultured HAFs for the first time.
Materials and methods
Collection and identification of the lichen sample
The sample was collected in 2014 from different localities within Erzurum province of Eastern Anatolian region of Turkey. After collecting materials, they were exposed to dry in room conditions. Herbaria of this lichen were made. Comparing the obtained macroscopic and microscopic data with literature, 21,22 it was identified as P. furfuracea (KKEF-801).
Isolation of lichen secondary metabolites
After air-drying lichen samples were powdered with liquid nitrogen. Then, 150 g of P. furfuracea was extracted by 250 mL of ethanol solvent using a Soxhlet extraction apparatus throughout 5 days at 80°C, respectively. Extraction of P. furfuracea yielded 20.26% (w/w) of lichen substances.
The crude extract of P. furfuracea was collected and subjected to silica gel (70–230 mesh) column chromatography by eluting it with n-hexane:ethyl acetate (90:10, 80:20, 70:30, 60:40, 50:50, 40:60, 30:70, 20:80, 10:90 and 0:100) and ethyl acetate:methanol (90:10, 80:20, 60:40 and 40:60) solvent systems. At the end of this process, OA and PA were obtained with a yield of 11.25% and 13.88% (w/w), respectively.
Secondary metabolites obtained from lichens were diluted to different concentrations (6.25, 12.5, 25, 50, 100, and 200 mg/L) before the experimental setup. Dimethyl sulfoxide (DMSO) + relevant cell culture medium (2% DMSO) was used as negative control (control−).
Chemical structures of secondary metabolites have been previously reported by Emsen et al., 19 Culberson, 23 and Asahina and Shibata. 24 Proton nuclear magnetic resonance and carbon-13 nuclear magnetic resonance spectrums utilized to determine the chemical structures (Figure 1) of the compounds were obtained in CDCl3. Tetramethylsilane was used as standard and the chemical shift values (δ) were expressed as ppm (Tables 1–2).
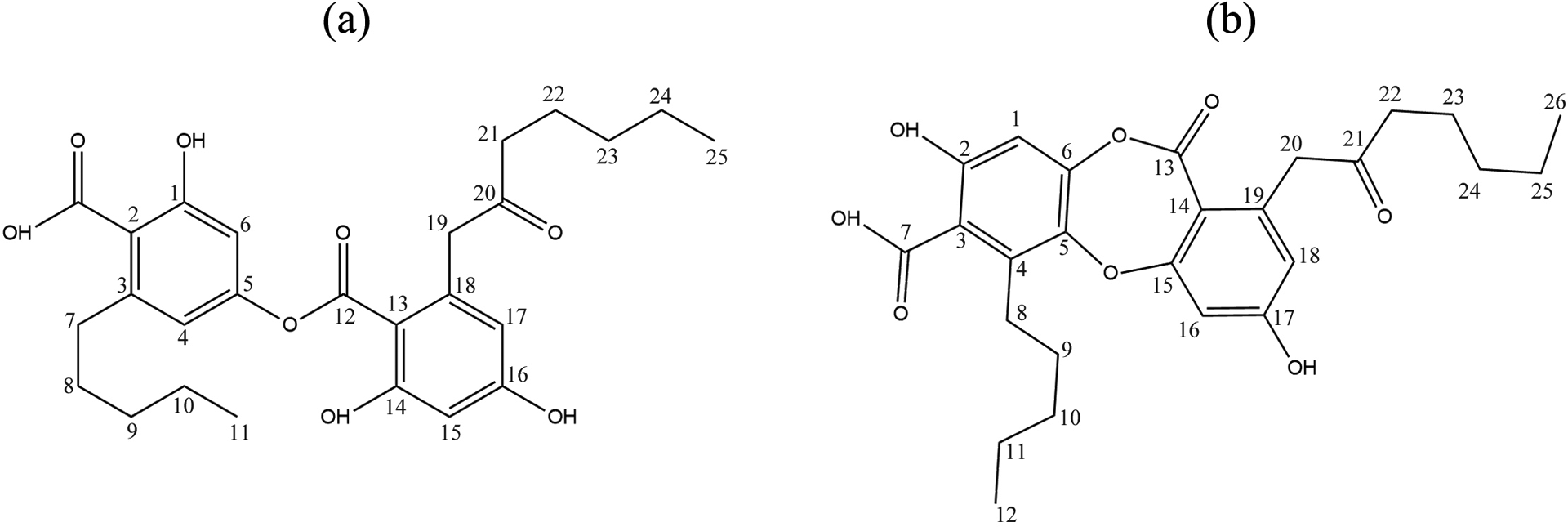
The chemical structures of the lichen secondary metabolites, (a) olivetoric acid and (b) physodic acid.
1H-NMR and 13C-NMR spectral data of OA.

1H-NMR: proton nuclear magnetic resonance; 13C-NMR: carbon-13 nuclear magnetic resonance; OA: olivetoric acid.
1H-NMR and 13C-NMR spectral data of PA.

1H-NMR: proton nuclear magnetic resonance; 13C-NMR: carbon-13 nuclear magnetic resonance; PA: physodic acid.
HAF cultures
Written informed consent was obtained from the patient for this study. Amniotic fluid was centrifuged at 1000 r/min for 10 min and supernatant was eliminated. After having thrown away the supernatant, fresh medium were added to the residue. While the medium was preparing, 10 mL BIOAMF-1 supplement (Biological Industries, Kibbutz Beit Haemek, Israel), 2 mL
MTT assay
The cells were seeded in 96-well plates. Cells were incubated at 37°C in a humidified 5% CO2/95% air mixture and treated with secondary metabolites at different concentrations (6.25, 12.5, 25, 50, 100, and 200 mg/L) for 48 h. MTT assay was carried out by commercially available kit (Cayman Chemical Company, Ann Arbor, Michigan, USA).
MTT reagent (10 µL) was added to the cell cultures. The plate was incubated in CO2 incubator at 37°C for 4 h and it was centrifuged at 400 g for 10 min. Approximately 100 µL of crystalline solvent solution was added to each well. The intensity of the formazan was measured at 570 nM wavelengths with Multiscan Go microplate reader (Thermo Scientific, Landsmeer, The Netherlands). In MTT assays, mitomycin-C (C15H18N4O5; Sigma, St Louis, Missouri, USA; at 10−7 M) chemotherapeutic agent was used as positive control.
LDH release assay
LDH assay was carried out in the culture medium by commercially available kit (Cayman Chemical Company).
Hundred microliters of LDH standard were added to relevant wells and 100 µL of medium on cells incubated for 48 h was added to other wells; 100 µL of LDH reaction solution was added to each well. The plate was incubated slightly for 30 min via orbital shaker (Labnet, Edison, New Jersey, USA) at room temperature. Spectrophotometric reading was carried out at 490 nM wavelengths. In LDH assays, mitomycin-C (10−7 M) was used as positive control.
TAC assay
TAC assay was carried out by commercially available kit (Rel Assay Diagnostics, Gaziantep, Turkey) on HAFs for 48 h. Kit assay is calibrated with a stable antioxidant of vitamin E analog called Trolox equivalent.
Cells incubated for 48 h were removed from the incubator. Medium on the precipitated cells was added to relevant wells. Standard solutions in kit were added to relevant wells. Reagent 1 solution was added to each well. First spectrophotometric reading was carried out at 660 nM wavelengths. After the first reading, reagent 2 solution was added to each well and the plate was incubated at room temperature for 10 min. Second spectrophotometric reading was carried out at 660 nM wavelengths. In TAC assays, ascorbic acid (Sigma-Aldrich; at 4 × 10−7 M) was used as positive control.
TOS assay
TOS assay was carried out by commercially available kit (Rel Assay Diagnostics) on HAFs for 48 h. Kit assay is calibrated with hydrogen peroxide (H2O2).
Cells incubated for 48 h were removed from the incubator. Medium on the precipitated cells was added to relevant wells. Standard solutions in kit were added to relevant wells. Reagent 1 solution was added to each well. First spectrophotometric reading was carried out at 530 nM wavelengths. After the first reading, reagent 2 solution was added to each well and the plate was incubated at room temperature for 10 min. Second spectrophotometric reading was carried out at 530 nM wavelengths. In TOS assays, H2O2 (Sigma-Aldrich, Darmstadt, Germany, at 5 × 10−4 M) was used as positive control.
Oxidative DNA damage assay
Oxidative DNA damage assay was carried out in the culture medium by commercially available DNA/ribonucleic acid oxidative damage kit (Cayman Chemical Company). The purpose of this assay is the determining of oxidative DNA damage in the cells via calculation of 8-OH-dG level. Experimental steps were performed in accordance with the kit procedure.
Statistical analyses
Mean values of different activities of secondary metabolites were compared with analysis of variance by Duncan’s multiple range test for comparing groups. A significance level of 5% was accepted for all comparisons. Median inhibitory concentration (IC50) values were calculated with probit regression analysis and associated 95% confidence limits for each treatment. Pearson’s correlation coefficients were used in order to determine relation levels among the variables. These calculations were carried out using Statistical Package for Social Sciences (SPSS, version 21.0; IBM Corporation, Armonk, New York, USA).
Results
Cytotoxicity testing
Cytotoxic activities of various concentrations of OA and PA on HAFs were assessed by MTT test. OA and PA showing cytotoxic effects depending on increase of the concentration caused toxicity on maximum 37.60% and 22.18% of HAFs, respectively (Figure 2). At the same time, IC50 values in Table 3 displayed that OA (571.27 mg/L) was more toxic compared with PA (3373.69 mg/L).

Viability rates in the HAFs exposed to OA and PA. Each value is expressed as mean ± standard deviation (n = 3). Values followed by different small letters differ significantly at p < 0.05. HAF: human amnion fibroblast; OA: olivetoric acid; PA: physodic acid.
IC50 values for HAFs exposed to different treatments (mg/L).a

IC50: median inhibitory concentration; PA: physodic acid; OA: olivetoric acid; HAF: human amnion fibroblast.
aValues followed by different superscript letters in the same column differ significantly at p < 0.05.
In addition to MTT test, LDH release analysis was performed in order to determine anti-proliferative activities on HAFs. Maximum LDH activities caused by OA and PA were defined at their maximum concentrations as 706.89 and 613.55 µU/mL. The differences among extracellular LDH activities of all treatments on HAFs were statistically (p < 0.05) significant (Figure 3).

LDH release levels in the HAFs exposed to OA and PA. Each value is expressed as mean ± standard deviation (n = 3). Values followed by different small letters differ significantly at p < 0.05. HAF: human amnion fibroblast; OA: olivetoric acid; PA: physodic acid; LDH: lactate dehydrogenase.
Anti-oxidative activities
Antioxidant capacities of different concentrations of OA and PA on HAFs were detected with TAC analysis. While ascorbic acid had the highest TAC level (29.55 mmol Trolox equivalent/L), minimum concentration of OA exhibited the lowest value (20.79 mmol Trolox equivalent/L). TAC level of ascorbic acid was statistically (p < 0.05) different from all other values. When investigated (Figure 4), a wavy appearance drew attention between concentration and TAC levels. High concentrations of OA and PA had low TAC. Considering maximum TACs, it was defined that concentrations of 25 mg/L of PA and OA significantly (p < 0.05) increased TAC level compared with control−.

TAC levels in the HAFs in the presence of OA and PA. Each value is expressed as mean ± standard deviation (n = 3). Values followed by different small letters differ significantly at p < 0.05. HAF: human amnion fibroblast; OA: olivetoric acid; PA: physodic acid; TAC: total antioxidant capacity.
Pro-oxidative activities
Oxidative stress levels caused by OA and PA on HAFs were examined via TOS analysis. It was revealed that TOS levels of control−, OA and PA were very close to each other. This case proved that OA and PA did not create oxidative stress on HAFs except for their maximum concentrations. Only concentrations of 200 mg/L of OA and PA significantly (p < 0.05) increased TOS level (28.32 and 27.84 µmol H2O2 equivalent/L, respectively) compared with control− (27.23 µmol H2O2 equivalent/L). H2O2 was separated from the other experiments with its TOS activity (53.66 µmol H2O2 equivalent/L) and this high value was statistically (p < 0.05) different from all TOS data (Figure 5).
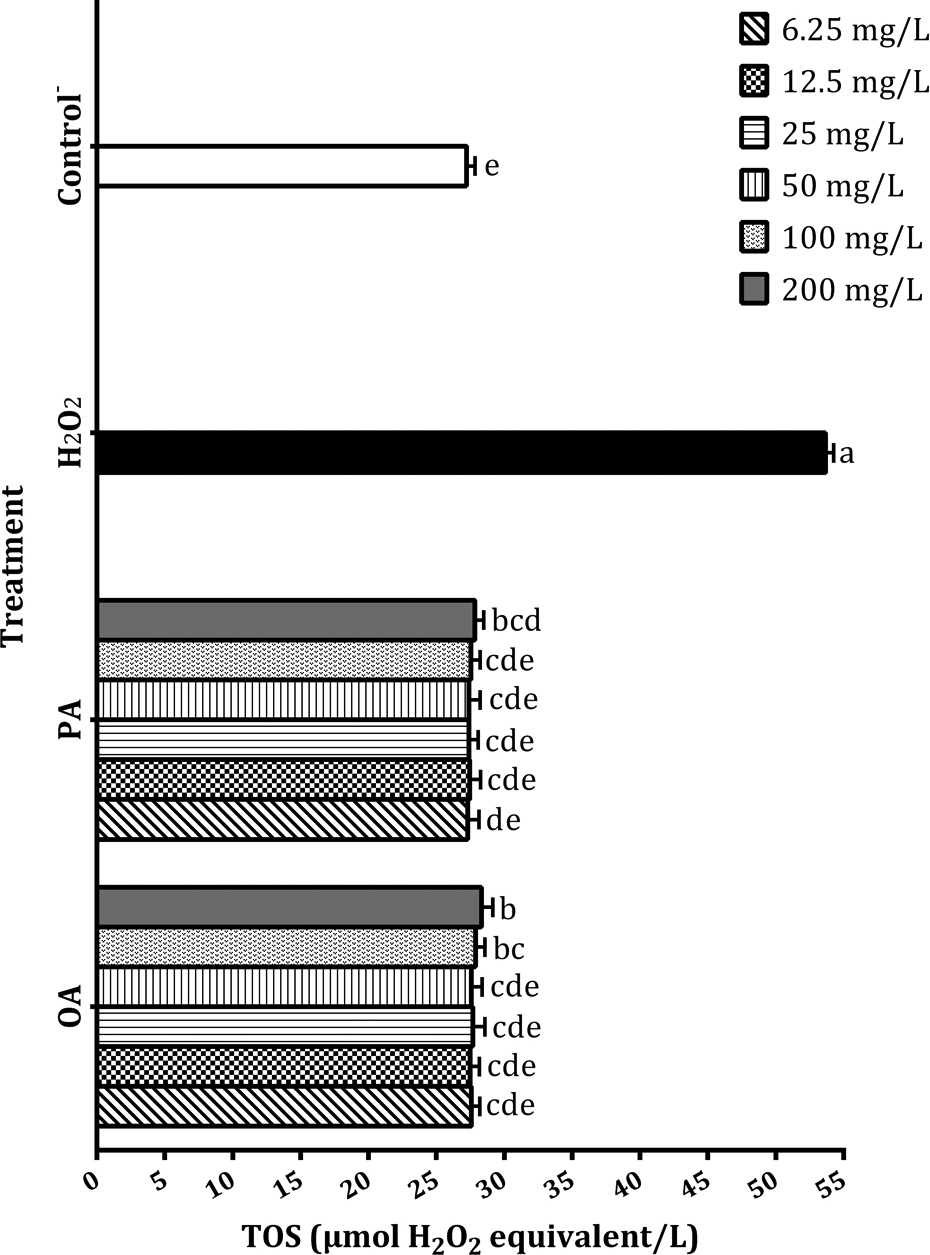
TOS levels in the HAFs in the presence of OA and PA. Each value is expressed as mean ± standard deviation (n = 3). Values followed by different small letters differ significantly at p < 0.05. HAF: human amnion fibroblast; OA: olivetoric acid; PA: physodic acid; TOS: total oxidant status.
Genotoxicity activities
Oxidative stress-induced DNA damage, emerged in HAFs by OA and PA, was measured with 8-OH-dG level occurring in the cells. It was observed that OA and PA treatments caused significantly (p < 0.05) low genotoxicity on HAFs as compared with mitomycin-C. It was also determined concentration-dependent genotoxic activity for OA and PA. Control− showing the lowest genotoxic effect (4.5 pg/mL) on HAFs was statistically (p > 0.05) indifferent from concentration of 6.25 mg/L of PA (5.03 pg/mL). Other OA and PA treatments significantly (p < 0.05) increased oxidative stress-induced DNA damage level compared with control−. Even so, these findings were too far and significantly (p < 0.05) different from the positive control value (15.2 pg/mL). Concentrations of 100 and 200 mg/L of OA among the tested metabolite experiments had great genotoxic activities (9.5 and 9.83 pg/mL, respectively) and the difference between these data was found insignificant (p > 0.05; Figure 6).

8-OH-dG adducts in the HAFs in the presence of OA and PA. Each value is expressed as mean ± standard deviation (n = 3). Values followed by different small letters differ significantly at p < 0.05. HAF: human amnion fibroblast; OA: olivetoric acid; PA: physodic acid; 8-OH-dG: 8-hydroxy-2′-deoxyguanosine.
Discussion
Plant-based products are preferred in the treatment of many diseases due to their natural components. Low level of side effects of herbal products was reflected in numerous studies. 25,26 Protective properties of some herbal medicines as well as therapeutic characteristics attracted attention. 27,28 Lichen metabolites are products that come into prominence because of aforementioned properties. 29
Relationship levels among various activities of OA and PA against HAFs were elucidated with correlation analyses. Based on cytotoxic activities of OA and PA on HAFs, it was detected that there were highly negative correlations between cell viability concentration and cell viability LDH activity. Pearson correlation coefficients (PCCs) for aforesaid binary variables ranged from −0.97 to −0.81. Another study about inhibition of proliferation of endothelial cells by OA depending on concentration was reported by Koparal et al. 30 Similarly, Emsen et al. 19 were determined that cytotoxic effect of OA increased in a concentration-dependent manner on U87MG glioblastoma cells. Inhibitory activities of OA against different organisms such as bacteria were also studied by some researchers. 31,32
Significantly (p < 0.01) positive correlations (PCCs = 0.93 and 0.91, respectively) between LDH activity and concentration for OA and PA were also remarkable (Tables 4 and 5). As shown in correlation between cell viability and LDH analyses, low concentrations caused minor toxicity on HAFs. Additionally, it was demonstrated that even high concentrations did not create high incidence of toxicity on HAFs (Figures 2 and 3).Towering IC50 data calculated for OA and PA also supported previously mentioned condition (Table 3).
Correlation between different variables for HAFs exposed to OA.

HAF: human amnion fibroblast; OA: olivetoric acid; TAC: total antioxidant capacity; LDH: lactate dehydrogenase; TOS: total oxidant status; 8-OH-dG: 8-hydroxy-2′-deoxyguanosine.
aPearson correlation coefficient.
bCorrelation is significant at the 0.01 level.
cCorrelation is significant at the 0.05 level.
Correlation between different variables for HAFs exposed to PA.

TAC: total antioxidant capacity; LDH: lactate dehydrogenase; TOS: total oxidant status; 8-OH-dG: 8-hydroxy-2′-deoxyguanosine; HAF: human amnion fibroblast; PA: physodic acid.
aPearson correlation coefficient.
bCorrelation is significant at the 0.01 level.
cCorrelation is significant at the 0.05 level.
In recent years, studies carried out in regard to proliferation inhibitory activities of PA were focused on the cancerous cells such as cervical cancer cells, 33 human melanoma, and colon cancer cells. 18 Oxidative stress and genotoxicity were important with regard to reasons of cytotoxicity caused by OA and PA. When considered OA metabolite, it was showed that TOS was negatively (at 0.05 level) and positively (at 0.01 level) correlated with cell viability and LDH activity, respectively. As for PA metabolite, it was detected a significant (p < 0.05) positive correlation between TOS and LDH activity (Tables 4 and 5). That is to say, anti-proliferative effects caused by OA and PA on HAFs might result from TOS. However, it was revealed that oxidative stress capacities of both metabolites on HAFs were not high level (Figure 5).
Oxidative stress levels occurred by different lichen secondary metabolites on various cells were investigated by many scientists. TOS of diffractaic acid, a lichen secondary metabolite was examined in human blood tissue and determined that this metabolite did not significantly increase TOS level. 34 Furthermore, it was pronounced that in the treatments performed in cultured human lymphocytes, many lichens such as Xanthoria elegans, 35 Bryoria capillaris, and Peltigera rufescens 36 might use as therapeutic agent by not creating oxidative stress.
Performed oxidative DNA damage test appeared that OA and PA had genotoxic effect on HAFs, albeit effect levels were low. It was indicated that genotoxic features of tested metabolites might give rise to HAFs anti-proliferation due to analyses carried out. Genotoxic increase depending on concentration for OA and PA was observed in correlation analyses (PCCs = 0.78 and 0.89, respectively). There were significantly (p < 0.01) negative correlations between 8-OH-dG level emerged in HAFs and cell viability for both tested metabolites. It was also reflected in correlation analysis results that 8-OH-dG level was associated with TOS activity (Tables 4 and 5). At the same time, high-degree positive correlations (PCCs = 0.89 and 0.94, respectively) between 8-OH-dG level and LDH activity, another cytotoxic effect indicator for OA and PA revealed genotoxicity–cytotoxicity relation (Tables 4 and 5).
There are many works reporting the genotoxic and anti-genotoxic activities of some lichens. Ceker et al. 37 reported that Usnea articulata and Usnea filipendula had anti-genotoxic potency against mutagenic agent, N-methyl-N′-nitro-N-nitrosoguanidine. A study about reducing of genotoxic effects of aflatoxin by methanol extract of Evernia prunastri was examined by Alpsoy et al. 38 Genotoxic effects of lichen secondary metabolites as well as extracts were also tested against various cell types. For example, it was measured in comet assay performed that the concentration of 120 µg/mL of (+)-usnic acid showed a genotoxic effect against V79 cells. However, it was determined that same concentration caused no genotoxicity in micronucleus test. 29 In the present study determined of cytotoxic, genotoxic, and oxidative status relationships of OA and PA, antioxidant capacities of forenamed metabolites on HAFs suggested about their protective properties. While TAC of concentrations from 50 mg/L to 200 mg/L of OA and PA were statistically (p > 0.05) indifferent from control−, concentration of 25 mg/L of both metabolites clearly raised TAC compared with control−. PA showed lower cytotoxic and genotoxic effect than OA against HAFs and also had high TAC with its concentration of 12.5 mg/L (Figure 4).
It was determined that many lichen species had high antioxidant capacity due to contained phenolic compounds. 39 Gastroprotective effects of rhizonaldehyde and rhizonyl alcohol isolated from Lobaria pulmonaria were detected in association with its antioxidant capacity. 40 2,2-Diphenyl-1-picrylhydrazyl (DPPH), superoxide anion capture, and reducing power activities in regard to anti-oxidative efficiency of PA from tested metabolites in the present study were investigated by some researchers. 18,41 In analogy to previous study, Mitrović et al. 31 found that OA used in the present study showed DPPH scavenging activity.
Taken together, data in the present study demonstrated that OA and PA had antioxidant features on HAFs when they used at low concentrations (6.25–25 mg/L). In conclusion, the tested lichen secondary metabolites, especially PA, promised sources of natural compounds with anti-oxidant and nongenotoxic activities and very low cytotoxic potential and warranted further researches.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
